Abstract
Background:
Esophageal cancer (EC) is the sixth leading cause of cancer mortality worldwide, with a poor prognosis and a 5-year survival rate of 5% in advanced cases.
Objectives:
To evaluate the efficacy of programmed death-1 (PD-1) and programmed death-ligand 1 (PD-L1) inhibitors in EC patients by analyzing phase III clinical trials.
Design:
A meta-analysis following the PRISMA Statement 2020 guidelines.
Methods:
PubMed/MEDLINE, Web of Science, and Embase were searched through 6 December 2022, and the analysis was conducted using Review Manager 5.4.3 (Cochrane).
Results:
Out of 387 studies, 13 phase III clinical trials with 6519 participants were pooled. Overall survival (OS) favored PD-1/PD-L1 inhibitors with a Cohen’s d of 0.28 (95% CI: 0.12–0.43; p = 0.0006), and the likelihood of achieving objective response also favored these inhibitors (OR: 2.04, 95% CI: 1.68–2.48; p < 0.0001).
Conclusion:
This meta-analysis provides strong evidence that PD-1/PD-L1 inhibitors combined with chemotherapy improve OS and objective response rate among patients with advanced EC but do not affect progression-free survival.
Trial registration:
Open Science Framework: osf.io/y27rx.
Plain language summary
Esophageal cancer is a significant global health concern with a low survival rate, especially in advanced cases. This study, following rigorous guidelines, analyzed data from 13 clinical trials involving over 6,500 participants to assess the effectiveness of PD-1 and PD-L1 inhibitors. The results suggest that combining these inhibitors with chemotherapy improves overall survival and the likelihood of positive treatment responses in advanced esophageal cancer patients. However, no significant impact on progression-free survival was observed. This meta-analysis provides valuable insights into the current state of immune checkpoint inhibitors for esophageal cancer, offering hope for improved treatment outcomes.
Introduction
Esophageal cancer (EC) is the sixth leading cause of cancer mortality and the seventh most common cancer globally. The two most common histological types of EC comprise adenocarcinoma (EAC) and squamous cell carcinoma (ESCC).1,2 Only 1–2% of all EC are small-cell carcinomas or sarcomas. 3 In rare cases, carcinoids, lymphomas, and melanomas may arise in the esophagus. 4 EC treatment is largely dependent on chemotherapy, radiotherapy, and surgery but the prognosis remains unfavorable in a majority of cases with a 5-year survival rate of 5% in advanced stages.5,6 The treatment strategies for EC are determined based on the pathological origin of the disease; however, therapeutic options are limited. 7 In early EC that is limited to the mucosa, the primary treatment option is endoscopic mucosal resection which has a 5-year survival rate of 41%. 8 For EC that is resectable with muscle invasion, the primary treatment option is esophagectomy along with lymphadenectomy; neoadjuvant chemotherapy, radiotherapy, or chemoradiotherapy is optional. 9 Immunotherapies have been tested across various cancer entities, specifically in head and neck cancers with proven efficacy that may share some mechanistic similarities with EC.6,10,11
Immunotherapy has rendered a shift in the concepts of tumor treatment and has become a third revolution in therapy following chemotherapy and targeted therapy measures. 12 Immunotherapy based on the use of immune checkpoint inhibitors (ICIs) has shown promising safety and efficacy in treating various immunogenic tumors and has led to sustained responses in diseases including malignant melanoma and non-small-cell lung cancer.13,14 Programmed cell death-ligand 1 (PD-L1) is an immune checkpoint molecule, also called leukocyte differentiation antigen 274 (CD274) or B7 homolog 1 (B7-H1). The CD274 gene encodes the PD-L1 protein. PD-L1 on tumor cells when combined with its receptor programmed death-ligand (PD-L1) disrupts the activity of Ras/MEK/ERK and PI3K/AKT signaling pathways; this leads to T-cell activation, proliferation, and impairment in the survival of tumor cells. 15 In addition to cancer cells, dendritic cells, macrophages, and cancer-associated fibroblasts also express PD-L1. These components together create an immunosuppressive microenvironment leading to tumor immune escape. PD-L1 also promotes the secretion of various cytokines including IL-1β, IL-6, IL-8, and tumor necrosis factor (TNF)-α, leading to sustained immune response. Immunotherapy with PD-1/PD-L1 inhibitors can restore the body’s anti-tumor response, thereby controlling and clearing tumor cells.16,17
In the past 10 years, various trials have tested immunotherapy in EC to improve therapeutic outcomes. This meta-analysis focuses on two key ICIs including PD-1 and PD-L1 designed to restore the anti-cancer response for EC. 18 Phase III clinical trials administering PD-1/PD-L1 inhibitors either alone or in combination with standard care are assessed. The objective of this report is to compile an updated meta-analysis that guides future directions for EC patients. By concentrating on overall survival (OS), progression-free survival, and objective response, this synthesis aspires to optimize outcomes and inform treatment decisions.
Materials and methods
Literature search
This meta-analysis adhered to PRISMA Statement 2020 guidelines. 19 The checklist is attached in Supplemental Materials. Three databases including PubMed/MEDLINE, Web of Science, and Embase were searched, along with an additional search of ClinicalTrials.Gov and WHO-ICTRP to locate any non-published studies, as well as ongoing trials (last searched on 31 July 2023). Keywords including PD-1, PD-L1, immunotherapy, esophageal, and/or cancer were applied across the databases and search engines. The keyword strings are attached in Table 1. A Boolean (and/or) logic was applied. The search was conducted through 6 December 2022. We did not apply any language restrictions; all non-English studies were translated into English using Google Translate.
Keyword strings.

PD-L1, programmed death-1; PD-L1, programmed death-ligand 1.
Eligibility criteria
Only phase III clinical trials were included administering PD-1/PD-L1 inhibitors either alone or in combination with standard care among adult patients with EC compared with placebo or standard care. Cohorts, case series/reports, previously conducted systematic reviews/meta-analytical studies, and brief reports were excluded.
Outcomes
There were three outcomes of this study. These comprised OS reported in months, progression-free survival (PFS) also reported in months, and objective response rate (ORR) reported as the proportion of patients with a response.
Data extraction and synthesis
Clinical trial data were tabulated and presented as author, year, title, intervention, participants and condition, and primary endpoint(s). Ongoing trials were tabulated as (NCT Number, Study Title, Interventions, Primary Outcome Measures, Phases, Enrollment, Study Design, Primary Completion Date, and Locations). The bibliographic information was stored in EndNote X9 (Clarivate Analytics, London, UK). For the quantitative analysis, data for OS, PFS, and ORR was entered into an Excel datasheet. Both OS and PFS outcomes were meta-analyzed and computed for standardized mean difference (SMD) applying 95% confidence intervals (CI), which was reported as Cohen’s d. Whereas ORR was reported as odds ratio (OR) applying 95% CI. A random effects model was applied for all outcomes. To quantify whether heterogeneity was present among the included studies, the I2 index was calculated. A funnel plot was generated to visually inspect for publication bias. The analysis was conducted in Review Manager 5.4.3. (RevMan, Cochrane, London, UK). Whereas the referencing software utilized for this study was Mendeley (Elsevier). A kappa score of inter-reviewer reliability was additionally measured to assess the level of agreement between the two independent raters along the inclusion process; this was conducted in Statistical Package for Social Sciences (SPSS, v24, Chicago, IL, USA).
Registration and role of funding
The protocol for this meta-analysis was registered in the Open Science Framework (OSF): (osf.io/y27rx). No funding was obtained for this study.
Results
Study selection and Kappa score
A total of 387 studies were identified from the databases. Of these, 36 were duplicates and were removed before the screening. We screened a total of 351 studies for potential inclusion in our meta-analysis. Out of these, 312 were excluded for various reasons. An estimated 87 studies were excluded as they were observational cohorts, case series, or individual case reports that did not fit the requirement for phase III clinical trials. In addition, 58 studies were previously conducted systematic reviews or meta-analyses, and as our focus was on original research, they were not included. Around 120 studies were brief reports or were unrelated to the specific administration of PD-1/PD-L1 inhibitors either alone or in combination with standard care among adult patients with EC, leading to their exclusion. The remaining 47 studies were removed for other miscellaneous reasons, such as lack of relevant data or not meeting other specified criteria. The remaining 39 studies were reviewed using their full texts. Of those, 13 phase III trials were included in this meta-analysis. During the identification of ongoing trial records via registers, a total of 198 records were identified. All were assessed for eligibility, after which 90 were excluded as they did not target PD-1/PD-L1 therapies for EC, whereas 17 were non-human studies; a total of 91 ongoing trial records were included. The PRISMA flowchart depicting the study selection process is illustrated in Figure 1. The kappa score of inter-reviewer reliability was computed to be 0.91 suggesting excellent agreement.

PRISMA flowchart depicting the study selection process.
Characteristics of the included completed trials
A total of 6519 participants were pooled across the 13 phase III trials included in this study. The key characteristics of all included trials are listed in Table 2.
Key characteristics of all included trials (N = 13).
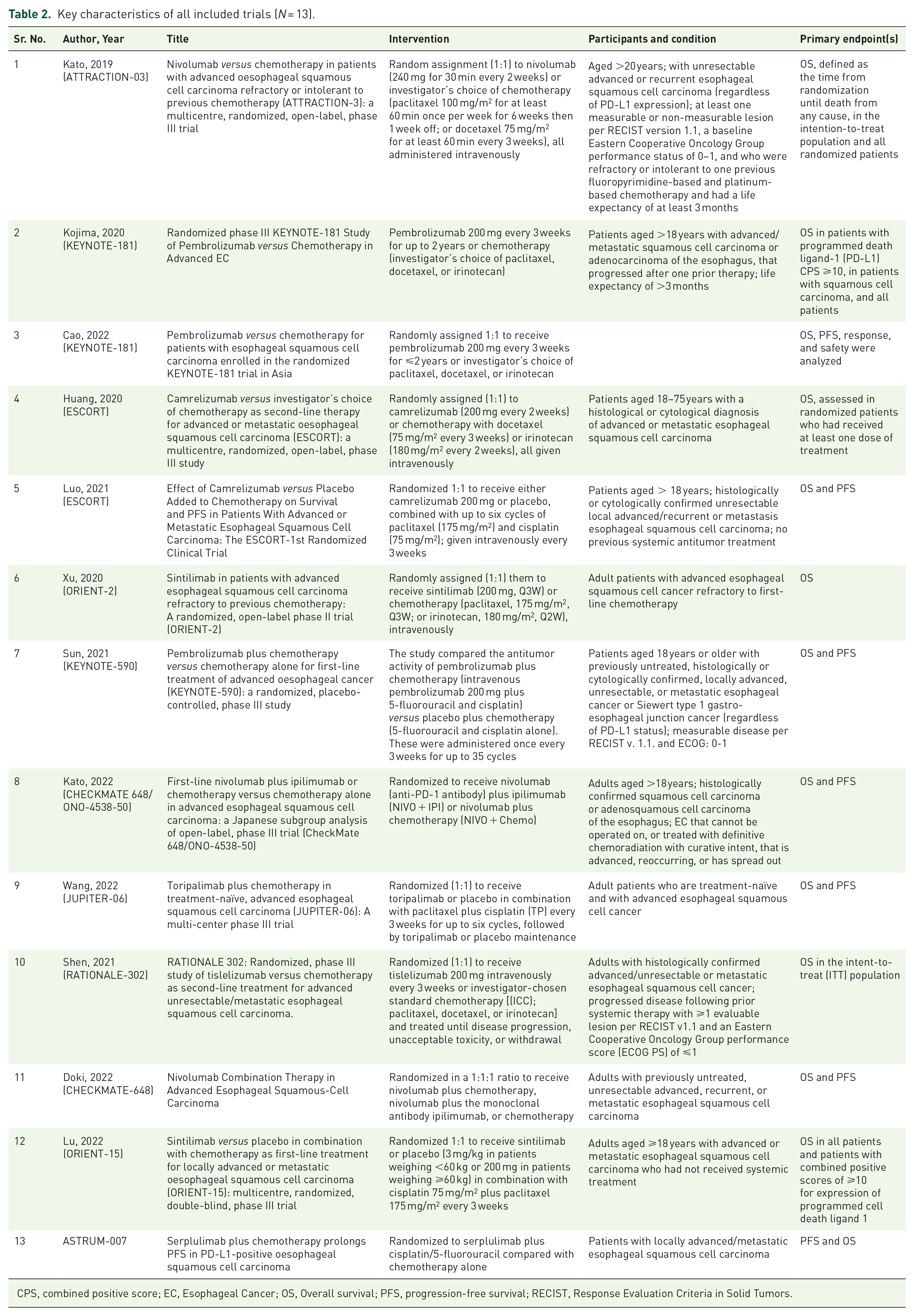
CPS, combined positive score; EC, Esophageal Cancer; OS, Overall survival; PFS, progression-free survival; RECIST, Response Evaluation Criteria in Solid Tumors.
Meta-analytical findings of participants with squamous cell carcinoma
This analysis incorporated data from 12 trials involving patients with squamous cell carcinoma (SCC). These trials comprised a total of 2876 participants in the ICI group and 2701 in the placebo or standard care group, and they were all evaluated for overall survival (OS) outcomes. The mean difference (MD) was determined to be 2.69 (95% CI = 2.33–3.06; p < 0.00001; I2 = 0%), indicating a statistically significant improvement in OS for the ICI group. The SMD, represented by Cohen’s d, was calculated as 0.28 (95% CI = 0.12–0.43). Despite being small, the effect size was statistically significant and favored the ICI group (p = 0.0006; I2 = 88%; Refer to Figure 2).

Forest plot depicting the outcome of OS computed as MD and SMD, which is reported as Cohen’s d. A random-effects model was applied with reporting of 95% CI, the I2 index value, and the p-value.
A sensitivity analysis was performed, in which data from three high-weight studies [Sun, 2021 (KEYNOTE-590); Huang, 2020 (ESCORT); and Shen, 2021 (RATIONALE-302)] were excluded. These three studies had initially contributed 73.5%, 5.3%, and 4.7% weights to the total effect size, respectively. After their exclusion, the recalculated MD was 2.52 (95% CI = 1.63–3.42), while Cohen’s d was 0.18 (95% CI = 0.12–0.25). These adjusted results remained statistically significant (p < 0.00001), aligned closely with the original findings, and displayed no heterogeneity (I2 = 0%) for both MD and Cohen’s d. This suggests that the overall conclusions from the original analysis were robust and not overly dependent on any single study.
In parallel, data from 12 trials encompassing 2992 participants in the ICI group and 2812 in the placebo or standard care group were analyzed for PFS outcomes. Unlike the findings for OS, the effect direction for PFS was unfavorable and not statistically significant. The MD was −0.17 (95% CI = −0.66−0.31; p = 0.49), indicating no significant difference in PFS between the ICI and placebo/standard care groups. This was coupled with high heterogeneity (I2 = 89%). The SMD, as represented by Cohen’s d, was calculated to be −0.01 (95% CI = −0.19–0.17; p = 0.93), reinforcing the lack of a significant effect. Heterogeneity remained high at I2 = 92% (refer to Figure 3).

Forest plot depicting the outcome of PFS computed as MD and SMD, which is reported as Cohen’s d. A random-effects model was applied with reporting of 95% CI, the I 2 index value, and the p-value.
Sensitivity analysis was conducted by excluding data from three high-weight studies [Sun, 2021 (KEYNOTE-590, 11.7%); Huang, 2020 (ESCORT, 11.3%); and Kojima, 2020 (KEYNOTE-181; 10.2%)]. After their exclusion, the recalculated MD was −0.13 (95% CI = −0.91–0.65; p = 0.75; I2 = 81%), and Cohen’s d was −0.04 (95% CI = −0.19–0.1; p = 0.54; I2 = 81%). These adjusted results still demonstrated no statistically significant difference in PFS between the two groups and the heterogeneity was somewhat reduced, but remained substantial. This sensitivity analysis confirms the robustness of the original findings for PFS.
For the outcome concerning ORR, data from 11 trials involving 2568 participants in the ICI group and 2561 participants in the placebo or standard care group were evaluated. The findings reveal a significant increase in the likelihood of achieving an objective response when ICIs were administered (OR = 2.04, 95% CI = 1.68–2.48), suggesting the efficacy of ICIs in improving ORR. The analysis exhibited moderate heterogeneity (p < 0.0001, I2 = 45%), indicating some variability across the included studies (refer to Figure 4).

Forest plot depicting the outcome of ORR computed as OR. A random-effects model was applied with reporting of 95% CI, the I2 index value, and the p-value.
In the sensitivity analysis, the three studies contributing the highest weights were excluded [Lu, 2022 (ORIENT-15; 13.7%); Doki, 2022 (CHECKMATE-658; 13.3%); and Luo, 2021 (ESCORT, 12.8%)]. Following their removal, the recalculated OR was 2.02 (95% CI = 1.52–2.69; p < 0.00001, I2 = 51%). This result maintained a significant effect favoring ICIs, albeit with slightly increased heterogeneity. These findings further substantiate the robustness of the original analysis for ORR outcomes.
Meta-analytical findings of patients with adenocarcinoma
The separate analysis of patients with adenocarcinoma involved a total of 201 patients, with 99 patients in the ICI arm and 102 in the placebo/standard care arm. The forest plots are depicted in Figure 5. The MD in OS for patients with adenocarcinoma treated with ICIs compared to placebo or standard care was 1.7 months (95% CI: 0.65–2.75), suggesting that ICIs extend the OS time by approximately 1.7 months compared to standard care or placebo. This effect was statistically significant, with a Z value of 3.18 (p = 0.001). The effect size calculated by Cohen’s d was 0.45 (95% CI: 0.17–0.73), indicating a medium effect size. This also showed statistical significance, with a Z value of 3.14 (p = 0.002).
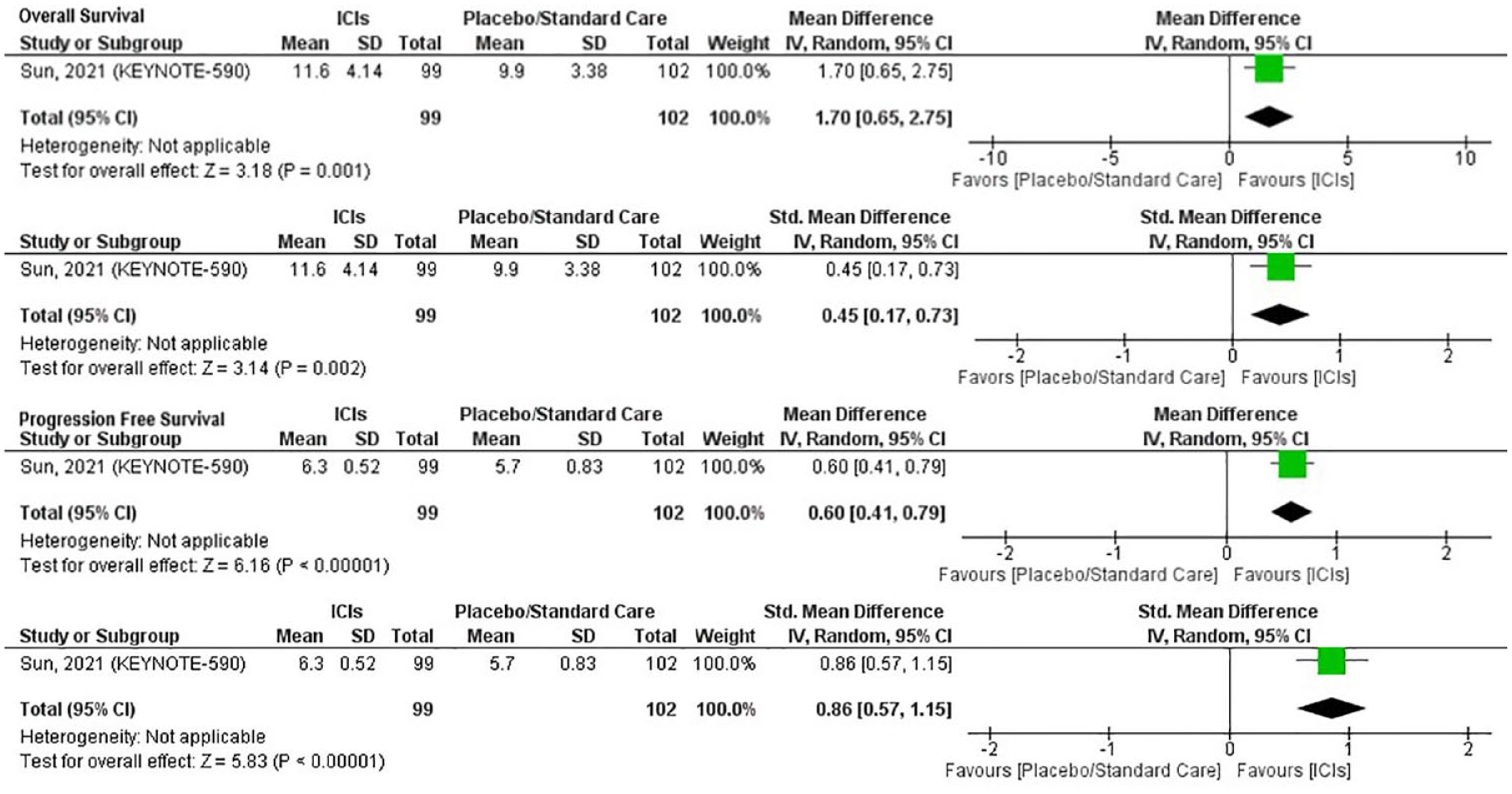
Forest plot depicting the outcomes in patients with adenocarcinoma of OS and PFS computed as MD, SMD, and OR. A random-effects model was applied with reporting of 95% CI, Z-value, and p-value.
The MD in PFS for patients with adenocarcinoma treated with ICIs compared to placebo or standard care was 0.6 months (95% CI: 0.41–0.79). This implies that ICIs increase the PFS time by roughly 0.6 months. This effect was statistically significant, with a Z value of 6.16 (p < 0.00001). Cohen’s d was calculated as 0.86 (95% CI: 0.57–1.15), suggesting a large effect size. This result was also statistically significant, with a Z value of 5.83 (p < 0.00001; refer to Figure 5).
Publication bias inspection
On visually inspecting the funnel plot, the studies appear balanced with no gross deviations. Overall, the plot is symmetric with a generally even distribution through the funnel shape. While there is a slight chance of publication bias due to external factors including the under-representation of populations, our synthesis suggests limited heterogeneity in the stipulated sample (Figure 6).

Funnel plot for the visual inspection of publication bias.
Synthesis of ongoing clinical trials
In total, 91 ongoing clinical trials enrolling 25,918 participants are discussed. The full data are tabulated in Supplemental Table 1. The trials span completion until December 2034. Among the ongoing trials, the largest number are in phase II, with a total of 45 trials. There are eight trials in phase III. Phase I trial number 10. In addition to these, 21 trials combine phases I and II. Notably, there is only one trial for which the phase is not specified.
The five most commonly used primary outcome measures in the 91 ongoing trials are as follows:
Number of participants with dose-limiting toxicities (DLTs): This is a measure to determine the toxicity and safety of a new drug or treatment regimen. A DLT is an adverse event or set of related events that are considered to be related to the drug and which prevent further escalation of the dose. This is typically evaluated during the first cycle of treatment (often 21 or 28 days).
Number of participants with adverse events (AEs): This is a measure of the number of participants who experienced undesirable experiences associated with the use of a medical product, whether or not they were considered related to the medical product. This is monitored throughout the study period.
Overall response rate (ORR): This is a measure of the proportion of patients who have a partial or complete response to treatment. It does not include patients who have stable disease or progressive disease. This is usually evaluated by the RECIST criteria (Response Evaluation Criteria In Solid Tumors), which give specific rules for when tumors in cancer patients improve (‘respond’), stay the same (‘stable’), or worsen (‘progression’) during treatment.
Incidence of treatment-emergent AEs: This is a measure of the number of participants who experience new, or a worsening of, AEs associated with the use of the study drug or treatment. These are typically reported from the time of the first administration of the study drug until a specified time after the last dose.
Maximum tolerated dose (MTD) or recommended phase II dose (RP2D): The MTD is the highest dose of a drug or treatment that does not cause unacceptable side effects. The determination of the MTD is a critical part of phase I studies. The RP2D is the dose or dose regimen that is recommended, based on safety and efficacy data, for evaluation in phase II clinical trials.
Out of the 91 ongoing trials, 41 of them do not specify an allocation method, 33 are randomized, and 17 are non-randomized. When it comes to the intervention model, the majority of the trials (41 in total) use a single-group model. Another 43 trials employ a parallel intervention model; six trials use a sequential model; and one trial uses a factorial model. As for masking, most trials (84 out of 91) do not use any masking. Three trials use a double masking method involving the participant and the investigator; another three trials utilize a quadruple masking method, which includes the participant, care provider, investigator, and outcomes assessor; and one trial employs a triple masking method involving the participant, investigator, and outcomes assessor. The primary purpose for almost all trials (90 out of 91) is treatment, with only one trial intended for screening.
The 91 ongoing trials span across multiple countries; the United States has the highest participation, being involved in 53 of the ongoing trials. China follows closely, with involvement in 19 trials. Taiwan is involved in nine trials, the United Kingdom in 11 trials, and Germany is involved in seven trials. Other trials are being conducted in Canada, France, Italy, Japan, the Republic of Korea (South Korea), Australia, Belgium, Brazil, Chile, Denmark, Hungary, Israel, the Netherlands, Poland, Russia, Spain, and Turkey.
Discussion
To our knowledge, this is the largest meta-analysis to date comparing outcomes of efficacy between immunotherapy and standard care or placebo groups for the treatment of EC. Our analysis encompassed 6519 participants across 13 trials. In the SCC group, we observed positive results for OS (SMD = 0.28, p = 0.0006) and ORR (OR = 2.04, p < 0.0001) but not for PFS (SMD = −0.01, p = 0.93). In the adenocarcinoma group, we noted favorable outcomes with immunotherapy. OS showed a medium effect size in favor of ICI treatment (SMD = 0.45, p = 0.002). For PFS, the adenocarcinoma group demonstrated a large statistically significant advantage for ICIs (SMD = 0.86, 95% CI: 0.57–1.15, p < 0.0001), in contrast to the SCC group. In our meta-analysis, we included the administration of nivolumab, pembrolizumab, camrelizumab, sintilimab, toripalimab, ipilimumab, and serplulimab. In almost all cases, the condition being treated was advanced EC of squamous origin with Keynote 590 treating esophageal adenocarcinoma.
Immune checkpoint signaling pathways are essentially comprised of PD-1/PD-L1 and CTLA-4 pathways.20–23 PD-1 gains relevance as it is a negative costimulatory receptor that is primarily expressed on activated T cells that can bind to PD-L1 and PD-L2 ligands inhibiting the effect and function of T cells.24,25 With 600,000 new cases and 540,000 deaths due to EC reported in 2020, the prognosis of the disease has been poor.26–28 Squamous cell pathology comprises an estimated 90% of EC cases; it also has a median survival of less than 1 year and treatment heavily relies on chemotherapy regimens.29–32 We see the landscape changing with immunotherapy playing a key role as a combination treatment. 26 Of key interest are the PD-1 and PD-L1 pathways because of the reproducible survival benefit being witnessed in differential trials. While trials have been ongoing for various investigational compounds, the US Food and Drug Administration approved nivolumab in combination with chemotherapy on 27 May 2022, as a first-line treatment for advanced or metastatic EC. Pembrolizumab and ipilimumab are also approved for EC. 33 In our meta-analysis, we find a positive associative relationship between anti-PD-1/PD-L1 immunotherapy with OS and ORR that can benefit the long-term survival of EC patients. We selected OS, PFS, and ORR as endpoints and found that PD-1 inhibitors significantly improved OS and ORR in EC compared to chemotherapy.
Overall, there has been suboptimal progress in the treatment of EC in the last two decades as the median OS in advanced disease is still 11 months. In our included data, many immunotherapeutic agents have improved survival rates among patients with advanced EC as both first-line and second-line treatments. Numerous phase III trials have been conducted recently that looked at various PD-L1 blockers and their efficacy for improved survival outcomes in EC. As a second-line treatment, ATTRACTION-03, 34 ESCORT, 35 and RATIONALE-03 36 trials reported significant improvement in median OS regardless of PD-L1 status, whereas KEYNOTE-181 enrolled only PD-L1-positive populations with EC. ATTRACTION-3 was the first phase III clinical trial of the immunotherapeutic agent Nivolumab across 8 high-income countries (HICs) given to 419 pre-treated patients with advanced EC (squamous cell) regardless of PD-L1 expression; modestly favorable outcomes were noted in the immunotherapy arm with a median OS of 10.9 versus 8.4 months in the chemotherapy group (p = 0.019). 34 The highest median OS was noted in the ESCORT trial with significant improvement in survival rates compared to other phase III trials for pre-treated EC patients. The ESCORT trial was a phase III trial of the immunotherapeutic agent Camrelizumab, given as second line to 596 patients in advanced EC across China. 35 Among patients who received both Camrelizumab and chemotherapy versus chemotherapy alone, the median OS was 15.3 versus 12 months (p = 0.001). In the RATIONALE 302 trial, immunotherapeutic agent Tislelizumab in pre-treated patients with ESSC showed improvement in median OS across the entire population (8.6 versus 6.3 months) and in the PD-L1 + population (10.3 versus 6.8 months) compared to chemotherapy group. 36 In KEYNOTE 181, second-line immunotherapy with Pembrolizumab was given to PD-L1 combined positive score (CPS) ⩾10 patients with advanced EC and there was a significant improvement in median OS in the pembrolizumab versus chemotherapy group of 9.3 versus 6.7 months (p = 0.0074). 37 Both the RATIONALE 302 and KEYNOTE 181 did find support for second-line immunotherapeutic agents in advanced EC; however, combination regimens with PD-1 blockers and chemotherapy demonstrated in the ESCORT trial provided important support for this treatment approach in EC.
In treatment-naïve patients with EC, the four phase III trials including Checkmate 648, 26 ORIENT-15, 38 JUPITER-06, 39 and ASTRUM-007 40 trials found improved survival outcomes among patients regardless of PD-L1 status among ESSC patients, whereas Checkmate 649 only treated EAC patients with PD-L1+ status. 40 Two trials, Checkmate 648 and 649, were conducted globally to understand the impact of nivolumab combination therapy as first-line therapy in advanced esophageal squamous cell carcinoma (ESSC) or esophageal adenocarcinoma (EAC), respectively. In Checkmate 648, Nivolumab combined with chemotherapy demonstrated a significant improvement in OS when compared to chemotherapy alone. In the overall population, the median OS was 13.2 months with Nivolumab plus chemotherapy, as opposed to 10.7 months with chemotherapy alone, with a 26% lower risk of death (hazard ratio, 0.74; 99.1% CI, 0.58–0.96; p = 0.002). Moreover, in the PD-L1-positive subgroup, the median OS was even more pronounced at 15.4 months with Nivolumab plus chemotherapy compared to 9.1 months with chemotherapy alone (p < 0.001). 26 In the Checkmate 649 trial, 40 first-line Nivolumab combined with chemotherapy had a better median OS than chemotherapy alone in advanced EAC (14.4 months versus 11.1 months, p < 0.0001) with PD-L1 CPS ⩾5 which was comparable to other PD-L1+ subgroup findings including the Checkmate 648 trial. In the ORIENT-15 trial as first line, Sintilimab plus chemotherapy had a median OS of 16.7 versus 12.5 months which was higher in patients with CPS ⩾10 (17.2 versus 13.6 months) yet the overall population also had significant improvement in survival rates regardless of PD-L1 expression levels. 38 In the JUPITER-06 trial, immunotherapeutic agent Toripalimab plus chemotherapy was given as first line to advanced ESSC patients irrespective of PD-L1 expression with a significant improvement in median OS for the Toripalimab group of 17.0 months versus 11.0 months for the chemotherapy group only (p < 0.001) which is promising. 39 In the ASTRUM-007 trial, the median OS was found to be significantly improved with first-line Serplulimab plus chemotherapy compared to chemotherapy alone in patients with CPS ⩾1 in advanced ESSC (15.3 versus 11.8 months, p = 0.002) which similarly suggests it to be an important immunotherapeutic agent against EC. 40
The PD-1/PD-L1 pathway works by many regulatory pathways including JAK/STAT3, NF-κB, WNT, Hedgehog (Hh), MAPK, and PI3K/AKT.41–44 All six pathways exert pivotal roles in tumorigenesis by regulating the expression of immune markers, activating the immune system, and/or regulating immune marker expression. Together, PD-1/PD-L1 inhibitors play a critical role in EC, thereby allowing the testing to be an impunity and challenge for therapy. However, the significance of PD-L1 expression levels for meaningful clinical response in EC patients is still unclear. While there are many unknowns in treatment, the ongoing and concluded phase III trials allow for a clear action plan for dosing, safety, durability, and efficacy.
It is crucial to consider homogenizing PD-1 assays in clinical trial settings as well as routine clinical examinations. Furthermore, the criteria for receiving PD-1 inhibitors require more insight as there are different metrics across clinical trials that have not been optimized for clinical practice. Further clinical trials can focus on exploring other potential biomarkers and their impact on immunotherapy treatment decisions such as human epidermal growth factor receptor 2 (HER2) amplification and mismatch repair deficiency/microsatellite instability (dMMR/MSI-H) which are being tested in clinical trials across gastric cancer.45–48 Patients with dMMR are more likely to express PD-L1 which may impact the efficacy of anti-PD1 treatments and is found; therefore, this biomarker can work synergistically with PD-L1 expression in further clinical trials of EC. 49 A study looked at the impact of tumor mutational burden (TMB), which may serve as a potential biomarker, across 62 patients with EC and those patients with higher TMB had significant improvement in survival. 50 Other studies have also shown improved outcomes with higher TMB among EC patients who received immunotherapy. 51 However, the implications of these biomarkers on the selection of treatment modalities and outcomes are still in the early stages. Overall, while many biomarkers including PD-1 have been selected as a metric for patients’ treatment eligibility, their effectiveness in better identifying the patient population with the highest odds to respond to PD-1 blockers, and other immunotherapy treatments must be explored with upcoming randomized clinical trials.52,53 It may be of contention to continue optimizing current biomarkers as well as explore new biomarkers in the context of EC with future research efforts.
Strengths and limitations
A challenge was observed which was the lack of homogeneous use of PD-L1 assays. The ESCORT trial used the Tumor Proportion Score (TPS) assay (6E8 antibody, Shuwen Biotech), whereas the CheckMate-648 trial used a different one, the Dako PD-L1 IHC 28-8 pharmDx assay. 26 The KEYNOTE-181 and ORIENT-15 trials used the PD-L1 IHC 22C3 pharmDx assay. 37 This variance in the assessment methods rendered the comparability of PD-L1 status across trials challenging and warrants careful consideration. This renders the comparability of PD-L1 status across trials challenging. The use of the Dako assay in categorizing EC is scarce. In addition, the Keynote 590 trial did not report ORR outcomes for subgroups, thereby limiting the analytical outcomes of this meta-analysis.
Moreover, as there was been an increase in the use of PD-L1 expression as a biomarker, it is worth considering the comparability of the TPS and CPS. Both scores have been used across the data in our study with a lack of clarity on agreement among both methods. In TPS, PD-L1 expression in tumor cells is evaluated by calculating the ratio of PD-L1-positive cells to the total number of viable tumor cells. In CPS, the number of PD-L1 staining cells including tumor cells, lymphocytes, and macrophages are noted relative to all viable tumor cells. However, TPS is preferred in routine clinical practice and has a high correlation with treatment response to anti-PD-1 therapy. It may be prudent to evaluate the inter-observer agreement between TPS and CPS before using them inter-changeably, which was similarly conducted in non-small-cell lung cancer (NSCLC). 54 Furthermore, there were no clear cutoffs used for either TPS or CPS across all trials for the level of PD-L1 expression which makes the interpretation of findings challenging.
At a broader level, the representation of different ethnical/racial groups is present but a higher representation of Chinese populations or Caucasians residing in HICs was noted across the trials. Therefore, this renders the findings of emerging data not entirely generalizable. Nevertheless, our strengths lie in robust data selection processes and inclusion into the study as well as a quantitative analysis found in our meta-analytical findings. Our study is the largest to the best of our knowledge that looks at all three endpoints including OS, PFS, and ORRs which are the most important primary endpoints in immunotherapy trial settings. Finally, we included all trials regardless of treatment-naïve/pre-treated or PD-L1+/PD-L1− status.
Conclusion
Our findings collate and synthesize strong evidence of PD-1/PD-L1 inhibitors when combined with chemotherapy, improving survival rates and ORR compared to chemotherapy in patients with advanced EC albeit with no effect on PFS. Our analysis offers insight into the outcomes of immunotherapeutic agents in combination with or without chemotherapeutic agents in EC. Additional trials are still required to further strengthen our findings and to explore alternative immunotherapy-based efficacious combinations with standard care interventions such as other ICIs. Finally, more sensitive biomarkers than PD-L1 expression allow for better patient identification of who is most likely to respond to immunotherapy-based regimens.
Supplemental Material
sj-docx-1-tam-10.1177_17588359231221339 – Supplemental material for Current evidence of PD-1 and PD-L1 immune checkpoint inhibitors for esophageal cancer: an updated meta-analysis and synthesis of ongoing clinical trials
Supplemental material, sj-docx-1-tam-10.1177_17588359231221339 for Current evidence of PD-1 and PD-L1 immune checkpoint inhibitors for esophageal cancer: an updated meta-analysis and synthesis of ongoing clinical trials by Saram Zafar, Rabbia Shehzadi, Hina Dawood, Moeez Maqbool, Azza Sarfraz and Zouina Sarfraz in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359231221339 – Supplemental material for Current evidence of PD-1 and PD-L1 immune checkpoint inhibitors for esophageal cancer: an updated meta-analysis and synthesis of ongoing clinical trials
Supplemental material, sj-docx-2-tam-10.1177_17588359231221339 for Current evidence of PD-1 and PD-L1 immune checkpoint inhibitors for esophageal cancer: an updated meta-analysis and synthesis of ongoing clinical trials by Saram Zafar, Rabbia Shehzadi, Hina Dawood, Moeez Maqbool, Azza Sarfraz and Zouina Sarfraz in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors wish to acknowledge the following for their contributions to the early drafting of the study: Syed Umair Shah Sherazi, MBBS; Amna Noor, MBBS; Muhammad Salahuddin, MBBS.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
