Abstract
The treatment of metastatic castration-resistant prostate cancer (mCRPC) remains a huge challenge. Newer radionuclides, such as 223Ra, 177Lu, 225Ac, etc., have shown efficacy in alleviating cancer-related pain and also in enhancing the prognosis of mCRPC patients. These novel radionuclides have emerged as important weapons in the treatment of mCRPC. This article reviews recent advances and key clinical trials in various radionuclides and their combinations in the application for treating mCRPC. Further directions of this new treatment strategy are also discussed in this review.
Keywords
Introduction
According to the Global Cancer Statistics 2022, prostate cancer is the second most common cancer among men worldwide. 1 For patients with metastatic hormone-sensitive prostate cancer, the condition often progresses to metastatic castration-resistant prostate cancer (mCRPC) after 18–24 months of hormone therapy. At this stage, treatment options include chemotherapy, novel hormonal therapies, PARP inhibitors, radionuclides, immunotherapy, and antibody–drug conjugates. 2 Recent years have seen significant advancements in radionuclide therapy. Traditional radionuclides like strontium-89 (89Sr) continue to play a role in treating mCRPC, while newer agents, such as 223Ra, 177Lu-PSMA-617, 225Ac-PSMA-617, etc., are developed and show promising results in clinical trials and real-world practice, further highlighting the importance of radionuclide therapy in the management of mCRPC. This article aims to review recent progress in the application of radionuclide therapy for mCRPC.
Basic concept of radionuclides
Radionuclides are metallic or non-metallic elements that can produce alpha, beta, or other forms of ionizing radiation and play a vital role in the treatment of tumors. Radiopharmaceuticals labeled with radionuclides can selectively accumulate in tumor lesions, where they emit radiation to damage tumor cells and achieve therapeutic effects. α particles, actually, are helium nuclei (⁴He²⁺), have high mass and strong ionizing capability, but extremely limited penetration. Due to this short range and concentrated energy deposition, α-emitters are unsuitable for imaging but possess high therapeutic potential by inducing lethal damage to nearby tumor cells while sparing distant normal tissue. 3 Commonly used α particle emitters include 225Ac, 223Ra, etc. β particles are either electrons (β−) or positrons (β+) emitted during radioactive decay. β−-decay commonly occurs in neutron-rich radionuclides and has moderate tissue penetration (typically up to several millimeters). 3 Common β particle emitters include 177Lu, 131I, etc. By contrast, β+ particles are emitted by proton-rich artificial radionuclides. After traveling 1–2 mm in tissue, they annihilate with electrons, producing two 511 keV photons emitted in opposite directions, which is the basis for positron emission tomography imaging. In addition to α and β particles, Auger electrons and internal conversion electrons also contribute to therapeutic efficacy. These low-energy, short-range electrons, emitted after electron capture or isomeric transition, can cause significant DNA damage when radionuclides are localized in the nucleus. 4 Moreover, they can kill targeted cancer cells by disrupting the cell membrane and may also affect non-targeted cells through cross-dose or bystander effects. 4 γ rays have strong tissue penetration but minimal ionization capability, resulting in weaker localized effects compared to β and α radiation. Therefore, they are generally unsuitable for therapeutic use but play a crucial role in nuclear imaging, such as in single-photon emission computed tomography.
The emission characteristics of α, β, and γ radiation and Auger electrons are summarized in Table 1, providing a comparative analysis of their physical properties and clinical applications.
Comparative analysis of α, β, and γ radiation and Auger electrons.

PET, positron emission tomography; SPECT, single-photon emission computed tomography.
Prostate-specific membrane antigen targeted radioligand therapy
Therapeutic mechanism of prostate-specific membrane antigen-radioligand therapy
Prostate-specific membrane antigen (PSMA) is a type II transmembrane carboxypeptidase, predominantly expressed in prostate tissue. Its expression in prostate cancer cells is remarkably higher, ranging from 100 to 1000 times, than in normal prostate tissue. 5 Studies have indicated a positive correlation between PSMA expression in prostate cancer tissue and both serum prostate-specific antigen (PSA) levels and Gleason scores. 6 We will primarily focus on two treatment methods based on 177Lu-PSMA-RLT and 225Ac-PSMA-RLT. Other new developing PSMA-radioligand therapies (PSMA-RLTs) are also briefly introduced.
PSMA-617, a widely used small molecule inhibitor that specifically targets PSMA and has a relatively short half-life in the blood, aiding in the efficient excretion of radionuclide drugs from non-target tissues. The two most researched PSMA-RLTs in mCRPC are 177Lu-PSMA-617 and 225Ac-PSMA-617. 177Lu emits β particles with a tissue penetration depth of approximately 1–3 mm and has a physical half-life of around 6.65 days, making it well-suited for targeted internal radiotherapy of tumors. 7 On the other hand, 225Ac emits α rays, which have higher energy but a shorter range, 7 with a half-life of about 9.9 days, is also a well-suitable internal radiotherapy. These radionuclides are administered intravenously and target the extracellular domain of PSMA on the membrane of prostate cancer cells. They are internalized into the tumor cells through endocytosis, accumulate inside the cells, and destroy the tumor cells through internal radiation emitted by the radioactive particles.
Advances of 177Lu-PSMA-617 in the treatment of mCRPC
Kratochwil et al. first reported an mPCa patient who achieved a complete response (CR) on imaging after two cycles of 177Lu-PSMA-617 treatment. 8 This case sparked a research boom in PSMA-RLT. A phase II clinical trial conducted a non-inferiority study involving 40 mCRPC patients with high PSMA expression who had previously received androgen deprivation therapy (ADT), bone protection agents, or novel hormone therapy treatments, but not docetaxel chemotherapy. Patients were randomly assigned to receive 177Lu-PSMA-617 treatment or docetaxel treatment at a ratio of 1:1. The study revealed a higher proportion of patients in the 177Lu-PSMA-617 group experiencing a ⩾50% decrease in PSA compared to those receiving chemotherapy (60% vs 40%). Furthermore, the occurrence of severe adverse events was lower in the 177Lu-PSMA-617 group compared to the chemotherapy group (30% vs 50%). 9
In patients who have received docetaxel chemotherapy, the phase II TheraP study compared 177Lu-PSMA-617 with cabazitaxel in patients previously treated with docetaxel. The results showed a higher PSA response rate and fewer grade 3 or 4 adverse events in the 177Lu-PSMA-617 group. 10 The 3-year follow-up results have been updated, although there was no significant difference in overall survival (OS) between the 177Lu-PSMA-617 group and cabazitaxel group (mOS 19.1 months vs 19.6 months, p = 0.99), but a substantial improvement in progression-free survival was noted in the 177Lu-PSMA-617 group (mPFS 7.1 months vs 5.0 months, p = 0.0028). 11 This result indicates that after progression on docetaxel, 177Lu-PSMA-617 RLT may be a more suitable treatment choice for mCRPC than cabazitaxel.
In the phase III VISION clinical trial (NCT03511664), 831 mCRPC patients who had undergone chemotherapy and novel hormone therapy were randomly assigned in a 2:1 ratio to receive 177Lu-PSMA-617 combined with standard treatment or standard treatment alone. The combined treatment group showed significantly prolonged overall survival (mOS 15.3 months vs 11.3 months; p < 0.001) and radiographic progression-free survival (mrPFS: 8.7 months vs 3.4 months; p < 0.001). 12
Following the VISION study results, in March 2022, the Food and Drug Administration (FDA) approved Pluvicto (177Lu-PSMA-617) for treating PSMA-positive mCRPC patients who have received docetaxel chemotherapy and novel hormone therapy, 13 marking it as the first RLT approved by the FDA for mCRPC patients. 13
The PSMAfore trial (NCT04689828) evaluated the effectiveness of 177Lu-PSMA-617 in taxane-naïve mCRPC patients. It randomly assigned 469 mCRPC patients who had previously received androgen receptor pathway inhibitors (ARPI) at a ratio of 1:1, with one group receiving 177Lu-PSMA-617 treatment every 6 weeks for a total of six cycles, and the other group using another ARPI. The results presented at the 2023 ESMO meeting showed that compared to the ARPI group, the 177Lu-PSMA-617 group demonstrated more than double the median radiographic progression-free survival time (median time: 12.0 months vs 5.6 months) and maintained good safety. 14 Based on the PSMAfore trial, on March 28, 2025, the FDA expanded the indication for 177Lu-PSMA-617 to include adults with PSMA-positive mCRPC previously treated with ARPIs and deemed suitable for delayed taxane-based chemotherapy.
Combination therapies based on 177Lu-PSMA-617
Due to the significant therapeutic efficacy of 177Lu-PSMA-617 monotherapy in patients with mCRPC, combination therapies involving 177Lu-PSMA-617 are also being widely explored.
ENZA-p (NCT04419402) is a phase II clinical trial for patients with mCRPC who have not received prior chemotherapy, aiming to evaluate the efficacy and safety of combining 177Lu-PSMA-617 with enzalutamide. According to the analysis of prostate-specific antigen progression-free survival (PSA-PFS), patients treated with enzalutamide in combination with 177Lu-PSMA-617 showed a significant advantage in PSA-PFS compared to enzalutamide alone (13 months vs 7.8 months). 15
Cabazitaxel has radiosensitizing properties that may enhance the cytotoxic effect of 177Lu-PSMA-617, and it is also useful in treating any PSMA-negative mCRPC. 16 LuCAB (NCT05340374) is an ongoing I/II clinical trial designed to evaluate the safety and efficacy of combining Cabazitaxel with 177Lu-PSMA-617 for the treatment of mCRPC, with the primary goal of determining the maximum tolerated dosage of both Cabazitaxel and 177Lu-PSMA-617. The trial is expected to be completed by 2026.
ANZUP2001 (NCT05150236) is an ongoing phase II clinical trial evaluating the efficacy of 7.4 GBq of 177Lu-PSMA-617 every 6 weeks for 6 cycles, in combination with ipilimumab and nivolumab for the treatment of mCRPC. The primary endpoint measures the 12-month PSA progression-free survival for patients. The primary completion date is expected to be December 2024.
The PRINCE study (NCT03658447), a phase I trial, evaluates the combination of 177Lu-PSMA-617 with pembrolizumab for mCRPC treatment. The latest median follow-up results presented at ASCO 2022 showed a PSA decline of ⩾50% in 76% of patients and an overall response rate (ORR) of 78%. The combination therapy did not exhibit a significant increase in adverse reactions compared to monotherapy. 17 This result indicates the feasibility of RLT and immunotherapy combinations in mCRPC, and further II/III phase studies are eagerly anticipated.
The LuPARP trial (NCT03874884) is a phase I dose-escalation and dose-expansion study aimed at evaluating the safety and tolerability of olaparib in combination with 177Lu-PSMA-617 for the treatment of mCRPC. Preliminary data from ASCO 2023 showed good tolerability and activity of this combination. 18
Progress in the treatment of mCRPC with 225Ac-PSMA
225Ac-PSMA is also a type of PSMA-targeted radioactive ligand therapy, in which 225Ac emits alpha particles. It is currently one of the most researched alpha-emitting isotopes used for targeted alpha therapy. Alpha particles have higher linear energy transfer, higher radiation energy, shorter range, and relatively higher biological effects, leading to stronger damage to tumor cells and lower toxicity to normal cells. 225Ac-PSMA-RLT has shown promising results in a wide range of studies.
Kratochwil et al. pioneered in the use of alpha-emitting radionuclide-labeled PSMA ligands in human therapy by administering 100 kBq/kg of 225Ac-PSMA-617 to two patients every 2 months. Subsequently, both patients experienced a decrease in PSA to below 0.1 ng/mL and achieved CR on imaging, without significant hematologic toxicities observed. 19 In another study, Kratochwil et al. continued clinical follow-up of 40 mCRPC patients receiving the same treatment regimen and observed that 87% of the patients experienced a PSA reduction, with 63% of patients achieving a PSA decrease of ⩾50%, further confirming the efficacy of 225Ac-PSMA-617 in mCRPC. 20 Sathekge et al. administered a total of 210 cycles of 225Ac-PSMA-617 treatment to 73 mCRPC patients. Their multivariate analysis indicated that a PSA reduction of ⩾50% post-treatment was significantly correlated with improved OS and PFS. 21 In a retrospective study reported in 2022, 53 mCRPC patients treated with 225Ac-PSMA-617 directly after ADT, a multifactorial analysis also found that a PSA decrease of over 50% was a predictor of OS and PFS. 22
Yadav et al. recruited 28 patients, of whom 15 (54%) had previously received 177Lu-PSMA-617 RLT, and the remaining 13 (46%) had not received 177Lu-PSMA-617 RLT. The study compared the response between these two groups of patients after receiving 225Ac-PSMA-617 targeted therapy. The results showed that 225Ac-PSMA-617 therapy induced moderate remission and improved survival (PFS/OS 11/13.7 months vs. 12/17 months) in 177Lu-PSMA-617 radioligand therapy refractory mCRPC patients. Despite the more modest response in the 177Lu-PSMA-617 refractory group compared to the naive group, similar OS and PFS validate that 225Ac can overcome the resistance of cancer cells to beta-emitting therapy (177Lu-PSMA-617 RLT) and trigger a response in refractory patients. 23
The AcTION phase I clinical trial (NCT04597411) is currently evaluating the efficacy and safety of 225Ac-PSMA-617 treatment in PSMA-positive prostate cancer patients, with or without prior 177Lu-PSMA-617 RLT, with maximum tolerated dose as the primary outcome, and is expected to be completed in 2025.
The WARMTH Act is a multicenter retrospective study that reviewed 488 mCRPC patients who were treated with one or more cycles of 8 MBq 225Ac-PSMA RLT. These patients had previously received treatment with ARPI, 177Lu-PSMA-617 (154.32%), or 223Ra (18.4%). The results showed that 225Ac-PSMA RLT showed good efficacy in mCRPC patients with a median overall survival of 15.5 months (95% CI 13.4–18.3) and a median progression-free survival of 7.9 months (6.8–8.9). 24
J591 is a monoclonal antibody targeting the external domain of PSMA, which has been extensively researched for radio-labeled isotopes. It is the most commonly used monoclonal antibody in clinical trials of anti-PSMA radioimmunotherapy. 225Ac-J591 is a novel radiopharmaceutical conjugate that combines J591 with the potent alpha-emitter 225Ac. In 2023, ASCO released the results of the first human dose escalation trial for 225Ac-J591 (NCT03276572, NCT04506567), showing its safety and preliminary efficacy. 25 The trial is still ongoing for further investigation.
In addition, the application of other 225Ac-PSMA-RLTs such as 225Ac-PSMA-I&T, 225Ac-PSMA-Trillium, and 225Ac-PSMA-R2 in the treatment of mCRPC is also emerging and being actively explored. TATCIST (NCT05219500) is an ongoing phase II study designed to assess the efficacy of 225Ac-PSMA-I&T in the treatment of mCRPC. PAnTHA (NCT06217822) is the first-in-human study aiming to evaluate 225Ac-PSMA-Trillium (BAY 3563254) in mCRPC, and is expected to commence recruitment in 2024. In addition, 225Ac-PSMA-R2 has also commenced phase I/II trials in patients with mCRPC (NCT05983198).
Currently, 225Ac-PSMA is still undergoing clinical trials and has not yet been recommended in major guidelines. Further extensive clinical trials are needed to confirm its efficacy in treating mCRPC.
Other PSMA-targeted RLT
131I can emit beta radiation and has long been used as a standard treatment for thyroid cancer. 131I-MIP-1095 is also the first PSMA RLT used for prostate cancer. 26 Some researchers have used it as a terminal treatment for metastatic prostate cancer, administering a 1-week course of 131I-MIP-1095. In the first 3 months after treatment, 60.7% of patients experienced a greater than 50% decrease in PSA levels, 25% of patients had a PSA decrease of over 75% and tumor size also significantly decreased. 26 However, with the continuous development of 177Lu-PSMA RLT, treatment related to 131I has been gradually replaced.
ECLIPSE (NCT05204927) is a phase III clinical trial using 177Lu-PSMA-I&T in patients with mCRPC who have previously received ARPI treatment and have not undergone taxane therapy. The trial began enrolling patients in November 2023. PNT2002 is an innovative, PSMA-targeted, 177Lu-based radionuclide therapy. In April 2023, the United States Food and Drug Administration granted fast-track designation to 177Lu-PNT2002, signaling its impending entry into clinical application. The SPLASH trial (NCT04647526) is a multicenter phase III clinical study aimed at evaluating the efficacy and safety of 177Lu-PSMA-PNT2002 in patients with mCRPC who have previously received second-line ARPI treatment. Abiraterone or enzalutamide is used as a control group. Data from the ongoing trial indicate that patients treated with 177Lu-PNT2002 had a median radiographic progression-free survival (rPFS) of 9.5 months, compared to 6.0 months in the control group. 27 Other 177Lu-PSMA drugs, such as 177Lu-PSMA-R2(NCT03490838), 177Lu-EB-PSMA(NCT04996602), and 177Lu-LNC1003(NCT06237491), are under extensive clinical trials.
212Pb is an emerging radionuclide that disrupts tumor cells by releasing alpha particles. Earlier studies by Banerjee et al. have demonstrated promising pharmacokinetics in a PCa mouse model. 28 Subsequently, Stenberg et al. found that 212Pb-NG001 exhibited PSMA-specific cancer cell targeting and delayed tumor growth in a C4-2 xenograft mouse model without the thymus. 29 These results indicate that 212Pb is a promising radionuclide for the treatment of mCRPC.
161Tb has decay properties similar to those of 177Lu, but emits a substantial number of conversion and Auger electrons. Compared to 177Lu, it may produce a greater therapeutic effect. Schaefer-Schuler et al. conducted a study on six patients with mCRPC who underwent treatment with 177Lu-PSMA-617 and subsequently with 161Tb-PSMA-617 to observe tumor absorbed dose. The results showed that 161Tb-PSMA-617 exhibited a significant advantage in tumor absorbed dose. 30 VIOLET (NCT05521412) is a phase I/II clinical trial evaluating the efficacy of 161Tb-PSMA-I&T in the treatment of mCRPC, with an expected completion date in 2026.
Radium-223
Therapeutic mechanism of radium-223
Radium-223 (223Ra) is the world’s first alpha-emitting radionuclide and has minimal impact on surrounding normal tissues. 31 It is primarily excreted through the gastrointestinal tract. 32 Acting similarly to calcium ions, 223Ra accumulates in areas of active bone metabolism, such as osteoblastic lesions, and emits high-energy α-rays into the nearby microenvironment. 223Ra reduces skeletal-related events (SREs) from bone metastasis while inhibiting cancer by inducing DNA double-strand breaks in nearby prostate cancer cells, osteogenic, and osteoblastic cells. It has also been proven to enhance the prognosis of mCRPC patients with bone pain. In May 2013, 223Ra (marketed as Xofigo) was approved by the FDA for therapy of bone metastases in patients with mCRPC.
Advances of radium-223 in the treatment of mCRPC
The ALSYMPCA study (NCT00699751), a pivotal phase III trial, first reported the use of 223Ra in treating mCRPC. The study involved 921 patients with symptomatic bone metastases but no visceral metastases, divided into two groups in a 2:1 ratio, with one group receiving intravenous injection of 223Ra and the other group receiving a placebo. The 3-year follow-up indicated that the median OS in the 223Ra treatment group was significantly longer than that in the placebo group (14.9 months vs 11.3 months, p < 0.001), and the time to first symptomatic skeletal event (SSE) was also extended by 5.8 months (15.6 months vs. 9.8 months, p < 0.001). 33 This study demonstrated the significant efficacy of 223Ra in prostate cancer bone metastases.
PARABO (NCT02398526) is a prospective, observational, non-interventional single-arm study with the primary endpoint being a clinically significant pain response (defined as an improvement of ⩾2 points in the most severe pain item on the Brief Pain Inventory). This study analyzed 354 patients and found that 223Ra reduced pain in mCRPC patients with symptomatic bone metastases, especially in those who received 5–6 injections. 34 The extent of metastatic disease did not impact the pain response. 34
The NCCN guidelines recommend 223Ra for treating symptomatic bone metastases in mCRPC patients without visceral metastases, 35 and the EAU guidelines list it as one of the second-line treatment options for mCRPC. 36 This therapy significantly improves OS, delays the first occurrence of SSEs, and enhances patients’ quality of life.
Combination therapies based on radium-223
Denosumab is a bone-targeted therapy. Combination therapy of 223Ra with denosumab can reduce the risk of SREs and improve patients’ prognosis.37,38 Saad et al. found that in mCRPC patients with bone metastases treated with a combination of 223Ra and denosumab had a longer median overall survival (mOS) compared to those without denosumab (15 months vs 13 months). 37
In trials involving novel hormone therapies, preliminary clinical results of ERA223 (NCT02043678) indicated that abiraterone in combination with 223Ra did not lead to an improvement in SSE-free survival compared to abiraterone monotherapy, but rather led to a higher frequency of fractures. 39 Consequently, the combination use of 223Ra with abiraterone is not currently recommended in clinical practice. REASSURE (NCT02141438) is a prospective observational study that enrolled 1474 patients, including 46 patients in the combination group (who initiated combined treatment of 223Ra and enzalutamide within 30 days) and 205 patients in the sequential group (who had received another treatment for more than 30 days prior to initiation of therapy). The results showed that the combined or sequential treatment of 223Ra and enzalutamide was safe and well tolerated, without increasing the risk of fractures. In addition, compared to the sequential group or all patients, the median overall survival was numerically longer in the combination group (22.2 months vs 16.5 months vs 15.6 months), indicating that the combination treatment of 223Ra and enzalutamide could significantly reduce the risk of disease progression 40 and offer a new approach for patients with mCRPC.
Bipolar androgen therapy (BAT) is an innovative hormone treatment method that involves periodic administration of high-dose testosterone to address PCa patients who have developed resistance to previous hormone treatment regimens. 41 This therapy aims to restore sensitivity to androgen signal suppression in patients and may lead to clinical responses. Currently, BAT-RAD (NCT04704505) is ongoing to evaluate the efficacy of combining BAT with 223Ra (RAD) in mCRPC patients. This study is expected to be completed by 2027.
Furthermore, a clinical trial investigating the combination of 223Ra and PARP inhibitors for the treatment of mCRPC is ongoing. The COMRADE trial (NCT03317392) is an open-label, multicenter, phase I/II trial aimed at evaluating the dose, safety, and effectiveness of combining olaparib with 223Ra in male patients with mCRPC and bone metastases. The COMRADE trial is divided into two stages: Stage 1 involves a dose escalation design using a 3 + 3 approach to determine the recommended phase II dose of olaparib in combination with a fixed dose of 223Ra (55 kBq/kg IV, administered every 4 weeks for a total of 6 doses). Preliminary results have shown a PSA response rate of 16.7% (n = 2) and an alkaline phosphatase response rate of 67% (n = 8). The median follow-up time was 6.5 months (range 2.8–11.8), with a 6-month rPFS of 57% (95% CI: 25%–80%). 42 Patients with BRCA2 mutations who received treatment with 223Ra and olaparib experienced a prolonged time to disease progression. The main endpoint of the phase II portion of the trial is rPFS assessed according to PCWG3 bone metastasis guidelines and RECIST v1.1 43 criteria for non-bone disease. The second stage of the trial is currently ongoing, and the results are eagerly anticipated.
With the popularity of immunotherapy, the combination of 223Ra with immunotherapy has also sparked interest. Studies such as the combination treatment of sipuleucel-T and 223Ra for mCRPC (NCT02463799), atezolizumab (ATZ) in combination with 223Ra in mCRPC (NCT02814669), the addition of pembrolizumab to 223Ra in mCRPC (NCT03093428), and the assessment of a better treatment approach among 223Ra alone, 223Ra in combination with M3814 (Nedisertib), or 223Ra in combination with M3814 (Nedisertib) and avelumab to reduce the chances of mCRPC growth or spread into the bones (NCT04071236). These studies are enriching the application of 223Ra in mCRPC.
Combination therapies based on different radionuclides
The combination of different RLTs has flourished in recent years and has shown promising results in clinical trials. In particular, the combination of 177Lu-PSMA-617 and 225Ac-PSMA-617 has obtained significant attention. Research indicates that 225Ac-PSMA-617 can enhance the clinical efficacy of 177Lu-PSMA-617 and reduce its toxic side effects. 42 A phase I/II clinical trial (NCT04886986) is assessing the combination use of 177Lu-PSMA-I&T and 225Ac-J591 in mCRPC patients, with the primary goal of assessing the proportion of patients with dose-limiting toxicity and a drop in PSA of more than 50%. It aims to evaluate the synergistic effects of alpha and beta particles in reducing PSA levels and minimizing adverse reactions. The study’s primary completion date is expected to be December 2024.
Furthermore, several studies also tried to combine 223Ra with RLTs to see whether this combination brings benefits in treatment with mCRPC. AlphaBet (NCT05383079) is a phase I/II study evaluating the safety of 223Ra in combination with 177Lu-PSMA-I&T in mCRPC, with an expected completion date in 2026. The RALU study evaluates the safety and efficacy of sequential 223Ra and 177Lu-PSMA therapies in mCRPC patients. An interim retrospective review of 49 patients revealed that those first treated with 177Lu-PSMA had a mOS of 12.6 months (95% CI 8.8–16.1), while the mOS for patients receiving 223Ra treatment as the first treatment was 31.4 months (95% CI 8.8–16.1). 44 This finding suggests that sequential treatment with these radionuclides is feasible, well-tolerated, and offers new insights for future radionuclide therapies.
Adverse effects of RLT
RLT has shown significant efficacy in the treatment of mCRPC, but it is also associated with several side effects. These adverse effects vary depending on the specific radionuclide used. 177Lu-PSMA, one of the most commonly used RLT, is associated with adverse events (AEs) such as renal toxicity, fatigue, and gastrointestinal symptoms, including xerostomia (occurring in up to 80%–87% of cases, 45 which is associated with PSMA binding in the salivary glands) and nausea/vomiting.46–49 In addition, hematologic toxicity, primarily thrombocytopenia and anemia, is frequently observed, with approximately 15%–30% of patients experiencing grade 3/4 toxicity.46–49 By contrast, 225Ac-PSMA may reduce myelosuppression to some extent (due to its shorter radiation range, which helps preserve bone marrow function), but its higher radiation energy increases the radiation burden on the salivary glands and kidneys, leading to a higher incidence of xerostomia and renal impairment.19,24 However, some studies suggest that dynamic dose-reduction strategies can help alleviate these adverse effects.19,50
223Ra, primarily used for the treatment of bone-metastatic disease, is mainly associated with myelosuppression, though the incidence of grade 3–4 bone marrow toxicity is relatively low, with thrombocytopenia and anemia being the most common AEs. Other AEs include gastrointestinal symptoms (such as nausea and diarrhea), exacerbation of bone pain, and spinal cord compression.33,44,51
Though these side effects of RLT seem unavoidable, they are relatively common, transient, and generally manageable. Strict monitoring and proactive management during treatment are essential to optimize therapeutic outcomes and maintain patients’ quality of life.
Conclusion
More and more clinical trials applied with RLT are explored in the treatment of mCRPC. We have listed key clinical trials in this field in Table 2. The advantage of PSMA-RLT lies in its ability to target PSMA, delivering localized radiation damage to tumor cells, thereby enhancing the selectivity and precision of the treatment. This therapy has been recommended for PSMA-positive mCRPC patients who have previously undergone androgen receptor inhibitors and taxane-based chemotherapy. 223Ra has also shown significant clinical efficacy in treating bone metastatic mCRPC, particularly in patients with symptomatic bone metastases and without known visceral metastases (Figure 1). Moreover, the combination of different radionuclide therapies with other treatment modalities, such as immunotherapy (NCT03658447, NCT03805594, et al.), chemotherapy (NCT05340374, NCT04343885), and PARP inhibitors (NCT03874884, NCT03317392), is also becoming a research hotspot. Furthermore, with the development of new radionuclide drugs, it is expected that RLT will play a vital role in the treatment of mCRPC.
Ongoing clinical trials of PSMA-targeted RLT and radium-223 in prostate cancer treatment.

ADT, androgen deprivation therapy; ARPI, androgen receptor pathway inhibitor; BAT, bipolar androgen therapy; DLT, dose-limiting toxicity; mCRPC, metastatic castration-resistant prostate cancer; MTD, maximum tolerated dose; OS, overall survival; PCa, prostate cancer; PSA-PFS, prostate-specific antigen-progression-free survival; PSMA, prostate-specific membrane antigen; RLT, radioligand therapy; RP2D, recommended phase II dose; rPFS, radiographic progression-free survival; SAE, serious adverse event; TEAEs, treatment emergent adverse events.
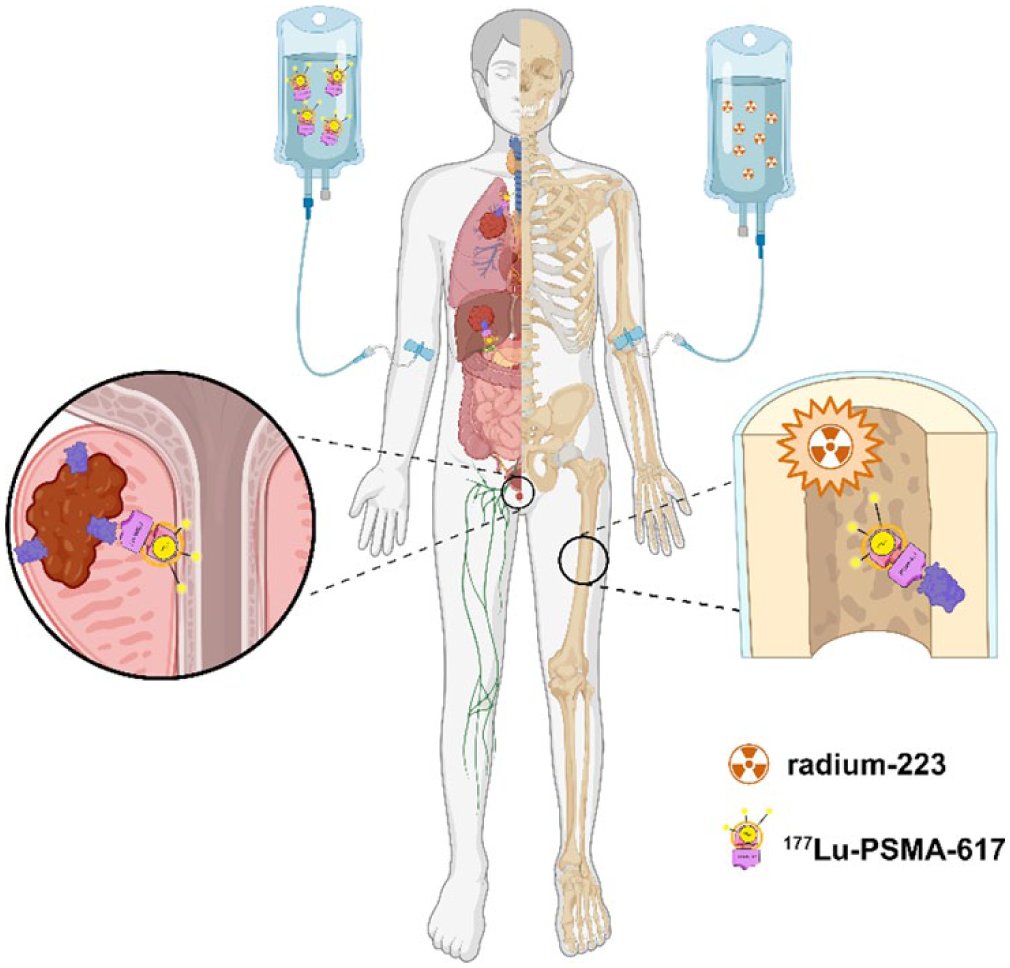
Treatment of mCRPC with 223Ra or 177Lu-PSMA-617.
