Abstract
Background:
Immune checkpoint inhibitors (ICIs) have transformed the treatment landscape of advanced or unresectable hepatocellular carcinoma (HCC). However, the comparative clinical efficacy and cost-effectiveness of various ICI-based combination therapies remain unclear.
Objectives:
This study aimed to evaluate the cost-effectiveness and clinical efficacy of the 12 first-line ICI-based therapies for advanced or unresectable HCC under two distinct healthcare systems—China and the United States.
Design:
A model-based pharmacoeconomic analysis.
Methods:
A network meta-analysis (NMA) was conducted to compare first-line ICI-based therapies in terms of overall survival (OS) and progression-free survival (PFS). A partitioned survival model with three health states (progression-free, progressive disease, death) was developed for cost-effectiveness analysis. Quality-adjusted life years (QALYs) and incremental cost-effectiveness ratio (ICER) were primary outcomes. One-way and probabilistic sensitivity analyses were performed to assess model uncertainty.
Results:
The NMA included 11 randomized clinical trials comprising 7289 patients. Four ICI-based regimens (atezolizumab plus bevacizumab, camrelizumab plus rivoceranib, sintilimab plus bevacizumab biosimilar, and penpulimab plus anlotinib) were associated with significantly improved OS and PFS compared with sorafenib. In China, camrelizumab plus rivoceranib and tislelizumab were cost-effective compared with sorafenib, with ICERs of $17,624.64 and $1971.14 per QALY, respectively. In the United States, no ICI-based therapy was cost-effective at a willingness-to-pay threshold of $150,000 per QALY; sorafenib remained the most cost-effective option. ICERs were most sensitive to drug costs, utility values, and discount rates.
Conclusion:
Camrelizumab plus rivoceranib represents a cost-effective first-line therapy for advanced or unresectable HCC in China. In the United States, current ICI-based therapies are not cost-effective at existing prices. These findings may inform treatment selection and health policy decision-making in different healthcare systems.
Keywords
Introduction
Hepatocellular carcinoma (HCC), the predominant histological subtype of primary liver cancer, is one of the most prevalent and deadliest cancers. 1 Although diagnosis at early stages can be cured by resection or liver transplantation, the majority of patients are diagnosed at an advanced stage and have a poor prognosis. 2 Oral multitargeted tyrosine kinase inhibitors were the standard first-line systemic treatments for advanced HCC for many years.3,4 Over the past several years, immune checkpoint inhibitors (ICIs) targeting the PD-1/PD-L1 or CTLA-4 pathway have revolutionized the management of HCC. ICIs combined with anti-angiogenic medications are a viable first-line therapy for advanced or unresectable HCC.5,6
In early 2020, the IMbrave-150 trial demonstrated that atezolizumab plus bevacizumab significantly improved overall survival (OS) and progression-free survival (PFS) compared to sorafenib. 5 Several other ICI-based regimens have also shown notable OS and PFS benefits, including sintilimab plus bevacizumab biosimilar (ORIENT-32), 6 tremelimumab plus durvalumab (HIMALAYA), 7 camrelizumab plus rivoceranib (CARES-310), 8 toripalimab plus bevacizumab (HEPATORCH), 9 nivolumab plus ipilimumab (CheckMate-9DW), 10 and penpulimab plus anlotinib (APOLLO). 11 These combinations represent promising first-line options for advanced or unresectable HCC. In addition, nivolumab, 12 tislelizumab, 13 pembrolizumab plus lenvatinib, 14 and atezolizumab plus cabozantinib 15 were not inferior to sorafenib or lenvatinib in terms of clinical activity and safety profile.
Currently, FDA-approved first-line therapies for advanced or unresectable HCC include atezolizumab plus bevacizumab, nivolumab plus ipilimumab, and tremelimumab plus durvalumab. In contrast, China’s NMPA has additionally approved sintilimab plus bevacizumab biosimilar and camrelizumab plus rivoceranib. Despite the growing number of treatment options, selecting an immunotherapy regimen with demonstrated efficacy and cost-effectiveness remains challenging due to regional approval differences, insurance constraints, and high drug costs. Accordingly, a systematic comparison of available regimens is essential to inform optimal treatment decisions under current access limitations.
To date, no study has comprehensively compared the efficacy and cost-effectiveness of all approved immunotherapy regimens for HCC. Prior research suggests that most approved immunotherapies offer limited economic value for HCC. Therefore, this study conducts a network meta-analysis (NMA) and cost-effectiveness analysis (CEA) to evaluate and compare the clinical and economic outcomes of ICI-based regimens for advanced or unresectable HCC, providing evidence-based guidance for clinical decision-making.
Methods
Network meta-analysis
Search strategy and selection criteria
This systematic review and NMA aimed to compare all published first-line immunotherapy approaches for advanced or unresectable HCC. This study was reported following the PRISMA statement (Table S1 in Supplemental File 1). 16 A comprehensive literature search was performed in Web of Science, Embase, PubMed, and Cochrane Library from their respective inception dates to May 30, 2025, restricting the search to English-language. The detailed search strategy is reported in Table S2 in Supplemental File 1. The search flow chart is shown in Figure S1 in Supplemental File 1. Finally, we manually screened and verified relevant literature to avoid missing articles.
Study selection and assessment of bias risks
We extracted the information from the identified articles (Supplemental Material, Supplemental File 2). We conducted ROB 2.0 and the Robvis package in R (version 4.2.1) to assess risk of bias for enrolled trials.17,18
Statistical analysis
The direct and indirect comparisons were performed with the Netmeta package in R. Due to the sparse star-shaped network structure and the lack of replicate comparisons between treatments, between-study heterogeneity (τ2) could not be reliably estimated. Therefore, a fixed-effects model was adopted as the primary analytic approach. To assess the robustness of results, both fixed- and random-effects models were fitted. Model fit was evaluated using the deviance information criterion, and the model with superior fit was selected for the primary analysis. p-Scores, ranging from 0 to 1, were calculated under the fixed-effects assumption to rank treatments based on their relative efficacy.
Cost-effectiveness analysis
Analytical overview
The hypothetical target population for this analysis comprised patients with advanced or unresectable HCC who had not previously received systemic therapy, consistent with the characteristics of patients enrolled in the trials. A partitioned survival model consisting of PFS, progression of disease (PD), and death was developed to evaluate the health and economic outcomes of immunotherapy regimens compared with sorafenib (see Figure 1). The cycle length was set at 21 or 28 days, as reported in the original research. The time horizon was set at 41 years, corresponding to the point at which 99% of the patients in all cohorts died. This study followed the Consolidated Health Economic Evaluation Reporting Standards reporting guideline (Table S3 in Supplemental File 1). 19
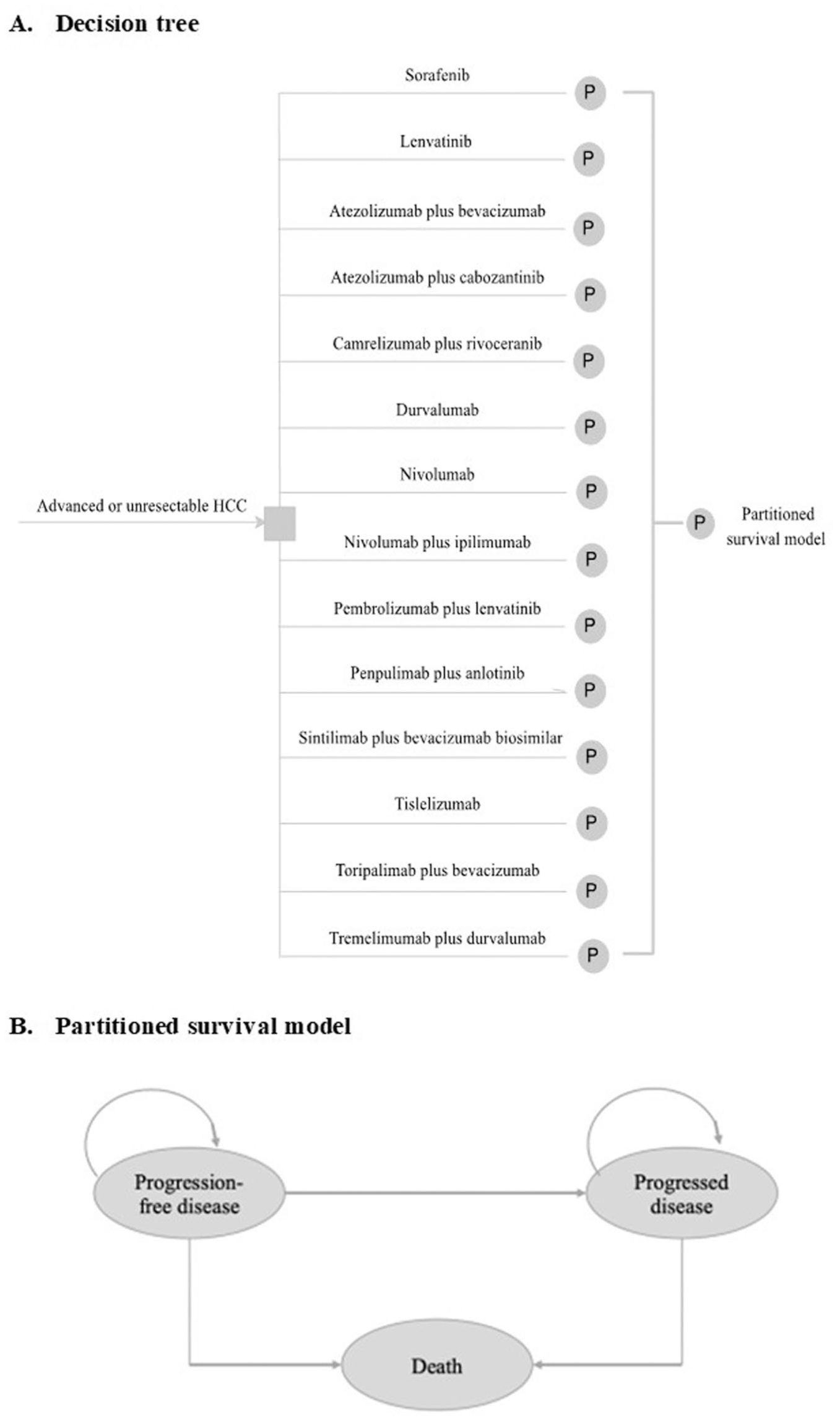
Model structure of a decision tree combining the partitioned survival model with the three health states. (a) Decision Tree. (b) Partitioned survival model.
Clinical data inputs
The probability of patients being alive with PFS was estimated from the PFS curves reported in the enrolled trials. Data points from the PFS and OS curves were extracted using GetData Graph Digitizer (version 2.26; developed by S. Fedorov) and used to reproduce the digitized Kaplan-Meier (KM) curves. The final survival distribution for all regimens is shown in Table S4 in Supplemental File 1.
Cost and utility inputs
Only direct medical costs were evaluated in this study (Table S5 in Supplemental File 1). The dosage and treatment cycle of the therapy regimen are shown in Table S6 in Supplemental File 1. The drug prices in China and the United States were collected from the bid-winning prices and published literature, respectively. To calculate the dosage of subsequent therapy, it was assumed that the patient had a body surface area of 1.72 m2 and a weight of 65 kg. The portion and costs for each subsequent treatment were shown in Table S7 in Supplemental File 1. The disutility value associated with grade 3 or higher AEs was considered in this study. 20 All adverse events (AEs) were assumed to be incurred during the first cycle.
Base-case analysis
The incremental cost-effectiveness ratio (ICER) was calculated as the incremental cost per additional quality-adjusted life-year (QALY) gained. It was assumed to be cost-effective when the ICER was lower than the prespecified willingness-to-pay (WTP) threshold. The WTP threshold was set at $150,000, and 3-times GDP per capita ($35,363.69) per QALY gained in the United States and China, respectively. Costs and QALYs were discounted at an annual rate of 5%. 21
Sensitivity analyses
One-way and probabilistic sensitivity analyses were performed to assess the robustness of the base-case results and to address the uncertainty in estimates of variables. A probabilistic sensitivity analysis (PSA) was conducted with 1000 iterations by Monte Carlo simulations, which simultaneously sampled from the distributions of all parameters. Survival curves were reconstructed and extrapolated with the survHE package in R, 22 and the partitioned survival model was developed in Microsoft Excel 2019.
Results
Network meta-analysis
The search identified 1266 citations, yielding 269 potentially relevant full-text articles (Figure S1 in Supplemental File 1). Eleven double-blind or open-label RCTs involving 7289 patients conducted between 2020 and 2025 were included. The evidence network comprised 13 treatment regimens primarily connected via sorafenib (Figure 2), forming 14 nodes and 13 direct pairwise comparisons in a predominantly star-shaped structure without closed loops. Most active immunotherapy regimens lacked direct head-to-head trials. The network was sufficiently connected with no isolated nodes.

Network meta-analysis of eligible comparisons for clinical efficacy. Each node represents a treatment regimen, with the node size proportional to the total number of randomized patients receiving that treatment. Lines represent direct head-to-head comparisons; line thickness corresponds to the number of trials (all = 1 in this network). The network exhibits a predominantly star-shaped structure with sorafenib serving as the common comparator.
Local consistency was evaluated using node-splitting analysis, revealing no significant inconsistency across pairwise comparisons (all p > 0.05), indicating agreement between direct and indirect evidence. However, global inconsistency assessment via the design-by-treatment interaction model was not feasible due to the network’s star-shaped structure and limited study design diversity.
Figure 3 shows the NMA’s results for the primary outcomes. In terms of OS, seven immunotherapy regimens prolonged OS more than sorafenib, with hazard ratios (HRs) ranging between 0.78 (95% confidence interval (CI): 0.65–0.93) for tremelimumab plus durvalumab and 0.42 (95% CI: 0.24–0.73) for nivolumab plus ipilimumab. Besides, penpulimab plus anlotinib, camrelizumab plus rivoceranib, atezolizumab plus bevacizumab, sintilimab plus bevacizumab biosimilar, and pembrolizumab plus lenvatinib had a significant statistical improvement over sorafenib in OS (Figure 3(a)). In terms of PFS, atezolizumab plus cabozantinib (HR: 0.74, 95% CI: 0.56–0.97), toripalimab plus bevacizumab (HR: 0.69, 95% CI: 0.53–0.91), atezolizumab plus bevacizumab (HR: 0.59, 95% CI: 0.46–0.75), sintilimab plus bevacizumab biosimilar (HR: 0.56, 95% CI: 0.45–0.69), camrelizumab plus rivoceranib (HR: 0.52, 95% CI: 0.41–0.65), and penpulimab plus anlotinib (HR: 0.52, 95% CI: 0.41–0.66) had significant statistical improvement than sorafenib (Figure 3(b)). To evaluate the differences between immunotherapy, we also synthesized head-to-head studies independently (Figure 3(c)). When all trials were considered, atezolizumab plus bevacizumab, camrelizumab plus rivoceranib, sintilimab plus bevacizumab biosimilar (IBI305), and penpulimab plus anlotinib seemed associated with greater survival benefits than sorafenib or other immunotherapy regimens.

Forest plots and head-to-head comparisons for overall and progression-free survivals of the first-line ICI-based therapies for HCC. (a) Forest plots for overall survival. (b) Forest plots for progression-free survival. (c) Head-to-head comparisons of the first-line ICI-based therapies for HCC.
In recent years, RCTs have increasingly tended to adopt open-label design methods rather than double-blind, which may lead to high-risk deviations from the intended intervention. Overall, 4 (36.4%) of 11 trials were rated as high risk of bias, 6 (54.5%) trials as some concerns, and 1 (9.0%) as low (Figure S2 in Supplemental File 1).
Cost-effectiveness analysis
Base-case analysis
In the base-case analysis, most immunotherapy regimens, except durvalumab monotherapy, increased life years for advanced HCC patients. From the Chinese perspective, tislelizumab and camrelizumab plus rivoceranib provided 0.19 and 0.12 additional QALYs compared to sorafenib, with ICERs of $1,971.14 and $17,624.64 per QALY, respectively (Table 1). Penpulimab plus anlotinib, toripalimab plus bevacizumab, nivolumab, and nivolumab plus ipilimumab gained 0.11, 0.37, 0.26, and 0.13 QALYs, with ICERs of $59,837.97, $155,722.08, $175,562.55, and $248,958.35, respectively. Atezolizumab plus bevacizumab, sintilimab plus bevacizumab biosimilar (IBI305), atezolizumab plus cabozantinib, and tremelimumab plus durvalumab were proven to have higher ICERs. Compared to lenvatinib, pembrolizumab plus lenvatinib, and nivolumab plus ipilimumab provided 0.60 and 0.36 additional QALYs, with ICERs of $221,482.93 and $152,471.54, respectively.
Summary of cost and outcome results in the base-case analysis.

The survival parameters of sorafenib were based on the survival data reported in all trials.
Anlotinib has not been available in the US, we assume that its cost was the same as cabozantinib in one cycle.
The survival parameters of lenvatinib were referenced from the LEAP-002 trial and CheckMate 9DW trial.
In CheckMate 9DW trial, lenvatinib was one of the control groups; the other control group was sorafenib. We compared the cost-effectiveness of nivolumab plus ipilimumab with lenvatinib and sorafenib, respectively, based on published survival data.
In LEAP-002 trial, pembrolizumab plus lenvatinib was the treatment arm, and lenvatinib was the control group.
ICER, incremental cost-effectiveness ratio; IncrC, incremental cost; IncrE, incremental effectiveness; NA, not applicable; QALY, Quality-adjusted life years.
Similarly, as shown in Table 1, from the perspective of the US health care payers, tislelizumab had the lowest ICER of $239,928.72/QALY, yielding an additional 0.19 QALYs. Nivolumab, nivolumab plus ipilimumab, atezolizumab plus bevacizumab, toripalimab plus bevacizumab, and penpulimab plus anlotinib gained 0.26, 0.13, 0.38, 0.37, and 0.11 QALYs, with ICERs of $276,988.18, $448,534.66, $797,378.34, $853,845.43, and $917,941.34 per QALY gained, respectively. Camrelizumab, sintilimab, and bevacizumab biosimilars are not commercially available in the US. Tremelimumab plus durvalumab and atezolizumab plus cabozantinib rank as the two most expensive options, with respective ICERs of $8,609,554.3 and $2,269,129.73. Compared to lenvatinib, pembrolizumab plus lenvatinib gained an ICER of $458,033.91, which was higher than from the Chinese perspective. Nivolumab plus ipilimumab provided an additional 0.36 QALYs than lenvatinib, resulting in an ICER of $257,169.88.
Sensitivity analysis
At a WTP threshold of $35,363.69 per QALY, the cost-effectiveness acceptability curves indicated that only tislelizumab and camrelizumab plus rivoceranib had a higher probability of cost-effectiveness than sorafenib from the perspective of the Chinese healthcare system (Figure 4(a)). The probability sensitivity analysis identified that tislelizumab monotherapy and camrelizumab plus rivoceranib were most likely to be cost-effective when compared with sorafenib (Figure 4(b)). Other immunotherapy options are not cost-effective under the current price system. Considering the clinical efficacy analyzed in NMA, camrelizumab plus rivoceranib currently seems to be the best choice for the first-line treatment of advanced or unresectable HCC patients in China. The majority of the ICERs in the sensitivity analysis fell into the first and fourth quadrants and were below the three times GDP per capita WTP threshold in China. This indicated a relatively high chance of being cost-effective and supported the robust results of the PSA. As shown in Figure 4(c), from the perspective of the US health care payers, none of the immunotherapy regimens demonstrated a higher probability of cost-effectiveness than sorafenib, either at WTP thresholds of $150,000 or $200,000 per QALY (Figure 4(c) and (d)). When the WTP thresholds were over $250,000 per QALY, nivolumab plus ipilimumab would be more cost-effective than sorafenib.

Cost-effectiveness acceptability curves and cost-effectiveness plan for the first-line ICI-based therapies for advanced or unresectable HCC. (a) Cost-effectiveness acceptability curves from the perspective of the Chinese healthcare system. (b) Cost-effectiveness plane from the perspective of the Chinese healthcare system. (c) Cost-effectiveness acceptability curves from the perspective of the United States healthcare system. (d) Cost-effectiveness plane from the perspective of the United States healthcare system.
The one-way sensitivity analyses revealed that the ICERs of immunotherapy were mostly sensitive to the cost of drugs, utility of PFS or PD, and discount rate (Figure S3 in Supplemental File 1). Not only the price of ICIs, but also the price of sorafenib or antiangiogenic agents greatly affects the cost-effectiveness of treatment. This factor is consistent between China and the United States.
Also, we generated a base-case analysis, PSA, and cost-effectiveness acceptability curves under scenarios where the costs of each drug were reduced to 25%, 50%, 75%, and 90% of current levels in both the US and China. The results, presented in Figure S4 (in Supplemental File 3), showed how cost reductions influence the probability of each regimen being cost-effective across a range of WTP thresholds.
Discussion
In this study, a comprehensive NMA and CEA were conducted to evaluate the cost and efficacy of the 12 first-line ICI-based therapies for advanced or unresectable HCC. The objective was to provide additional information regarding the clinical efficacy and cost-effectiveness of treatment options. The latest evidence was synthesized, and the most recent clinical trials of first-line immunotherapies were enrolled, including COSMIC-312, CARES-310, HEPATORCH, CheckMate 9DW, and APOLLO.8,10,11,15,23 These RCTs provided new immunotherapy options that had not been evaluated by previous studies. On the other hand, the survival curves of all studies were reconstructed to fit the optimal distribution, avoiding the assumptions used in previous studies.24 –26 In recent years, frequent price adjustments of ICIs have introduced considerable variability in cost-effectiveness outcomes, underscoring the need for timely research. This study responds to the current gap in clinical and economic evaluation of first-line immunotherapy for advanced or unresectable HCC. In our analysis, sorafenib was used as a comparator because it served as the control arm in all pivotal first-line RCTs, ensuring consistency with trial evidence. To improve clinical relevance, we also assessed cost-effectiveness against lenvatinib for regimens supported by direct comparative data.
The present study indicated that, in comparison with sorafenib, four kinds of ICI-based therapy regimens have significantly improved OS and PFS, namely penpulimab plus anlotinib, camrelizumab plus rivoceranib, atezolizumab plus bevacizumab, and sintilimab plus bevacizumab biosimilar. However, it was only the combination of camrelizumab and rivoceranib that was found to yield comparatively superior health benefits while maintaining cost-effectiveness from the perspective of the Chinese healthcare system. From the US perspective, atezolizumab plus bevacizumab was the only immunotherapy that was superior to sorafenib in prolonging OS and PFS. Nevertheless, the regimen was not cost-effective at a WTP threshold of $200,000 per QALY. Sorafenib continues to be a cost-effective treatment option as the first-line therapy for the American population. It was determined that when the WTP threshold exceeded $250,000 per QALY, nivolumab plus ipilimumab would be deemed cost-effective in comparison to sorafenib. The cost of acquiring drugs (ICIs or sorafenib), utility of PFS or PD, and discount rate were found to be the most significant factors in determining whether an immunotherapy regimen is cost-effective.
As stated in previous studies, NMA provides substantial evidence supporting the efficacy of immunotherapy as a first-line treatment for advanced HCC.24 –26 In summary, the combination of sintilimab and bevacizumab biosimilar, camrelizumab and rivoceranib, and atezolizumab and bevacizumab has demonstrated superior effectiveness in prolonging OS and PFS. These findings were consistent with conclusions previously reported in our study. However, a paucity of studies has thus far precluded the establishment of a consensus on the economic evaluation of these measures.24,25 In early studies, sintilimab plus IBI305 and atezolizumab plus bevacizumab were the two most commonly used treatment options for comparison. Li et al. 24 considered that, only under the patient assistance program (PAP), sintilimab plus bevacizumab biosimilar is cost-effective as the first-line therapy for unresectable HCC in China at a $33,500 WTP threshold. Nevertheless, whether or not atezolizumab PAP is taken into account, the atezolizumab plus bevacizumab combination is not cost-effective. Wang et al. 27 found that sintilimab plus IBI305 is more cost-effective than atezolizumab plus bevacizumab in China, without mentioning WTP. Similarly, Gong et al. 25 found that lenvatinib, sintilimab plus bevacizumab biosimilar, and atezolizumab plus bevacizumab were not cost-effective at a WTP of $36,600. However, given that only four treatment options were compared in these studies—namely, the combination of sintilimab and bevacizumab biosimilar, atezolizumab plus bevacizumab, lenvatinib, and sorafenib—these conclusions still require further exploration and refinement. 25
Subsequent studies incorporated a more extensive array of treatment modalities, including oral multi-kinase inhibitors and immunotherapy. Wang et al. 27 examined the cost-effectiveness of eight initial treatment plans for unresectable HCC, though they did not compare the clinical effects of these treatments. Liu et al. 26 conducted an NMA and CEA to compare 16 immunotherapy or targeted therapy options as initial strategies for unresectable HCC. This systematic review represents the most comprehensive evaluation to date of first-line treatments for HCC. However, its early publication limited access to detailed data and survival curves from several RCTs, particularly those involving ICIs. Notably, the authors reconstructed only the survival curve for sorafenib and applied the fitted Weibull model parameters to all treatment groups. This uniform application introduces a critical assumption that likely compromises the accuracy of effectiveness estimates and QALY calculations.
Tislelizumab is the most economical first-line treatment option for unresectable HCC in China in the study conducted by Liu et al. 26 However, owing to the lack of a clinical efficacy advantage of tislelizumab, the acceptability curve proved that for different WTP levels, the most cost-effective methods are likewise varied. Tislelizumab, for instance, is a better option when it falls below China’s WTP threshold of $37,653/QALY. When the WTP value is between $37,653 and $80,000/QALY, camrelizumab plus rivoceranib is the better choice. When WTP is larger than $80,000/QALY, sintilimab plus a bevacizumab biosimilar is a superior choice.
In our study, survival curves for all treatment groups were individually reconstructed and fitted based on original RCT data. We identified Lognormal or Loglogistic, or Weibull distributions as the best fit for PFS or OS (Table S5 in Supplemental File 1). In addition, unlike the prior study that assumed uniform use of regorafenib as second-line therapy per NCCN and CSCO guidelines, published RCTs indicate that tyrosine kinase inhibitors—most commonly sorafenib—were more frequently used, followed by locoregional therapies. These differing assumptions on subsequent treatment significantly impact cost estimations and, consequently, cost-effectiveness outcomes. Such methodological limitations reduce the accuracy and reliability of the previous study’s QALY and ICER estimates, contributing to discrepancies with our findings.
The relatively low price of ICIs enhances the cost-effectiveness of immunotherapy. In China, pharmacoeconomic evidence is used to support national insurance negotiations with pharmaceutical companies. According to the 2024 national negotiation results, the average price reduction for listed drugs was 63%. For instance, the price of camrelizumab decreased from $2723.52/200 to $402.75/200 mg after its inclusion in the National Medical Insurance in March 2021, representing an 85.2% reduction. In contrast, the high cost of pharmaceuticals in the United States remains a major concern. The rising expenses of innovative cancer therapies are driven by factors such as the high cost of drug development and clinical trials, complex manufacturing processes, and industry pricing strategies.28,29 By 2030, national cancer care costs are projected to reach $245 billion. 28 This pricing disparity largely stems from the fragmented US healthcare system, which lacks centralized price negotiations. The high cost of ICIs places a substantial financial burden on both patients and the healthcare system. Medicare reimbursement for the same drug varies across regions and plans, further exacerbating cost issues. These factors collectively limit access to immunotherapy for HCC. To address this, policy measures such as stronger drug price regulation, improved transparency in pricing, standardized Medicare reimbursement, and promotion of generic and biosimilar competition are essential. A comprehensive approach is needed to ensure equitable access to advanced immunotherapy for HCC patients.
In this study, we conducted country-specific CEAs for the United States and China. The aim was not to compare cost-effectiveness across the two countries, but rather to evaluate the relative value of first-line immunotherapy regimens within each healthcare system. This approach aligns with international pharmacoeconomic guidelines, such as ISPOR, NICE, and WHO-CHOICE guidelines, which recommend that CEAs incorporate local drug prices, treatment patterns, and WTP thresholds, given the substantial differences across jurisdictions. The marked variation in pricing structures between the US and China further underscores the need for separate national evaluations, as payers depend on evidence generated under their own cost environment. Therefore, the two models are presented as independent analyses designed to inform decision-making in their respective settings, rather than as a direct economic comparison between countries.
Limitation
This study has several limitations. First, the NMA predominantly included comparisons versus sorafenib, resulting in a star-shaped network with few closed loops, thereby limiting both global and local inconsistency assessments. The absence of direct head-to-head trials between active regimens restricted formal evaluation of consistency assumptions. Second, model inputs for clinical efficacy and safety were derived from clinical trials, with long-term outcomes extrapolated using parametric functions, introducing potential bias relative to real-world scenarios. Third, due to the lack of direct comparisons, economic evaluations between first-line ICI-based regimens could not be conducted. Sorafenib, used as the comparator in this study, was no longer a preferred first-line therapy for advanced HCC, which may limit the direct applicability to current clinical practice. Despite these limitations, the study provides a robust estimation of the clinical and economic value of immunotherapy regimens for advanced or unresectable HCC in China and the United States.
Conclusion
Penpulimab plus anlotinib, atezolizumab plus bevacizumab, camrelizumab plus rivoceranib, and sintilimab plus bevacizumab biosimilar have all demonstrated significant improvements in OS and PFS compared to sorafenib as first-line treatments for advanced or unresectable HCC. Among these, camrelizumab plus rivoceranib appears to be a cost-effective and clinically beneficial option in the Chinese setting. However, under current pricing, none of the ICI-based regimens are cost-effective within the US healthcare system, where sorafenib remains the most cost-effective option.
Supplemental Material
sj-docx-1-tam-10.1177_17588359261417630 – Supplemental material for Immune checkpoint inhibitor-based first-line therapies for advanced or unresectable hepatocellular carcinoma: a network meta-analysis and cost-effectiveness analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359261417630 for Immune checkpoint inhibitor-based first-line therapies for advanced or unresectable hepatocellular carcinoma: a network meta-analysis and cost-effectiveness analysis by Huijie Qi, Yuxin Huang, Wenxin Zhang, Jiyifan Li, Tianxiao Wang, Yinqing Ying, Bicui Chen, Feng Miao and Qunyi Li in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359261417630 – Supplemental material for Immune checkpoint inhibitor-based first-line therapies for advanced or unresectable hepatocellular carcinoma: a network meta-analysis and cost-effectiveness analysis
Supplemental material, sj-docx-2-tam-10.1177_17588359261417630 for Immune checkpoint inhibitor-based first-line therapies for advanced or unresectable hepatocellular carcinoma: a network meta-analysis and cost-effectiveness analysis by Huijie Qi, Yuxin Huang, Wenxin Zhang, Jiyifan Li, Tianxiao Wang, Yinqing Ying, Bicui Chen, Feng Miao and Qunyi Li in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-3-tam-10.1177_17588359261417630 – Supplemental material for Immune checkpoint inhibitor-based first-line therapies for advanced or unresectable hepatocellular carcinoma: a network meta-analysis and cost-effectiveness analysis
Supplemental material, sj-pdf-3-tam-10.1177_17588359261417630 for Immune checkpoint inhibitor-based first-line therapies for advanced or unresectable hepatocellular carcinoma: a network meta-analysis and cost-effectiveness analysis by Huijie Qi, Yuxin Huang, Wenxin Zhang, Jiyifan Li, Tianxiao Wang, Yinqing Ying, Bicui Chen, Feng Miao and Qunyi Li in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
