Abstract
Metastatic breast cancer (BC) is considered an incurable disease and is usually treated with palliative intent. However, about 50% of metastatic BCs present with only a few metastatic lesions and are characterized by longer overall survival. These patients, defined as oligometastatic, could benefit from a multimodal approach, which combines systemic therapy with metastasis-directed treatment (stereotactic ablative therapy or surgery). The current definition of oligometastatic seems incomplete since it is based only on imaging findings and does not include biological features, and the majority of relevant data supporting this strategy comes from retrospective or non-randomized studies. However, the chance of reaching long-term complete remission or even a cure has led to the development of randomized trials investigating the impact of combined treatment in oligometastatic BC (OMBC). The SABR-COMET trial, the first randomized study to include BC patients, showed promising results from a combination of stereotactic ablative radiotherapy and systemic therapy. Considering the randomized trial’s results, multidisciplinary teams should be set up to select OMBC patients who could achieve long-term survival with aggressive multimodal treatment.
Introduction
The usual classification of breast cancer (BC) distinguishes between locoregionally confined disease, amenable to local therapy and potentially curable, and metastatic disease, generally considered an incurable-condition candidate for palliative therapy. 1 Metastatic BC usually occurs as a relapse of early BC at distant sites, although, in about 6% of cases, it can also arise de novo. 2
Despite metastatic BC being classified as a unique setting (stage IV according to the American Joint Committee on Cancer staging system), it covers a large disease spectrum ranging from a single metastatic site to numerous lesions in different organs with different biological traits. 1
About 50% of metastatic BC presents with less than three metastatic lesions. 3
Clinical evidence of longer overall survival (OS) in patients with a limited metastatic burden4,5 and advances in local technique modalities, such as surgery and stereotactic ablative radiotherapy (SABR), as well as in systemic therapy (Figure 1), has generated interest in the multimodal approach for oligometastatic BC (OMBC) aimed at long-term complete remission at the cost of added toxicity. 6
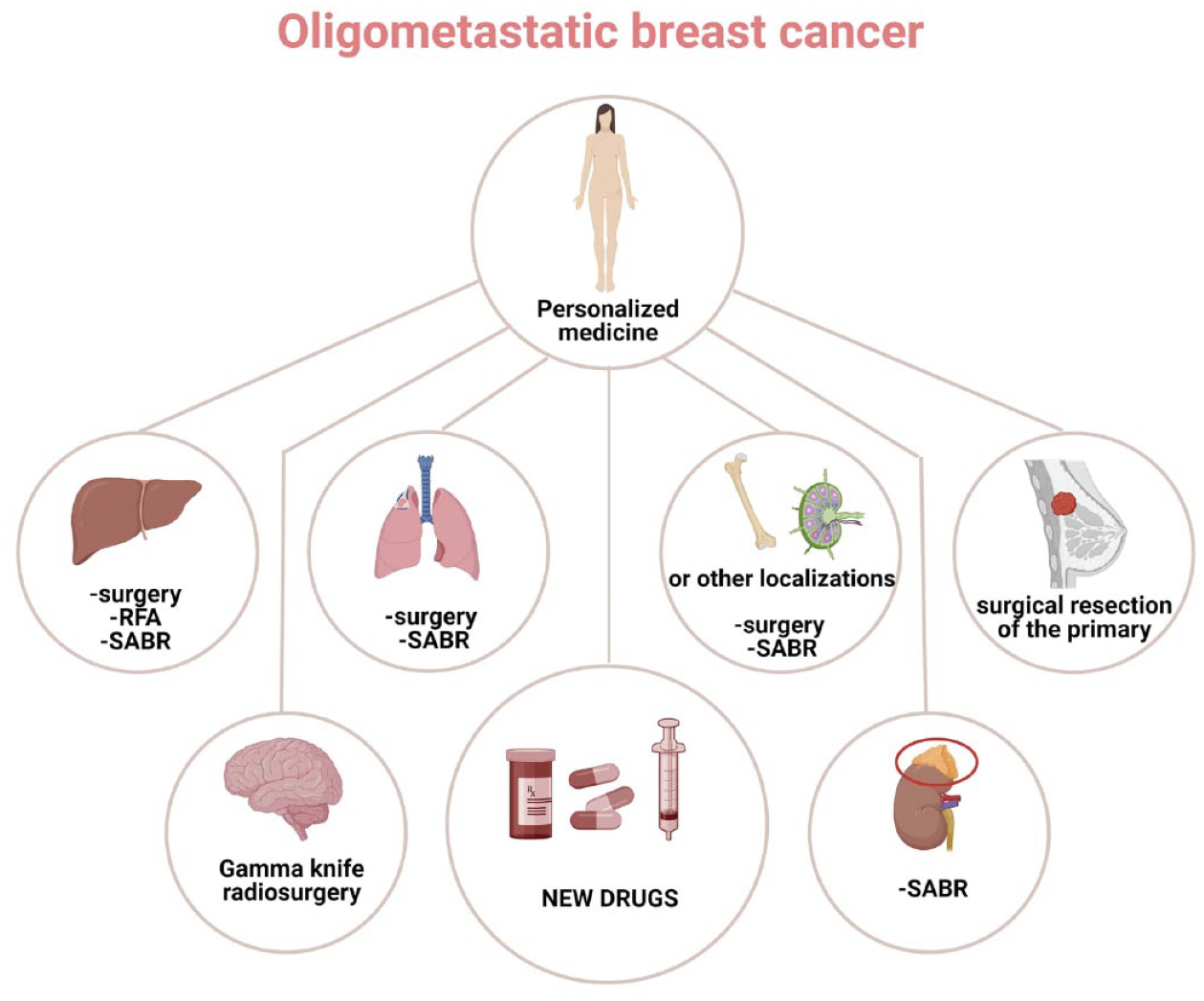
Tailored multimodal approach for OMBC combining modern systemic therapies and metastasis-directed treatments such as surgery, SABR, gamma knife radiosurgery, and RFA based on metastatic sites.
However, the data supporting this treatment strategy refer mostly to retrospective or prospective non-randomized studies, with a heterogeneous definition of oligometastatic disease, the inclusion of various tumor histologies, and a lack of BC subtype stratification. 7
Randomized controlled trials demonstrated a benefit in progression free-survival (PFS) 8 and OS 9 with the use of local ablative therapy after first-line systemic therapy in oligometastatic non-small-cell lung cancer patients and gave an advantage to PFS in oligometastatic prostate cancer patients with metastasis-directed treatment (MDT). 10 The SABR-COMET trial was the first randomized trial in an oligometastatic setting to include BC patients. 11
In this review, we discuss the present understanding of OMBC, the available data regarding multimodal strategy, which could guide decision-making in current clinical practice, and the ongoing trials that may eventually establish this aggressive approach as the gold standard for OMBC.
Definition of oligometastatic disease
The term ‘oligometastatic’ was coined in 1995 by Hellman and Weichselbaum to define an intermediate state between locoregional and widespread disease in which the full metastatic biological potential is not expressed and circulating tumor cells have metastasized in limited and suitable sites.12,13
This theory implies that an aggressive local therapy of the metastatic sites could potentially eradicate the disease. 6 Therefore, it is necessary to identify the right candidate for multimodal treatment to avoid potential over-treatment.
Despite the definition of an oligometastatic state as heterogeneous across the literature, a recent consensus statement by the European Society of Radiotherapy and Oncology (ESTRO) and the American Society of Radiation Oncology (ASTRO) suggested a threshold of 1–5 metastatic lesions safely treatable with local therapy; furthermore, the oligometastatic state must be assessed with high-resolution imaging. 14
However, this imaging-based classification includes a group of patients with different outcomes, 15 given that in some cases, the presence of a small number of lesions does not reflect the real widespread of the disease. The oligometastatic state could represent only the tip of the iceberg. 6 The understanding of the biological behavior of the disease should be integrated into the oligometastatic definition. In support of this concept, some studies highlighted a different micro-RNA (miRNA) expression in aggressive metastatic diseases compared to slower ones,16,17 even though validated biomarkers are still lacking.
Achieving an exhaustive definition of the oligometastatic state is also hampered by its potential presentation at different timepoints of the disease course. Recently, the ESTRO and the European Organisation for Research and Treatment of Cancer (EORTC) obviate this problem by creating a dynamic oligometastatic state model. Based on five questions regarding the disease-free interval (DFI), the pre-existent disease burden, and the use of active systemic therapy at the time of oligometastatic disease diagnosis. They identified different patient subgroups characterized by diverse treatment strategies, goals, and the possibility of achieving a cure: for instance, the term ‘induced oligopersistence’ represents patients with the poly-metastatic disease who reached oligometastatic state after systemic treatment, while ‘de novo oligometastatic disease’ defines patients with no history of metastatic disease. 18 The adoption of this classification could harmonize the interpretation of study results and simplify study comparisons.
Surgical intervention
The studies which investigated the role of surgical resection of the metastases are summarized in Table 1.
Retrospective and prospective studies of metastasis-directed therapy for OMBC patients.

BC, breast cancer; IMRT, intensity-modulated radiation therapy; mOS, median overall survival; mPFS, median progression-free survival; OS, overall survival; PFS, progression-free survival; RT, radiotherapy; SABR, stereotactic ablative radiotherapy; SOC, standard of care.
Liver
The most successful example of surgical treatment for oligometastatic disease is the resection of liver metastasis from colorectal cancer, which has significantly improved the survival of these patients. 32
However, there is no evidence from randomized trials of metastatic BC patients benefitting from surgical treatment. Even if some retrospective and prospective non-randomized studies pointed out potential survival advantages, definitive conclusions are limited by population heterogeneity and different trial designs.
A systematic review by Chua et al., 33 including 19 studies and a total of 553 patients, revealed a median OS of 40 months and a median 5-year survival rate of 40% in metastatic BC patients with hepatic lesions treated with surgery.
In 2017, another systematic review published its investigations on the surgical approach to BC liver metastasis. It examined 43 retrospective studies involving 1686 patients, the majority of whom were estrogen receptor (ER) positive. Similar to the previous review, the median OS was 36 months, and the 5-year survival rate was 37%. The perioperative risk was acceptable, having a morbidity rate of 20% and a 30-day mortality rate of 0.7%. 19
Radiofrequency ablation (RFA) could be an alternative to hepatic resection. 7 This technique is safe and can provide local control, especially in patients with lesions smaller than 3 cm, a single metastasis, and ER-positive status. 34 However, a systematic review and meta-analysis published in 2018 highlighted a better survival rate for hepatic resection than RFA, suggesting limiting this method to unresectable liver metastases or patients with high surgical risk. 35
Lung
A retrospective study conducted by Yoshimoto et al. 20 examined the potential benefit of solitary lung BC metastasis resection. A total of 90 patients were included in the analysis. The overall 5- and 10-year cumulative OS rates were 54% and 40%, respectively. A disease-free survival (DFS) longer than 3 years and stage I disease at diagnosis were associated with significantly longer survival (p = 0.013).
A meta-analysis of 16 cohort studies including patients with pulmonary metastases who underwent metastasectomy showed pooled 5-year survival rates of 46%, the number of pulmonary metastases higher than one, negative hormone receptor (HR) status, and DFS lower than 3 years were determined negative prognostic factors. 36
Brain
Surgery can also have a role in brain metastases, especially in cases concerning solitary lesions needing pathologic confirmation.
A prospective study, conducted between 1985 and 1988, randomized 54 BC patients with single brain metastasis (18 of whom also presented extracranial sites of disease) to its surgical removal, followed by radiotherapy (RT) or needle biopsy plus RT. Local recurrence was more frequent in the radiation group than in the surgical group (52% versus 20%), and median OS (mOS) was significantly longer in the surgical group (40 weeks versus 15 weeks). 21
In a retrospective study published in 1997 involving 70 patients with BC brain metastases who underwent surgical resection, 2-year, 3-year, and 5-year survival rates were 25.7%, 18.6%, and 7%, respectively. The median survival was 11 months in patients with brain metastases ⩾4 cm and 16.3 months in patients with smaller ones. Adjuvant whole brain RT after surgery was correlated with longer survival at multivariate analysis. 22
Other retrospective series have also reported an association between postoperative RT and survival improvement. 23
Bone
Bone metastases are usually treated with RT, and data regarding surgical resection in OMBC are lacking.
However, surgery can play an interesting part in solitary bone metastasis.
A retrospective study investigated the effect of bone metastases resection in 70 patients: 19 had a single osseous lesion, 19 had multiple bone lesions, and 32 also presented visceral involvement.
The OS rate was 36% after 2 years and 13% after 5 years for the entire population, while patients with solitary bone metastasis showed an OS rate of 39% after 5 years. 24
In a retrospective study (published in 2012) of 115 BC patients who underwent surgery for bone metastases, wide resection and the absence of visceral metastases were associated with longer survival at univariate analysis. 37
Nevertheless, surgical decompression of symptomatic spinal metastases is still crucial to preserve or restore neurologic function, enforce spinal stability, and reduce pain. 38
Primary tumor surgery
Approximately 6% of metastatic BC arises de novo. 2 The indication of local treatment (surgery and eventual adjuvant RT) of the primary in addition to systemic therapy is still a matter of debate since data regarding its impact on survival are conflicting. 39
A retrospective study using the 1988–2011 Surveillance, Epidemiology, and End Results database included 29,916 metastatic BC patients, showed an association between primary tumor resection and improved median OS (34 versus 18 months), especially for OMBC. 40
The BOMET MF 14-01 is a multicenter, prospective registry study initiated in 2014, including de novo stage IV BC patients with bone-only metastases. Patients were divided into two subgroups based on surgical removal of the primary. The results published in 2021 showed a 5-year OS rate of 72% for patients who underwent locoregional treatment of the primary in addition to systemic therapy and a 5-year OS rate of 33% for systemic therapy-only patients. Surgical removal of the primary improved survival for both HR-positive/HER2-negative and HER2-positive patients, but no survival benefit was reported for triple-negative patients. 41 However, some prospective studies pointed out the absence of survival benefits deriving from the surgery of the primary. 42 Randomized studies showed mixed results as well.
A Turkish multicenter, phase III study randomized 274 de novo metastatic BC patients to locoregional treatment in addition to systemic therapy versus systemic therapy alone. The median OS was significantly improved for the locoregional treatment group (46 months versus 37 months); a lower risk of death was noted for HR-positive and HER2-negative status and patients with solitary bone metastasis. 43
An open-label randomized controlled trial compared the locoregional treatment of the primary versus no locoregional treatment in a population of 350 patients affected by de novo metastatic BC. Most patients were pre-treated with chemotherapy, and almost 25% had less than four metastases. In this study, no improvement in OS was observed between the two groups, not even in OMBC patients. 44
The preliminary results of the phase III trial ECOG-ACRIN E2108 were presented at ASCO 2020. This trial randomized 256 metastatic BC patients who did not progress during 4–8 months of optimal systemic treatment to the locoregional treatment of the primary or not. At a median follow-up of 59 months, no significant difference in OS was observed. 45 However, it should be noted that no subgroup analysis was performed according to the metastatic site, even for bone-only metastases patients, which accounted for 38% of the study population. Although these results seem to rule out the role of surgical resection of the primary as a gold standard for de novo metastatic BC, it could be offered to selected patients in clinical practice, such as patients with limited metastatic burden, bone-only disease, and HR-positive or HER2-positive disease. Further trials focusing on OMBC patients receiving also local treatment for metastatic lesions should be encouraged as they could of this patient group.
Stereotactic ablative body radiotherapy
SABR consists of a highly focused radiation beam on an extracranial site localized through internal imaging. This technique limits the dose to the surrounding organs and requires only one to five treatment sessions to reach the effective dosage, providing an attractive option instead of surgery.
The role of SABR in OMBC is supported mainly by retrospective or prospective randomized studies (Table 1). However, a phase II randomized controlled trial (SABR-COMET) was published in The Lancet in 2019. This study randomized 99 oligometastatic cancer patients with up to five metastatic lesions and controlled primary malignancies between standard-of-care (SOC) treatment and SOC with the addition of SABR. The primary endpoint was OS; PFS toxicity and quality of life were secondary endpoints. BC was one of the most represented primary tumors (18%). At a median follow-up of 51 months, the 5-year OS rate was significantly better in the SABR arm (42.3%) compared to the control arm (17.7%). Using SABR was not associated with an increase in grade 2–5 events, and a detrimental effect on the quality of life was not observed.
The limited benefit in median PFS (11.6 months for the SABR arm versus 5.4 months for the control arm) compared to the marked improvement in OS together with the fact that 30% of long survivors were treated with SABR against new metastatic lesions seem to support the use of salvage SABR. 25
Regarding the non-randomized evidence, a recent retrospective multicenter analysis including a total of 49 recurrent oligometastatic HR-positive/HER2-negative BC patients showed an improvement in PFS for those who underwent local treatment (30 versus 18 months) without any difference in terms of OS. This result seems in contrast with the SABR-COMET study. However, uncontrolled post-progression therapy and an inadequate follow-up period and sample size (considering the favorable prognosis of HR-positive/HER2-negative patients) might explain this discrepancy. 26
A single institution prospective trial investigated the role of single fraction SABR in 15 BC patients (the majority of whom were HR positive) with up to three bone-only metastases and controlled primary. The distant and local PFS at 2 years of follow-up were 67% and 100%, respectively. 27
A phase II Italian trial conducted by Trovo et al. included 54 OMBC patients with less than six lesions. Metastases were treated with SABR or fractionated intensity-modulated radiotherapy concomitantly to systemic therapy. About 85% of patients had less than three lesions, and 80% presented HR-positive disease. After a median follow-up of 30 months, PFS (the primary endpoint) at 1 and 2 years was 75% and 53%, respectively, superior to historical controls. Two-year OS was 95%, and no grade 3 toxicity was documented. 28
BC appears to be one of the most suitable candidates for SABR in an oligometastatic state.
The results of a prospective observational study, funded by National Health Service England’s Commissioning through Evaluation program, have been recently published. This study recruited 1422 patients with up to three metastatic lesions of mixed histology, absence of intracranial disease, and a DFI ⩾ 6 months from the primary tumor. These patients with different tumor histologies (5.5% BC) have been treated with SABR (24–60 Gy in three to eight fractions). The 2-year OS for BC patients was 83.2%, inferior only to prostate cancers. 29
Klement et al. 30 assessed how histology influences metastases control in 363 patients with liver lesions treated with SABR, collected in a multicenter database from the German Society of Radiation Oncology. BC metastases proved more radiosensitive than other histologies with a favorable tumor control probability at 2 years.
This assumption was also supported by the long-term follow-up results of a prospective mixed histology trial, which enrolled 121 patients with up to five lesions, and with no more than three organ sites involved, treated with SABR. The 6-year OS for BC patients was 47%, compared to 9% in other histologies. 31
Prognostic factors
The results available suggest a survival benefit for OMBC patients by adding locoregional treatment to systemic therapy. However, the lack of data from randomized trials precludes definitive conclusions. Therefore, it is necessary to identify prognostic factors which can guide clinicians in case-by-case management because patients with good prognoses could be ideal candidates for multimodal treatment.
Patients with isolated sternal metastasis and contralateral axillary metastases are classified as oligometastatic but demonstrate good survival. 1 In a retrospective cohort study including 35 de novo metastatic BC patients with isolated sternal or mediastinal disease treated with metastasis-directed radiotherapy, and surgical resection of the primary and systemic therapy, a 5-year OS of 63% was reached. When this cohort was compared to a group of stage IIIB BC patients, statistically significant differences were not reported. 46 The favorable prognosis of this subgroup is also confirmed by case series of BC patients with resected sternal metastasis.47,48 Isolated sternal metastasis may be linked with regional lymphatic spread rather than hematogenous dissemination, explaining a good prognosis. 49
Similarly, contralateral axillary metastases could be associated with lymphatic spread due to aberrant lymphatic drainage. A systematic review published in 2015 including 24 articles describing 48 patients with contralateral lymph node recurrence reported an OS of 82.6% after a mean follow-up time of 50.3 months. 50 Therefore, contralateral axillary metastases should be treated with a curative approach as a regional disease, with lymph node dissection followed by radiotherapy and systemic therapy. 1
In a recently published population-based study of 3447 patients affected by de novo metastatic BC identified by the Netherlands Cancer Registry between January 2000 and December 2007, the presence of up to three metastases was related to better survival, with a 10-year OS estimate of 14.9% compared to 3.4% for patients with more than three metastases. Premenopausal and perimenopausal status and the absence of lung lesions were associated with a better prognosis in terms of OS and PFS. In contrast with other studies, single-organ involvement was not associated with favorable survival. 51
A systematic review including 20 studies reporting data on OMBC patients treated with multimodal therapy identified five potential prognostic factors for this subgroup: the presence of a solitary metastasis, a DFI ⩾ 24 months, HR status positivity were associated with better OS and PFS, while axillary lymph node metastases at primary diagnosis and HER2 positivity were found to be negative prognostic factors. However, it must be clarified that anti-HER2 therapy use was limited in selected studies. 6
As regards metastatic BC patients undergoing pulmonary metastasectomy, a systematic review and meta-analysis highlighted DFI ⩽ 3 years, incomplete resection of metastases, more than one lung lesion, and HR-negative status as the main negative prognostic factors. 43
Among patients with brain metastases, those with HER2-positive and Luminal/HER2-positive disease appear to be the ones to benefit the most from Gamma knife radiosurgery based on a multi-institutional retrospective study conducted in Japan. 52
Even though confirmation of these prognostic factors and the identification of new ones are warranted, they can already be helpful for the management of OMBC patients (Figure 2).

OMBC patients’ selection for metastasis-directed therapy based on prognostic and predictive factors.
Circulating tumor cells, circulating tumor DNA (ctDNA), inflammatory indexes, tumor tissue, and functional imaging were studied as potential prognostic and predictive biomarkers in locally advanced and metastatic BC.53–59 Their impact on OMBC needs to be further elucidated, in particular, to select patients for locoregional treatment.
New systemic therapies
The evolution of systemic therapy is paving the way to a progressive survival improvement in all subtypes of BC, and it could modify the definition of the oligometastatic state as even more patients could be considered a good prognosis. The studies cited in this review mainly concern BC patients treated with outdated systemic therapies. The introduction of cyclin-dependent kinase 4/6 inhibitors and hormonal therapy increased both PFS and OS in HR-positive/HER2-negative metastatic BC patients, 60 and the combination with local treatment could even prolong survival in these patients.
Adding pertuzumab to docetaxel plus trastuzumab increased the number of HER2-positive long survivors as assessed by the CLEOPATRA study, which reported an 8-year survival rate of 37% for patients treated with dual HER2 blockade therapy. 61 These results encourage using aggressive treatment, for example, metastasis-directed therapy, as a cure for HER2-positive metastatic BC patients no longer seems unachievable.
Moreover, implementing immune checkpoint inhibitors (ICIs) for PD-L1-positive triple-negative metastatic BC 62 has opened the door to the potential synergistic effect between RT and immunotherapy. RT can cause the secretion of cytokines and chemokines with consequent antitumor immune response enhancement. This synergy is proving effective in other malignancies, such as non-small-cell lung cancer,63,64 and looks promising even for BC. In addition to preclinical evidence of RT plus ICIs effectiveness in murine models, 65 in a phase I trial, 73 metastatic patients, six of whom were affected by BC, were treated with hypofractionated image-guided RT and subsequently with pembrolizumab. The overall response rate (ORR) was 13.2%, but it was not stratified by tumor histology. Grade 3 toxicity was observed for only six patients, and the combination was well tolerated. 66
A phase II trial investigating the combination of palliative RT and pembrolizumab in 17 pretreated metastatic BC patients showed an ORR of 17.6%, which proved durable. Notably, one of these patients was free from evaluable disease at 108 weeks of follow-up. 67 Despite early evidence, these results appear promising, and phase II trials focused on RT and ICI combinations in metastatic BC are already ongoing. 68
Despite the advances in conventional chemotherapies, their use is limited by a narrow therapeutic window based on toxicities. The breakthroughs of nanotechnology and antibody–drug conjugates represent an accomplishment in the selective targeting of tumor sites, thus increasing the therapeutic potential. They constitute an elegant means of tackling the limitations of conventional chemotherapeutics by selectively delivering a highly toxic payload with subsequently increased efficacy.69–71
Ongoing trials
The previously reported data do not provide definitive conclusions but inspired growing interest in OMBC management. Several mixed histologies and breast-specific prospective and phase II/III randomized controlled trials are ongoing and will clarify the role of multimodal treatment in this population (Table 2).
Ongoing clinical trials of metastasis-directed therapy for OMBC patients.

BC, breast cancer; EFS, event-free survival; HR, hormone receptor; OS, overall survival; PFS, progression free survival; QoL, quality of life; RT, radiotherapy; SABR, stereotactic ablative radiotherapy; SOC, standard of care.
Based on the promising survival benefit reported in the phase II SABR-COMET trial regarding mainly oligometastatic patients with up to three metastatic lesions, two phase III randomized studies were designed with OS as the primary endpoint.
The SABR-COMET-3 trial aims to confirm the phase II study results by recruiting cancer patients with one to three metastatic lesions. Patients are randomized between SOC treatment with or without SABR and stratified by histology and DFI (⩽2 versus > 2 years). 72
Whereas the goal of the SABR-COMET-10 trial is to assess the role of SABR in patients with 4–10 metastatic lesions. Randomization is stratified by histology and pre-specified systemic therapy (immunotherapy/targeted therapy versus cytotoxic therapy versus observation). 73
Both SABR-COMET-3 and SABR-COMET-10 trials also assess the circulating tumor cells, cell-free DNA, and tumor tissue as potential prognostic and predictive biomarkers.
Another example of a randomized controlled study comparing SOC and SOC plus SBRT is the phase II/III CORE trial. Eligible patients must be affected by breast, prostate, or non-small-cell lung oligometastatic cancer with up to three metastases and a maximum of two organ systems involved. The primary endpoint is PFS. 74
Other randomized mixed histology trials that will contribute to determining the role of SABR and systemic therapy combination are ongoing (STEREO-OS, PROMISE-005).75,76
Regarding non-randomized studies, E2-RADIatE (EORTC-ESTRO Radiation Infrastructure for Europe) is a platform that was launched in June 2019, which aimed at collecting prospective real-world data of cancer patients treated with RT. OligoCare is the first project on the E2-RADIatE platform. It is a pragmatic observational cohort study collecting data from OMBC, prostate, lung, and colorectal cancer patients (both synchronous and metachronous) treated with radical RT. This study will elucidate how patient, tumor, and treatment characteristics influence OS and will also test the prognostic value of EORTC-ESTRO oligometastatic state classification. 81 The preliminary results presented at ESTRO 2021 congress did not report data on survival; however, it showed that prostate cancer was the most frequent primary (42%), while BC patients represented 17% of the 328 cases considered for this analysis.
Some breast-specific trials are also ongoing. The OLIGOMA trial is a multi-national confirmatory study in which OMBC patients with up to five metastatic lesions are randomized to standard systemic therapy with or without RT to all metastatic sites. Patients with a maximum of three brain metastases are eligible for this study, and locoregional recurrence is allowed as the target lesion. PFS and quality of life constitute the primary endpoints, while OS is a secondary endpoint. 77
The STEREO-SEIN trial, a phase III study, is randomizing BC patients between systemic therapies and systemic therapies with associated SABR. In contrast with the OLIGOMA trial, only ‘de novo’ HR-positive patients are eligible, and brain metastases are not allowed. 78
The CLEAR trial (phase II, single arm) will assess the survival impact of RT and/or surgery in addition to first-line endocrine treatment in ER-positive/HER2-negative BC patients with low-burden oligometastatic disease (⩽2 lesions in a single organ). 79
A phase III randomized study sponsored by The Netherlands Cancer Institute will investigate a different aggressive approach for OMBC. This study is intended to assess the survival impact of a tandem high-dose alkylating chemotherapy regimen with high doses of carboplatin, thiotepa, and cyclophosphamide in OMBC HER2-negative patients harboring homologous recombination deficiency and not previously treated with chemotherapy in metastatic setting. High-dose chemotherapy remains an investigational strategy in selected BC patients. 82 In this protocol, the oligometastatic disease is defined as presenting up to three distant metastatic lesions, with or without a primary tumor. Patients in the control arm are treated with three cycles of standard chemotherapy. 80
Another breast-specific randomized study is the NRG BR002. This phase II/III trial was designed to evaluate the survival benefit gained with SABR and/or surgical resection. Eligible patients must have less than five lesions and controlled primary; the presence of brain metastases is an exclusion criterion. In the case of median PFS improvement, the study could have continued as a phase III trial with 5-year OS as the primary endpoint. 83
However, the PFS result was recently presented at the 2022 ASCO Annual meeting showing no benefit with the addition of metastasis-directed therapy to systemic therapy (mPFS of 23 months for SOC arm versus 19.5 months for SOC + ablation arm), thus blocking accrual for the phase III part of the trial.
Discussion
Using MDT and systemic therapy for oligometastatic disease is an attractive option, especially for BC patients, where long-term survival seems achievable.11,31 However, this assumption is mainly based on retrospective or prospective non-randomized studies (except for the SABR-COMET trial), which have inherent limitations and biases. The variability of oligometastatic disease definition among the reported studies hampers interpretation and comparisons of the results. Furthermore, the efficacy of the multimodal approach may have been underestimated given that a large part of the available data refers to an era before modern imaging, with potentially under-staged patients included in oligometastatic series, even if polymetastatic. 84
Randomized trials which will eventually validate this aggressive approach in clinical practice are still ongoing; currently, the application of local treatment for metastatic lesions varies widely between clinicians and different hospital departments and is often considered for palliative purposes rather than curative intent. The first issue in clinical practice is detecting the right candidate for multimodal therapy. Even though local treatment appears to be relatively safe, with less than 10% incidence of grade 3–5 toxic effects in patients treated with SABR, 85 correct patient selection is crucial to avoid useless toxicity. Identifying some prognostic factors, such as the presence of solitary metastasis, a DFI ⩾ 24 months, and HR status positivity, could be helpful for clinicians in the selection of the best candidate for aggressive multimodal treatment. 6
The choice to perform RT or surgery as a local ablative technique is also a matter of debate.
SABR has fewer side effects, is more suitable for multiple lesions treatment than surgery, and is the treatment modality with more supporting evidence. On the other hand, surgery allows the complete removal of the metastases and the acquisition of histological data.
Unmet points
The optimal timing of MDT remains a matter of debate as well. Should we treat metastases upfront, or should we prioritize systemic therapy and subsequently consider MDT? In the latter case, how long should we wait to initiate MDT? Based on current data, these questions remain unanswered, and ongoing trials seem not designed to solve this issue.
However, the most controversial topic remains the definition of the oligometastatic state itself. ESTRO-ASTRO’s proposal to consider the disease as oligometastatic in the presence of 1–5 lesions, safely treatable with local therapy, has certainly been a first step toward a homogeneous understanding of this setting, 14 even though the determination of the number of the metastatic sites can sometimes be misleading, with particular regard to lymph nodes involvement. Only a few studies specify counting each metastatic lymph node separately.73,82
The oligometastatic definition evolved further thanks to ESTRO-EORTC dynamic classification based on the moment of oligometastatic state presentation in the patient’s history. While patients with ‘de novo oligometastatic disease’ represent the ideal candidate for aggressive multimodal therapy, those with ‘induced oligometastatic disease’ have poor chances of achieving the cure by the addition of local treatment. 18
These classifications will surely be helpful for future trials and clinical practice, but the oligometastatic state cannot be defined only by imaging findings. It should also be considered from a biological point of view. Some studies demonstrated different miRNA expressions in patients with imaging-defined oligometastatic disease, which could potentially distinguish between patients prone to develop a poly-metastatic disease and those in a real oligometastatic state. 16 Furthermore, the SABR-COMET-3 and SABR-COMET-10 randomized trials will assess the role of circulating tumor cells and ctDNA as biomarkers for oligometastatic disease outcomes, and, hopefully, this will contribute to a more accurate insight into this setting.
Implementing advanced imaging for more accurate staging could also improve the imaging-based oligometastatic state definition. The use of whole-body magnetic resonance imaging (WB-MRI), for example, demonstrated better performance in the detection of bone and lymph nodes metastases at staging than other standard imaging techniques in prostate cancer patients. 86 Moreover, in a retrospective study including advanced BC, 58 patients underwent both whole-body MRI and standard imaging (PET-TC or CT); in 23 of these patients WB-MRI detected additional sites of disease and in 17 of 28 patients, progressive disease was reported only by WB-MRI. 87
The oligometastatic state is a hardly classifiable dynamic entity that could eventually change depending on systemic therapies innovations. The achievement of better outcomes and the potential synergy between novel therapies (such as ICIs) and RT could justify the use of an aggressive multimodal therapy aimed at long survival in a larger population of metastatic patients, extending the definition of oligometastatic.
The phase II part of the NRG-BR002 study, recently presented at the 2022 ASCO Annual Meeting, failed to show a signal for PFS improvement in OMBC patients treated with metastases-directed therapy. This result raises questions about the metastases-ablation role in this setting but it is not sufficient to abandon the multimodal approach. This trial has some limitations, for example, only 129 OMBC patients have been enrolled, the HER2-positive BC subtype was poorly represented and the vast majority of patients in the ablation arm were treated with SABR, while only 2% of them underwent surgical resection.
In conclusion, looking forward to other randomized trials and disease-specific study results, oligometastatic patients should be discussed by a multidisciplinary team. We already know that isolated sternal and contralateral axillary metastasis must be candidates for a local approach in addition to systemic therapy, but other OMBC patients could achieve long OS or could be cured with an aggressive multimodal approach, and we cannot miss this opportunity.
