Abstract
Lung cancer is one of the most common malignancies and causes the most cancer deaths in the United States. Targeted therapies have improved the survival of patients with advanced disease. Neurotrophic tropomyosin receptor kinase (NTRK) fusions are a rare oncogenic driver that has been targeted with the tumor-agnostic drug, larotrectinib. There are limited data on the treatment of non-small-cell lung cancer (NSCLC) with larotrectinib because of the rarity of this fusion in this population. We present the case of a patient who was diagnosed with SQSTM1-NTRK1 fused NSCLC with polymetastatic disease involving the brain and subsequently treated with a multidisciplinary approach via neurosurgical resection, radiotherapy, and larotrectinib. The combination of aggressive local treatments and systemic therapy is a relatively new treatment paradigm and represents a new area of research to optimize local control of metastatic lesions and potentially improve progression-free survival compared to the trials that show the efficacy of systemic monotherapies. The patient has experienced a sustained complete response to treatment almost 3 years later, and he has tolerated the drug without any significant adverse effects. The combination of systemic therapy with larotrectinib and aggressive local treatments could benefit patients with targetable fusions even with multiple metastatic lesions.
Plain language summary
Lung cancer is one of the most common malignancies and causes the most cancer deaths in the United States. Specific medications have been made to target cancer-causing mutations in the tumor, which have improved our ability to treat cancer. Larotrectinib is one of these targeted medications but has not been largely used in lung cancer. We present a patient who had widespread lung cancer and was treated with larotrectinib, surgery, as well as a highly accurate form of radiation to the brain. The patient is doing well with no sign of disease almost three years after beginning treatment.
Keywords
Introduction
Lung cancer is the third most commonly diagnosed malignancy and accounts for the highest number of cancer deaths in the United States each year. 1 Advances in novel systemic therapies, including tyrosine kinase inhibitors targeting oncogenic drivers, have improved survival outcomes for locally advanced and metastatic non-small-cell lung cancer (NSCLC). 2 A major example is the epidermal growth factor receptor mutations, which are now targeted with medications such as osimertinib, which has become a viable adjuvant therapy in patients with stage 1b-IIIa NSCLC who have this actionable mutation. 3 Other oncogenic drivers include mutations in anaplastic lymphoma kinase (ALK), ROS proto-oncogene (ROS-1), rearranged during transfection (RET), and neurotrophic tropomyosin receptor kinase (NTRK).
Central nervous system (CNS) metastases are relatively common among NSCLC patients, with up to 28.6% harboring CNS disease at presentation, and a 13% per-year incidence of CNS metastases. 4 Furthermore, NSCLC patients with tumor gene alterations in ALK or RET have a higher prevalence of brain tumors at presentation, suggesting a higher propensity of CNS spread with certain genetic alterations.
NTRK genes encode the proteins tropomyosin receptor kinase (TRK) A, B, and C. These proteins are integral in the development of both the central and peripheral nervous systems. They can be found in neuronal tissue as well as smooth muscle. Once activated by the binding of neurotrophin, they act through the Ras/MAPK and the PI3K/Akt pathways. NTRK fusions can cause the ligand-independent activation of intracellular pathways and lead to uncontrolled cell growth. There are a multitude of different fusions; however, common fusions in NSCLC are ETV6-NTRK3, TPM3-NTRK1, and SQSTM1-NTRK. These fusions have been implicated in multiple malignancies including NSCLC, thyroid cancer, cholangiocarcinoma, glioblastoma, and sarcomas. 5
Larotrectinib is a potent TRK inhibitor that acts as an adenosine triphosphate competitive inhibitor with activity against TRKA, TRKB, and TRKC. 6 It was approved by the Federal Drug Administration as the first NTRK fusion targeted therapy and the second tissue-agnostic therapy. 7 Given the rarity of NTRK fusion with only 0.1% of NSCLC patients harboring a clinically impactful variant, there is a paucity of data on managing these patients, especially in the metastatic setting. 8 Other case reports have described the treatment of various NTRK fusion NSCLC patients with larotrectinib or entrectinib alone with durable response, one of which harbored an NTRK2 fusion and a malignant pleural effusion, and the other harbored a novel NTRK3 fusion who was started on entrectinib after progression of their metastatic disease.9,10 Herein, we present a case of polymetastatic NTRK1-fused NSCLC treated with metastasis-directed therapy, with a durable and sustained complete response.
Methods
This case report was prepared according to the CARE guidelines. 11 The completed CARE checklist is shown in Supplemental File A. Patient consent was obtained prior to the submission of this article. All information was de-identified to protect the patient’s identity. Patient blood was collected and submitted to Guardant Health to perform the Guardant 360 cell-free DNA sequencing. Technical notes for Guardant 360 are shown in Supplemental File B. For next-generation sequencing and PD-L1 testing, formalin-fixed paraffin-embedded tissue was submitted. PD-L1 testing was performed at NeoGenomics utilizing immunohistochemistry. Technical notes for PD-L1 testing and HopeSeq NGS are shown in Supplemental File C.
Case report
The patient is a 66-year-old male with a past medical history of coronary artery disease status post-coronary artery bypass graft, congestive heart failure, diabetes, extensive smoking history, and choroidal melanoma who presented to a local emergency department with chief complaints of visual disturbances and left eye pain. Physical examination was significant for visual field deficits in the left eye. Initial workup including computed tomography (CT) chest, abdomen, and pelvis revealed a left upper lobe hilar mass, left upper lobe nodules, and lytic lesion in the fifth lumbar vertebrae. Subsequent positron emission tomography-computed tomography (PET/CT) showed fluorodeoxyglucose-18 (FDG) avidity in the corresponding areas found on CT. Initial bronchoscopy showed a left upper lobe bronchial obstruction, but pathology was not diagnostic. Magnetic resonance imaging (MRI) of the brain revealed at least 25 enhancing brain lesions, the largest of which was a partially cystic lesion in the left occipital lobe measuring up to 4 cm (Figure 1). Repeat bronchoscopy with endobronchial ultrasound and biopsy revealed the diagnosis of lung adenocarcinoma. This pathological diagnosis, in conjunction with the imaging findings suggesting CNS metastatic disease, established a diagnosis of polymetastatic lung adenocarcinoma. Guardant 360 testing further revealed an SQSTMI-NTRK1 fusion. Please see Supplemental File B for the full report.
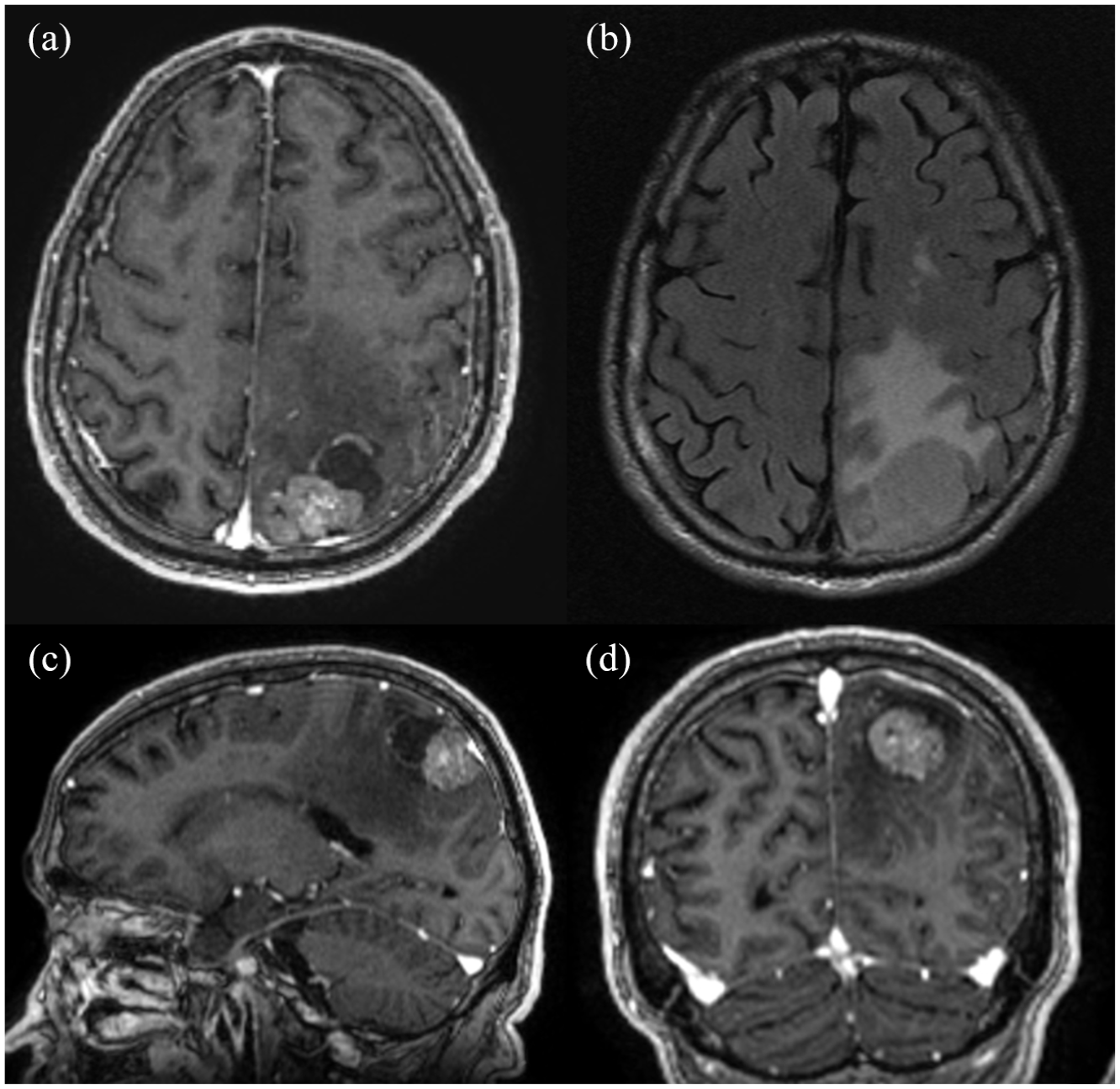
Preoperative magnetic resonance imaging brain with and without intravenous gadolinium contrast shows brain lesions from non-small-cell lung cancer. There is a partially cystic enhancing lesion measuring 4 cm in the left occipital lobe seen on T1c on axial slice (a). There is a moderate amount of surrounding edema seen on T2/FLAIR, also on axial slice (b). Sagittal (c) and coronal (d) sections are also shown in T1c. Not shown are scattered supratentorial small enhancing lesions consistent with brain metastases measuring 1 cm or less.
His case was presented at a multidisciplinary tumor board, and, given his medical comorbidities and SQSTM1-NTRK1 fusion, he was offered larotrectinib 100 mg twice daily therapy. Entrectinib was also discussed but felt to be more risky given risks for elevated creatinine levels and more musculoskeletal issues including edema. 12 After multidisciplinary discussion, he underwent craniotomy with gross total resection of a symptomatic 4 cm left occipital lobe mass. Postoperative MRI showed expected post-surgical changes with nonspecific mild residual enhancement along the inferior resection margin. Subsequently, he met with radiation oncology, and he was offered postoperative stereotactic radiosurgery (SRS) to the 12.5 cm 3 resection cavity. In addition, he was recommended SRS to 11 intact brain metastases, with the largest measuring 1.2 cm. The cavity and all intact lesions received 27 Gy in three fractions delivered every other day. Figure 2 shows the radiation treatment fields and corresponding dose volume histogram. Pathology from the craniotomy specimen was consistent with metastatic poorly differentiated adenocarcinoma, and HopeSeq performed on both the craniotomy and lung samples confirmed the SQSTM1-NTRK1 fusion. Please see Supplemental File C for the full report.
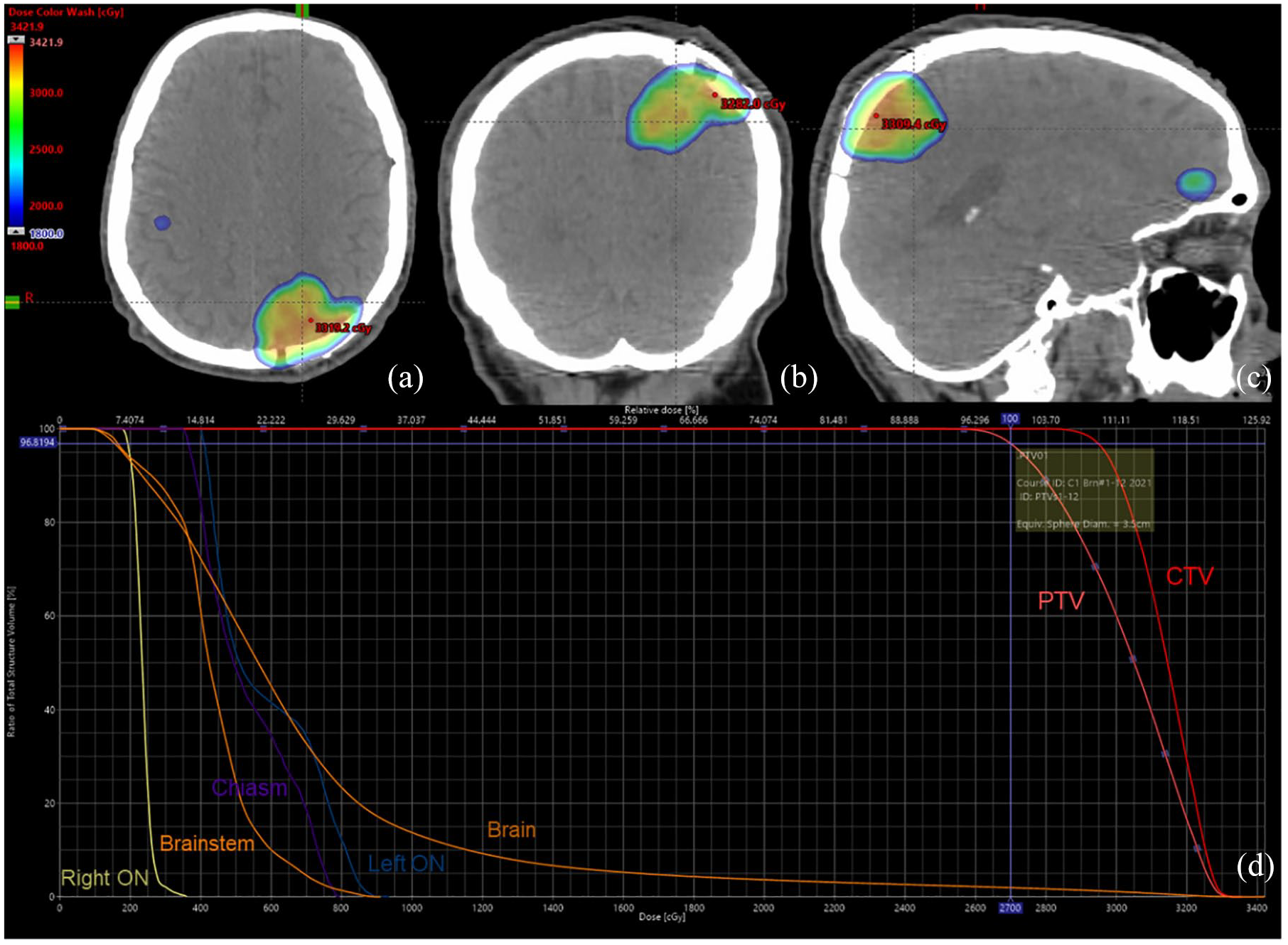
Treatment planning and dosimetric evaluation for fractionated SRS. Computed tomography scans from the simulation are shown with dose color wash overlay in the axial (a), coronal (b), and sagittal (c) views. DVH shows dose metrics for target structures and critical OARs. CTV (red) and PTV (salmon) are target structures of the left occipital lobe postoperative cavity. The prescription dose is 27 Gy in three fractions delivered every other day to the PTV. Crosshair on the DVH highlights that the volume of PTV receiving 100% of the prescription dose is 97%. OARs shown include brain (brown), left optic nerve (ON; blue), optic chiasm (purple), brainstem (orange), and right ON (yellow). In panels a and c, there are other smaller brain lesions seen, and there were a total of 12 areas treated in this SRS plan all treated to 27 Gy in three fractions every other day.
Follow-up MRI of the brain, performed 5 months postoperatively, showed partial response in his brain metastases and expected post-surgical changes (Figure 3). No evidence of radiation necrosis was observed. Five months after initiating larotrectinib, PET/CT showed resolution of FDG avidity in the lung and bone metastases (Figure 4). He remains disease-free over 3 years following SRS and continues on larotrectinib with regular surveillance brain and systemic imaging. His most recent MRI showed no evidence of disease (Figure 5). He cooks and prepares healthy food for himself and his family to manage his type 2 diabetes. He is active and enjoys going on walks with his grandkids. He has tolerated larotrectinib with no appreciable toxicities. See Figure 6 for a graphical depiction of the events from diagnosis to follow-up.
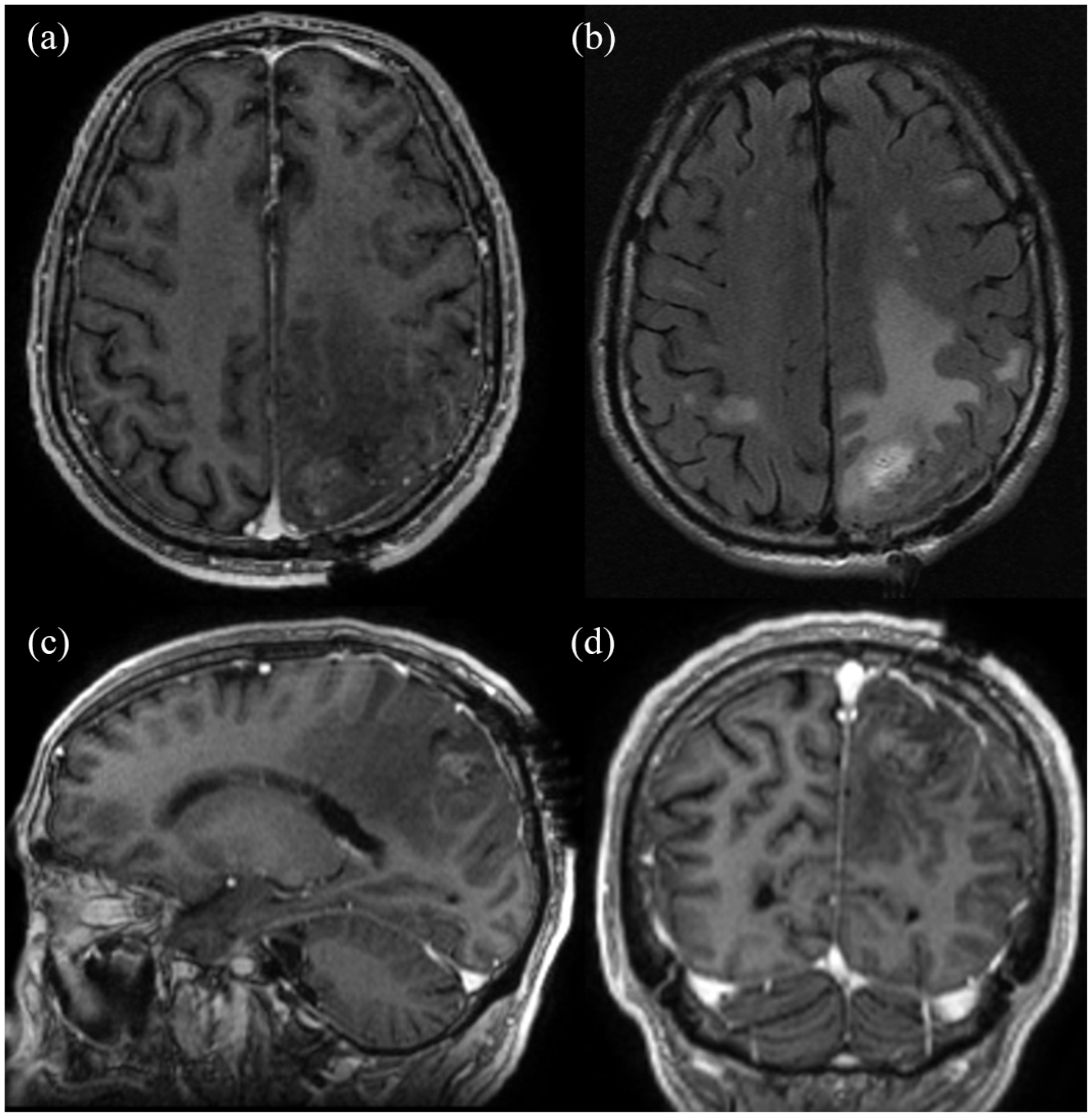
Postoperative brain MRI with and without intravenous contrast following left occipital craniotomy. There is interval resection of a left occipital lobe lesion with residual blood products, post-surgical changes, and residual enhancement suggestive of possible residual disease on T1c in the axial plane (a). There is evidence of residual moderate edema without any midline shift on T2/FLAIR (b). Sagittal (c) and coronal (d) slices are shown as well using T1c. Again, not shown is a redemonstration of numerous scattered enhancing bilateral parenchymal brain lesions consistent with metastases, overall stable in size and number.

(a) Initial PET/CT showing FDG avidity in the hilum and L5 and (b) post-treatment PET/CT showing interval resolution of FDG avidity.
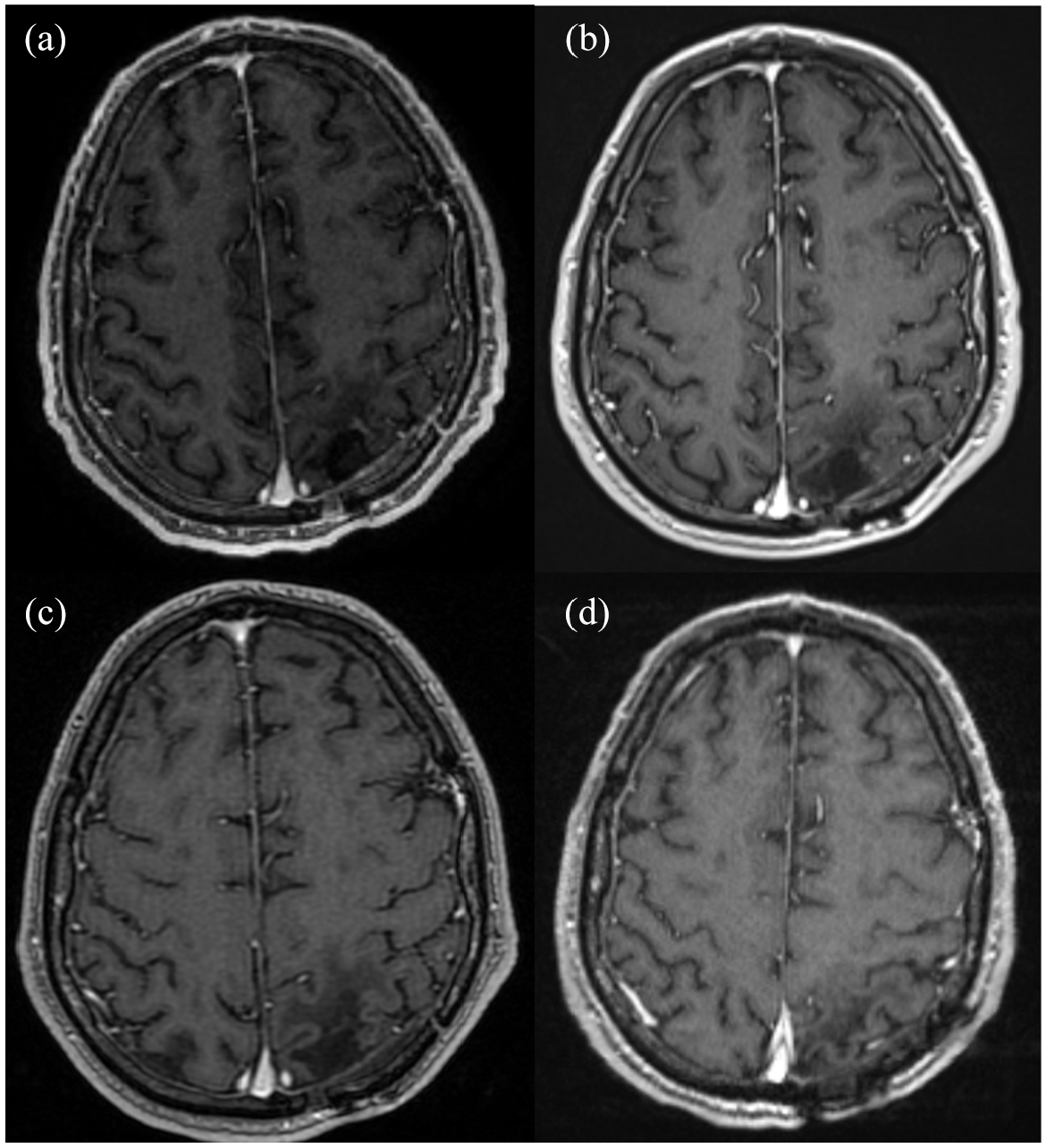
Follow-up brain magnetic resonance imaging scans in T1 sequence with intravenous contrast. At 3 months after stereotactic radiosurgery (a), there was complete resolution of contrast enhancement. There were some residual cystic and inflammatory changes consistent with the post-treatment effect. His additional 11 lesions showed interval resolution as well. This was consistently demonstrated as his follow-up scans did not show any evidence of disease at 6 months (b), 16 months (c), and 20 months (d).

Timeline of pertinent events, procedures, and diagnostic tests from the patient presentation to the most recent follow-up.
Discussion
Our patient demonstrates a multidisciplinary approach in the management of a rare entity in NSCLC, which continues to evolve over time given the ambiguity of treatment paradigms when combining new targeted therapies with intracranial penetration and radiation therapy. There are some data to suggest that NSCLC patients with brain metastases may respond to monotherapy with larotrectinib or entrectinib.13,14
In a pooled analysis of 20 TRK fusion-positive NSCLC patients, larotrectinib treatment resulted in an overall response rate (ORR) of 73%, with 10 patients (67%) having a partial response (PR) and 1 patient (7%) achieving complete response (CR). 13 Among the eight patients with CNS metastases at baseline, ORR was 63%, and all five patients had PR. In addition, the initial pooled analysis of the phase I and II trials that led to the larotrectinib approval showed that 9 out of 12 patients with CNS metastases responded to the drug. 14 Molecular data characterized 29 upstream fusion partners from this analysis, with NTRK3 (55%) and NTRK1 (40%) as the most common downstream fusions.
These data suggest that larotrectinib may induce intracranial tumor response with the potential to cross the blood–brain barrier (BBB). Given its robust ORR, larotrectinib may penetrate the CNS to therapeutic levels to produce an antitumoral effect in TRK-fused NSCLC brain metastases. However, neither study investigated CNS response as a primary or secondary endpoint, and additional research is needed to fully uncover the intracranial efficacy of larotrectinib. Interestingly, larotrectinib was specifically designed to have a greater affinity for the efflux pumps of the BBB endothelial layer, to decrease the neurological side effects that often affected patients taking entrectinib. 15
For our patient, because of the large symptomatic lesion, it was decided to pursue multimodal treatment with upfront resection followed by radiotherapy. To date, there are no reported cases of trimodal therapy with larotrectinib, neurosurgery, and radiotherapy in metastatic NSCLC. In the present case, our patient impressively had complete resolution of all systemic diseases following larotrectinib therapy. Intracranially, concurrent larotrectinib with postoperative and definitive SRS also ultimately generated CR, and radiotherapy may further enhance the previously reported therapeutic benefit of larotrectinib.13,14 Our studies demonstrate acceptable outcomes with a complete and durable response to treatment both intracranially and extracranially without any reported toxicity including radiation necrosis.
Another option for this patient, especially as observed in some practices in more recent years, would be for systemic therapy alone followed by surgery or SRS if there was no intracranial response. For our patient, one could have considered local treatment for the largest symptomatic lesion and monitored the additional smaller ones to see if they would respond to the larotrectinib. Potential benefits of monitoring may be to reduce radiation exposure to the brain and minimize long-term radiation necrosis risks and cognitive changes. Potential risks, however, could be additional growth, leading to neurologic events including new-onset seizures, paresthesias, or other potential neurologic changes that, if not treated urgently, could be permanent or even life-threatening.
Moreover, our patient did not experience any grade 3 or higher treatment-related adverse events based on the Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. Larotrectinib is overall well-tolerated, and the most common adverse events in the initial trials of larotrectinib were fatigue, cough, constipation, anemia, neutropenia, and increased alanine aminotransferase levels. 13 In comparison, entrectinib can predispose patients to more CNS-related side effects, such as cerebellar dysfunction, cognitive disorders, and memory impairment. 12 Additional treatment-related adverse events can include left ventricular dysfunction, pleural effusion, and fluid retention. Dose discontinuations were seen in up to 39% of patients in initial trials. 6 Fortunately, our patient overall tolerated their treatment well without any treatment breaks or dose modifications despite their medical comorbidities, including congestive heart failure and diabetes. Furthermore, by avoiding entrectinib, they were spared from any unnecessary potential cardiac and neurologic side effects. 12
The advent of targeted therapies and the durable responses that they may provide has created a frontier in the treatment of intracranial metastatic disease. The landscape is evolving as we define more patients in terms of an oligo disease state, allowing more flexibility to treat aggressively, which has been shown to improve overall survival in some studies. 16 When a patient, such as our case, presents with numerous brain metastases, it is paramount to proceed in a multidisciplinary manner and consider the various possibilities of medical therapies and local modalities. Though challenging because of the rarity of this mutation in the NSCLC population, prospective data are needed to further define the effect of combining multimodal treatment in patients in this disease state. Since medical treatments and radiation technologies have improved so dramatically, treatments can be tailored to the patient and whole brain radiotherapy does not have to be relied upon to treat these patients with extensive disease.
Conclusion
Our case depicts a successful multimodal approach to a rare disease entity. The treatment of NSCLC is evolving, and, in the case of our patient, there seems to be a benefit of aggressive upfront control of intracranial metastases without notable toxicity including radiation necrosis. This case could guide further research into the addition of radiotherapy and surgery to metastatic NSCLC with targetable mutations.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251317134 – Supplemental material for Metastatic SQSTM1-NTRK1 fused non-small-cell lung cancer treated with larotrectinib and stereotactic radiosurgery resulting in durable complete response: a case report
Supplemental material, sj-docx-1-tam-10.1177_17588359251317134 for Metastatic SQSTM1-NTRK1 fused non-small-cell lung cancer treated with larotrectinib and stereotactic radiosurgery resulting in durable complete response: a case report by Colton Betts, Ari Kassardjian and Arya Amini in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251317134 – Supplemental material for Metastatic SQSTM1-NTRK1 fused non-small-cell lung cancer treated with larotrectinib and stereotactic radiosurgery resulting in durable complete response: a case report
Supplemental material, sj-docx-2-tam-10.1177_17588359251317134 for Metastatic SQSTM1-NTRK1 fused non-small-cell lung cancer treated with larotrectinib and stereotactic radiosurgery resulting in durable complete response: a case report by Colton Betts, Ari Kassardjian and Arya Amini in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-3-tam-10.1177_17588359251317134 – Supplemental material for Metastatic SQSTM1-NTRK1 fused non-small-cell lung cancer treated with larotrectinib and stereotactic radiosurgery resulting in durable complete response: a case report
Supplemental material, sj-pdf-3-tam-10.1177_17588359251317134 for Metastatic SQSTM1-NTRK1 fused non-small-cell lung cancer treated with larotrectinib and stereotactic radiosurgery resulting in durable complete response: a case report by Colton Betts, Ari Kassardjian and Arya Amini in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
