Abstract
Gastroenteropancreatic (GEP) neuroendocrine tumors (NETs) represent the most common subtype of NETs. The incidence of all NETs, and specifically GEP NETs, has risen exponentially over the last three decades. Only within the past several years have these tumors been appropriately classified, allowing for meaningful drug development. Broadly, some of the most exciting drug classes being developed for patients with well-differentiated GEP NETs include newer types of peptide receptor radionuclide therapy (PRRT) or combinations which increase the potency of lutetium-177 (177Lu)-Dotatate, novel multi-target receptor tyrosine kinase inhibitors (RTKIs) and immunotherapy modalities, beyond checkpoint inhibitors, which seek to unleash the immune system against NETs. Specifically looking at newer types of PRRT, somatostatin receptor antagonists and alpha-emitter radionuclides each have demonstrated the ability to elicit greater DNA damage than 177Lu-Dotatate in preclinical models. Early clinical experiences with each of these agents suggest they may be more cytotoxic than 177Lu-Dotatate. Other approaches seeking to build upon the DNA damage created by 177Lu-Dotatate include combinations of PRRT with radiosensitizers such as heat shock protein 90 inhibitors, hedgehog inhibitors, chemotherapy combinations, and triapine. Many of these combinations have just begun to be tested clinically. With regards to novel RTKIs, some of the ones which have demonstrated potent cytoreductive potential include cabozantinib and lenvatinib. Other RTKIs which are further along the clinical development spectrum and have demonstrated benefit in randomized trials include surufatinib and pazopanib. And though single-agent immune checkpoint inhibitors have not demonstrated significant anti-tumor activity in patients with GEP NETs, outside of certain biomarker selected subsets, somatostatin receptor-directed chimeric antigen receptor (CAR) T cells and vaccines such as SurVaxM, which targets survivin, represent two means through which NET-directed immunity may be modulated. The potential of these agents, if clinically realized, will likely improve outcomes for patients with well-differentiated GEP NETs.
Introduction
Gastroenteropancreatic (GEP) neuroendocrine tumors (NETs) are a heterogenous group of neoplasms which represent the most common subtype of NETs. 1 Though considered to be rare, they represent the second most prevalent gastrointestinal malignancy after colorectal cancer in the United States. 2 Well-differentiated GEP NETs pose unique management challenges for providers because they are often detected at advanced stages, are characterized by diverse molecular profiles without obvious targetable mutations, and can cause patient morbidity directly (e.g. organ compromise, small-bowel obstructions) or indirectly (e.g. carcinoid syndrome, carcinoid heart disease).3,4 As such, improving treatments for patients with well-differentiated GEP NETs has become an increasing priority for the oncologic community. Though a number of therapeutics are currently approved for the treatment for patients with GEP NETs, many of them are limited by a lack of cytoreductive capacity.5–9 Herein we describe novel therapeutics being developed for the treatment of advanced well-differentiated GEP NETs. We specifically focus on agents with relevant preclinical or clinical studies published within the last 5 years to provide a contemporary review of systemic therapy-focused research developments within the field.
Peptide receptor radionuclide therapy (PRRT)
PRRT has been a transformative therapy for patients with somatostatin receptor (SSTR)-expressing advanced GEP NETs. First developed in Europe in the early 1990s, PRRT with lutetium-177 (177Lu)-Dotatate garnered European Medicines Agency and Food and Drug Administration (FDA) approval for patients with GEP NETs in 2017 and 2018, respectively.10,11 The regulatory approvals were based upon findings from the NETTER-1 trial and the Erasmus Medical Center Study.12,13 In the NETTER-1 trial, while patients receiving PRRT experienced marked improvements in progression-free survival (PFS) and overall survival (OS) compared with patients receiving a high dose of somatostatin analog (SSA) alone, the objective response rate (ORR) was only 18%. A recent post-hoc analysis from the study suggests that patients with bulky tumors (defined as >3 cm in size) in any location experience reduced tumor cytoreduction and PFS compared with patients without such bulky tumors. 14 Given the advanced disease burden of most patients, there exists a clear need to improve the cytoreductive ability of PRRT. Several novel approaches to build upon the ORR of PRRT, including alpha particle therapy, albumin-bound radionuclide carriers, SSTR antagonists, radiation sensitizers, and DNA damage repair inhibitor combinations, will be discussed in the subsequent paragraphs. The role for surgery prior to PRRT, to remove bulky lesions which are less likely to demonstrate tumor shrinkage from the therapy, is being discussed at various NET trials planning meetings. While this approach is intriguing, given the post-hoc NETTER-1 trial findings, it has not yet been tested prospectively.
177Lu and yttrium-90 (90Y) are β-emitting radionuclides which represent the two most common therapeutic radionuclides utilized in PRRT. In comparison with β-emitting radionuclides, α emitters possess a higher linear energy transfer. α emitters have demonstrated an ability to elicit greater amounts of DNA double-strand breaks in a cell cycle-independent manner compared with β emitters and overcome resistance to β emitters in vitro.15–17 Bismuth-213 (213Bi), actinium-225 (225Ac), and lead-212 (212Pb) are three experimental α emitters which are currently being tested in clinical trials in patients with well-differentiated GEP NETs. 213Bi was the first α emitter tested in seven NET patients (five of whom possessed GEP NETs) who were refractory to 90Y or 177Lu-based PRRT, with previously chronicled results demonstrating cytoreduction and prolonged disease control in treated patients. 18 Early clinical results with 225Ac were reported in 32 patients with GEP NETs with progressive (56%) or stable disease (44%) after 177Lu-Dotatate therapy. 19 Patients received 225Ac 100 kilobecqeurel (kBq)/kg every 8 weeks up to a cumulative dose of 55,5000 kBq. Among 24 patients assessable for response, 15 (62.5%) achieved partial responses and nine (37.5%) achieved stable disease; no patient deaths were documented by the investigators after a median follow-up period of 8 months. With regards to toxicity, 20 (62.5%) of all patients experienced grade 1/2 hematologic adverse events; no patients experienced grade 3/4 neutropenia, thrombocytopenia, or lymphopenia, or grade 3/4 non-hematologic adverse events. 212Pb-Dotatate is being tested in an ongoing phase I study in patients with SSTR-avid NETs. 20 In contrast to the previously reported studies with α emitters, this study will test the efficacy of the agent class in PRRT-naïve patients, thus providing a baseline ORR for possible future comparative studies. One of the potential disadvantages with α emitters is the release of daughter radionuclides from the chelator, which can result in circulation of radionuclides with a long half-life, possibly causing severe toxicity. Longer-term follow-up is needed with α emitters before the toxicity profile of these radioligands is fully understood.
Novel carriers for 177Lu such as EB-TATE have also been explored based upon promising preclinical data which suggest that the albumin-bound entity can prolong radionuclide circulation times, increase tumor/kidney dose ratio, and improve tumor regression in SSTR-expressing xenografts. 21 In the first in human study, five patients received a single dose of 177Lu-EB-TATE while three patients received a single dose of 177Lu-Dotatate. 22 Patients treated with the novel carrier demonstrated increased circulation time and a 7.9-fold increase in tumor dose. However, dose delivery to the kidney and bone marrow was increased by 3.2 and 18.2 fold, respectively, compared with patients receiving 177Lu-Dotatate. While it remains unclear whether agents of this class can be safely administered to patients with NETs, a phase I study of 177Lu-EB-TATE is ongoing in this patient population (NCT03478358).
Current forms of PRRT rely on SSTR agonist molecules to bind to target receptors on NETs; however, preclinical evidence suggests SSTR antagonist molecules can occupy more receptor sites on tumor tissue with lower rates of dissociation, leading to greater tumor and reduced normal organ uptake of radionuclide, respectively. 23 177 Lu-OPS201 (also known as 177Lu DOTA-JR11 or Satoreotide tetraxetan) is one of the antagonists that is now being tested in a clinical trial. In vivo experiments in xenograft models demonstrated that mice treated with 177Lu-OPS201 compared with 177Lu-Dotatate experienced longer periods of tumor stabilization and longer median survival times. 24 A subsequent pilot study tested four patients who received treatment with both 177Lu-OPS201 and 177Lu-Dotatate. 25 Patients underwent whole-body imaging and SPECT/CT imaging post-treatment to measure biodistribution and underwent gallium-68 ( 68 Ga)-Dotatate scans to assess response. Patients were found to have a tumor dose which was 1.7–10.6 times greater with the SSTR antagonist compared with the SSTR agonist. These findings prompted a phase I study of 177Lu-OPS201 in well-differentiated NET patients who were PRRT naïve. 26 The total study sample size is 40 patients; however, preliminary results were reported after 20 patients (90% GEP NET) were evaluable for outcome assessment. Patients were treated with two cycles of the agent at 3-month intervals with six patients receiving one cycle and 14 receiving two cycles. ORR was 45%, disease control was 85%, and median PFS was 21 months in treated patients. Grade 4 myelosuppression was observed in four (57.1%) patients after cycle two, necessitating a protocol amendment to limit total bone marrow exposure to one gray and reduce the cycle two dose by 50%. Full study results need to be reported prior to determining the further clinical development of 177Lu-OPS201, and potentially other SSTR antagonists, to ensure safety of the treatment modality.
Adding radiation sensitizers which target DNA damage repair, DNA damage induction, and cell cycle signaling pathways to PRRT are some of the combinatorial strategies which have demonstrated the most preclinical promise. 27 Poly(ADP-ribose) polymerase inhibitors (PARPi), which interfere with base excision repair, a type of single-strand DNA repair, have been combined with 177Lu-Dotatate in NET models. The rationale for this combination is that single-strand breaks elicited by 177Lu-Dotatate can no longer be repaired by NET cells in the presence of PARPi, creating double-strand breaks and, ultimately, cell death. 28 Investigators published results from experiments of 177Lu-Dotatate in combination with PARP is (DHQ, veliparib) in BON-1 (pancreatic NET) cell lines. 29 In BON-1 monolayer experiments, cell viability was markedly decreased in cells treated with the combination therapy compared with those treated with either therapy alone. This was confirmed in BON-1 spheroid models with proliferation assays. The cell-line studies confirmed that PARPi augmented the DNA damage elicited by 177Lu-Dotatate by increasing double-strand DNA breaks as measured by γ-H2AX accumulation. Based on this preclinical rationale, a phase I/II study of 177Lu-Dotatate plus olaparib in PRRT-naïve progressive GEP NETs will begin enrollment shortly (NCT04086485). Patients who enroll on the study will begin the olaparib 2 days prior to the first infusion of 177Lu-Dotatate and will continue the PARPi twice daily until 4 weeks after the final PRRT administration.
Heat shock protein 90 (Hsp90) represents a promising target for GEP NETs because of its ubiquitous overexpression in these tumors. 30 Hsp90 inhibitors such as onalespib are well-established radiosensitizers, increase DNA damage and apoptosis in treated cancer cells, and have demonstrated preclinical promise in NET models both in vitro and in vivo. 31 In BON1 xenograft models, mice treated with onalespib on days 1–4 (30 mg/kg intraperitoneal administration) and 177Lu-Dotatate on days 2–4 [0.0037 Gigabecquerel (GBq) IV] demonstrated improved tumor growth slowing, survival, and tumor regression compared with monotherapy with either agent. Specifically, complete responses were increased from 8% in xenografts treated with 177Lu-Dotatate monotherapy to 29% in xenografts treated with onalespib and 177Lu-Dotatate. 32 The cytoreductive potential of the combination is intriguing; however, it remains to be seen whether this can be translated to patients.
The hedgehog cellular signaling pathway is dysregulated in GEP NETs of multiple types, thereby making it an attractive drug target. 33 Hedgehog inhibitors as monotherapy have demonstrated anti-tumor effect in NET models, and investigators have examined combining it with 177Lu-Dotatate in GOT1-bearing (small intestinal NET cell line) BALB/c nude mice. 34 Mice were treated with either sonidegib (80 mg/kg twice weekly administered orally), a single injection of 30 MBq 177Lu-Dotatate IV, or a combination of both. Mice treated with a combination of sonidegib and 177Lu-Dotatate demonstrated greater than 3-fold tumor regression and had longer time to progression compared with mice treated with either monotherapy. On pharmacodynamic studies, differential pathway activation (Wnt/B-catenin, PI3K/AKT/mTOR) was observed in tumors of mice treated with the combination therapy compared with tumors treated with 177Lu-Dotatate monotherapy. While promising preclinically, this combination has also not been tested clinically yet.
Another radiation sensitizer being combined with PRRT is the ribonucleotide reductase inhibitor triapine. Ribonucleotide reductase is the rate-limiting enzyme in DNA synthesis and repair, as it is the sole enzyme responsible for the conversion of ribonucleotide to deoxyribonucleotide diphosphates. 35 Theoretically, by inhibiting the enzyme, the DNA damage elicited by PRRT would not be able to be repaired and lead to NET cell death. Based on this rationale, an ongoing phase I study of 177Lu-Dotatate plus triapine in PRRT-naïve patients with well-differentiated GEP NETs has been initiated. 36
Though not novel treatments, novel approaches combining chemotherapy with PRRT also have been recently reported. The CONTROL NETS trial is an ongoing randomized parallel assignment study in which patients with pancreatic and small intestinal NETs are randomized in a 2:1 fashion to the combination of capecitabine plus temozolomide (CAPTEM) and177Lu-Dotatate or either 177Lu-Dotatate or CAPTEM monotherapy. CAPTEM has well-established cytoreductive activity in pancreatic NETs and less established cytoreductive activity in non-pancreatic NETs; however, it is a natural partner for PRRT due to its DNA-damaging mechanism of action. 37 In the report from the small intestinal NET cohort of the CONTROL NETS study, after a median follow-up time of 35 months, median 15-month PFS time was 90% and 92% in combination therapy and monotherapy arms, respectively. 38 The ORR with the combination was 31% compared with 15% with 177Lu-Dotatate monotherapy, at the cost, however, of increased toxicity. Among patients treated with the combination and monotherapy, 75% and 38%, respectively, experienced at least one grade 3 adverse event. In the pancreatic NET cohort, after 34 months of follow-up, 76% and 67% of pancreatic NET patients treated with the combination and 177Lu-Dotatate monotherapy experienced 12-month PFS, respectively. The ORR with the combination was 68% compared with 33% with CAPTEM monotherapy. In the pancreatic NET cohort, the rates of grade 3 adverse events were not significantly different between the two arms (28% versus 33%). Longer-term follow-up is needed to see if any OS difference is observed with the combination treatment in both cohorts. In patients with pancreatic NETs, however, the ORR observed with the combination is intriguing and may represent a cytoreductive option for patients with particularly bulky disease.
Results from phase I/II study of everolimus in combination with 177Lu-Edotreotide in patients with GEP NETs and lung NETs were reported. 39 The rationale for this study stemmed from lung NET xenograft data where the combination of everolimus and177Lu-Dotatate elicited improved tumor shrinkage compared with monotherapy with either agent, and early clinical data in patients with GEP NETs where the combination appeared to be cytoreductive (ORR of 44%). 40 In the study, 11 patients (10 GEP NET) patients received four doses of 177Lu-Edotreotide (3.7 GBq per dose) concurrently with everolimus in the trial. The maximum tolerated dose (MTD) of the mTOR inhibitor was determined to be 10 mg; however, the ORR of the combination was only 9%. After a median follow-up of 18.9 months, median PFS in the treated cohort was 23.3 months. A total of 36% of patients experienced grade 3 toxicities including infection, fatigue, pneumonitis, and neutropenia, while no patients experienced grade 4 toxicities. Based on only the modest cytoreduction elicited by the treatment combination, along with the toxicity profile, it is unlikely that this combination is promising enough to be further developed in patients with GEP NETs. The activity of this combination strategy will likely need to be explored in patients with lung NETs to see if it merits further study. A summary of ongoing PRRT studies with several of the previously discussed novel therapeutics is presented in Table 1.
Ongoing or soon to be activated studies of novel PRRT compounds or combinations in patients with gastroenteropancreatic NETs.

DOR, duration of response; GEP, gastroenteropancreatic; GI, gastrointestinal; MTD, maximum tolerated dose; NET, neuroendocrine tumor; ORR, objective response rate; OS, overall survival; PFS, progression-free survival; PRRT, peptide receptor radionuclide therapy; RP2D, recommended phase II dose; SSTR, somatostatin receptor.
Multi-targeted receptor tyrosine kinase inhibitors (RTKIs)
Anti-angiogenic agents have been a cornerstone of therapy for NETs given their vascular nature and vascular endothelial growth factor (VEGF)-driven oncogenesis. 41 Though sunitinib has been approved for patients with progressive pancreatic NETs, the ORR elicited by the compound is modest. 5 As such, newer anti-angiogenic agents have been developed which offer promising activity in patients beyond those with pancreatic NETs.
Cabozantinib is a multi-targeted RTKI which targets VEGF receptor 2 and tyrosine-protein kinase Met (c-Met). The drug was tested in a two-cohort parallel phase II study in third-line and beyond pancreatic NET and carcinoid (non-pancreatic NET) patients. 42 Cabozantinib was administered at a starting dose of 60 mg daily; however, 80% of patients required a dose modification to 40 mg daily. Most patients with non-pancreatic tumors possessed tumors of suspected gastrointestinal origin (78%). The ORR in both patients with pancreatic NETs and non-pancreatic NETs was 15%. The PFS in patients with pancreatic NETs and non-pancreatic NETs was 21.8 months and 31.4 months, respectively. The most common grade 3/4 adverse events observed were hypertension (13%), hypophosphatemia (11%), diarrhea (10%), and fatigue (5%), consistent with the adverse event profile of the drug seen in other diseases.43,44 Based on these study findings, the randomized phase III CABINET trial has been initiated. This study is comparing cabozantinib with placebo in patients with well-differentiated pancreatic or carcinoid NETs who have progressed on at least one prior systemic therapy; a recent amendment has removed the need for patients to have progressed on everolimus prior to being eligible to enroll on the study (NCT03375320). Other combinatorial regimens with cabozantinib are also being explored in patients with progressive well-differentiated GEP NETs to build upon the cytoreductive activity seen with the single agent. 45
Lenvatinib is a multi-targeted RTKI which targets VEGF receptors 1–3 and fibroblast growth factor receptor (FGFR) 1–4. The TALENT study was a multi-cohort phase II study which tested lenvatinib in patients with well-differentiated pancreatic and gastrointestinal NETs with progression on at least one prior line of treatment. 46 In this trial, 55 patients with pancreatic NETs and 56 with gastrointestinal NETs received treatment with the agent. The ORR in patients with pancreatic NETs and gastrointestinal NETs was 42.3% and 16.4%, respectively. Amongst the pancreatic NET cohort, the median PFS was 15 months while median OS was 29.2 months. Amongst the gastrointestinal NET cohort, the median PFS was 15.4 months and median OS was not reached. The most common grade 3/4 adverse events were hypertension (22%), fatigue (11%), and diarrhea (11%). Though the utility of cross-trial comparisons between RTKIs is limited, the ORR elicited in either patient cohort with lenvatinib was the highest reported to date.
Surufatinib is a RTKI which targets VEGF receptors 1–3, FGFR1, and colony-stimulating factor 1 receptor (CSF1R). Based upon promising findings from a phase I/II study, in which surufatinib demonstrated an ORR of 19% and 15% in patients with well-differentiated pancreatic and extra-pancreatic NETs, respectively, two phase III studies with the drug were initiated in China. 47 One of these trials was SANET-ep where patients with well-differentiated extra-pancreatic NETs were randomized in a 2:1 fashion to 300 mg daily of surufatinib or placebo. 48 Most patients (60.6%) had suspected gastrointestinal primary tumors; 83.8% of tumors were grade 2. Median PFS of surufatinib-treated patients was 9.2 months, while median PFS of placebo-treated patients was 3.8 months [hazard ratio (HR) 0.33, p < 0.0001]. The most common adverse events ⩾grade 3 were hypertension (36.4%), proteinuria (19.4%), and anemia (7.0%) in the surufatinib-treated patients. The other study was SANET-p, in which patients with well-differentiated pancreatic NETs were randomized in a 2:1 fashion to 300 mg daily of surufatinib or placebo. 49 Most patients possessed grade 2 tumors (86.6%). Median PFS of surufatinib-treated patients was 10.9 months, while median PFS of placebo-treated patients was 3.7 months (HR 0.49, p = 0.001). The most common adverse events ⩾grade 3 were hypertension (38%), proteinuria (10%), and hypertriglyceridemia (7%). As these two phase III studies were done in Chinese populations, a recent phase I/II study reported interim results with surufatinib in a US population. 50 In this study, 16 patients with pancreatic NETs and 16 patients with extra-pancreatic NETs (median lines of prior therapy 3) were treated with surufatinib. The recommended phase II dose (RP2D) of the compound was determined to be 300 mg, identical to the dose identified in the Chinese studies. The median duration of treatment in all patients was 19 weeks, with an ORR of 9.4%. Of the treated patients, 50% reported ⩾grade 3 adverse events; the most common adverse events reported were hypertension, fatigue, diarrhea, proteinuria, and nausea. Based on the activity from these studies, surufatinib has been granted FDA Fast Track Designations for treatment of advanced pancreatic and extra-pancreatic NETs.
Pazopanib is a RTKI with activity against VEGF receptors 1–3, platelet-derived growth factor (PDGF) receptor α and β, and c-KIT, which has demonstrated single-agent activity in extra-pancreatic NETs and combinatorial activity in pancreatic NETs. The single-agent study with pazopanib was a randomized phase II study of pazopanib 800 mg daily versus placebo in 171 patients with well-differentiated extra-pancreatic NETs (66% with small-bowel NETs). 51 The median PFS in the pazopanib and placebo arms was 11.6 and 8.5 months, respectively (HR 0.53, p < 0.0005). The ORR in the pazopanib arm was only 2.1%. Of pazopanib-treated patients, 60.7% experienced treatment-related grade 3/4 adverse events. The most common grade ⩾3 adverse events in patients in the treatment arm were hypertension (26.9%), transaminase increases (18%), fatigue (7.9%), nausea (4.5%), and diarrhea (4.5%). Pazopanib has been combined with temozolomide in a phase I/II study in patients with well-differentiated pancreatic NETs. Temozolomide was administered on days 1–7 and 15–21 with pazopanib at a flat dose of 400 mg daily. The MTD for the combination was established at a temozolomide dose of 75 mg/m2 and pazopanib dose of 400 mg. The ORR was 25% (all partial responses), median PFS was 12.1 months, and median OS was 36.5 months. The most common treatment-related adverse events were hepatic toxicity (16%), nausea (5%), and fatigue (5%). The phase II portion of this study remains ongoing, and if found to confirm the early results may represent a valuable treatment combination for patients with pancreatic NETs.
As noted previously, sunitinib is an established therapeutic for pancreatic NETs and is a RTKI which targets VEGF receptors 1–3, PDGFR α and β, CSF-1R and fms-like tyrosine kinase 3 (FLT-3), among other targets. 5 Given the hypoxia induced by the agent in the tumor microenvironment, investigators have combined it with evofosfamide, a DNA alkylator prodrug which is activated specifically under hypoxic conditions. In the phase II single-arm SUNEVO trial (GETNE-1408), patients received treatment with evofosfamide 340 mg/m2 days 8, 15, and 22 every 4 weeks and sunitinib 37.5 mg/day continuously. 52 The primary endpoint was ORR using a Simon’s two-stage design. The prespecified ORR was not met after stage 1, with only 11.8% of patients achieving a response. Median PFS was 10.4 months in study patients. Grade 3/4 adverse events occurred in 64.7% of patients, with neutropenia (33.3%), fatigue (16.7%), thrombocytopenia (11.1%), and hand-foot syndrome (5.6%) occurring most commonly. Given the toxicity profile of this combination, along with its lack of activity, it does not appear this combination will be further studied in patients with pancreatic NETs.
Axitinib is a RTKI which targets VEGF receptors 1–3, PDGF receptor, and c-KIT, and has been tested in patients with well-differentiated extra-pancreatic NETs. In a single-arm phase II study of 30 patients (77% with gastrointestinal primaries) pretreated with one prior line of therapy, study patients were treated with axitinib 5 mg twice daily continuously. 53 The primary endpoints of the study were PFS and 12-month PFS rate. The primary endpoints were met with a median PFS of 26.7 months and 12-month PFS rate of 74.5%. The ORR was 3% with one partial response. Grade 3/4 hypertension, however, occurred in 63% of patients, leading to treatment discontinuation in 20% of patients. Though this agent prolonged disease progression in patients with extra-pancreatic NETs, its toxicity profile may make it a challenge to utilize, particularly in patients with baseline sympathetic dysregulation (e.g. carcinoid syndrome).
Several novel RTKIs have demonstrated promising single-agent and combinatorial anti-tumor activity (ORR), in addition to prolonging PFS for patients with GEP NETs. It remains to be seen how these agents will fare in randomized studies against current standard-of-care treatments. A summary of the reported monotherapy trials with RTKIs is shown in Table 2.
Reported monotherapy studies of RTKIs in patients with well-differentiated gastroenteropancreatic NETs.
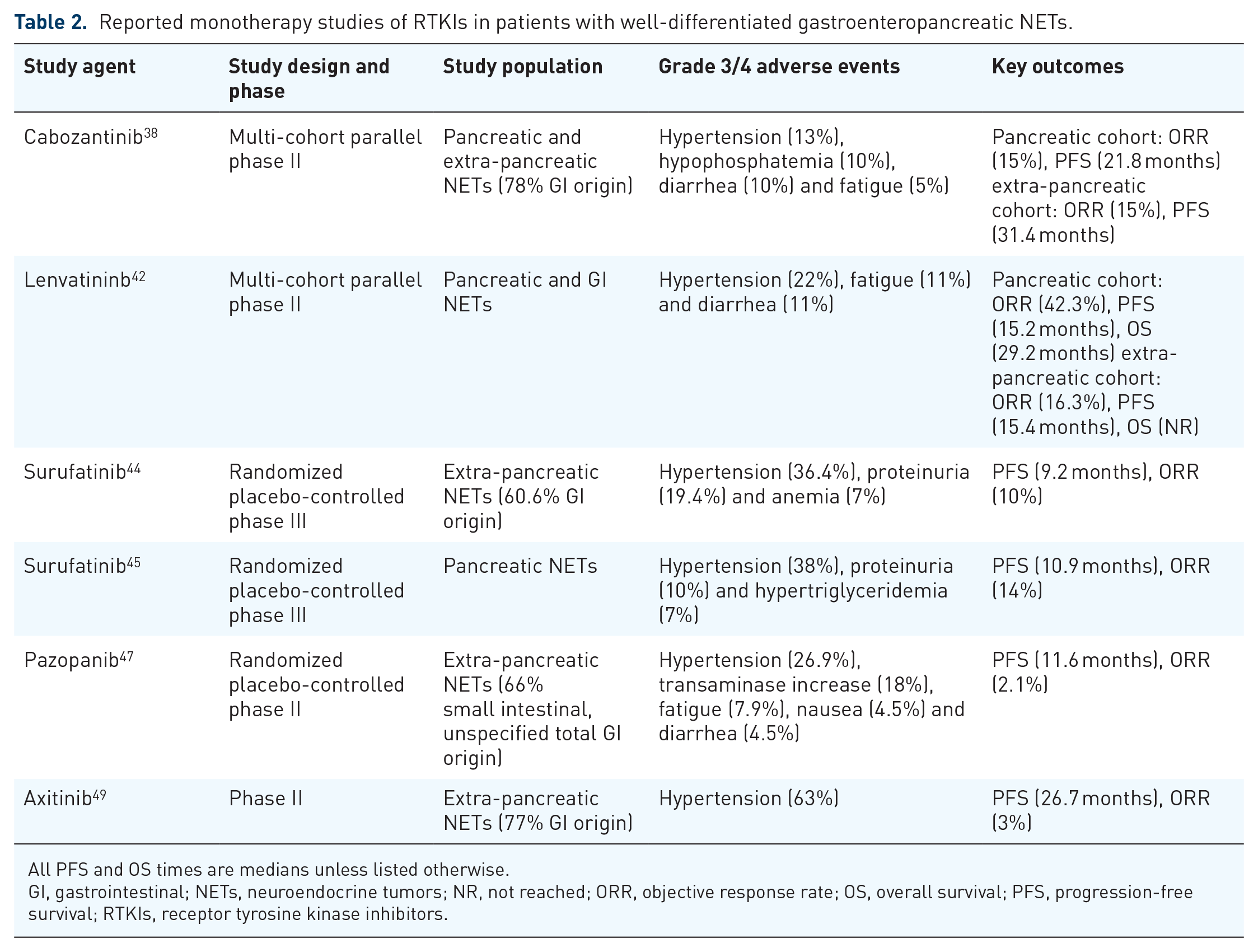
All PFS and OS times are medians unless listed otherwise.
GI, gastrointestinal; NETs, neuroendocrine tumors; NR, not reached; ORR, objective response rate; OS, overall survival; PFS, progression-free survival; RTKIs, receptor tyrosine kinase inhibitors.
Immunotherapy agents
Immunotherapy with immune checkpoint inhibitors (ICIs) and chimeric antigen receptor (CAR) T cells has transformed the treatment landscape for patients with certain tumors.54–59 The durability of responses achieved with these agents in patients with refractory tumors is striking, and as such a tremendous interest exists to incorporate some of these approaches into the treatment armamentarium for patients with well-differentiated NETs. In the subsequent paragraphs, we will discuss ongoing or presented efforts to translate the benefit of immunotherapy to patients with well-differentiated GEP NETs.
Single-agent treatment with ICIs has not produced meaningful anti-tumor effects for patients with well-differentiated GEP NETs, apart from the rare subset of patients with microsatellite instability-high (MSI-H) tumors. However, among patients with microsatellite-stable GEP NETs, there may be specific subsets in whom the agents may be more effective (e.g. those with tumors with elevated Ki-67 indexes). The initial study to test activity of the anti-programmed cell death protein 1 (PD-1) ICI pembrolizumab was the Keynote-028 study. 60 In this phase Ib trial, patients with well-differentiated pancreatic NETs and extra-pancreatic NETs with programmed death-ligand 1 (PD-L1)-positive tumor expression were treated with pembrolizumab 10 mg/kg every 2 weeks. The primary endpoint of the study was ORR. Ultimately, 16 patients with pancreatic NETs and 25 patients with extra-pancreatic NETs (56% GEP NET total) received treatment. The ORR in patients with pancreatic NETs was 6.3%, and was 12% in patients with extra-pancreatic NETs; only partial responses were observed. Median PFS in the pancreatic NET and extra-pancreatic NET cohorts was 4.5 months and 5.6 months, respectively. In the well-differentiated NET cohort of the Keynote-158 phase II study, 107 patients (77.5% GEP NET, 16.1% PD-L1 positive) were treated with 200 mg pembrolizumab every 3 weeks. 61 The primary endpoint of the study was ORR. The ORR was 3.7% (all partial responses) and all responses occurred in PD-L1-negative patients. All the responders in this study had GEP NET tumors (three pancreatic, one rectal) and three of four patients had Ki-67 indexes of ⩾10–15%.
Spartalizumab is a second-generation anti-PD-1 antibody whose activity has been explored in patients with NETs. In a phase II study of 116 patients (56% well-differentiated GEP NETs), patients were treated with spartalizumab 400 mg every 4 weeks. 62 Only one patient with a GEP NET experienced a partial response; all other responses were observed in lung NET patients. No causality was observed between PD-L1 expression and response to the ICI. Toripalimab is another second-generation anti-PD-1 antibody which has been tested in patients with NETs. A phase Ib study of the agent was conducted in 23 patients with NETs with a Ki-67 index ⩾10% and positive PD-L1 expression (⩾5% tumor cell or >1% immune cell expression), with a primary objective of ORR. 63 Only five patients possessed well-differentiated NETs, while the remainder possessed neuroendocrine carcinomas (NECs) or mixed adenoneuroendocrine carcinoma (MANEC). Among the patients with NETs, the ORR was 40%; both patients who experienced partial responses possessed PD-L1 positive disease.
The anti-PD-1 antibody nivolumab was tested in combination with temozolomide in a phase II multi-cohort study of patients with well-differentiated NETs and small-cell lung cancer. 64 The scientific basis behind this combination arises from the antigen-presenting cell priming capacity of temozolomide, in certain dose administrations. 65 Theoretically, this improved activation of antigen-presenting cells would synergize with anti-PD-1 checkpoint inhibitors which improve T-cell infiltration into the tumor microenvironment. Among 12 efficacy-evaluable patients with well-differentiated NETs, 25% demonstrated a partial response while 67% demonstrated stable disease. All three responding patients possessed tumors with Ki-67 indexes ⩾15%, and two of three patients possessed GEP NETs. Though preliminary efficacy has been suggested from the interim analysis from the well-differentiated NET cohort, the final study results remain pending.
Adding anti-angiogenic agents to ICIs appears to be another means of improving anti-tumor activity of ICIs. Preclinical studies suggest that anti-angiogenic agents such as bevacizumab may modulate the tumor immune microenvironment to improve anti-tumor immune cell/pro-tumor immune cell ratio and decrease the expression of regulatory checkpoints on tumor-infiltrating lymphocytes. 66 Based upon this rationale, the anti-PD-L1 antibody atezolizumab was combined with bevacizumab in a basket study in rare tumors; two of these cohorts included patients with well-differentiated pancreatic NETs and extra-pancreatic NETs. 67 Patients received atezolizumab 1200 mg and bevacizumab 15 mg/kg every 3 weeks, and the primary endpoint of the study was ORR. The ORR in the pancreatic NET cohort and extra-pancreatic cohort was 20% and 15%, respectively. Median PFS in the pancreatic NET and extra-pancreatic NET cohorts was 19.6 months and 14.9 months, respectively. The most common grade 3/4 adverse events were hypertension (20%) and proteinuria (15%). Pretreatment and on-treatment biopsies were obtained from all study patients and correlative analyses remain pending; transcriptomic analysis from these biopsies may identify certain patient subsets who derive the optimal benefit from the treatment combination.
Given the modest activity observed with single-agent ICIs in patients with well-differentiated NETs, several studies have explored combinatorial ICI strategies. One of these was the DUNE trial, a multi-cohort phase II study of the anti-PD-L1 antibody durvalumab plus the anti-cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) antibody tremelimumab. 68 Cohorts two and three in the study included grade 1/2 pancreatic NETs and grade 1/2 gastrointestinal NETs, respectively. Cohorts one and four in the study included lung NETs and grade 3 GEP NETs, respectively. Durvalumab was administered 1500 mg every 4 weeks while tremelimumab was administered 75 mg every 4 weeks. After the first four cycles during which both drugs were co-administered, durvalumab was continued as a monotherapy. The primary endpoint for cohorts 1–3, 9-month clinical benefit rate, was not met in a recent presentation of the study. 69 Another study testing combined CTLA-4 and PD-1 blockade is the phase II DART trial, which explored the activity of nivolumab plus ipilimumab in patients with NETs; outcomes from the extra-pancreatic NET cohort of this study were recently published. 70 Among 32 included patients, 18 possessed grade 3 NETs while 14 possessed grade 1/2 NETs. The ORR in patients with grade 1/2 NETs and grade 3 NETs was 0% and 44%, respectively; it has not been reported whether patients with grade 3 disease possessed well-differentiated grade 3 tumors or poorly differentiated NEC. At this juncture, it is unclear whether the addition of anti-CTLA-4 antibodies to anti-PD-1 or anti-PD-L1 antibodies offers much benefit over single-agent ICI therapy in patients with well-differentiated NETs.
Other novel immunotherapy approaches being developed for patients with GEP NETs involve SSTR-directed CAR T cells and vaccines. Preclinical data with a second-generation CAR T cell construct were presented at NANETs 2019. 71 This specific construct included two octreotide molecules as an extra-cellular moiety and Cluster of Differentiation 28 (CD28) as a co-stimulatory signal. The compound was tested in CM, BON1, and QGP1 cell lines; CM and BON1 cell lines overexpressed SSTR 2/5 compared with QGP1 cell lines by flow cytometry. Compared with untreated T cells, CAR T cells secreted significantly higher levels of IFN-γ, IL-2, and TNF-α after co-incubation with CM or BON1 cells (p < 0.01). Anti-SSTR CAR T cells infiltrated target tumors and significantly reduced the growth of subcutaneous CM and BON1 xenografts in mouse models (p = 0.01). Clinical studies with this SSTR-directed CAR have not been initiated. SurVaxM is a survivin-targeted vaccine which is now being tested clinically in NET patients; it had originally received an orphan disease designation for glioblastoma in 2017. Survivin belongs to the Inhibitor of Apoptosis Protein (IAP) family and is overexpressed in NETs and in other tumor types; overexpression of the protein has been shown to be a negative prognostic marker in NETs. 72 Three NET patients have been treated thus far in an ongoing phase I study of the agent plus octreotide. The study is open to patients with GEP NETs and lung NETs (NCT03879694). More mature data with the vaccine and the clinical translation of anti-SSTR-directed CAR T cells are needed to assess whether these may be meaningful options for patients with GEP NETs. A summary of ongoing and reported immunotherapy studies in patients with GEP NETs is shown in Table 3.
Reported or ongoing immunotherapy studies in patients with well-differentiated gastroenteropancreatic NETs.

CBR, clinical benefit rate; DCR, disease control rate; DOR, duration of response; GEP, gastroenteropancreatic; NECs, neuroendocrine carcinoma; NETs, neuroendocrine tumors; ORR, objective response rate; OS, overall survival; PFS, progression-free survival, TTP, time-to-progression; WD, well differentiated.
Miscellaneous agents
PEN-221 represents a miniaturized antibody–drug conjugate which combines a SSTR agonist molecule with a cytotoxic payload. The advantage of the miniaturized construct is that it can readily perfuse into hypoxic and poorly vascularized tumor environments. 73 In the case of the PEN-221, the microtubule targeting agent is the maytansinoid emtansine (DM1). PEN-221 targets SSTR2-expressing cells, and based on anti-tumor activity in SSTR2-expressing NET xenografts was moved into the clinical arena. 74 An ongoing phase I/II clinical trial is testing PEN-221 in GEP NETs and small-cell lung cancer. The phase I results from this study were presented at ASCO 2018. 75 Of the initial treated cohort of 23 patients, 61% possessed GEP NETs. The patients included were those that were naïve to PRRT. The MTD was established at 18 mg every 3 weeks. The most frequent adverse events were fatigue (43%), nausea (43%), vomiting (26%), abdominal pain (22%), and anorexia (22%). No patients experienced an ORR; however, three of seven efficacy-evaluable GEP NET patients did experience a minor response. Expansion cohort enrollment in patients with small intestinal and pancreatic NETs is ongoing.
Fosabretabulin is a phosphorylated prodrug of the natural compound combretastatin A4, which is a tubulin depolymerizing agent. Mechanistically, the agent acts as a vascular-disrupting agent, targeting and destroying well-established tumor blood vessels. 76 Fosabretabulin has demonstrated single-agent activity in GEP NETs and thus was combined with everolimus in a phase I study. 77 In this study, 17 patients with GEP NETs were treated with escalating doses of everolimus daily and fosabretabulin either 60 mg/m2 every 3 weeks or weekly. The RP2D was everolimus 10 mg with weekly fosabretabulin 60 mg/m2; only five patients experienced grade 3 toxicities. Among the 16 patients who completed the 12-week study, only one patient progressed; all other patients experienced stable disease. It is unclear given the cytostatic effect of the combination, rather than cytotoxic effect, to what extent further development will be pursued.
In an analysis of pancreatic NET tumor samples, cyclin-dependent kinase 4 (CDK4) and its downstream target phosphorylated-Rb1 were found to be overexpressed in 58% and 68% of samples, respectively. 78 In this same study, the CDK4/6 inhibitor palbociclib reactivated Rb1, induced cell cycle arrest, inhibited BON1 and QGP1 cell lines in vitro, and demonstrated tumor regression in a QGP1 xenograft model. Based on these findings, a single-arm phase II study of palbociclib was initiated in patients with well-differentiated pancreatic NETs (PALBONET). 79 In this trial, patients with pancreatic NETs were treated with a flat dose of palbociclib at 125 mg daily for 3 weeks on, 1 week off; 21 patients enrolled and 19 were evaluable for response. Among the initial 11 patients, the ORR was 0%. Because the specified efficacy threshold for activity was not met after stage I of the study, further accrual was halted. Median PFS and OS in all study patients were 2.6 months and 18.7 months, respectively. Five patients (23.8%) developed grade 3/4 neutropenia and two (9.5%) experienced grade 3/4 thrombocytopenia. Another CDK4/6 inhibitor ribociclib has been trialed in patients with foregut NETs (50% pancreatic NETs), and despite not demonstrating any cytoreduction (0% ORR) did demonstrate a more promising PFS (median 10.4 months). 80 An ongoing study is exploring the combination of ribociclib with everolimus in patients with NETs of foregut origin based on preclinical rationale which suggests synergy between the combination (NCT03070301).
A rare subset of NET tumors harbor fusion mutations in NTRK genes which encode the neurotrophin receptors TRK A, TRKB, and TRKC. A large comprehensive gene-profiling study identified these mutations in 0.3% of all NETs. 81 The NTRK fusions create unique susceptibility to the TRK inhibitors larotrectinib and entrectinib. 82 A case report of a GEP NET patient with a NTRK3 fusion and bulky progressive disease responding to entrectinib for 12 months highlights the power of such targeted strategies. While rare, the discovery of actionable mutations such as NTRK fusions and MSI-H status may make next-generation sequencing compulsory testing for all patients with progressive GEP NETs.
Conclusion
The agents discussed in the preceding paragraphs represent some of the drug development efforts aimed at expanding the treatment armamentarium for patients with well-differentiated GEP NETs. Though many agent classes were covered, our list is by no means exhaustive. Other agents which were not discussed and are being tested in patients with GEP NETs include those that have been repurposed from other disease indications, such as metformin and tamoxifen.83–85 Amongst the drugs covered, we believe some of the most intriguing include novel types of PRRT such as alpha emitters and SSTR antagonists, along with radiation sensitizers which seek improve the potency of 177Lu-Dotatate. Newer RTKIs such as lenvatinib, cabozantinib, and surufatinib seem to possess more potent anti-tumor activity than prior RTKIs and may be utilized as monotherapy or backbones upon which to add additional agents. Targeted immunotherapy approaches such as SSTR-targeting CAR T cells and the SurVaxM vaccine may unleash the immune system against GEP NETs in a way that checkpoint inhibitors have not been able to do. Penultimately, comprehensive next-generation sequencing efforts may reveal additional Achilles’ heels of GEP NET tumors which can be targeted, such as NTRK fusions. Finally, though many of these treatments have been tested in settings when patients are no longer candidate for surgical debulking, it remains to be seen whether surgical debulking prior to some of these therapies may enhance patient outcomes. We believe the direction of drug development efforts for patients with GEP NETs is promising, and will ultimately lead to quantity and quality of life gains for patients afflicted by these tumors.
Footnotes
Conflict of interest statement
Dr. Das previously received Speakers’ Bureau fees from Ipsen and is an unpaid consultant for Ipsen.
Dr. Dasari has previously served in a consulting or advisory role for Ipsen, AbbVie, Stemcentrx, Novartis, Voluntis, and Lexicon.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr. Das’s research efforts are funded by the NET Research Foundation Investigator Award.
