Abstract
Background:
Advanced biliary cancer (ABC) is still regarded as an incurable condition. However, the improved depth and duration of response enabled by chemo-immunotherapy may foster intensified strategies, including surgical procedures, radiotherapy and intra-arterial techniques.
Objectives:
The prevalence, clinical features and treatment outcomes of ABC receiving multimodality treatment in the immunotherapy era are unknown.
Design:
Newly diagnosed ABC treated with chemo-immunotherapy and loco-regional procedures from February 2022 to December 2024 were retrospectively identified at 10 tertiary referral cancer centres in Italy.
Methods:
Categorical variables were compared using the chi-squared test, and the inverse probability of treatment weighting analysis was performed to reduce selection biases.
Results:
Of 241 ABC receiving first-line treatment, 12 (4.9%) fulfilled the inclusion criteria. The median age was 69 (range 36–80), and 9 patients (75%) had intrahepatic cholangiocarcinoma (iCCA). Ten (83.3%) presented with de novo unresectable ABC: 3 (25%) had locally advanced, and 7 (50.3%) had metastatic disease. The median number of metastatic sites was one, with lymph nodes being the most commonly involved location (41.6%, n = 5). Patients treated with intensified therapy were more likely to have low tumour burden (p = 0.03) and iCCA (p = 0.06). Overall, four patients underwent surgery, three transarterial radioembolization, three stereotactic body radiotherapy and two liver transplants. As of data cut-off, 11 patients were alive (91.6%), and five patients were disease-free (41.6%). After adjusting for age, gender, Eastern Cooperative Group Performance Status (ECOG PS), primary site, disease status and number of metastases, the overall survival for the multimodality strategy versus systemic treatment alone was not reached versus 14.1 months (p < 0.001).
Conclusion:
This study, the first on multimodality treatment of ABC in the immunotherapy era, suggests that a highly selected subset of patients may achieve long-term disease control with an intensified approach. Oligometastatic patients and those affected by iCCA appear as the best candidates. Future studies are needed to confirm these preliminary findings.
Keywords
Introduction
Biliary tract cancers are clinically and biologically heterogeneous tumours arising from the biliary ducts within and outside the liver and the gallbladder. 1 They make up 20% of all primary liver cancers, and their incidence and mortality are increasing worldwide. Approximately two-thirds of patients present with de novo unresectable advanced biliary cancer (ABC), and among operable cases, relapse rates are as high as 50%–60%. 2 Thus, the vast majority of patients require systemic treatment during their disease aimed at prolonging survival, delaying disease progression, controlling symptoms and improving quality of life. Historical studies of standard-of-care chemotherapy showed marginal efficacy for cisplatin–gemcitabine combination with most ABC patients not surviving beyond a year from the diagnosis. 3 In 2022, after more than a decade of therapeutic stagnation, the first-line treatment paradigm of ABC has evolved with the advent of immunotherapy. Indeed, the addition of an anti-PD(L)1 agent to cisplatin–gemcitabine backbone has produced unprecedented survival outcomes in two randomized controlled phase III trials,4,5 extending the overall survival (OS) of ABC beyond 12 months. Despite an overall limited survival gain, the benefit was sustained over time in a proportion of patients. In the TOPAZ-1 trial, the combination of the anti-programmed death ligand-1 (PDL-1) durvalumab with cisplatin and gemcitabine resulted in 14.6% of patients alive at 36 months compared to 6.9% in the control arm. In the same trial, the chemo-immunotherapy combination led to a significantly higher objective response rate than conventional chemotherapy (26.7% vs 18.7%). 4 Hence, this improvement has been considered by the medical community as clinically relevant, and chemo-immunotherapy is currently approved and recommended by international guidelines, as the reference regimen for newly diagnosed biomarker-unselected ABC. 6 Together with extending survival, achieving tumour shrinkage is a clinically relevant goal in the treatment of ABC. This may improve cancer-related symptoms in patients with high tumour burden, may have a positive prognostic impact, and, more interestingly, may allow for the secondary use of intensified treatment modalities, including both surgical and non-surgical modalities.
In the pre-targeted therapy and immunotherapy era, the poor activity of conventional chemotherapy and its short duration of response resulted in a low chance of effectively downsizing the tumour to a curative-intent surgical approach. Cases of ABC downsized to liver resection or transplantation following chemotherapy are sparsely reported in the literature, including only patients with locally advanced disease.7,8
In recent years, the observation that among ABC, selected patients present with liver-limited disease and have a better prognosis, 9 prompted the study of locoregional treatment (LRT) to improve patients’ outcomes. A recently published systematic review and pooled analysis in initially unresectable intrahepatic cholangiocarcinoma (iCCA) reported objective response rates in the range of 23.4%– 41.3% and a pooled mean OS in the range of 14.1–21.3 months for first-line chemotherapy combined with LRT, including transarterial radioembolization (TARE; also known as selective internal radiotherapy, SIRT) and hepatic arterial infusion of chemotherapy (HAIC). Interestingly, 14.6% of patients achieved a secondary resection following these intra-arterial therapies. 10 Although encouraging, these data come from retrospective single-centre studies with high heterogeneity in terms of both patient populations and treatment modalities.
With the advent of newly available chemo-immunotherapy regimens, what is currently unknown is whether LRTs are administered concomitantly with or sequentially to durvalumab, cisplatin and gemcitabine, and whether this multimodality treatment (MMT) may alter the natural history of ABC. The improved depth and duration of response of novel first-line options, coupled with the use of surgical and non-surgical LRTs, have the potential to achieve long-term disease control and even a chance of cure in highly selected patients with upfront unresectable disease.
As such, we reviewed all consecutive cases of patients affected by ABC and treated with first-line durvalumab, cisplatin and gemcitabine in a real-world multicentre setting to describe the frequency, characteristics, treatment patterns and survival outcomes of patients receiving MMT.
Materials and methods
Patient selection
Newly diagnosed patients with ABC treated in clinical practice with chemo-immunotherapy from February 1st, 2022 to December 1st, 2024 were retrospectively identified at 10 tertiary referral cancer centres in Italy, including University Hospital of Modena, University Hospital of Marche (Ancona), University Hospital of Bologna, University Hospital of Parma, Piacenza General Hospital, “Vito Fazzi” Hospital (Lecce), Ospedale del Mare (Naples), IRCCS “S. de Bellis” Research Hospital (Bari), Istituto Tumori “Giovanni Paolo II” (Bari) and Ca’ Granda Ospedale Maggiore Policlinico (Milan).
Chemo-immunotherapy consisted of durvalumab, cisplatin and gemcitabine administered according to the schedule of the TOPAZ-1 trial. Durvalumab (1500 mg) was given on day 1 of each cycle, in combination with gemcitabine (1000 mg/m2) and cisplatin (25 mg/m2), which were administered on days 1 and 8 of each 21-day cycle, for up to eight cycles. After completion of gemcitabine and cisplatin, maintenance durvalumab was administered once every 4 weeks until disease progression, until unacceptable toxicity or patient/physician decision. Main inclusion criteria were as follows: (i) cyto/histologically confirmed diagnosis of BTC, including intrahepatic and extrahepatic cholangiocarcinoma and gallbladder cancer according to the latest International Classification of Diseases 11th; (ii) judgment of unresectability upon multidisciplinary hepato-pancreato-biliary discussion based on updated guidelines,11,12 (iii) MMT with LRT as induction or consolidation to front-line chemo-immunotherapy, including liver surgery, liver transplantation, intra-arterial therapies (i.e. TARE/SIRT and HAIC) and external beam radiotherapy (EBRT). A minimum follow-up period of 8 months from LRT was required. The disease was staged according to the eighth edition of the American Joint Committee of Cancer (AJCC)/Union for International Cancer Control TNM cancer staging manual. Data on patient and disease characteristics, treatment patterns and survival outcomes were collected and analysed.
The study was conducted in accordance with the 1975 Declaration of Helsinki and the protocol was reviewed and approved by the Area Vasta Emilia Nord Ethics Committee (n. 183–2019/OSS/AOUMO). Informed consent was waived because of the retrospective nature of the study and the use of anonymized clinical data.
Statistical analysis
In descriptive statistics, continuous variables were reported as mean and standard deviation, while categorical variables were reported as absolute and percentage frequencies. The association between covariates was assessed using the Student t test or Wilcoxon–Mann–Whitney test for continuous numerical variables, whereas Pearson’s chi-square test was used to compare categorical variables. The primary endpoint of the study was the overall survival (OS), defined as the time interval from the date of systemic treatment initiation to the date of disease progression, death or last follow-up, whichever occurred first. Treatment response when assessable was evaluated by computed tomography and categorized as complete response (CR), partial response (PR), stable disease (SD) or progressive disease (PD) according to Response Evaluation Criteria in Solid Tumors (RECIST) v1.1. Treatment-related adverse events were classified and graded according to the common terminology criteria for adverse events (CTCAE) version 5.0.
Inverse probability of treatment weighting analysis
The inverse probability of treatment weighting analysis (IPTW) was applied to balance the baseline characteristics between the group of patients who received or not receive MMT, including the following covariates: gender, age, ECOG PS, disease site, prior surgery, disease status and number of metastatic sites. IPTW was based on energy balancing using formulas described by Huling and Mak. 13 IPTW weights were truncated at the 2.5th and 97.5th percentiles to limit the influence of extreme weights. Standardized mean differences before and after weighting were assessed to check for adequate covariate balancing, considering absolute values below 0.1 as indicative of adequate balance. The difference in OS between the two groups was analysed using the weighted pseudo-population by means of a univariable Cox proportional hazards model. Robust variance estimation was applied to account for the weighting, and the hazard ratio (HR) was reported with 95% confidence interval and p-value.
The Kaplan–Meier estimators were used to calculate the survival probability, and the log-rank test was used to make comparisons between curves. The median follow-up time was calculated using the reverse Kaplan–Meier method. All tests were two-sided, and p values <0.05 were considered statistically significant. The statistical analyses were performed using the SPSS software (version 26.0; SPSS Inc., Chicago, IL, USA). The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement 14 (Supplemental Data).
Results
Patient demographics, disease characteristics and treatment modalities
Overall, out of 241 ABC patients treated with first-line chemo-immunotherapy during the study period, 19 (7.8%) received MMT, and 12 (4.9%) fulfilled the inclusion criteria, being thus included in the present analysis (Figure 1).

CONSORT diagram with patients’ selection and attrition.
Among them, the median age at diagnosis of advanced disease was 69 years (range 36–80), the vast majority were females (n = 8, 66.6%), with an ECOG PS of 0 in half of the cases (50%). In total, 9 (75%) patients were affected by iCCA, while 2 (16.6%) had gallbladder cancer (GBC) and 1 (0.8%) had perihilar cholangiocarcinoma (pCCA). While 10 patients (83.3%) presented with de novo unresectable ABC, 2 (16.6%) relapsed following previous curative-intent surgery. Within the former group, 3 (25%) patients had locally advanced disease and 7 (50.3%) had metastatic disease. Among the latter, most commonly involved metastatic sites were the lymph nodes (n = 5, 41.6%), followed by the liver (n = 3, 25%). The median number of distant metastases was one.
Overall, 11 (83.3%) patients received LRT during systemic treatment and 16.6% (n = 1) before starting chemo-immunotherapy. With regards to treatment modalities, 3 (25%) patients received stereotactic body radiation therapy (SBRT), 3 (25%) with TARE, 4 (33%) underwent radical surgery and 2 (16.6%) underwent liver transplantation (Table 1).
Patient characteristics, treatment pattern and outcome.
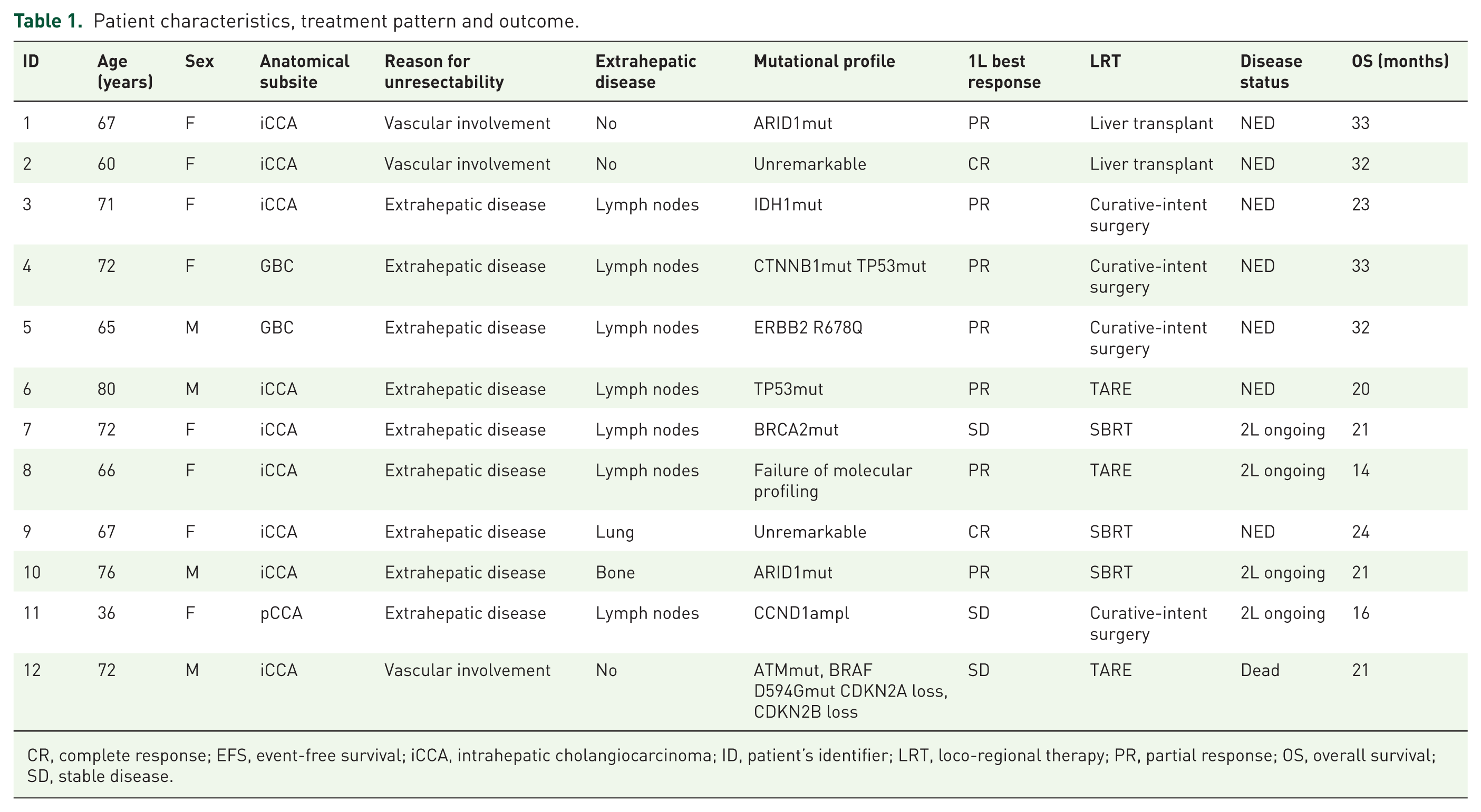
CR, complete response; EFS, event-free survival; iCCA, intrahepatic cholangiocarcinoma; ID, patient’s identifier; LRT, loco-regional therapy; PR, partial response; OS, overall survival; SD, stable disease.
When compared to ABC receiving chemo-immunotherapy alone, patients treated with intensified therapy were more likely to have low tumour burden (p = 0.03) and to be affected by iCCA (p = 0.062; Table 2).
Baseline patient and disease characteristics in the whole ABC cohort.

ABC, advanced biliary cancer; iCCA, intrahepatic cholangiocarcinoma.
Treatment outcomes and tolerability
As of data cut-off (January 25th, 2025), the median duration of follow-up was 21.2 months (95% CI, 17.8–24.1). Overall, 11 (91.6%) patients were alive and among them 5 (41.6%) patients were disease-free. Of note, the only occurring death was not disease- nor treatment-related.
In total, 7 (58.3%) patients were on systemic anticancer treatment: 3 (42.8%) were receiving durvalumab maintenance, and 4 (57%) were receiving second-line chemotherapy.
All patients experienced disease control with first-line treatment, and the best response to chemo-immunotherapy included the following: 2 (16.6%) complete responses, 7 (58.3%) partial responses and 3 (25%) stable disease (Figure 2). The mean duration of first-line treatment before LRT was 7 months (Figure 3).
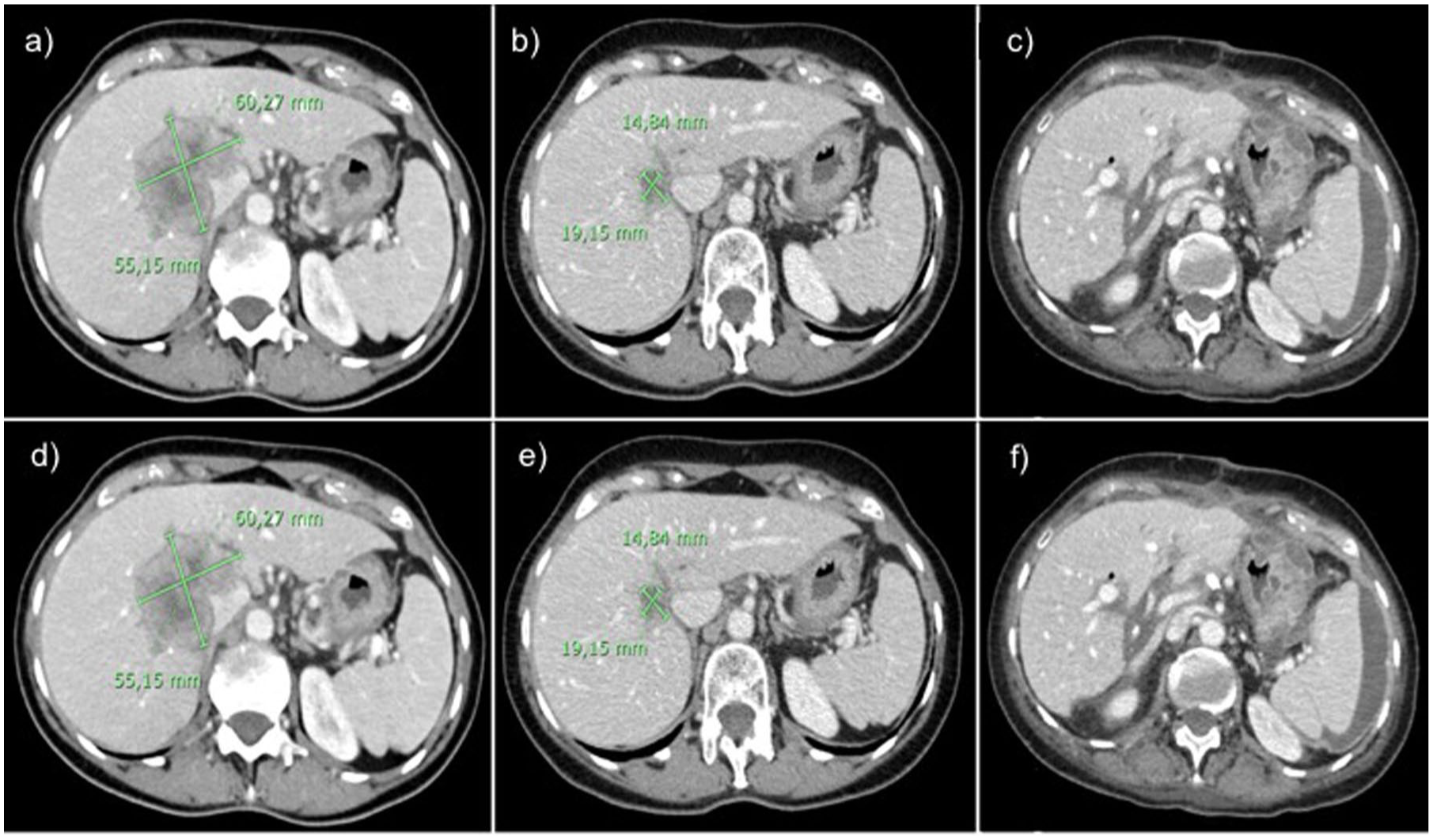
Representative images of ABC cases receiving multimodality management. Axial section of abdominal CT scan of patient ID1 showing at baseline a large, centrally located, iCCA measuring 60.2 × 55.2 mm, infiltrating major vascular and bile duct structures (a). (b) Depicts a dramatic response to chemo-immunotherapy occurring after eight cycles of treatment, with the tumour mass reduced below 2 cm in its longest diameter. Post-transplant CT scan showing no evidence of disease at a 30-month follow-up (c). Axial section of abdominal CT scan of patient ID3 showing at baseline a 60.4 × 43 mm paracaval iCCA, located at the VIII segment with vascular invasion and a 20.5 × 15.6 mm celiac adenopathy (d). CT assessment after eight cycles of chemo-immunotherapy demonstrates a partial response on both disease locations (e). At a 20-month follow-up, the patient was disease-free after surgical resection (f).

Treatment course of ABC patients undergoing a multimodality strategy.
Regarding the safety profile, treatment-related adverse events of any grade (G) were recorded in 11 (91.6%) patients during first-line chemo-immunotherapy. Among them, G1-2 neutropenia (66.6%) and asthenia (41.6%) were the most frequently occurring side effects. The most common grade 3 adverse event was neutropenia (25%). Grade 1 itching and grade 1 hypothyroidism were the only immune-related toxicities, occurring in one case (8%), respectively.
Overall, only one patient experienced an adverse event to LRT: grade 1 cholangitis. No treatment-related deaths occurred due to systemic treatment nor to LRT.
The 1-year and 2-year OS of the study population was 100% and 91.6%, respectively. The 1-year and 2-year EFS was 83.3% and 58.3%, respectively. The median EFS was 20.3 months (95% CI 8.7–31.8), while the median OS was not reached. After adjusting for potential confounding factors with IPTW (Figure 4), the OS was significantly longer in ABC patients treated with MTT compared to those receiving systemic treatment alone (HR = 0.025, 95% CI = 0.004–0.173, p-value = 0.0002, effective sample size = 99.17; Figure 5).
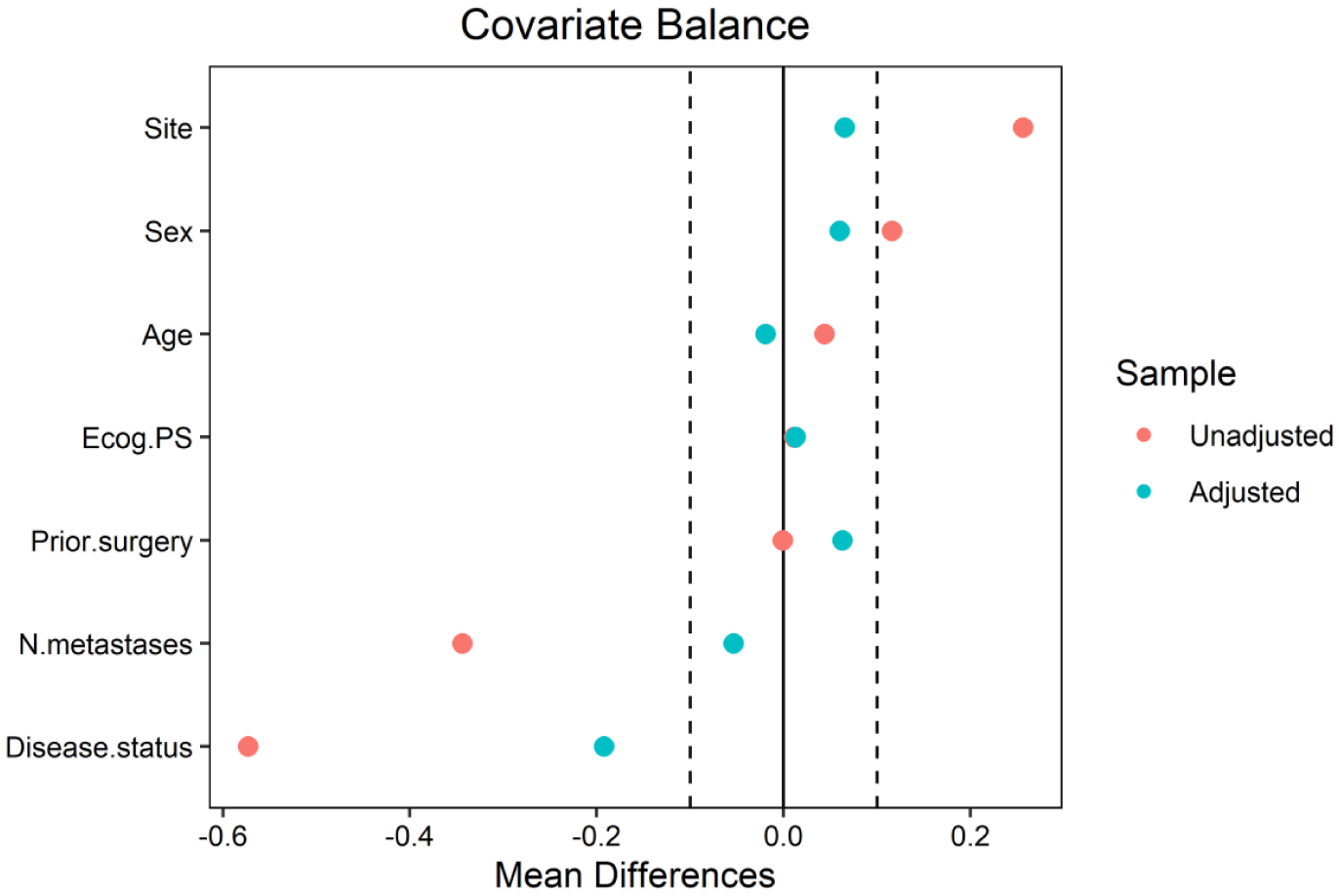
Plot balance showing the covariates’ balance before and after IPTW.
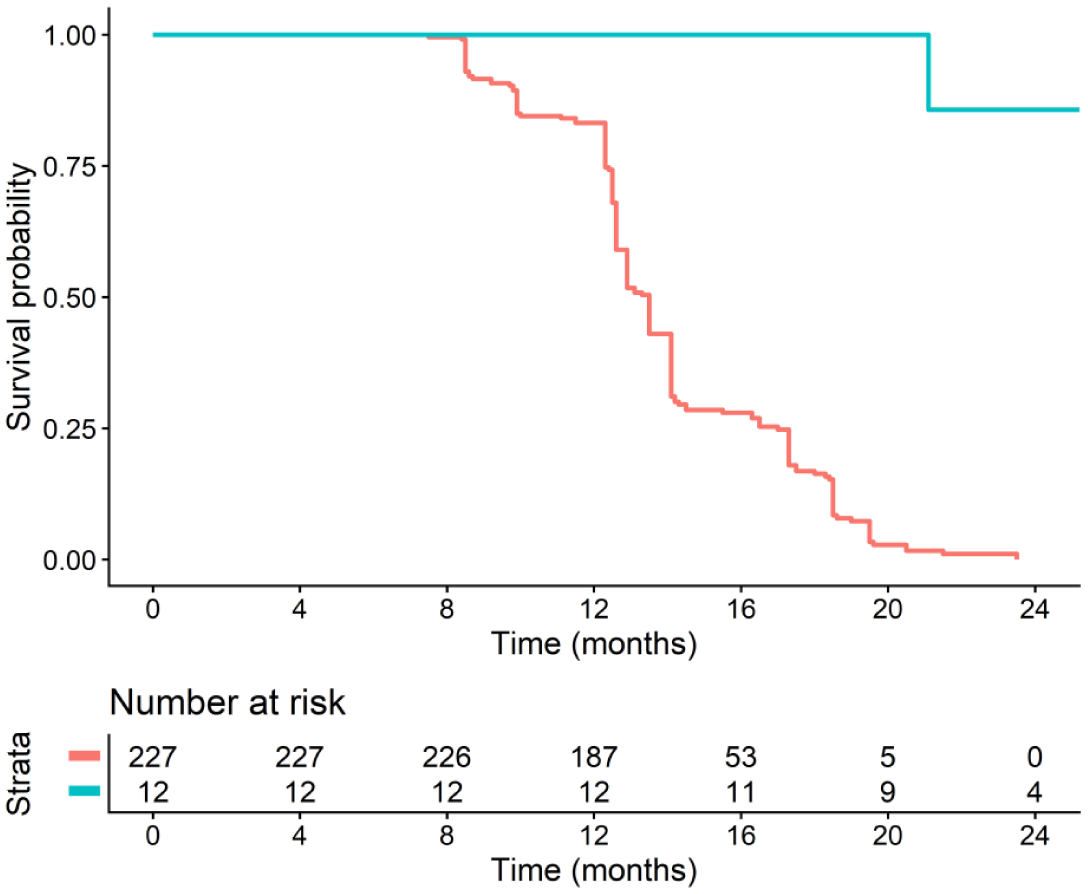
Kaplan–Meier curve of overall survival for ABC undergoing multimodality treatment and the matched cohort after weighting.
Discussion
In this preliminary report, we described the first multicentre series of ABC receiving an MMT consisting of front-line chemo-immunotherapy associated with LRTs in a real-world scenario. Excellent outcomes have been recorded without major complications, showing the feasibility and effectiveness of MMT in carefully selected cases with initially unresectable disease. Notably, patients who received MMT had significantly longer survival than those receiving chemo-immunotherapy alone.
Advanced biliary cancer is, by definition, an incurable condition hallmarked by a very poor prognosis. Historical data on palliative-intent cisplatin–gemcitabine combination showed a median overall OS hardly exceeding 12 months. While long-term survivors have been described in surgical series of primarily resectable BTC, ABC experiencing durable disease control with chemotherapy doublet is much more rare. Among 410 patients enrolled in the ABC-02 trial, only 21 (5%) survived at least 36 months, most of whom featured locally advanced tumours. 15 Recently, the TOPAZ-1 and the KEYNOTE-966 trials reported practice-changing data for the combination of an immune checkpoint inhibitor targeting the anti-PD(L)1 axis with cisplatin and gemcitabine. Interestingly, the depth and duration of response enabled by chemo-immunotherapy have the potential to foster intensified strategies, including surgical and non-surgical loco-regional procedures, to achieve durable disease control and even cure in highly selected cases. In this study, after analysing the whole cohort treated with first-line cisplatin–gemcitabine–durvalumab, we identified 12 (4.9%) patients who received LRTs in addition to systemic treatment. Half of the cases received surgical procedures: four patients underwent curative-intent surgery and two underwent liver transplantation. In the pre-immunotherapy era, cases of BTC converted to surgery were limited to single case reports and small studies. Le Roy et al. reported that among 74 patients with nonmetastatic unresectable iCCA, 39 (53%) underwent secondary resection following a median of six chemotherapy cycles. 7 However, two-thirds of patients had an R1-resection and nearly 80% of them developed disease recurrence, the vast majority within 24 months. Our results compare favourably with those from Le Roy et al., especially in light of the higher rate (75%) of patients with metastatic disease in our analysis and the inclusion of poorer prognosis anatomical subsites (including two GBC, one pCCA, and one iCCA). More importantly, after nearly 2 years of follow-up, the four patients converted to surgery were alive, three of whom are still receiving anti-PD-L1 maintenance treatment, with no evidence of disease. The data highlight how newly available systemic options may act as conversion therapy for a subset of selected ABC and suggest a role for adjuvant immunotherapy in resected high-risk disease. Although low-level evidence exists, and it is currently not a standard therapy, recently published guidelines recognize that liver transplantation may advantage ABC patients with good and prolonged responses to neoadjuvant chemotherapy. To this end, MacMillan et al. reported an encouraging survival of 57% at 5 years for 18 patients ultimately transplanted after 6 months of disease control while on neoadjuvant chemotherapy. 16 In our experience, two locally advanced iCCA cases remained unresectable despite a remarkable and sustained response to systemic treatment due to their central location and vascular invasion. After a careful multidisciplinary discussion, the decision was made to list and finally transplant them. Interestingly, both patients are currently disease-free with a survival of more than 30 months after transplantation.
The second half of patients in our study, consisting of only iCCA, were given non-surgical LRT, including TARE and SBRT in three cases, respectively. In recent years, LRT has been increasingly studied, particularly in the subgroup of advanced liver-only iCCA, which is known to have a better prognosis compared with all-comers ABC. The reported resection rates of 11% and 22% in phase II trials exploring chemotherapy combined with hepatic arterial infusion of floxuridine and Y90 radioembolization, respectively, are encouraging findings for initially unresectable CCA.17,18 Moreover, specific to TARE, a systematic review of mostly small, single-centre, heterogeneous, retrospective studies reported ORR ranging from 0% to 36%, and mOS ranging from 8.7 to 32.3 months. 10 More interestingly, a comparative analysis through target trial emulation recently suggests improved OS and PFS for the combination of TARE and chemotherapy over chemotherapy alone in liver-limited iCCA. 19 The results presented are limited to a highly selected cohort of patients with locally advanced intrahepatic cholangiocarcinoma (iCCA). However, we find our findings particularly noteworthy because they also encompass patients with non-iCCA subtypes and those with metastatic disease, thereby broadening the applicability and potential impact of our observations across a more diverse patient population.
Our results add novel insights to the existing evidence, showing the potential of sequencing TARE as a consolidation modality in patients experiencing disease control with chemo-immunotherapy induction. Lesser evidence is available on the role of SBRT in ABC. SBRT has been shown to provide a high local control rate (pooled 1-year local control rate 83%), yet with unsatisfactory survival outcomes (pooled 1-year OS rate 58.3%). 20 However, in our experience, the synergism between immunotherapy-based combination and SBRT looks promising for patients with unresectable ABC. We treated three metastatic iCCA with this multimodality approach, two on distant sites and one on the primary tumour, achieving an encouraging survival exceeding 20 months in all cases.
Importantly, the OS achieved in our cohort compared favourably with that reported for systemic treatment alone in ABC both in the literature and in the real-world. To this end, MMT produced a markedly longer 24-month OS compared to the TOPAZ-1 trial (91.6% vs 24.9%) and resulted in prolonged OS with respect to patients treated with upfront cisplatin–gemcitabine–durvalumab in clinical practice.
Selecting patients more likely to benefit from an intensified approach is challenging. In fact, beyond microsatellite instability high and/or tumour mutational burden high status, occurring in less 1% of cases, currently, no biomarkers may accurately predict immunotherapy benefit. As a consequence, the major drivers of our treatment decision remain clinical: (i) primary tumour site, (ii) disease extent and (iii) response to systemic treatment. When analysed collectively and compared with the population of ABC receiving cisplatin–gemcitabine–durvalumab alone, our cohort was enriched in the intrahepatic subsite and low-burden tumours. This came as no surprise since these are both established prognostic factors in ABC, though they contribute to narrowing the field of potential candidates to the MMT. iCCA, particularly those presenting with liver-predominant disease, have been shown to have a better prognosis than all-comers BTC when treated with chemotherapy according to a recent pooled analysis of the ABC-01, -02 and -03 trials. 9 Moreover, of the same importance are dynamic parameters such as the achievement of sustained disease control with systemic treatment since this can be considered a surrogate measure of favourable disease biology. Indeed, all patients undergoing MMT in our cohort benefitted from chemo-immunotherapy and stayed on systemic treatment for at least 6 months before receiving intensified modality.
The present study has several limitations to be acknowledged. Firstly, the retrospective nature of the study, along with the minimum threshold of 8-month follow-up, does not preclude the risk of biases. Secondly, treatment modalities are heterogeneous, preventing us from comparing each approach separately. Thirdly, the relatively short follow-up, along with the small sample size, makes it necessary to further confirm these preliminary findings on a longer time frame with the inclusion of more patients.
Conclusion
We provided herein the first evidence that, in the era of immunotherapy, a multimodality approach could produce excellent long-term survival in highly selected ABC. While awaiting predictive biomarkers to assist patient selection, those with sustained response to chemo-immunotherapy and upfront locally unresectable or oligometastatic disease appear to be the best candidates for treatment intensification. Importantly, these data also highlight the importance of centralizing patients to experienced high-volume centres and discussing all newly diagnosed BTC, including patients with ABC, in a multidisciplinary tumour board.
In the rapidly evolving treatment landscape of ABC, the upfront use of chemo-immunotherapy has the potential to impact the course of disease like never before.
Supplemental Material
sj-docx-1-tam-10.1177_17588359261426823 – Supplemental material for Preliminary report on advanced biliary cancer patients receiving multimodality treatment in the immunotherapy era: a real-world multicentre experience
Supplemental material, sj-docx-1-tam-10.1177_17588359261426823 for Preliminary report on advanced biliary cancer patients receiving multimodality treatment in the immunotherapy era: a real-world multicentre experience by Massimiliano Salati, Eleonora Borghi, Riccardo Cuoghi Costantini, Alessandro Parisi, Alessia Lancianese, Andrea Palloni, Chiara Ricci, Ingrid Garajova, Elena Orlandi, Ina Valeria Zurlo, Angelica Petrillo, Anna Diana, Angela Dalia Ricci, Alessandro Rizzo, Martina Manni, Ornella Garrone, Massimo Dominici and Michele Ghidini in Therapeutic Advances in Medical Oncology
