Abstract
Background:
Immunotherapy combined with intra-arterial therapy (IAT) has shown great potential in the treatment of unresectable hepatocellular carcinoma (uHCC). However, there are currently no available biomarkers that can predict the prognosis of immune-based combined therapy.
Objectives:
To establish a scoring method to predict prognosis in uHCC patients undergoing IAT plus immunotherapy.
Methods:
Between March 2019 and August 2022, uHCC patients undergoing IAT in combination with programmed cell death (ligand) 1 (PD-1)/PD-L1-based immunotherapy were retrospectively analyzed.
Results:
Among 1046 patients included, 780 patients were enrolled into hepatic arterial infusion chemotherapy immunotherapy cohorts (training set: n = 546, one center; external testing set: n = 234, three centers) and 266 patients were treated with trans-arterial chemoembolization (TACE) plus immunotherapy were enrolled into TACE immunotherapy cohort (validation set: n = 266). We developed the easy-to-apply alpha-fetoprotein (AFP), C-reactive protein (CRP), and platelet-to-lymphocyte ratio (PLR) in immunotherapy (AFCRPLITY) score and investigated the prognostic value of baseline variables on the disease control rate (DCR) and progression-free survival (PFS). HCC patients with low AFCRPLITY scores would have better PFS and DCRs than patients with high AFCRPLITY scores (AFCRPLITY 0: vs AFCRPLITY 1: vs AFCRPLITY 2: vs AFCRPLITY 3: p < 0.001 for PFS, p = 0.001 for DCRs) in the training set, which was confirmed in the external testing set and validation set. The highest level of CD8+ T cells was in the AFCRPLITY score = 0 group than the other two groups.
Conclusion:
The AFCRPLITY score is associated with PFS and DCR in uHCC patients receiving IATs plus immunotherapy. This score may be helpful for counseling, but prospective validation is needed.
Design:
A retrospective, multi-institutional study.
Trial registration:
The study has been retrospectively registered at the Chinese Clinical Trial Registry (https://www.chictr.org.cn/, ChiCTR2300075828).
Keywords
Introduction
Hepatocellular carcinoma (HCC), which is the most common primary liver cancer, usually develops in patients who have underlying cirrhosis.1,2 In China, the main etiological risk factor for HCC is hepatitis B virus (HBV) infection, as individuals with HBV are more prone to disease progression. Due to hidden symptoms, plenty of HCC patients are already in an advanced stage at diagnosis. 3 In recent times, there has been a growing use of several tyrosine kinase inhibitors (TKIs) and immune checkpoint blockers (ICBs) for the treatment of advanced hepatocellular carcinoma (Ad-HCC).4–6 Atezolizumab and bevacizumab have been recommended as first-line treatments for Ad-HCC in the 2022 Barcelona Clinic Liver Cancer (BCLC) guidelines. 7
Hepatic arterial infusion chemotherapy (HAIC) is a standard intra-arterial therapy (IAT) that could effectively reduce the intrahepatic tumor burden by directly delivering chemotherapeutic agents into the arteries that nourish tumors.8,9 Lyu et al. 10 reported that the efficacy and safety of HAIC using the FOLFOX regimen (oxaliplatin plus fluorouracil and leucovorin) were superior to the efficacy and safety of sorafenib. Chemotherapeutic agents have been shown to exert synergistic anticancer effects with TKIs and ICBs. Our team designed a combination protocol using HAIC combined with Apatinib plus Carrilizumab (TRIPLET protocol) to treat Ad-HCC, and the preliminary results reported that HAIC-based triple therapy has a longer progression-free survival (PFS) and overall survival (OS) to Ad-HCC. 11
Previously, several biomarkers have been identified as predictors of response during the immunotherapy process, including activated Wnt/b-catenin signaling and programmed death ligand 1 (PD-L1) expression. In addition, the CheckMate 040 study demonstrated that an inflammatory gene profile strongly influences the prediction of nivolumab treatment responses in Ad-HCC, 12 indicating that inflammatory biomarkers may be useful in identifying patients who will benefit from immunotherapy. Based on these results, Scheiner et al. 13 developed a simple and easy-to-apply scoring method to predict OS after immunotherapy, thus helping with patient counseling and treatment decision-making. Molecular markers such as ctDNA are also under exploration, which can effectively predict the prognosis and survival of HC. At present, most studies are focused on the prediction of monotherapy in HCC, while combined therapy has become the first-line treatment in most cases. Currently, there is a lack of validated biomarkers to guide decisions for Ad-HCC patients who underwent IATs combined with immunotherapy. In the current study, we established a simple and easy-to-apply scoring method to predict PFS in HCC patients undergoing HAIC/TACE plus immunotherapy.
Materials and methods
This retrospective, multi-institutional study protocol was conducted by the principles of the 1975 Helsinki Declaration. The STROBE statement for observational cohort studies was followed.
Study design and patient enrollment
HAIC plus immunotherapy cohorts
The American Association for the Study of Liver Disease and the European Association for the Study of Liver guidelines were used to diagnose all HCC patients.14,15 Data were reviewed from March 2019 to August 2022 from 2289 consecutive patients with HCC who underwent initial HAIC combined with anti-PD-1/PD-L1 at four tertiary hospitals in China: Shandong Cancer Hospital and Institute, The First Affiliated Hospital of Sun Yat-Sen University (FAHSUSY), Sun Yat-sen University Cancer Center (SUSYCC), and The Third Affiliated Hospital of Sun Yat-Sen University (Supplemental Methods E1.1–1.3). The distribution of clinical data sources is shown in Supplemental Table 1. The inclusion criteria were as follows: (a) aged 18–75 years; (b) Child-Pugh class A or B; (c) Eastern Cooperative Oncology Group (ECOG) performance status 0 or 1; (d) the patients with HCC were either pathologically or clinically confirmed according to American Association for the Study of Liver Disease 3 ; (e) C-reactive protein (CRP), alpha-fetoprotein (AFP), and platelet-to-lymphocyte ratio (PLR) were test within 1 weeks before therapy. The following individuals were excluded: (a) history of any antitumor treatments; (b) those who combined with other cancers; and (c) incomplete clinical and follow-up data. The training set included patients from SUSYCC from March 2019 to August 2022. Thirty-five patients from the phase II clinical trial (NCT04191889) of SUSYCC were included in the training set. 11 The patients who received HAIC combined with immunotherapy in the other three hospitals were assigned to the validation set from January 2020 to December 2021. Figure 1 shows the enrollment procedures for qualified patients. Supplemental Methods E1.1–1.3 include details on the HAIC processes, the combined protocol of immunotherapy, and the criteria for protocol treatment discontinuation.

Enrollment pathway of patients with unresectable HCC who received HAIC and TACE combined with PD-(L)1-based immunotherapy.
TACE plus immunotherapy cohorts
From May 2019 to June 2022, data from a total of 2432 consecutive patients with HCC who underwent initial TACE combined with immunotherapy at 8 tertiary hospitals were reviewed. The distribution of clinical data sources is shown in Supplemental Table 1. Similar to the HAIC plus immunotherapy cohorts, only patients who had data regarding their baseline serum levels of AFP, platelets, lymphocytes, and CRP were eligible. The exclusion criteria were the same as those for the HAIC plus immunotherapy cohorts. The TACE procedure is described in Supplemental Methods E1.4. Retrospective data collection was used to gather demographics, oncological features, and laboratory results for each cohort. Immunotherapy was started at the baseline.
Assessments and follow-up
In this research, enrolled HCC patients were censored on October 1, 2023, the last follow-up date. After thorough IATs were accomplished, the serum AFP and dynamic contrast-enhanced images (e.g., CT or MRI) were examined again at 3- to 6-month intervals during the follow-up period and at approximately 3-month intervals in the first year. If disease progression was not found sequentially, follow-up was performed at 6-month intervals. Using the modified Response Evaluation Criteria in Solid Tumors, which includes complete response (CR), partial response (PR), stable disease (SD), and progressing disease (PD), the responses were evaluated using dynamic contrast-enhanced CT or MRI. 16 Two radiologists (reader 1, C.A., and reader 2, W.L., with 10 years of experience) conducted the assessment every 4–6 weeks following the start of treatment. The primary endpoints were PFS and extrahepatic PFS (EPFS). The date from the first IATs to the date of PD or the end of the follow-up was applied for the calculation of the PFS. The objective response rate (ORR) and disease control rate (DCR) were the second set of outcomes. The proportion of patients with CR and PR lasting longer than 4 weeks following the initial radiological confirmation was referred to as ORR. The fraction of patients with CR, PR, and SD was known as the DCR.
Analysis of multiplex immunofluorescence staining
The tumor tissues were obtained from 60 HCC patients, collected from both SUSYCC and FAHSUSY between 2019 and 2022. These tissues were properly preserved using formalin fixation and paraffin embedding techniques to create 4-μm paraffin sections. The Mantra System (PerkinElmer, Waltham, MA, USA) was used to capture the multispectral immunofluorescence images with the fluorescence spectra at 20-nm wavelength intervals from 420 to 720 nm with the same exposure time, which were then composited to establish a single stack image. To capture images of sections without autofluorescence, we extracted the spectrum of autofluorescence of TMAs and each fluorescein from the images of unstained and single-stained sections, which were used to establish the spectral library for multispectral unmixing using inForm image analysis software (PerkinElmer, Waltham). Two independent pathologists analyzed and counted single-positive cells and the expression of these genes in each tissue at 200× magnification in a blinded manner. The nucleated stained cells were quantified and expressed as the number of cells. Tumor tissues verified by hematoxylin and eosin staining were incubated successively with primary antibodies, including cytokeratin 8 (CK8)/18 (Novus, Los Angeles, America, NBP2-44929, 1:500), CD57, also named beta-1,3-glucuronyltransferase 1 (abcam, ab82749, 1:200) and CD8 (abcam, ab237709, 1:200), and peroxidase-conjugated secondary antibodies. The positive rate of the single index and single index intensity score was employed to evaluate the expression and distribution of the identified genes in cancer, which were calculated by multiplication of the multiplex immunofluorescence staining intensity (percentage of single index % = number of positive cells/total number of cells; the 25% staining was taken as the threshold of the strength score; 25%–49%, strength I; 50%–74%, strength II; 75%–100%, strength III; single index strength score = ((strength I * positive rate of single index) * 1 + (strength II * positive rate of single index) * 2 + (strength III * positive rate of single index) * 3) * 100).
Statistical analysis
Mann–Whitney U and Kruskal–Wallis tests were used to compare the quantitative variables with mean ± standard deviation (SD) or median with interquartile range (IQR). The qualitative variables with frequency were compared using the χ2 test. The univariate and multivariate Cox regressions were used to identify the independent prognostic variables. The Kaplan–Meier technique and log-rank test were employed to evaluate the prognosis.
This approach was used to provide an easy-to-apply scoring method that is simple to use and offers extra prognostic information on elements that are previously known. Dose–response relationships and tests for nonlinear associations were carried out for the originally continuous variables (PLR, CRP, and AFP) using restricted cubic spline (RCS) regression with three knots (positioned at the 5th, 50th, and 95th percentiles) based on multivariate Cox proportional hazard models. These findings led to the selection of simple-to-remember cutoff values for PLR, AFP, and CRP. These levels were the first to be recommended for uHCC patients receiving HAIC in addition to PD-(L)1-based immunotherapy. The simple score was named AFCRPLITY (AFP, CRP, and PLR in ImmunoTherapY), and it was validated and tested to stratify the PFS of HCC patients who received immunotherapy. We verified the score in an external, independent cohort to prevent findings from being too optimistic, given the score was developed and tested on the same dataset. The bootstrap approach was used to validate the model internally. Statistical analysis was performed using SPSS version 25.0 and R software version 4.3.0.
Results
Baseline characteristics
In total, 780 HCC patients (93 females and 687 males; median age, IQR: 51.00, 42.00–60.00) who received HAIC plus immunotherapy were enrolled. To develop and validate the AFCRPLITY score, these enrolled patients were divided into two datasets: a training dataset (n = 546) from SUSYCC Hospital and an external testing dataset (n = 234) from the other three hospitals. Moreover, a total of 266 HCC patients (37 females and 229 males; median age, IQR: 53.50, 43.00–62.00) who underwent TACE plus immunotherapy SUSYCC Hospital were assigned to the test dataset to determine the robustness of this AFCRPLITY score. The baseline characteristics of the HCC patients in the training, validation, and test datasets are summarized in Table 1. Except for ascites, tumor diameter, vascular invasion, metastasis, and BCLC stage, the distribution of other clinical variables showed no significant difference among the three datasets (all p > 0.05). The types of immunotherapeutic agents used are shown in Supplemental Table 2. Immunotherapy and TKIs were used synchronously and asynchronously in 879 (84%) and 167 (16%) patients, respectively. The majority of patients (62.9%) had advanced-stage HCC and Child-Pugh class A (94.3%). The median duration of estimated follow-up was 17.43 months (IQR, 10.00–26.63) in the whole cohort, including 16.58 months (9.98–25.81 months) in the training dataset, 18.00 months (IQR, 10.23–25.40 months) in the test dataset, and 18.38 months (IQR, 10.12–30.23 months) in the validation dataset.
Baseline characteristics of HAIC and TACE combined with immunotherapy cohorts.
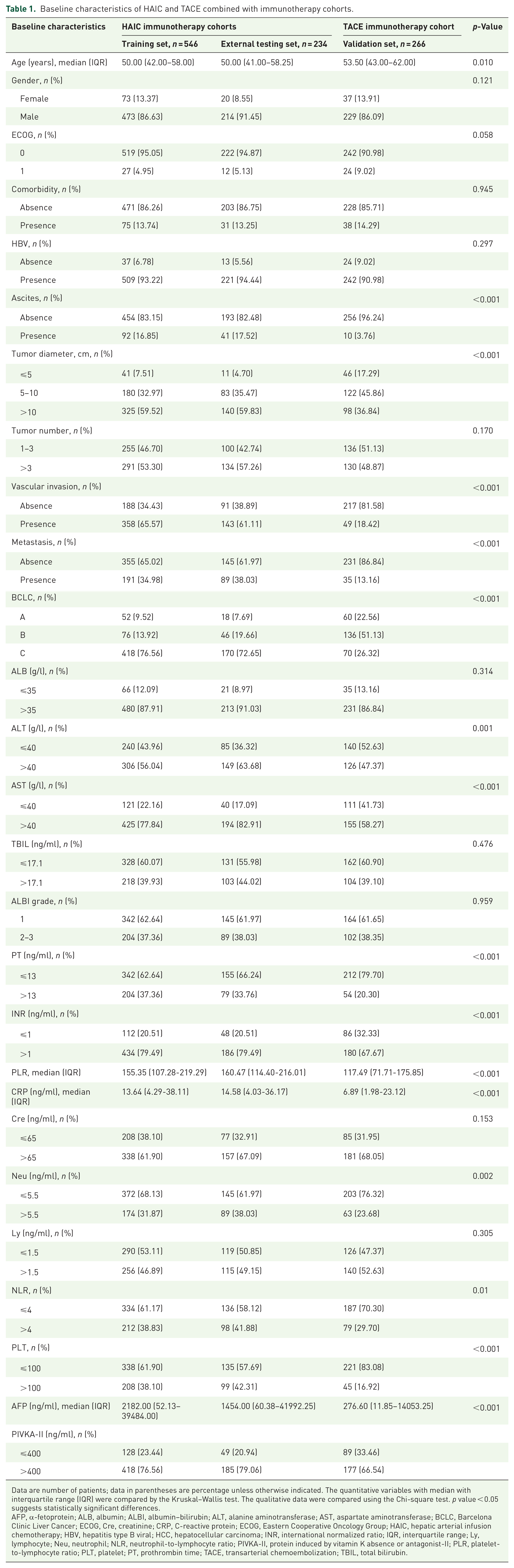
Data are number of patients; data in parentheses are percentage unless otherwise indicated. The quantitative variables with median with interquartile range (IQR) were compared by the Kruskal–Wallis test. The qualitative data were compared using the Chi-square test. p value < 0.05 suggests statistically significant differences.
AFP, α-fetoprotein; ALB, albumin; ALBI, albumin–bilirubin; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BCLC, Barcelona Clinic Liver Cancer; ECOG, Cre, creatinine; CRP, C-reactive protein; ECOG, Eastern Cooperative Oncology Group; HAIC, hepatic arterial infusion chemotherapy; HBV, hepatitis type B viral; HCC, hepatocellular carcinoma; INR, international normalized ratio; IQR, interquartile range; Ly, lymphocyte; Neu, neutrophil; NLR, neutrophil-to-lymphocyte ratio; PIVKA-II, protein induced by vitamin K absence or antagonist-II; PLR, platelet-to-lymphocyte ratio; PLT, platelet; PT, prothrombin time; TACE, transarterial chemoembolization; TBIL, total bilirubin.
Efficacy
In the training dataset, the median PFS was 11.17 months (IQR, 5.96–21.00) months. The risk factors for PFS were assessed by univariate analyses (Supplemental Table 3). Age, tumor size, tumor number, AFP, PLR, and CRP were all substantially correlated with PFS in the univariate analysis (Supplemental Table 3).
Every patient who underwent at least two follow-up imaging evaluation sessions had their radiological tumor response evaluated. In the HAIC plus immunotherapy cohort (n = 780), 37 patients (4.74%) achieved CR, and 337 (43.21%) patients achieved PR, resulting in an ORR of 47.9%. A total of 269 (34.49%) patients had SD, resulting in a DCR of 82.4%. In the TACE plus immunotherapy cohort (n = 266), 16 patients (6.02%) achieved CR, and 105 (39.47%) achieved PR, resulting in an ORR of 45.49%. A total of 115 (43.23%) patients had SD, resulting in a DCR of 88.72%. There was no significant difference between these two cohorts (p = 0.308) (Supplemental Table 4). Besides, we compared the survival of HAIC plus immunotherapy and TACE plus immunotherapy groups for HCC patients in all cohorts. There was no significant difference between HCC patients receiving HAIC and TACE plus immunotherapy (Supplemental Figure 1).
The AFCRPLITY score predicts PFS in patients with HCC receiving HAIC plus immunotherapy
The next goal was to create an objective, laboratory-based score to forecast the PFS of HCC patients using immunotherapy in addition to HAIC. Prognostic variables for PFS included AFP, PLR, and CRP; their regression coefficients in the univariate Cox analysis were identical (Supplemental Table 5). RCS analyses supported the cutoff values for AFP, CRP, and PLR (Figure 2). The results of RCS models that were adjusted for multiple covariates and all significant reproductive factors in Table 2 revealed a significant linear association between AFP–PLR–CRP and PFS in patients with HCC receiving HAIC plus immunotherapy (AFP: p-overall < 0.001, p-nonlinear = 0.733; PLR: p-overall = 0.005, p-nonlinear = 0.538; CRP: p-overall < 0.001, p-nonlinear = 0.606). The 50th quintile of the level of AFP was 2234 ng/ml, the level of PLR was 157, and the level of CRP was 13.66 mg/dl (approximately equal to 14 mg/dl). When using CRP (14 mg/dl), PLR (157), and AFP (2234 ng/ml) as the reference, the hazard ratios (HRs) of PFS related to AFP, PLR, and CRP levels increased sharply when AFP levels >2234 ng/ml or CRP levels >14 mg/dl or the PLR >157 (Figure 2).

Cubic spline graph of the adjusted HR (represented by a solid line) and 95% CI (represented by the blue area) for the association between AFP–CRP–PLR and PFS. (a) Log hazard ratio function and 95% pointwise confidence band estimated by an RCS function for quantifying the effect of log10 (AFP) on PFS. Smaller log hazard ratios indicate better survival. A reference value of 2234 ng/ml was used (i.e., a value of 3.34 on the decadic log scale). (b) Log hazard ratio function and 95% pointwise confidence band estimated by an RCS function for quantifying the effect of log10 (CRP) on PFS. Smaller log hazard ratios indicate better survival. A reference value of 14 g/l was used (i.e., a value of 2.19 on the decadic log scale). (c) Log hazard ratio function and 95% pointwise confidence band estimated by an RCS function for quantifying the effect of log10 (PLR) on PFS. Smaller log hazard ratios indicate better survival. A reference value of 157 was used (i.e., a value of 1.14 on the decadic log scale).
Prognostic factor analysis for PFS in train set.
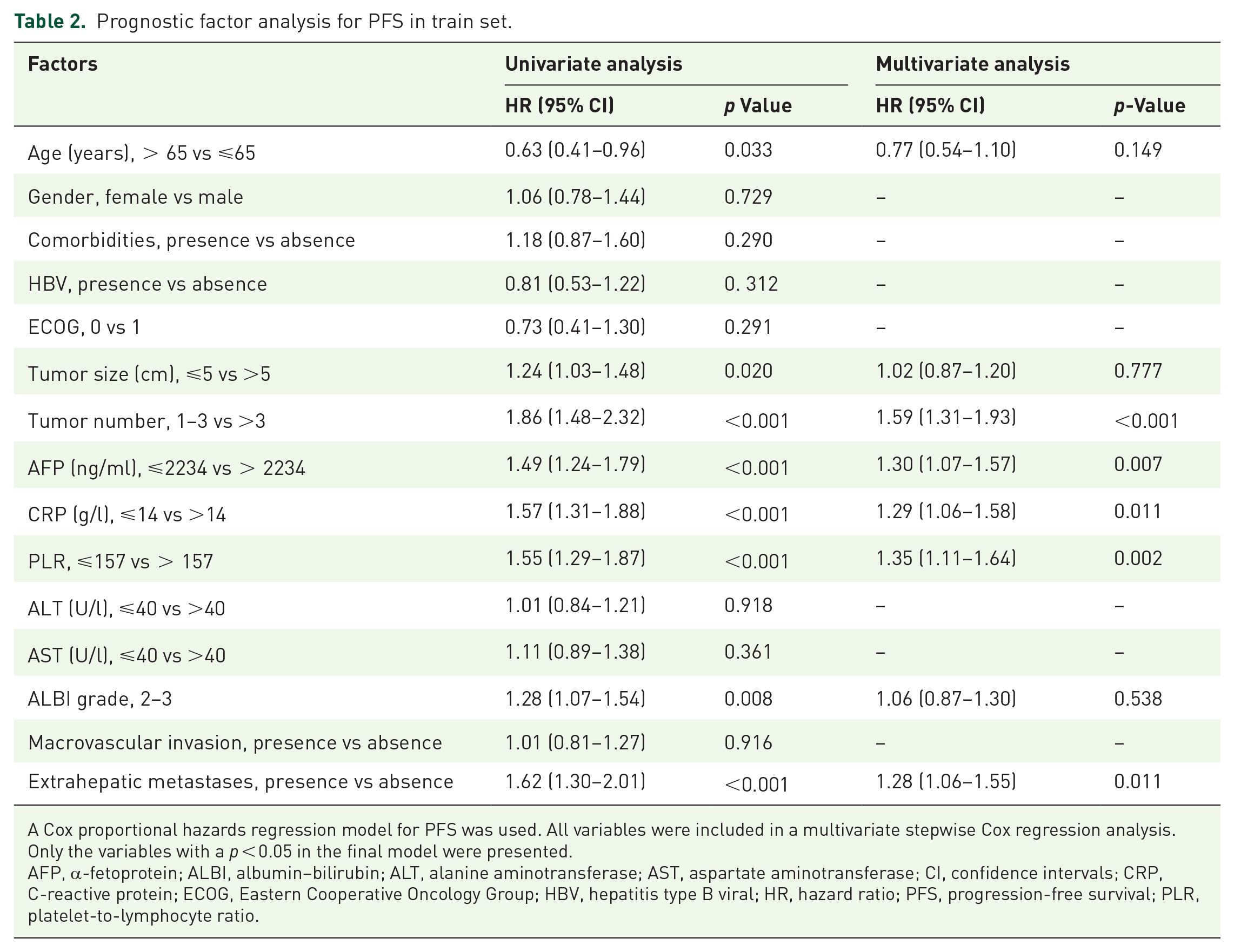
A Cox proportional hazards regression model for PFS was used. All variables were included in a multivariate stepwise Cox regression analysis. Only the variables with a p < 0.05 in the final model were presented.
AFP, α-fetoprotein; ALBI, albumin–bilirubin; ALT, alanine aminotransferase; AST, aspartate aminotransferase; CI, confidence intervals; CRP, C-reactive protein; ECOG, Eastern Cooperative Oncology Group; HBV, hepatitis type B viral; HR, hazard ratio; PFS, progression-free survival; PLR, platelet-to-lymphocyte ratio.
As a result, we created a straightforward score based on those three factors, awarding one point for each of the following: AFP levels >2234 ng/ml, CRP levels >14 mg/dl, and PLR >157. Therefore, a patient could receive zero points (AFP ⩽2234 ng/ml, CRP ⩽14 mg/dl, and PLR ⩽157), one point (AFP >2234 ng/ml or CRP >14 mg/dl or PLR >157), two points (AFP >2234 ng/ml and CRP >14 mg/dl, or both PLR >157 and CRP >14 mg/dl, or both AFP >2234 ng/ml and PLR >157), or three points (AFP >2234 ng/ml and CRP >14 mg/dl and PLR >157) (Figure 2).
The HCC patients were stratified into groups based on their scores: AFCRPLITY score = 0, AFCRPLITY score = 1, AFCRPLITY score = 2, and AFCRPLITY score = 3. The cumulative 1-, 3-, and 5-year PFS rates were 78.2%, 20.1%, and 10.0% in the AFCRPLITY score = 0 group, 62.5%, 38.0%, and 16.6% in the AFCRPLITY score = 1 group, 49.4%, 26.3%, and 0.0% in the AFCRPLITY score = 2 group, and 37.2%, 13.2%, and 9.9% in the AFCRPLITY score = 3 group, respectively. There was a significant difference in the training dataset (Figure 3(a)–(c), AFCRPLITY score = 3 vs AFCRPLITY score = 0 HR, 2.68; 95% CI, 1.84–3.90; p < 0.001, Supplemental Tables 6–8).

Comparing the survival of AFCRPLITY scores for uHCC patients in all cohorts. Kaplan–Meier curves for the (a) OS, (b) PFS, and (c) EPFS of uHCC patients in training cohorts receiving HAIC plus immunotherapy. Kaplan–Meier curves for the (d) OS, (e) PFS, and (f) EPFS of uHCC patients in external testing set receiving HAIC plus immunotherapy. Kaplan–Meier curves for the (g) OS, (h) PFS, and (i) EPFS of uHCC patients in the validation set receiving TACE plus immunotherapy.
Our findings were verified using a testing dataset from other sources, including 234 individuals receiving immunotherapy and HAIC. Table 1 describes the baseline characteristics. The HCC patients were stratified into AFCRPLITY score, AFCRPLITY score, AFCRPLITY score = 2, and AFCRPLITY score = 3 groups. The cumulative 1-, 3-, and 5-year PFS rates were 83.6%, 44.7%, and 33.5% in the AFCRPLITY score = 0 group, 55.8%, 29.7%, and 29.7% in the AFCRPLITY score = 1 group, 42.5%, 13.2%, and 9.9% in the AFCRPLITY score = 2 group, and 40.2%, 24.4%, and 24.4% in the AFCRPLITY score = 3 group, respectively. There was a significant difference in the test dataset (Figure 3(d) and (e), AFCRPLITY score = 3 vs AFCRPLITY score = 0 HR, 2.87; 95% CI, 1.59–5.21; p = 0.001, Supplemental Table 9). The performance of the AFCRPLITY score in the HAIC plus immunotherapy cohorts is shown in Supplemental Figures 2 and 3.
The AFCRPLITY score predicts PFS in patients with HCC undergoing TACE and immunotherapy
We next tested our results in an independent cohort of 266 patients who underwent TACE plus immunotherapy. The baseline characteristics are described in Table 1. The HCC patients were stratified into groups based on their AFCRPLITY score: AFCRPLITY score, AFCRPLITY score = 1, AFCRPLITY score = 2, and AFCRPLITY score = 3. The cumulative 1-, 3-, and 5-year PFS rates were 74.7%, 48.8%, and 31.3% in the AFCRPLITY score = 0 group, 45.0%, 25.7%, and 14.3% in the AFCRPLITY score = 1 group, 40.5%, 13.5%, and 4.5% in the AFCRPLITY score = 2 group, and 31.2%, 4.8%, and 4.8% in the AFCRPLITY score = 3 group, respectively. There was a significant difference in the validation set (Figure 3(g)–(i), AFCRPLITY score = 3 vs AFCRPLITY score = 0 HR, 3.35; 95% CI, 2.03–5.52; p < 0.001).
The AFCRPLITY score predicts DCR and ORR in patients with HCC patients
The DCR rates were 94.57% in the AFCRPLITY score = 0 group, 86.84% in the AFCRPLITY score = 1 group, 82.00% in the AFCRPLITY score = 2 group, and 69.61% in the AFCRPLITY score = 3 group, respectively. And there was a significant difference among different groups in the training dataset (p < 0.001, Table 3). The ORRs were 55.44% in the AFCRPLITY score = 0 group, 64.26% in the AFCRPLITY score = 1 group, 50.00% in the AFCRPLITY score = 2 group, and 40.20% in the AFCRPLITY score = 3 group, respectively (p < 0.001, Table 3). Similar results were revealed in the external testing set and validation set (p < 0.001).
Efficacy according to AFCRPLITY risk score in the training set.

Data are number of patients; data in parentheses are percentage unless otherwise indicated. A Cox proportional hazards regression model for disease-free survival was used. The qualitative data were compared using the Chi-square test.
p-Value < 0.05 suggests statistically significant differences.
AFCRPLITY, AFP, CRP, and PLR in ImmunoTherapY; CI, confidence intervals; CR, complete response; DCR, disease control rate; HR, hazard ratio; ORR, objective response rate; PD, progression disease; PR, partial response; SD, stable disease.
The AFCRPLITY score predicts outcomes in different subgroups
We combined the training and testing sets to increase the number of patients for exploratory subgroup analysis (n = 780). In the pooled cohort, the median PFS was 20.0 (median, 13.1–28.8) months for the AFCRPLITY score = 0 group (n = 129), 17.4 (95% CI 9.1–21.4) months for the AFCRPLITY score = 1 group (n = 280), 16.9 (95% CI 10.2–25.9) months for the AFCRPLITY score = 2 group (n = 222), and 12.8 (95% CI 8.4–21.1) months for the AFCRPLITY score = 3 group (n = 149). Subgroup analyses of OS, PFS, and EPFS were conducted based on important variables, and the forest plots are shown in Figure 4. Most of the subgroup analysis suggested that HCC patients with higher AFCRPLITY scores could have poorer prognosis signatures Figure 4(a)–(c).

Subgroup analyses of AFCRPLITY scores for unresectable HCC patients in HAIC plus immunotherapy cohorts. Forest plot showing the factors associated with OS, PFS, and EPFS in the HCC patients with different AFCRPLITY scores. (a) OS in all patients. (b) PFS in all patients. (c) EPFS in HCC patients.
Efficacy of the AFCRPLITY score to identify the tumor microenvironment of HCC patients
It is still unclear exactly how ICB immunotherapy, which targets the PD-1/PD-L1 axis, works. According to earlier research, CD8+ T cells function as core effector cells and modulate the effectiveness of ICB. 17 Natural killer (NK) cells are innate lymphocytes that produce cytokines and chemokines to cause cytotoxic action against cancer cells. 18 Because NK cells can identify the distinctive molecular patterns of stressed cells, they take part in immune responses against hematopoietic malignancies and may increase the survival time of patients undergoing ICB treatment. 19 Evaluating the level of tumor infiltrates CD8+ T cells and NK cell clouds helps to predict the immunotherapy response in HCC patients. We collected 60 samples from the training set (20 samples from the AFCRPLITY score = 0 group, 20 samples from the AFCRPLITY score = 1/2 groups, and 20 samples from the AFCRPLITY score = 3 group) (Supplemental Table 9). We evaluated the levels of tumor cells (CK8), CD8+ T cells (CD8), and NK cells (CD57) with multiplex immunofluorescence staining. The results showed that the highest level of CD8+ T cells was in the AFCRPLITY score = 0 group; furthermore, the level of CD8+ T cells was higher in the AFCRPLITY score = 1/2 groups than in the AFCRPLITY score = 3 groups (p < 0.05, Figure 5). There were no significant differences in tumor cell and NK cell levels among the three groups (p > 0.05, Figure 5). An increase in the levels of tumor-infiltrating CD8+ T cells could explain the better prognosis observed among HCC patients undergoing TACE plus immunotherapy in the AFCRPLITY score = 0 group than in the other groups.

Efficacy of the AFCRPLITY score to identify the tumor microenvironment of HCC patients. Representative images of multiplex immunofluorescence in 20 samples from the AFCRPLITY score = 0 group, 20 samples from the AFCRPLITY score = 1/2 groups, and 20 samples from the AFCRPLITY score = 3 group from HAIC plus immunotherapy cohort. Quantification of tumor cells (CK8), CD8+ T cells (CD8), and NK cells (CD57) as a proportion of total cells. Scale bars, 50 μm.
Discussion
IATs combined with immunotherapy have greater advantages for treating HCC with a high tumor burden; these advantages mainly include repeated and continuous chemotherapy effects and a larger ORR.20–22 More antitumor immunity is produced by a combination of antiangiogenic and anti-PD-1/PD-L1 treatment than by HAIC or TACE alone since it has been demonstrated to activate immunological checkpoints and stimulate T-cell activity in tumor cells. However, post-recurrence and excessive progress after ICB are ongoing challenges for physicians. It is necessary to develop an accurate and noninvasive method for the preoperative estimation of HCC patient status, thus helping to identify optimal candidates who can achieve disease control after receiving IATs combined with immunotherapy.
Notably, our study first addressed the associations of AFP, CRP, and PLR with PFS among patients with HCC who received HAIC plus ICBs. In this study, we developed and validated a simple, easy-to-use score based on the serum levels of AFP, serum levels of CRP, and PLR; this score predicts the probability of successful immunotherapy and improved disease control. We used the RCS model to identify the linear associations of AFP, PLR, and CRP with PFS in patients with HCC receiving HAIC plus immunotherapy and evaluated whether the HR is positively correlated with the levels of AFP, levels of CRP, and the PLR when AFP levels >2234 ng/ml or CRP levels >14 mg/dl or the PLR >157. Accordingly, when we defined the cutoff values of AFP, CRP, and PLR as 2234 ng/ml, 14 mg/dl, and 157, respectively, patients with lower scores had better DCR and PFS than patients with higher scores. Although our proposed cutoff values are inconsistent with previously reported results, these inconsistencies may be due to the enrolled patients having a greater tumor burden and biological activity in this study. Furthermore, these findings were validated in a separate validation cohort of HCC patients undergoing TACE plus ICBs, and accurate recurrence risk categorization and DCR and PFS prediction are still achievable.
The combined use of AFP, CRP, and PLR to forecast the prognosis for patients with HCC receiving different treatment modalities makes sense. Serum AFP is a commonly utilized biomarker for the management of HCC, and the reaction to AFP is one of the key indicators for predicting radiological response and efficacy.23,24 CRP and PLR, two inflammation-based parameters, have been suggested to play a role in the development and progression of tumors.25,26 A meta-analysis by Zheng et al. 27 demonstrated that a high PLR can increase the risk of HCC recurrence, even in a multivariate model. In addition to PFS, our score was linked to attaining radiological disease control (stabilization or response) during the ICB procedure. Because both objective response and disease stability are linked to better OS, achieving any of these outcomes might be seen as a sign of success. In total, 88.4% of patients in our study who received HAIC plus immunotherapy showed DCR at radiological assessment. This rate is higher than the IMbrave150 trial, which used the combination of atezolizumab and bevacizumab (80%), but comparable to other phase II studies that used the TRIPLET protocol (87.1%).7,11 This discrepancy may arise from the fact that most of our patients received HAIC in addition to TKIs and ICBs.
AFP, CRP, and PLR were shown to be closely associated with the immunosuppressive tumor microenvironment (TME). Previous studies proved that patients with a high level of AFP were more likely to have a larger tumor diameter and more vascular invasion than patients with a low level of AFP. 28 In addition, HCC samples with high levels of AFP exhibited altered immunity-related pathways, including a suppressive effect on NK cells and T cells and an impaired effect of dendritic cells (DCs) in stimulating antigen-specific T-cell activation and proliferation. 29 AFP has been associated with intricate immunological dysfunction, marked by a reduction in different subgroups of T cells and an increase in tumor-associated macrophages. 30 In addition, AFP promotes the upregulation of Vascular Endothelial Growth Factor (VEGF) signaling, which hinders the function and maturation of effector T cells and leads to an increase in immune checkpoint molecules. 31 CRP is a quintessential acute-phase protein in humans that has a role in the regulation of immune cells. It is well known that CRP participates in innate immunity by binding to complement components to facilitate opsonization and phagocytosis 32 and slows immunological synapses from forming and blocks the early stages of T-cell receptor interaction, which ultimately prevents T-cell proliferation in individuals with melanoma. 33 There is mounting evidence that CRP can restrict the growth of MART-1 antigen-specific CD8+ T cells, depress the proliferation and effector activities of activated CD4+ and CD8+ T cells, and decrease the expression of costimulatory signals on mature DCs. A recent research established the suppressive impact of CRP in CD8+ T cells in vivo by demonstrating that it lowered cytotoxic activity, decreased T-cell proliferation in a dose-dependent manner with modulation of the p38MAPK-ROS pathway, and raised the expression of senescent markers in CD8+ T cells. 34 Recent research has shown that peripheral immune cell activation is necessary for tumor elimination with immunotherapy, 35 and the systemic hematological PLR parameter was associated with tumor immune infiltrate features in breast cancer.31,36 There was a relationship between decreased CD3+ T-cell infiltration and increased tumor neutrophil infiltration as well as a relationship between increased peripheral blood neutrophil levels and increased PLRs. 37 PLR is a poor predictor of PFS that activates transcription factors such as tumor necrosis factor-alpha, activator of transcription 3, hypoxia-inducible factor 1-alpha, and nuclear transcription factor-kappa B. As a result, PLR is crucial for angiogenesis, metastasis, and treatment response.
The AFCRPLITY score could identify a specific HCC patient who underwent TACE or HAIC plus immunotherapy with a homogeneous recurrence risk, indicating the robustness and accuracy of the score for these received IAT immunotherapy and contributing to the design of targeted trials and interventions. We further evaluated this score in numerous relevant subgroups and included AFP, CRP, PLR, and other pertinent factors as candidate covariates in multivariate analysis to account for any selection bias. HCC patients were successfully stratified into several risk categories by the AFCRPLITY score, which also assisted in identifying the best candidates for immunotherapy. Furthermore, risk scores may be useful in determining an HCC patient’s TME. Because it made it possible to identify a small but possibly controllable subset of patients who were at high risk of recurrence and received HAIC plus immunotherapy or TACE plus immunotherapy, the AFCRPLITY score had clinical significance. We either altered the treatment plan beforehand for these patients, who were classified as high-risk according to the AFCRPLITY score, or we put a postoperative preventive and monitoring strategy into place.
Even though our study has several advantages, such as a sizable cohort and a multicenter study design, there are still certain restrictions. First off, there may have been selection bias because this study included patients from a variety of institutions throughout the nation. These cohorts varied widely in terms of liver function, course of treatment, and kind of immunotherapy. Second, as HBV infection is the most common cause of HCC in China, the majority of patients with large HCC were included in this study. It is unclear if the findings would be broadly applicable in Western nations, where the majority of patients had alcoholic liver cirrhosis as the primary cause of a low tumor load. Third, it is essential to recognize that the study’s retrospective design precluded a planned radiological evaluation. To test the AFCRPLITY score as a radiological response predictor, a prospective cohort including predetermined and uniform imaging evaluations is required. Fourth, more experiments are needed to verify the relationship between the prediction model and the pathological mechanisms.
Conclusion
In conclusion, we developed and validated a simple score named AFCRPLITY based on AFP, CRP, and PLR, which are factors that are known to promote immunosuppression. The AFCRPLITY score developed herein predicts the outcomes of IATs plus immunotherapy for Ad-HCC, independent of age and tumor size. The AFCRPLITY score merits prospective validation in a large clinical study, ideally with an active control group that is not treated with IATs plus immunotherapy. In addition, it might improve clinical practice decision-making and aid in the selection of patients for participation in clinical trials.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241297080 – Supplemental material for The AFCRPLITY score for predicting the prognosis of immunotherapy combined with local–regional therapy in unresectable hepatocellular carcinoma
Supplemental material, sj-docx-1-tam-10.1177_17588359241297080 for The AFCRPLITY score for predicting the prognosis of immunotherapy combined with local–regional therapy in unresectable hepatocellular carcinoma by Mengxuan Zuo, Ran Wei, Da Li, Wang Li and Chao An in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359241297080 – Supplemental material for The AFCRPLITY score for predicting the prognosis of immunotherapy combined with local–regional therapy in unresectable hepatocellular carcinoma
Supplemental material, sj-docx-2-tam-10.1177_17588359241297080 for The AFCRPLITY score for predicting the prognosis of immunotherapy combined with local–regional therapy in unresectable hepatocellular carcinoma by Mengxuan Zuo, Ran Wei, Da Li, Wang Li and Chao An in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
