Abstract
Objectives:
To identify useful biomarkers by reviewing laboratory data for a predictor of the clinical course following treatment with radium-223 dichloride (Ra-223) in patients with metastatic castration-resistant prostate cancer.
Methods:
Eighteen metastatic castration-resistant prostate cancer patients who were administered Ra-223 at our hospital were retrospectively enrolled in this study. Prostate-specific antigen doubling times before and after the administration of Ra-223 were evaluated as prognostic factors for metastatic castration-resistant prostate cancer patients treated with Ra-223 using the Kaplan–Meier method and Log-rank test.
Results:
Four patients failed to complete the planned six-time Ra-223 treatments with the exacerbation of their condition. In the 14 patients who completed the planned Ra-223 treatment, before the Ra-223 treatment, no significant differences were observed in overall survival between patients with prostate-specific antigen doubling time of 6 months or less and those with prostate-specific antigen doubling time of more than 6 months or stable (
Conclusion:
Prostate-specific antigen doubling time after the Ra-223 treatment is a useful predictor of the clinical course following treatment in metastatic castration-resistant prostate cancer patients.
Keywords
Introduction
Prostate cancer is the most diagnosed tumor type in the male population and is one of the main causes of cancer-related death worldwide, including Japan. 1 The natural history of prostate cancer has different phases, such as hormone-sensitive prostate cancer and metastatic castration-resistant prostate cancer (mCRPC). Skeletal involvement has been reported in up to 90% of cases of metastatic prostate cancer. 2
The bone-targeting agent radium-223 dichloride (Ra-223), an alpha-emitting agent, plays a significant role in the treatment of the skeletal metastases of prostate cancer. Ra-223 was tested in the Alpharadin in Symptomatic Prostate Cancer Patients (ALSYMPCA) trial. 3 Overall survival (OS), the primary endpoint, and the time to the first symptomatic skeletal-related event, one of the secondary endpoints, were longer in the Ra-223 group than in the placebo group. Furthermore, Ra-223 was associated with few adverse events (AEs) and a low rate of myelotoxicity.
Despite the benefits of the Ra-223 treatment, there is currently no marker to monitor the clinical course of patients treated with Ra-223, such as the prostate-specific antigen (PSA) blood test. 4
Therefore, we herein investigated potentially useful biomarkers by reviewing laboratory data for the therapeutic effects of Ra-223. We examined the relationship between the prostate-specific antigen doubling time (PSADT) and the clinical course following treatment with Ra-223 in mCRPC patients. The results obtained demonstrated that PSADT after the Ra-223 treatment was a useful predictor of the clinical course in mCRPC patients following treatment.
Methods
We investigated potentially useful biomarkers by reviewing laboratory data for the therapeutic effects of Ra-223 in an observational study.
Patients
All patients had mCRPC (Table 1). CRPC (castration-resistant prostate cancer) was defined as disease progression at a serum testosterone level <50 ng per deciliter during maintenance treatment, which consisted of androgen deprivation therapy (ADT) with a luteinizing hormone-releasing hormone agonist or antagonist. Inclusion criteria were CRPC patients with bone metastases and any lymph node metastases less than 2 cm without visceral metastases that were treated with Ra-223 at our hospital between November 2018 and December 2020. Exclusion criteria were previous blood transfusions or the use of erythropoietin-stimulating agents within the previous 4 weeks. All patients were observed until April 2022 or death. The present study was approved by the Ethical Committee of the Uonuma Institute of Community Medicine, Niigata University Medical and Dental Hospital (approval no. 03-008). Written informed consent from the subjects and from the legally authorized representatives of the deceased subjects for the publication of this study was waived by the Ethical Committee of the Uonuma Institute of Community Medicine, Niigata University Medical and Dental Hospital. This study was conducted in accordance with the Declaration of Helsinki. All patients were pathologically diagnosed with prostate cancer before the initial treatment. In all patients, two or more bone metastases were detected on skeletal scintigraphy or CT (computed tomography), and there was no known visceral metastasis other than lymph nodes metastases of less than 2 cm before the administration of Ra-223.
Patient characteristics.

EOD classification: 0, normal or abnormal due to benign bone disease; 1, fewer than six bony metastases, each of which is less than 50% the size of a vertebral body (one lesion approximately the size of a vertebral body was counted as two lesions); 2, between 6 and 20 bone metastases, sized as described above; 3, more than 20 metastases, but fewer than those observed in a superscan; and 4, a superscan or its equivalent (more than 75% of the ribs, vertebrae, and pelvic bones).
Systemic therapies other than ADT before the Ra-223 treatment included bicalutamide, flutamide, estramustine phosphate sodium, enzalutamide, abiraterone with prednisolone, docetaxel, cabazitaxel, and denosumab or zoledronic acid. The bone-protecting agents, denosumab and zoledronic acid, were continued before, during, and after the Ra223 treatment. Systemic therapies other than ADT and bone-protecting agents before the Ra-223 treatment were discontinued before its initiation.
Systemic therapy
All patients received ADT in addition to systemic therapies before, concomitant with, and after the Ra-223 treatment throughout the study (Table 1). Systemic therapies other than ADT before the Ra-223 treatment included bicalutamide, flutamide, estramustine phosphate sodium, enzalutamide, abiraterone with prednisolone, docetaxel, cabazitaxel, and denosumab or zoledronic acid. The administration of the bone-protecting agents, denosumab and zoledronic acid, was continued before, during, and after the Ra223 treatment. Systemic therapies other than ADT and bone-protecting agents before the Ra-223 treatment were discontinued before its initiation.
Administration of Ra-223
All patients were scheduled to receive up to six injections of Ra-223 (Bayer Yakuhin Ltd., Osaka, Japan) (55 kBq/kg), which were administered at a rate of one every 4 weeks (Table 1). AEs occurring in the follow-up for up to 4 weeks after the final Ra-223 injection were only reported if they were considered to be related to Ra-223. AEs were graded according to Common Terminology Criteria for AEs, version 4.0. In all patients, blood collection was performed before each administration of Ra-223 and after the final administration. In patients who had progressed to mCRPC and started the Ra-223 treatment, current treatments, namely, ADT and bone-protecting agents, were continued concomitant with Ra-223.
Evaluation
The following background characteristics were examined between patients who received six injections of Ra-223, did not die, and did not have visceral metastasis during the observation period and those who died during the observation period: age, the performance status, treatment history, PSA, PSADT, and alkaline phosphatase (ALP) and lactate dehydrogenase (LDH) levels before the Ra-223 treatment and the extent of bone disease. Changes in the blood levels of biomarkers were compared between the two groups. Laboratory data and other clinical information were collected from medical records.
PSADT
PSADT predicts changes in PSA levels over time and may be used as an indicator of biochemical and clinical progression. PSADT was calculated as the natural log of 2 (0.693) divided by the slope of the linear regression of the natural log of PSA levels versus the time of the PSA measurement in months. 5 If the slope of the PSA level was 0 (constant PSA level) or negative (decreasing PSA level after an initial increase), PSADT was arbitrarily set to 100. 6
Statistical analysis
The serum data of patients who received six injections of Ra-223 were investigated before and after the Ra-223 treatment using the paired Wilcoxon signed-rank test. The serum data of patients who were alive and did not have visceral metastasis during the observation period and those who died during the observation period were examined using the Mann–Whitney
PSADT in patients who received the planned six Ra-223 injections were divided into two groups (PSADT of 6 months or less and PSADT of more than 6 months or stable) and compared. PSADT before and after the Ra-223 treatment were evaluated as prognostic factors for mCRPC patients treated with Ra-223. The Kaplan–Meier method with the Log-rank test was used to compare time-to-event analyses from the beginning of the Ra223 treatment to death using PSADT (PSADT of 6 months or less and PSADT of more than 6 months or stable) before and after the Ra223 treatment. A
IBM SPSS Statistics Ver. 27 was used as statistics software.
Results
Patient characteristics and outcomes of the Ra-223 treatment (Table 1).
Between the start of the Ra-223 treatment at our hospital (November 2018) and December 2020, 18 CRPC patients with bone metastases were treated with Ra-223. ADT was continued before, during, and after the Ra-223 treatment. Patients received multiple systemic therapies other than ADT prior to the Ra-223 treatment. The number of additional treatments was four (
At the time of the analysis, 10 (55.6%) patients were alive, while eight (44.4%) had died of cancer progression.
Number of patients treated with Ra-223
Four out of 18 patients failed to complete the Ra-223 treatment. Two patients discontinued after the administration of Ra-223 once, while another two discontinued after its administration five times with the exacerbation of their conditions.
Fourteen patients received the planned six Ra-223 injections. Ten patients were alive and did not have visceral metastasis within the observation period and four died during the observation period. Four out of 14 patients received cabazitaxel after completing the Ra223 treatment, while the other 10 were not treated with drugs that would improve their prognosis within the observation period.
Markers before the Ra-223 treatment
Ten patients who were administered the six injections of Ra-223 and were alive and did not have visceral metastasis within the observation period were compared with eight who died during the observation period (Table 2). Pretreatment PSA levels (
Number of Ra-223 injections and markers before the Ra-223 treatment.
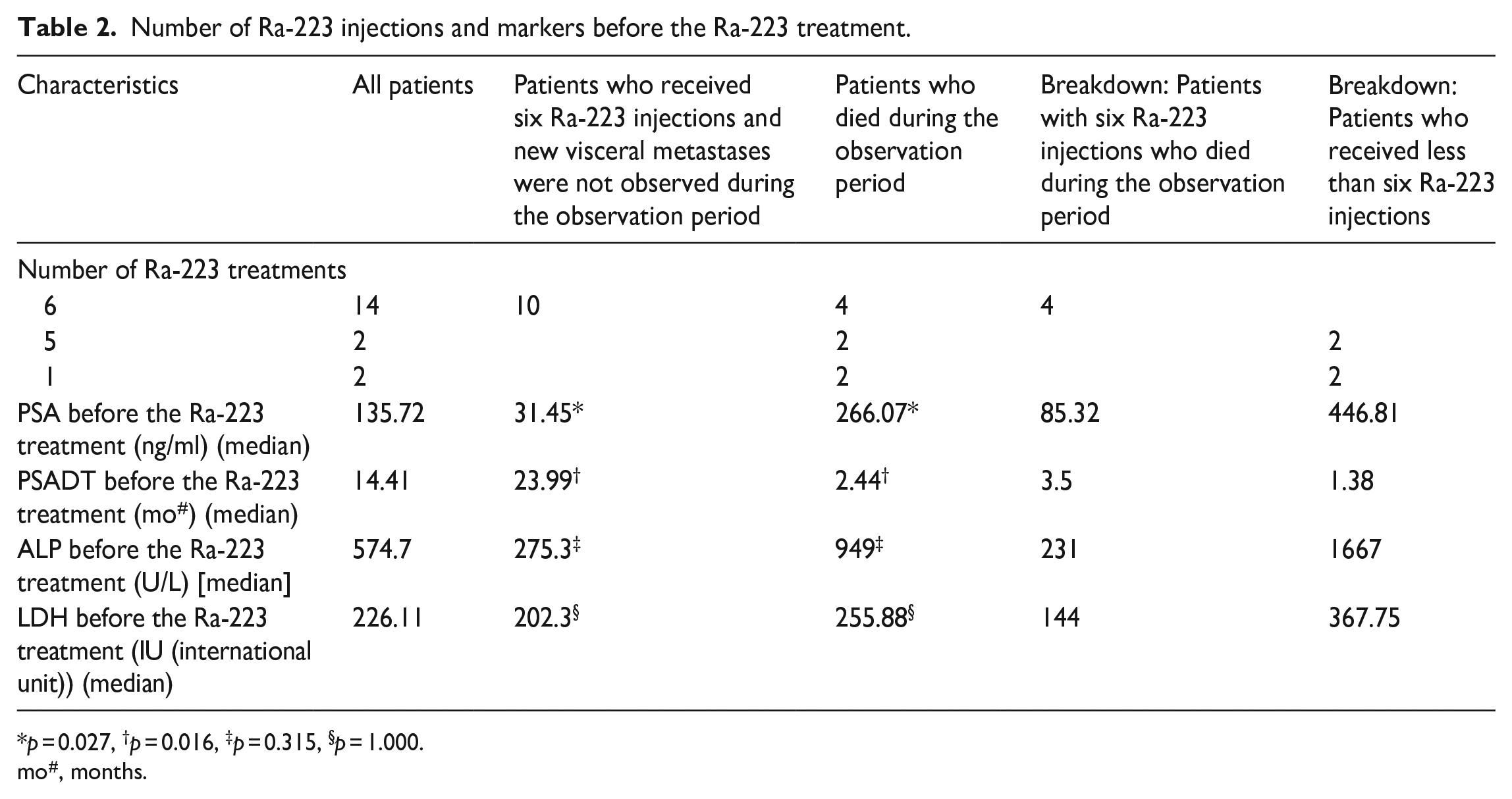
mo#, months.
Patients who completed the Ra-223 treatment
Patients who received the planned six Ra-223 injections had significantly lower Hb levels (
Among the 14 patients who received the planned six injections of Ra-223, 10 patients who did not die and whose cancer did not metastasize to viscera during the observation period were compared with four who died during the observation period (Tables 3 and 4). No significant differences were observed in pretreatment PSA levels (
Markers before and after six Ra-223 injections in patients who completed the Ra-223 treatment.

mo#: months.
PSA and PSADT before and after six Ra-223 injections.
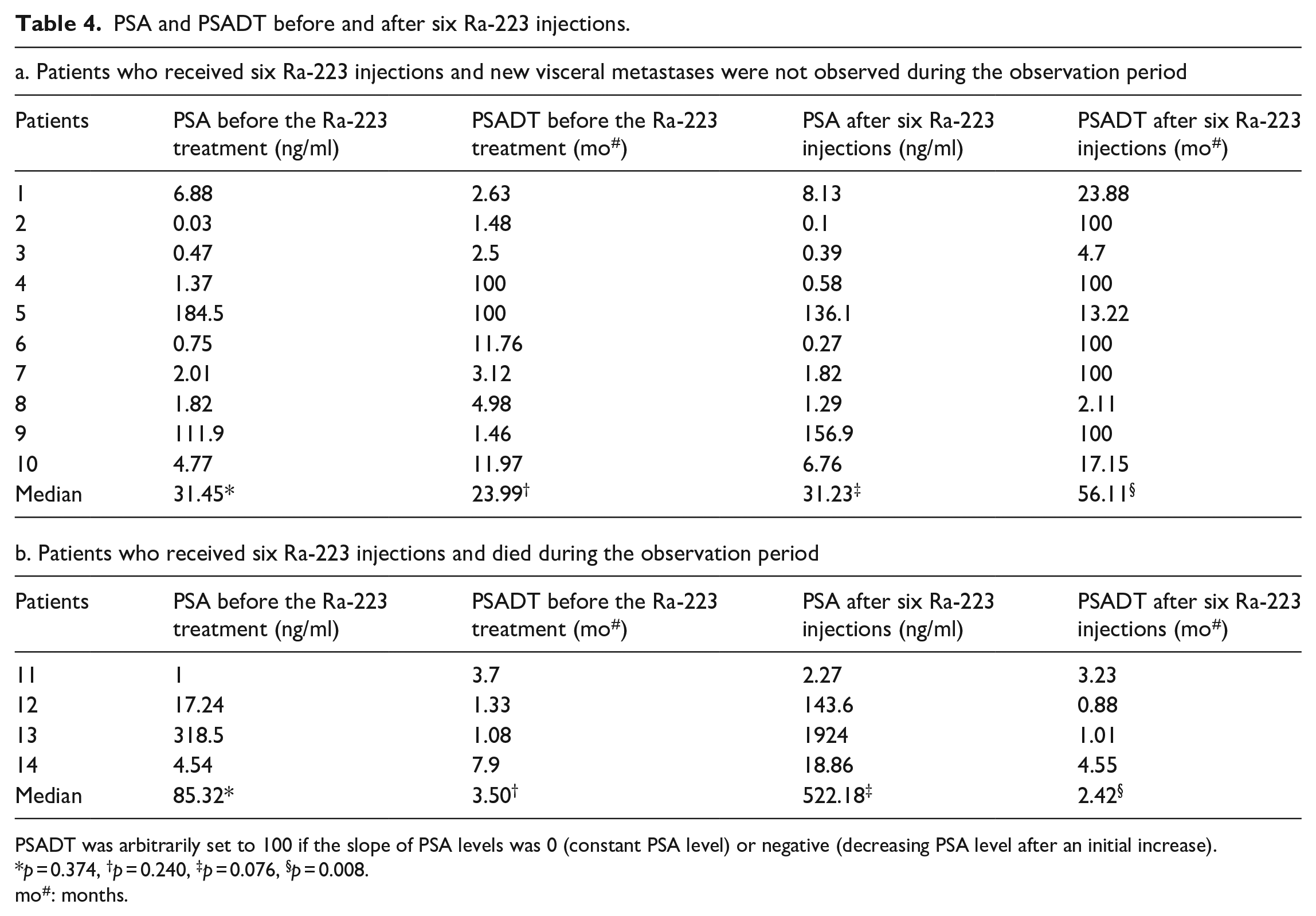
PSADT was arbitrarily set to 100 if the slope of PSA levels was 0 (constant PSA level) or negative (decreasing PSA level after an initial increase).
mo#: months.
PSADT in patients who received the planned six Ra-223 injections were divided into two groups (PSADT of 6 months or less and PSADT of more than 6 months or stable) and compared. Before the Ra-223 treatment, there was no significant difference in OS between patients with PSADT of 6 months or less and those with PSADT of more than 6 months or stable (
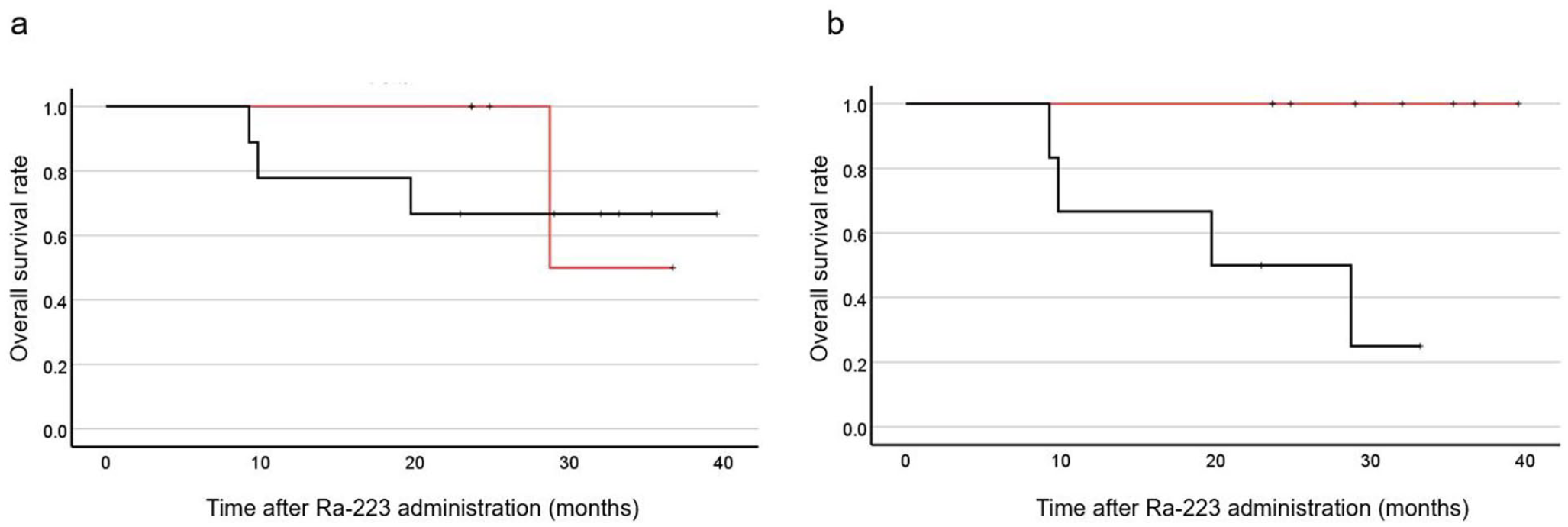
OS curve of 14 mCRPC patients who received six Ra-223 injections by a Kaplan–Meier analysis: (a) Before Ra-223 injections, there were no significant differences in OS between patients with PSADT of 6 months or less and those with PSADT of more than 6 months or stable (
The Ra-223 treatment was not discontinued due to AEs, such as myelosuppression associated with Ra-223.
Discussion
The present study revealed that following the completion of the Ra-223 treatment, OS was significantly shorter in patients with PSADT of 6 months or less than in those with PSADT of more than 6 months or stable (
Ra-223, an alpha-emitting bone-targeted therapeutic agent, is selectively taken up by bone lesions, emits high-energy, short-range (<100 μm) alpha rays in tissues, and exerts strong cell-killing effects on tumor cells. 7
The ALSYMPCA trial examined the benefits of Ra-223 in mCRPC patients (
In an open-label single-arm study, a group without metastatic bone pain achieved greater OS benefits than a group with symptomatic pain. 9 Therefore, patients with bone metastatic CRPC are the ideal candidates for Ra-223 therapy.
The potential of various biomarkers, such as ALP and PSA, to estimate the impact of Ra-223 has been examined; however, difficulties are associated with assessing their therapeutic effects. Changes in PSA and ALP during a treatment with Ra-223 were previously demonstrated to be useful indicators of the likelihood of completing treatment with Ra-223 and the prognosis of patients.4,10–13 The bone scan index has also been used as a metastatic bone disease quantifier and predictor of Ra-223 biochemical responses; however, we did not collect data on the bone scan index in the present study.14–16 PSA levels were previously shown to sometimes become elevated during the first three cycles of Ra-223 due to the flare phenomenon; however, the flare phenomenon was not detected in the present study. 17 The present results showed that Hb, leukocyte, and ALP levels were significantly reduced in patients after the planned six Ra-223 injections; however, it was considered to be difficult to evaluate them individually.
PSADT is a commonly used indicator of distant metastasis among patients treated with ADT and radiation therapy; however, it currently remains unclear whether PSADT after treatment with Ra-223 reflects therapeutic effects and the subsequent clinical course.4,11–16 Based on the present study, PSADT after the Ra-223 treatment was a useful indicator for estimating the clinical course following treatment.
Since the present study examined a small number of cases at our institution and we acknowledge the lack of power calculation as a limitation of this study, future analyses of more cases are needed to demonstrate the usefulness of PSADT for estimating the clinical course following treatment with Ra-223.
Conclusion
PSADT following the Ra-223 treatment is a predictor of the clinical course after treatment in patients with mCRPC. Although the present study had a small number of cases, the results obtained suggest that PSADT after the Ra-223 treatment is a useful indicator for estimating the clinical course following treatment.
Footnotes
Authors’ contributions
TN is the corresponding author, managed the study with the design and implementation of the research, analyzed data, and oversaw medical procedures. GK collected data, managed the study with the design and implementation of research, analyzed data, and oversaw medical procedures. KA performed the statistical processing and analysis of data. TI, YI, and NH contributed to the design, implementation of the research, and data analysis. All authors reviewed and edited the manuscript and approved the final version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
This study was approved by the Ethical Committee of the Uonuma Institute of Community Medicine, Niigata University Medical and Dental Hospital (approval no. 03-008). This study was conducted in accordance with the Declaration of Helsinki.
Informed consent
Written informed consent from the subjects and from the legally authorized representatives of the deceased subjects for the publication of this study was waived by the Ethical Committee of the Uonuma Institute of Community Medicine, Niigata University Medical and Dental Hospital (approval no. 03-008).
Trial registration
Not applicable.
