Abstract
Background:
Colorectal cancer liver metastases (CRLM) represent a major cause of mortality in advanced colorectal cancer, with intra-arterial interventional therapy (IAIT) playing an increasingly important role in multidisciplinary management. This study aims to develop a machine learning (ML)-based prognostic model to predict survival outcomes in unresectable colorectal cancer liver metastases (uCRLM) patients undergoing IAIT treatment, enabling improved risk assessment.
Design:
A retrospective study.
Objectives:
This study aims to explore the effect of IAIT on the survival of patients with uCRLM.
Methods:
Retrospective data were obtained from patients with CRLM who visited Luhe Hospital and Peking University Cancer Hospital from January 2018 to January 2023. The study population was divided into two groups: one group received IAIT sequence by systemic standard of care (SOC) therapy group (ISOC; n = 340), while the other group received systemic SOC therapy alone (n = 234). To reduce potential selection bias between the two groups, propensity score matching (PSM) was employed. The primary outcome measured was overall survival (OS). A prognostic model for IAIT was then constructed using five supervised ML models. The performance of the model was assessed by calculating the area under the receiver operating characteristic curve (AUC) and decision curve analysis. Kaplan–Meier analysis was used to reveal the OS risk stratification of the ML. To assess the prognostic nature of our models, we will include interaction terms between treatment modalities and key prognostic factors, followed by likelihood ratio tests to evaluate their significance.
Results:
After PSM 1:1, 574 patients were divided into two groups. The median OS of patients who received ISOC was significantly higher than those who received systemic SOC therapy alone (40 vs 25 months, p = 0.036). Among the five ML models, the Random Survival Forest model demonstrated the most robust prognostic performance with 1-year, 2-year, and 3-year AUCs of 0.899 (95% confidence interval (CI): 0.858–0.939), 0.903 (95% CI: 0.864–0.943), and 0.873 (95% CI, 0.828–0.919). In the external validation cohort, the AUCs for 1, 2, and 3 years were 0.665 (95% CI: 0.455–0.875), 0.737 (95% CI: 0.636–0.837), and 0.730 (95% CI: 0.640–0.821), respectively. Kaplan–Meier curve analysis confirmed the model’s prognostic power for the ISOC treatment strategy. We tested for interaction effects between treatment modalities (e.g., ISOC vs SOC) and the ML model’s risk strata, but no significant interaction was observed (P for interaction p > 0.05).
Conclusion:
In this study, ISOC significantly improved the prognosis of patients. The ML model provides accurate prognostic stratification for uCRLM patients, which may aid in risk-based clinical decision-making.
Keywords
Introduction
Colorectal carcinoma (CRC) is one of the leading causes of cancer-related deaths worldwide, with a rising incidence in several Asian countries, notably China. 1 The liver is the most common site for metastasis in CRC patients, with approximately 50% developing liver metastases—15%–25% synchronously and 20% metachronously.2–7 Liver metastasis is a critical prognostic factor, with a 5-year overall survival (OS) rate of only 25%–40% in resectable colorectal cancer liver metastases (CRLM) patients.8,9 Unfortunately, only 10%–15% of CRLM cases are initially considered resectable, underscoring the importance of comprehensive treatment for unresectable CRLM.10,11
Current guidelines recommend a multidisciplinary treatment approach, including systemic therapy, surgery, and intra-arterial interventional therapies (IAITs) to manage CRLM and improve survival. 12 Systemic standard of care (SOC) therapy group for unresectable colorectal cancer liver metastases (uCRLM) involves (e.g., irinotecan 13 and oxaliplatin 14 ), modern combination regimens (e.g., FOLFOX (leucovorin fluorouracil, and oxaliplatin) 15 and FOLFIRI (LV, fluorouracil, and irinotecan)), and FOLFOXIRI (fluorouracil, oxaliplatin, and irinotecan) with or without targeted therapies (e.g., anti-epidermal growth factor receptor (EGFR; for KRAS wild-type tumors) and anti-vascular endothelial growth factor (VEGF) agents)16,17 to improve the conversion resection rate. 18 Approximately 20% of patients with initially CRLM may become candidates for surgical resection following various treatments such as chemotherapy, targeted therapy, or locoregional therapies like ablation or embolization. 19 This conversion from unresectable to resectable status can significantly improve outcomes for these patients.
IAITs, such as Hepatic Arterial Infusion Chemotherapy (HAIC) and Transcatheter Arterial Chemoembolization (TACE), are often used in palliation and conversion settings to target liver metastases from colorectal cancer.20,21 Studies indicate that HAIC can significantly benefit patients with initial uCRLM by potentially extending median OS and downsizing tumors to a resectable status.22–24 Intra-arterial administration of drugs with high first-pass hepatic extraction (such as fluorouracil (FUDR)) limits systemic toxicity and allows for simultaneous systemic chemotherapy at near full doses. 25 Modern chemotherapy drugs such as irinotecan and oxaliplatin are also used for arterial delivery, mainly in Europe and Asia.26,27 TACE, especially Drug-Targeted Arterial Chemoembolization is a treatment modality that combines chemotherapy and embolization to treat liver metastases. It involves the selective delivery of chemotherapy agents directly into the tumor through the hepatic artery, followed by embolization of the blood vessels supplying the tumor, which limits the tumor’s blood supply and enhances the local concentration of the drugs. TACE can also be combined with other local therapies such as percutaneous thermal ablation or systemic chemotherapy. 28 Based on current European Society for Medical Oncology guidelines, TACE should be considered a possible treatment option when patients with metastatic liver-limited disease do not respond to systemic chemotherapy. 29 However, studies examining prognostic factors for determining survival and treatment efficacy in patients with CRLM treated with DEBIRI TACE are rare.30,31 IAITs are one of several locoregional options for unresectable CRLM, though their use varies by institutional expertise. It is still an exploratory treatment rather than a standard protocol.
On the other hand, some studies indicate that IAIT treatment can improve progression-free survival (PFS) and OS in patients, controversy remains, as other research has failed to show significant benefits. These research results regarding ISOC for uCRLM are inconsistent. It is primarily due to the small sample sizes of phase II and retrospective studies, as well as the heterogeneity in tumor burden, molecular characteristics of liver metastases, and differences in antitumor treatment regimens, all of which complicate the data and weaken the reliability of the conclusions. To date, accurately assessing the potential benefits of ISOC for uCRLM is crucial for improving long-term survival outcomes. However, a noninvasive, convenient, and reliable model to guide physicians in ISOC decision-making is still lacking. This study aimed to develop and validate a machine learning (ML) prognostic model utilizing real-world data from patients with uCRLM who underwent ISOC. The ultimate goal is to provide robust evidence that guides physicians to identify uCRLM patients most likely to benefit from ISOC.
Methods
Study design and data selection
All a total of 574 uCRLM patients were screened and enrolled in this study, with 234 in the systemic standard of care (SCO) group and 340 patients in the IAIT sequence by SOC (ISCO)therapy group. A prognostic model based on ML was established using retrospective clinical data from the ISCO group and validated with data from separate institutions. The inclusion criteria were as follows: (a) aged 18–75 years, (b) had an Eastern Cooperative Oncology Group (ECOG) performance status <2, (c) No treatment received before admission; the exclusion criteria were as follows: (a) patients who received any treatment prior to inclusion in this study (e.g., ablation or radiation therapy), (b) had colorectal cancer (CRC) combined with other malignancies, and (c) were lost to follow-up for more than 6 months. Candidate predictors, such as demographics, clinical, dietary, and oncological factors, were obtained from medical records and patient interviews. The selection of prognostic criteria for locoregional interventional treatment was based on disciplinary expertise and a review of existing literature. Demographic data included age and sex, while clinical data encompassed illness complications, lymphocyte and leukocyte counts, hemoglobin levels, and BMI. Oncological data comprised information on the tumor’s location, histological type, carcinoembryonic antigen, gene status, tumor burden (tumor size and number of tumors) extrahepatic, metastasis, and T and N stages. The flowchart of the selection process for the uCRLM is presented in Figure 1.
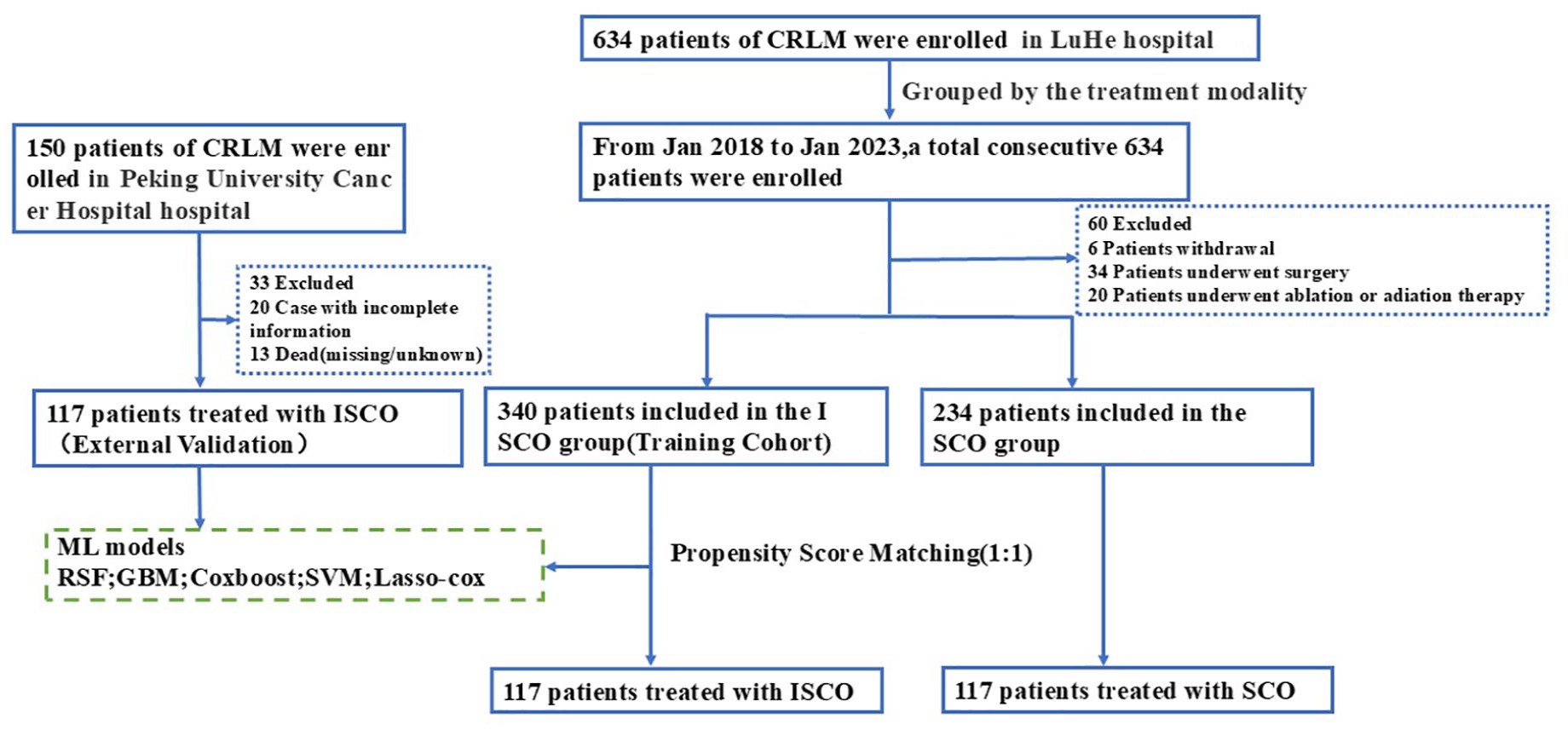
Flow diagram illustrating recruitment of patients.
Patients eligible for the study had an ECOG performance status ranging from 0 to 2, sufficient organ function, and a measurable tumor, evaluated using multi-detector spiral CT scanning according to Response Evaluation Criteria in Solid Tumors (RECIST) version 1.1 guidelines. According to the treatment method, we divided the patients into two groups. Namely, the ISOC group and SOC group through Multidisciplinary Treatment, The ISOC group received sequential intervention therapy after 4–6 cycles of first-line systemic treatment, with at least 1 intervention every 3 weeks. The SOC group received maintenance therapy after eight cycles of systemic treatment and was observed for responses and survival outcomes. Tumors were classified based on the AJCC TNM staging system, 8th edition for colorectal cancer.
Follow-up protocol and endpoint definition
Patients were routinely followed until death or the end of the study (December 31, 2023). Patient evaluations were scheduled prior to the first treatment or followed up every three cycles. Assessments of safety were performed according to the Common Terminology Criteria for Adverse Events, version 5.0, published by the US Department of Health and Human Services. Patients received follow-up with contrast-enhanced CT/magnetic resonance imaging (MRI) every 8–12 weeks. For tumor response assessment, objective responses after treatment were evaluated on the basis of contrast-enhanced or MRI (CT/MRI) scans, which were independently reviewed and interpreted according to the RECIST criteria by two physicians with extensive experience in abdominal imaging as described above. In case of disagreement between the two evaluations, a consensus conclusion was reached.
ML construction
Five representative supervised ML algorithms (Random Survival Forest (RSF), Gradient Boosting Machines (GBM), Cox Proportional Hazards Model Boosting (COXboost), Support Vector Machine, and Lasso-Cox) were applied and compared in this study. The ML-based model with optimal discrimination was used for building a simpler restricted system for guiding the selection of ISOC schemes, which was derived from clinical information.
Dynamic prognostic risk based on ML model
This study integrates multiple factors (RAS mutation, tumor size, Neutrophil-to-Lymphocyte Ratio (NLR), tumor site, Hb levels, Extrahepatic metastasis, and T-stage) and quantifies individual outcome probabilities using algorithmic tools such as ML. Second, the ML model was used to help physicians decide whether to adjust the sequential schemes during the follow-up monitoring process based on response to IAITs and tumor recurrence. In addition, death predictions for 1, 2, and 3 years were analyzed. A total of 17 variables closely related to clinical outcomes were collected to construct the ML model.
Statistical analysis
Continuous data were presented as the median with interquartile range, while categorical variables were reported as frequency and proportion. The Mann–Whitney U test was applied for continuous variables, and the χ2 test was used for categorical variables. All statistical analyses were conducted using R software version 4.3.1 (http://www.r-project.org/). The performance of ML models was assessed by calculating the area under the receiver operating characteristic curve (AUC) and decision curve analysis (DCA). The models were further evaluated using receiver operating characteristic (ROC) curves and the concordance index (C-index). Time-dependent ROC curves and AUC values, measured at different time points from 12 to 36 months, were employed to evaluate OS prediction accuracy over time. Model prediction error was quantified using the integrated Brier score. To interpret the causal relationships indicated by the ML models, survival curves were generated using the Kaplan–Meier method and compared with the log-rank test. All significance tests were two-sided, with a p-value of less than 0.05 considered statistically significant.
Results
Demographics and clinical characteristics
Table 1 lists the baseline characteristics of the patients stratified according to treatment modality; there are 124 females and 216 males. The age of the patients had a median value of 66 years. Out of the 574 patients, 340 (59.23%) received local interventional treatment. The rectum accounted for 36.76% of the tumors, making it the most common site. The left colon accounted for 41.11% of the tumors, while the right colon accounted for 22.13%. The majority of patients with colon cancer presented with advanced clinical stages, with T4 being the most common (33.10%), followed by T3 (31.53%), T2 (12.37%), and T1 (22.30%). Before matching the first two groups based on the BMI, Diabetes, Cardiovascular-comorbidity, T stage, and Time to metastases, the difference was statistically significant (all, p < 0.05), and the balance of the distribution of the general clinical information between the groups was poor. After using the propensity score matching (PSM; 1:1) to match patients with the most similar propensity scores of the two groups, the result was that the two groups were successfully matched to 117 pairs of patients, and all the variables were not significantly statistically significant (p > 0.05) after matching. In the validation cohort, patient demographics and pathological characteristics are shown in Supplemental Table 1.
Baseline characteristics of the patients with CRLM who received IAIT and SCO therapy before and after PSM.

CEA, carcinoembryonic antigen; CRLM, colorectal cancer liver metastases; IAIT, intra-arterial interventional therapy; PSM, propensity score matching; SD, standard deviation.
Comparison of the prognostic performance of the ML models
For local interventional therapy ML models, five ML algorithms—Lasso-cox, RSF, COXboost, GBM, and Survivalsvm—were utilized to create models, and their performance was evaluated using ROC curves (Figure 2). The RSF model exhibited the utmost precision in forecasting the influence of local treatment on survival, (Figure 2(a)–(c)) with a 1-year AUC of 0.899 (95% confidence interval (CI): 0.858–0.939), a 2-year AUC of 0.903 (95% CI: 0.864–0.943), and a 3-year AUC of 0.873 (95% CI, 0.828–0.919). In the validation cohort, the AUCs for 1, 2, and 3 years were 0.665 (95% CI: 0.455–0.875), 0.737 (95% CI: 0.636–0.837), and 0.730 (95% CI: 0.640–0.821), respectively (Figure 2(d)–(f)). The number of predictors and AUC, Brier score, and C-index of each ML model based on clinical variables for the prediction of 3-year death are shown in Tables 2 and 3.

ROC curves of five machine learning algorithm models predicting the survival risk of colorectal cancer patients receiving local intervention treatment in the training set (a–c) and validation set (d–f) at 1, 2, and 3 years. The RSF model shows the best AUC performance.
OS in the training cohort.

CI, confidence interval; OS, overall survival.
OS in the external validation cohort.
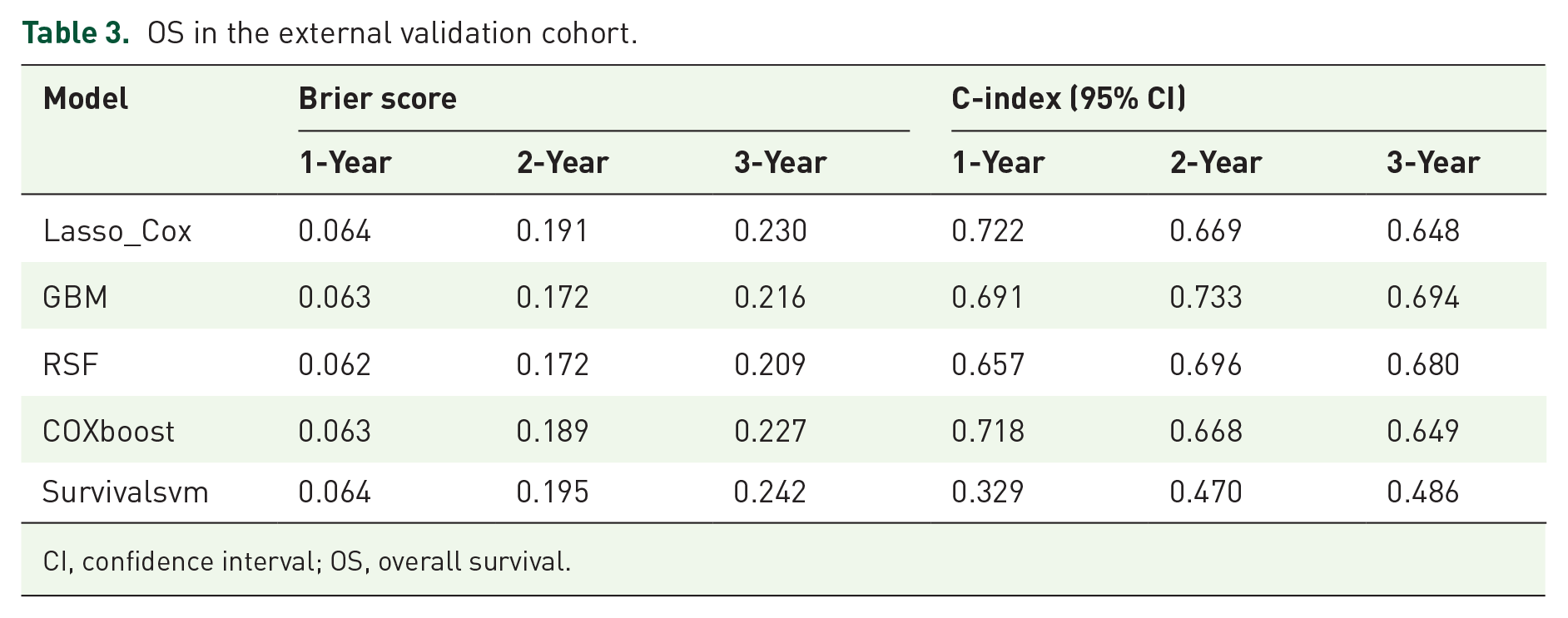
CI, confidence interval; OS, overall survival.
In this study, the models were evaluated with the external verification set. The RSF model outperformed the other ML model. Model performance was evaluated on the training and validation set by C-index (Figure 3). The RSF model C-index of the training set during follow-up was analyzed with time-to-event analysis, which proved the good accuracy of the OS prediction. The verification of the RSF model of C-index is also higher than that of the other model, which indicates that the RSF model C-index is well calibrated, meaning that the prediction probability of each subgroup is close to the observation probability. The DCA for the ML is presented in Figure 4((a)–(f)). The decision curve showed that if the threshold probability of the patient is between 0.3 and 0.95, using both the IAIT and SOC in the current study to predict OS added more benefit than SOC, the DCA of the RSF model showed fair clinical net benefits for the 1-, 2-, and 3-year survival prediction both internal and external validation sets. In the calibration curve of the five ML models, RSF has higher capability (Supplemental Figures 1–5).
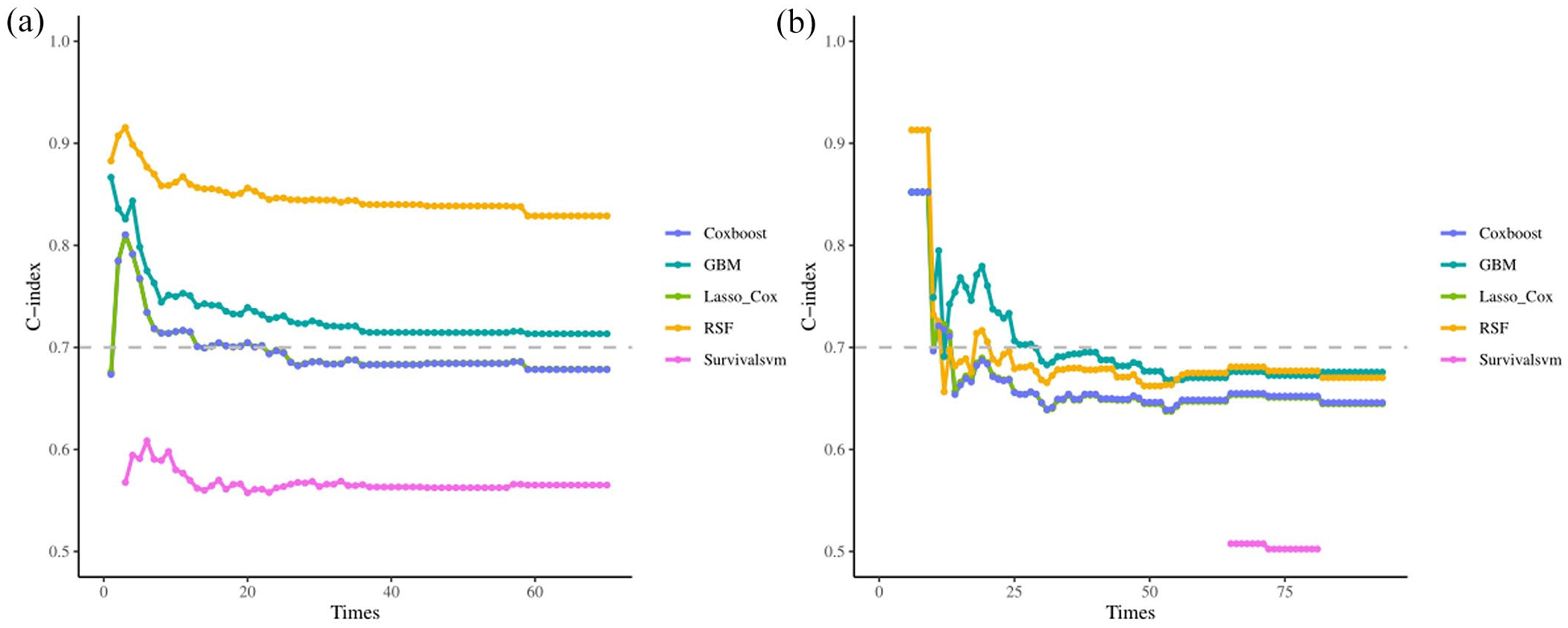
The C-index trends over time in both training (a) and validation (b) sets.
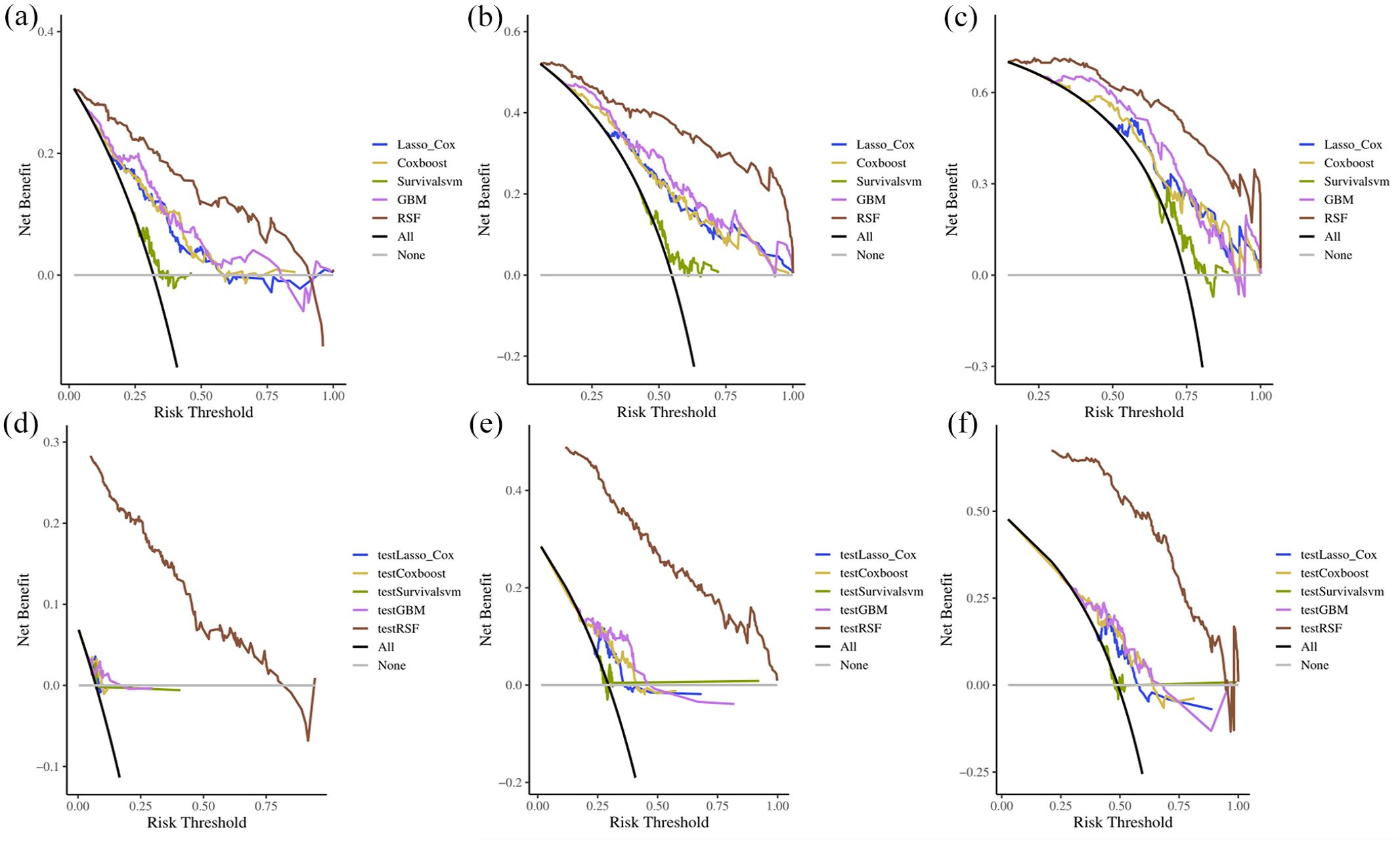
The DCA of the ML model in the train sets (a–c) and validation sets (d–f). (a) The 1-year decision analysis curve of the ML model. (b) The 2-year decision analysis curve of the ML model. (c) The 3-year decision analysis curve of the ML model. In the decision analysis curve, the x-axis represented the threshold probability, while the y-axis represented the clinical net benefits.
A RSF classifier was used to develop a risk prediction model that includes these factors. After adjusting the parameters, a consistent error rate was observed when the number of trees (ntree) was set to 100. The Gini coefficient showed that RAS mutation had the greatest impact on predicting the effect of local treatment on survival, followed by tumor size, NLR, site, Hb, extratropical metastasis, and T-stage (Figure 5).
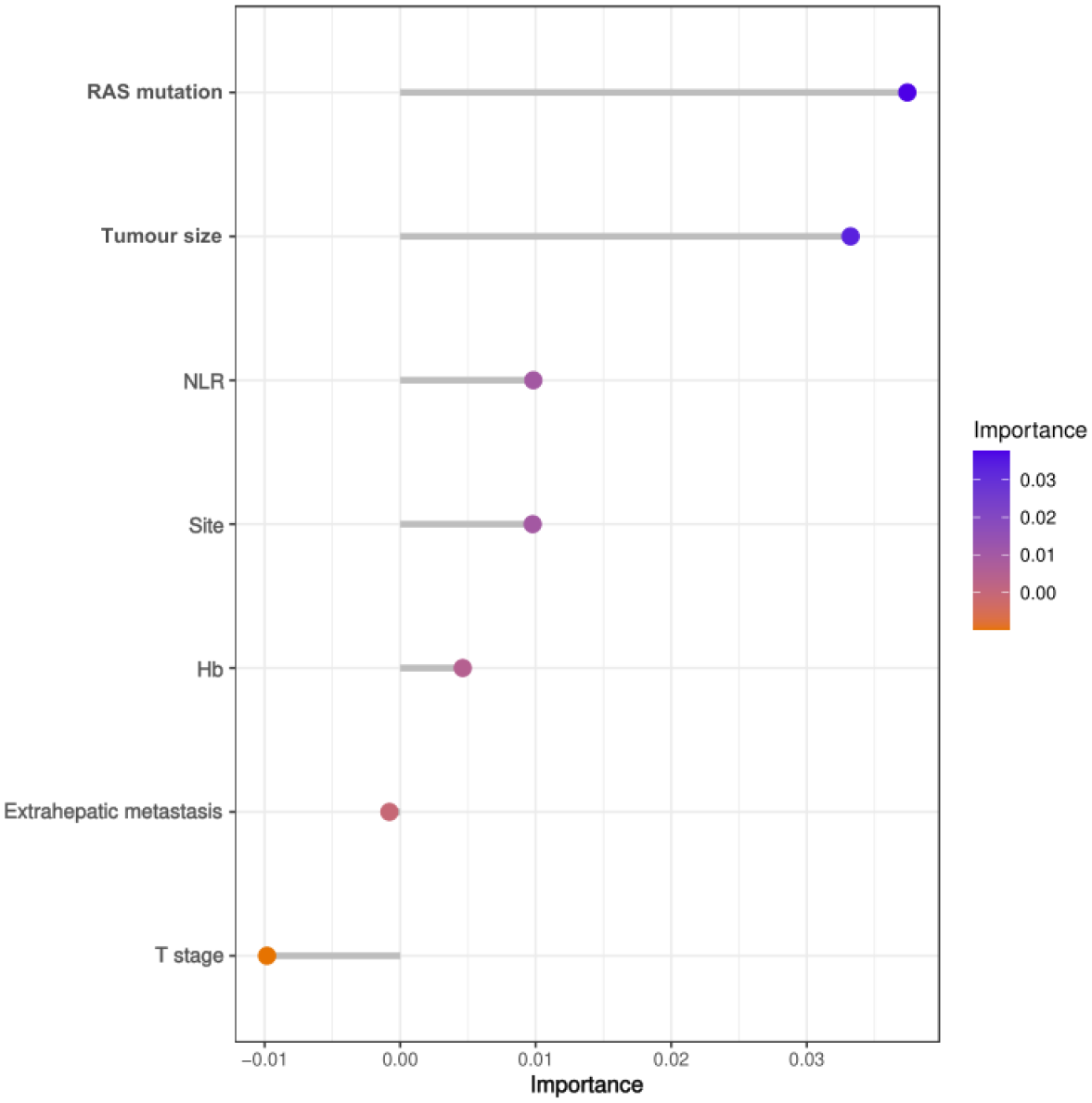
Variable importance based on random forest, ranking of out-of-bag variable importance.
ML model for identifying survival benefits
Effective patient management requires appropriate risk stratification. Generate a risk score by summing up the risk scores of each patient’s RSF. According to the risk score of RSF in the training cohort, CRLM patients were divided into two groups: high-risk group (>31.01 points) and low-risk group (⩽31.01 points; Figure 6(a) and (b)). Based on the prognostic model scores, we stratified patients in both the ISOC and SOC cohorts into low-risk and high-risk subgroups. Our analysis revealed that low-risk patients in the ISOC group demonstrated significantly longer median OS compared to their SOC counterparts (p = 0.013), whereas no statistically significant survival difference was observed between treatment groups in the high-risk stratum (p = 0.12; Figure 7(a) and (b)). These findings highlight the model’s further reinforcing its potential role in guiding therapeutic decision-making. Critically, our results demonstrate that the model effectively stratifies patients by prognostic risk, with the low-risk subgroup appearing to derive benefit from locoregional therapy. Most importantly, our data suggest that high-risk patients may not gain significant survival advantages from aggressive locoregional interventions, supporting the judicious avoidance of overtreatment in this specific patient population.
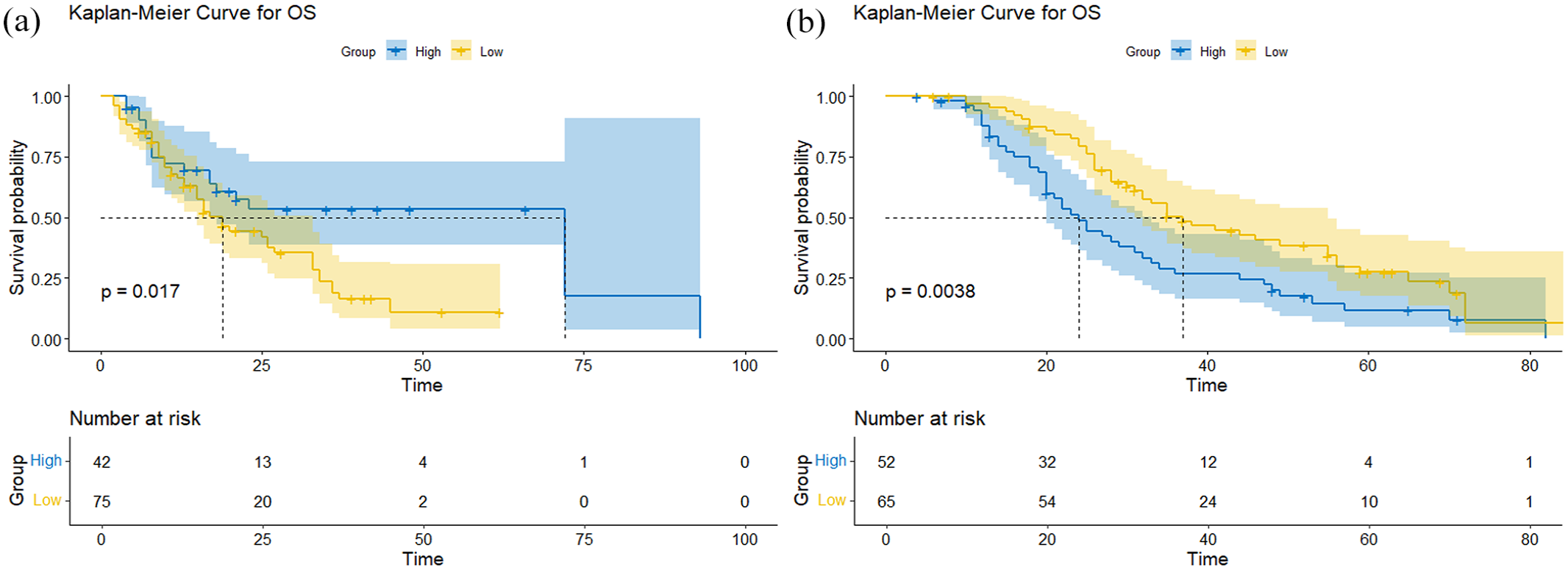
(a, b) Kaplan–Meier curve shows the comparison of overall survival rates between the low-risk and high-risk groups predicted based on RSF in the (a) ISOC of the development cohort, and (b) ISOC of the validation cohort.

(a, b) Kaplan–Meier analysis demonstrates the comparative overall survival between ISOC and SOC groups in (a) the low-risk cohort and (b) the high-risk cohort.
Test of interaction for ML model
We then tested for effect modification by RAS mutation, Primary Site, T-stage, Extrahepatic metastasis, NLR, Hb, and Tumor size. Interaction on the multiplicative scale was assessed by conducting likelihood ratio tests, and on the additive scale by calculating the relative excess risk caused by interaction. The results of these interaction analyses (Figure 8) showed no statistically significant interactions (all P for interaction >0.05). Our model stratifies patients into distinct prognostic groups, with high-risk patients showing poorer OS, regardless of treatment type.

Interaction forest plot for treatment modality subgroups.
Discussion
In the current study, we investigated the impact of the ISOC regime on the prognosis of unresectable colorectal cancer and identified it as an independent prognostic factor for uCRLM. ISOC has emerged as a promising treatment option for uCRLM, with accurate and well-informed decision-making playing a crucial role in achieving long-term survival benefits. Identifying the patients who are most likely to benefit from this treatment is essential. Studies investigating prognostic factors for survival and treatment efficacy in patients with uCRLM undergoing IAIT sequences by systemic SOC are rare. This study is the first to utilize ML models to predict the impact of ISOC on the survival of patients with uCRLM. Five ML prediction methods were evaluated using patients’ clinical characteristics. Among these, the RSF model exhibited the best prognostic performance, achieving the highest area under the curve (AUC) value. Key factors such as RAS mutation, tumor size, NLR, tumor site, Hb levels, extratropical metastasis, and T-stage were identified as the key factors most significantly impacting outcomes. Importantly, these variables can be easily obtained from routine medical records, offering clinicians accessible and actionable insights. This information plays a crucial role in understanding precision therapy strategies and optimizing treatment sequences to improve patient outcomes.
The study demonstrates improved prognostic stratification, enabling better risk-based clinical decisions. In this study, ISCO significantly improved OS in uCRLM compared to SCO, indicating IAIT’s potential as an effective treatment option for uCRLM. As has been reported previously in potentially resectable CRLM, IAIT significantly enhances survival and quality of life.32–35 There remains a lack of robust research data on the role of IAIT combined with systemic therapy in patients with uCRLM. Most existing studies are limited to phase II trials with small sample sizes, largely due to the absence of clearly defined population characteristics that benefit from the combination of IAIT and systemic therapy. Accurate and reasonable IAIT decision-making is essential for achieving long-term survival benefits. Therefore, we developed an ML prognostic model to guide clinical decision-making for the IAIT sequence by SOC treatment. The ML model effectively stratifies CRLM patients who have undergone IAIT into two distinct risk groups, each demonstrating significantly different long-term OS outcomes. Consequently, this model may enhance personalized decision-making while reducing mortality rates, treatment-related side effects, and the economic burden for more uCRLM patients undergoing ISOC.
This study has several notable strengths and novel aspects. First, the ML model was developed and externally validated using data from two hospitals in China. The dataset included 574 patients, with 17 readily available clinical variables and a relatively long median follow-up time of 4.2 years. These factors collectively ensure the model’s comprehensiveness, robustness, and accuracy from multiple perspectives. Second, the ML model was constructed using a diverse set of variables, encompassing demographic characteristics, tumor features, laboratory test results, and treatment regimens, thereby ensuring a holistic approach to patient data. Third, we evaluated the performance of five representative ML models and identified the RSF model as the most suitable. The RSF model, incorporating the most rational selection of variables, was subsequently employed to develop a robust ML framework for stratifying prognostic risk. Furthermore, we developed a prognostic risk score for patients with uCRLM sequence by ISCO treatment based on the optimal ML model. By relying on the pretreatment risk scores generated by the ML model, patients received more precise and individualized IAIT treatment. Our model identified a subgroup of patients (low-risk) with significant survival benefits from ISCO, while high-risk patients did not benefit beyond chemotherapy. This supports personalized treatment allocation. Our ML model improves prognostic stratification in CRLM patients, aiding personalized clinical management.
Our study demonstrates a significant correlation between RAS mutations and poor prognosis in patients, consistent with previous studies on the metastasis and survival of patients with initially uCRLM-administrated systemic and hepatic arterial chemotherapy as characterized by different genomic profiles. 36 In recent years, genomic biomarkers have been identified as key factors in the prognosis of CRLM patients undergoing resection. For example, RAS mutations are associated with an increased risk of postoperative recurrence. In the current study, we also found that patients with the RAS wild-type gene may benefit more from ISCO treatment than from SCO treatment for uCRLM.
Previous studies have shown that tumor morphology factors, such as tumor size and quantity, are important indicators affecting the prognosis of CRLM patients.37–39 Sljivic M’s study indicated that patients with four or fewer liver metastases had better survival outcomes than those with more. However, given the considerable variation in lesion size, metastatic volume may be a more reliable prognostic factor. 29 In our ML model, both tumor size and number were incorporated. However, Zakaria et al. 39 evaluated the general applicability of several main methods using risk-scoring systems based on tumor size and quantity and concluded that these models are only slightly better than pure chance in predicting disease-specific survival rates. One potential reason for the poor performance of these scoring systems may be that they were developed before the widespread use of modern chemotherapy. Many CRLM patients now receive modern chemotherapy, and tumor morphology is influenced by the acceptance and response to preoperative chemotherapy.
The NLR is increasingly recognized as a useful inflammatory biomarker for the prognosis of various cancers, including CRC.40,41 Ueno A et al. 42 reported that human neutrophil peptides are upregulated in CRC patients and are closely associated with lymph node and liver metastasis. Further research has established a correlation between an increased NLR and poor prognosis in individuals with CRLM. 40 Our study indicated that the NLR is correlated with prognosis in uCRLM patients treated with the ISCO strategy, as determined through the application of ML techniques.
The ML model incorporates simple, readily available clinical variables that can effectively identify uCRLM patients at high risk. Low-risk patients should receive a combination of systemic and local comprehensive treatment. The model efficiently classified uCRLM patients into two subgroups based on different risk levels and helped identify the most suitable candidates for the IAIT sequence by SOC. Within the training cohort, 45% of the uCRLM patients belonged to the high-risk group, which had a 5-year death rate of 75.6%. In contrast, the low-risk group had a 5-year death rate of 26.8%. The median survival time for patients classified as high-risk by the ML model is worse than that of low-risk patients in uCRLM (p < 0.001), and they do not benefit from the ISOC treatment strategy.
Our research has some limitations. Although it provides valuable insights into the treatment outcomes that affect the prognosis of uCRLM patients, this study is limited by its relatively small sample size, which may limit its statistical power and the generalizability of the results. The potential for selection bias and uncontrolled confounders inherent in retrospective studies remains a concern. To address this, we employed PSM to minimize between-group variability. Second, to verify the accuracy of the study data, many patients were excluded from the sample, and reducing the sample size may have introduced bias. In the future, we will conduct prospective clinical studies based on the ML model risk scores to guide treatment decisions for patients with uCRLM, aiming to explore the decision-making value of the ML model risk scores in the comprehensive treatment of uCRLM patients. The applicability of the model to patients outside of China remains uncertain. While the risk score stratified OS, it did not differentially predict SOC benefit. Future studies should incorporate treatment-specific features (e.g., tumor response to SOC) to improve prognostic power.
Conclusion
This model aims to identify key prognostic factors and enhance treatment decision-making. The model helped identify patients at risk, allowing early intervention to reduce the poor prognosis. While this model does not predict treatment-specific benefits, it provides robust prognostic stratification that could aid in risk-adapted clinical management.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251353084 – Supplemental material for A machine learning-based prognostic stratification of locoregional interventional therapies for patients with colorectal cancer liver metastases: a real-world study
Supplemental material, sj-docx-1-tam-10.1177_17588359251353084 for A machine learning-based prognostic stratification of locoregional interventional therapies for patients with colorectal cancer liver metastases: a real-world study by Jing Wang, Kai Wang, Kangjie Wang, Baogen Zhang, Siyu Zhu, Xuyang Zhang, Li Wang, Yingying Tong, Aiwei Feng, Haibin Zhu, Ting Xu, Xu Zhu and Dong Yan in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251353084 – Supplemental material for A machine learning-based prognostic stratification of locoregional interventional therapies for patients with colorectal cancer liver metastases: a real-world study
Supplemental material, sj-docx-2-tam-10.1177_17588359251353084 for A machine learning-based prognostic stratification of locoregional interventional therapies for patients with colorectal cancer liver metastases: a real-world study by Jing Wang, Kai Wang, Kangjie Wang, Baogen Zhang, Siyu Zhu, Xuyang Zhang, Li Wang, Yingying Tong, Aiwei Feng, Haibin Zhu, Ting Xu, Xu Zhu and Dong Yan in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
