Abstract
Introduction:
The combination of immune checkpoint inhibitors (ICIs) and chemotherapy is a promising first-line therapy for patients with advanced non-squamous non-small cell lung cancer (NSCLC). The cost-effectiveness of combinations with different ICIs is yet to be compared.
Methods:
We utilized Bayesian network meta-analyses for the comparisons of overall survival, progression-free survival, and incidence of adverse events of the included treatments in the total population and subgroups with different programmed death-ligand 1 tumor proportional scores (TPS). The cost-effectiveness of the treatments from the perspectives of the US and Chinese healthcare systems was assessed using Markov models.
Results:
Three combinations, including pembrolizumab + chemotherapy (PembroC), nivolumab + ipilimumab + chemotherapy (NivoIpiC), and atezolizumab + chemotherapy (AteC), were included in our study. In terms of efficacy, PembroC was most likely to be ranked first for extending progression-free survival (PFS) (93.16%) and overall survival (OS) (90.73%). Nevertheless, from the US perspective, NivoIpiC and PembroC showed incremental cost-effectiveness ratios (ICERs) of $68,963.1/quality-adjusted life-years (QALY) and $179,355.6/QALY, respectively, compared with AteC. The one-way sensitivity analysis revealed that the results were primarily sensitive to the hazard ratios for OS or the cost of immunotherapy agents. At a willingness-to-pay (WTP) threshold of $150,000/QALY, NivoIpiC had the highest probability of being cost-effective (63%). As for the Chinese perspective, NivoIpiC and PembroC had ICERs of $145,983.4/QALY and $195,863.3/QALY versus AteC, respectively. The results were primarily sensitive to the HRs for OS. At a WTP threshold of $38,017/QALY, AteC had the highest probability of cost-effectiveness (94%).
Conclusion:
Although PembroC has the optimal efficacy, NivoIpiC and AteC were the most favorable treatments in terms of cost-effectiveness for patients with advanced non-squamous NSCLC from the US and Chinese perspectives, respectively.
Keywords
Introduction
The 2022 Cancer Statistics report revealed that lung cancer was the most common cause of cancer-related death, accounting for almost a quarter of all cancer deaths. 1 The disease burden of lung cancer is also high, according to the Global Burden of Disease Study in 2019, which ranks lung cancer as one of the leading causes of disability-adjusted life-years (DALYs). 2 Non-small cell lung cancer (NSCLC) is the predominant histological subtype, representing about 85% of all lung cancer cases, with non-squamous cell carcinoma accounting for over 50% of cases. 3 Due to the limited testing techniques, NSCLCs are usually diagnosed at an advanced stage, making effective first-line treatments crucial. 4
Currently, for advanced NSCLC patients with negative driver gene mutations, first-line regimens include platinum-based doublet therapy plus immune checkpoint inhibitors (ICIs) with or without bevacizumab or ICIs monotherapy for patients with a programmed death-ligand 1 (PD-L1) tumor proportional score (TPS) ⩾50%. 5 KEYNOTE 189, a phase III randomized controlled trial (RCT), paved the way for ICI plus chemotherapy (CT) as a standard first-line treatment for advanced NSCLCs without sensitizing targetable mutations. In this trial, pembrolizumab plus CT led to improvements in overall survival (OS) of non-squamous NSCLCs, especially in those who have PD-L1 TPS ⩾50% (27.7 versus 10.1 months). 6 Similarly, IMpower 132, a randomized phase III trial showed that non-squamous NSCLC patients, regardless of PD-L1 expression, can benefit from the combination of atezolizumab and CT (median OS: 17.5 versus 13.6 months). 7 Besides, the phase III CheckMate 9LA study revealed that nivolumab, a PD-1 inhibitor, plus ipilimumab, a cytotoxic T lymphocyte-associated antigen-4 inhibitor, plus CT as first-line treatments for non-squamous NSCLC cases also could prolong the OS compared with CT alone (CheckMate 9LA: 17.0 versus 11.9 months). 8
In determining the most suitable treatment for patients, factors such as efficacy, safety, and cost-effectiveness must be considered. While previous studies have assessed the cost-effectiveness of ICIs plus CT versus CT alone, there is a lack of direct comparison of ICI combination therapies.9–13 Therefore, this study aims to compare the cost-effectiveness, efficacy, and safety of different combination regimens with ICIs for untreated non-squamous NSCLCs by using a network meta-analysis (NMA) from the perspective of US healthcare payers. This NMA and cost-effectiveness analysis enables a comprehensive evaluation of the available evidence from clinical trials, which can inform healthcare decision-makers on the best treatment options.
Materials and methods
Network meta-analysis
Study selection
This NMA was finished following the Preferred Reporting Items for Systematic Review and Meta-Analyses extension statement and not registered online (Supplemental Material 2). 14 Two authors (LN and WT) individually searched the Pubmed, Cochrane Library, EMBASE, and Web of Science for RCTs from 1 January 2000 to 24 February 2022, using terms of ‘Carcinoma, Non-Small-Cell Lung’, ‘Immunotherapy’, ‘Immune Checkpoint Inhibitors’, ‘Pembrolizumab’, ‘Nivolumab’, ‘Atezolizumab’, and ‘Durvalumab’. Detailed search strategies are provided in Supplemental Table S1. Literature titles, abstracts, and full text were screened by the two authors independently, and then we combined the results and deleted duplicated results. In case of disagreement, a third researcher was required. Details about the inclusion and exclusion criteria and study selection are in the Supplemental, respectively, in Figure 1.
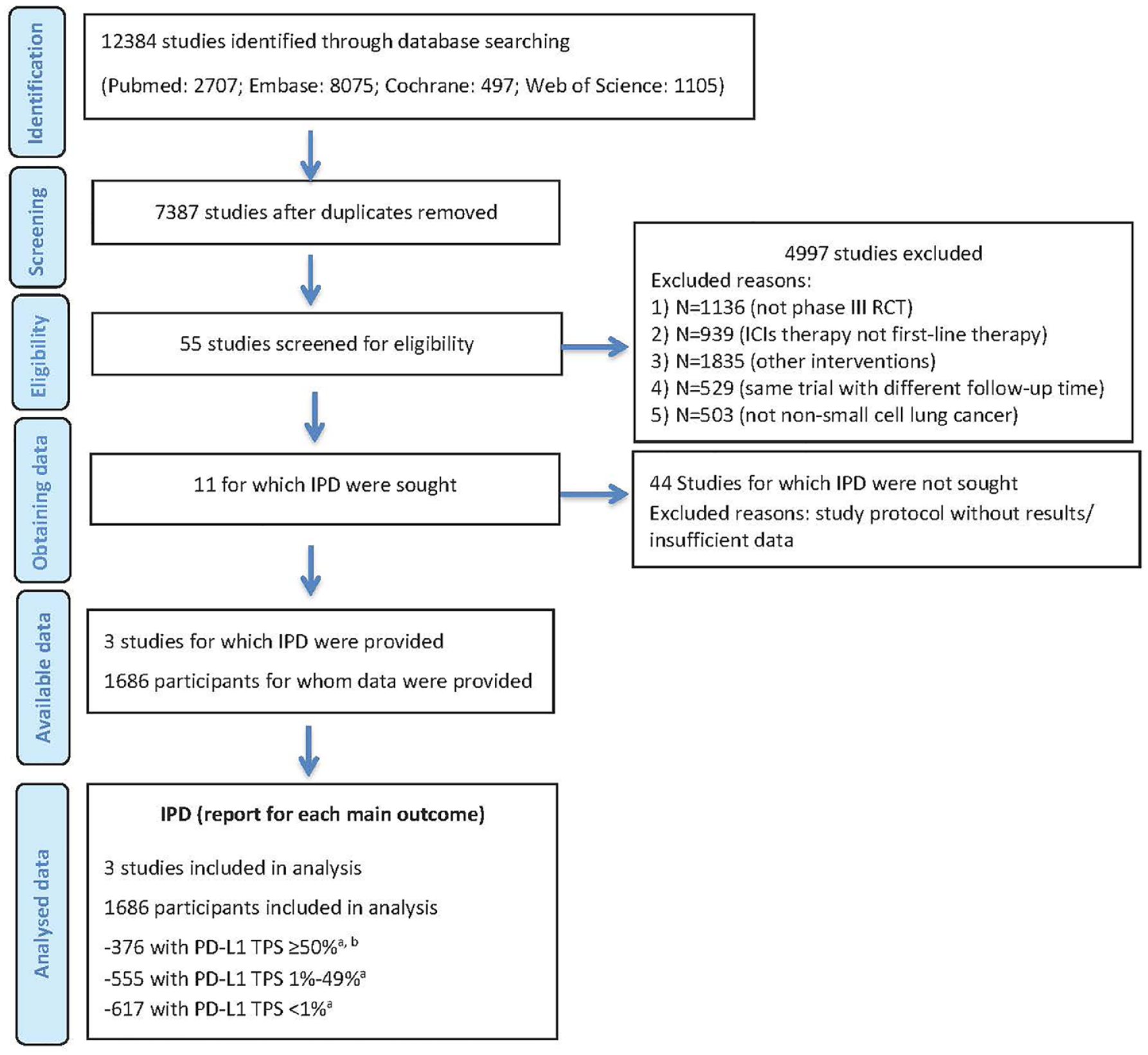
Flow diagram of study selection.
Data extraction and assessment of risk of bias
Two investigators (LN and WT) extracted data independently by browsing the full text of the studies. The following information was extracted: (1) authors and publication year; (2) median follow-up time; (3) interventions; (4) the number of patients with non-squamous NSCLC; (5) PD-L1 expression; (6) OS (the time from randomization to patient’s death of any cause); (7) progression-free survival (PFS) (the time from randomization to objective tumor progression or all-cause death); (8) the number of patients with treatment-related adverse events (TRAEs) or TRAEs of grade ⩾3 specifically. The methodological qualities (risk of bias) of included studies were assessed by the two authors independently in RevMan, version 5.4, according to the guidance provided in the Cochrane handbook.
Statistical analysis
We assessed the primary outcomes, including hazard ratios (HRs) for PFS and OS and odds ratio (OR) for AEs. Markov Chains Monte Carlo sampler was used to establish three Markov Chains that run 5000 burn-ins and 20,000 subsequent simulations to finish posterior summaries. The Brooks–Gelman–Rubin plots method was used to assess the model convergence. A fixed-effect model was used for the analysis due to the absence of data to assess the heterogeneity between trials. In order to meet the requirement of consistency, we included phase III RCTs with similar basic characteristics of patients and compared the distribution of clinical or methodological variables. The Bayesian method can provide treatment ranking probabilities by calculating surfaces under the cumulative ranking curves. Therefore, each treatment in a network can get a particular placement in ordering treatment effects from best to worst. Statistical analysis was performed using R software (version 4.0.4) with the package ‘gemtc’.
Cost-effectiveness analysis
The cost-effectiveness analysis follows the Consolidated Health Economic Evaluation Reporting Standards Value of Information guideline (Supplemental Material 3). 15
Model structure
A Markov model was needed to simulate patient-level data to compare the cost-effectiveness of different ICIs as first-line treatments for advanced non-squamous NSCLC from the perspectives of the United States and China. This model was constructed using TreeAge Pro 2021 (TreeAge Software, LLC, Williamstown, Massachusetts) with a 21-day cycle length and three mutually independent health states: PFS, progressive disease (PD), and death (Figure 2). Initially, all patients were enrolled in the PFS health state and subsequently received various first-line ICI treatments until either disease progression or the development of intolerable toxicity. Patients who experienced disease progression during the first-line treatment were transitioned to the PD health state, where they received subsequent anticancer therapies. Ultimately, patients in any of the states had the potential to directly transition to the state of death.

Framework of the decision tree and the Markov model.
In the model, the analytic time horizon was set to 15 years since 99% of the cohort died at this time point. The primary outcomes consisted of the life-years (LYs), quality-adjusted life-years (QALYs), overall costs, and incremental cost-effectiveness ratios (ICERs). We applied a half-cycle correction. A 3% discount rate per year and a 5% discount rate per year was used for both cost and health outcomes in the United States and in China, respectively. 16 The willingness-to-pay (WTP) threshold in this study was set to $150,000 per QALY and $38,017 per QALY for the US perspective and the Chinese perspective, respectively. 17
Clinical data
The Kaplan–Meier (K–M) curves, including PFS and OS, were obtained from KEYNOTE 189, CheckMate 9LA, and IMpower 132. Subgroup analysis was conducted in patients categorized by PD-L1 expression level. Survival data points were extracted from curves using GetData Graph Digitizer (version 2.26, GetData Pty Ltd., Kogarah, NSW, Australia). The individual patient data were recreated using the method of Hoyle et al. 18 For reconstructing the individual patient time-to-event data, these data points are used to fit parametric models, including exponential, Weibull, log-logistic, lognormal, genGamma, and Gompertz distributions. Subsequently, according to the values of the Akaike Information Criterion (AIC) and Bayesian Information Criterion (BIC) combined with visual inspections, the best-fit distribution models were chosen, and K–M curves were evaluated by using R software (version 4.0.4) with the package ‘survival’ and ‘flexsurv’. For some curves, the fitting curves with the lowest AIC/BIC were not selected because of tailing or PFS being higher than OS. The AIC and BIC values of each distribution are shown in Supplemental Table S2, and the comparisons between K–M and fitting curves of all non-squamous NSCLCs and patients with different levels of PD-L1 expression are shown in Supplemental Figure S5. The chosen estimated curves are displayed in Supplemental Figure S6. The HRs of PFS and OS among the three interventions were calculated using NMA and were needed to estimate the PFS and OS rates. According to the National Comprehensive Cancer Network (NCCN) guidelines, pembrolizumab plus CT is the preferred recommendation regardless of PD-L1 expression, so it was chosen as the baseline treatment. OS and PFS probabilities of the intention-to-treat (ITT), PD-L1 1–49%, and PD-L1 < 1% patients in the atezolizumab plus CT group were also calculated based on the those in the pembrolizumab plus CT group and the HRs. 19 Due to the absence of PD-L1 subgroup survival data of non-squamous patients in CheckMate 9LA, we made reasonable assumptions based on relevant data and examined them in the sensitivity analyses.
Cost and utility data
Only direct medical costs were considered, which consisted of first-line and subsequent drug costs, costs of intravenous infusion, imaging trimonthly, routine follow-up, end-of-life (6 months), best supportive care (BSC), and management of AEs (Table 1). According to the trial design, the experimental groups were given pembrolizumab 200 mg triweekly combined with CT, nivolumab 360 mg triweekly plus ipilimumab 1 mg/kg every 6 weeks plus CT, or atezolizumab 1200 mg triweekly plus CT, respectively, in KEYNOTE 189, CheckMate 9LA, and IMpower 132. After induction treatment with ICIs plus CT, maintenance therapy was given until disease progression or unacceptable toxicity. Furthermore, patients would receive subsequent treatment and the BSC until death. All details of subsequent treatment options were derived from relevant clinical trials (Supplemental Table S3).6–8 To estimate the dose of agents, we assumed a typical patient had 70 kg in weight and 1.79 m 2 in body surface area (BSA), and creatinine clearance rate is 70 ml/min.20,21 All values of costs were from the Centers for Medicare & Medicaid Services and some published articles.22–28 The utility-scale was from 0 (death) to 1 (perfect health), and different utilities represented particular health states. Our study’s survival and health state utilities were all obtained from previously published articles.29–31 We also collected some utilities related to AEs, and the details of the utilities are shown in Table 1.
Parameters input to the model.

AteT, atezolizumab + chemotherapy; BSA, body surface area; CMS, Centers for Medicare & Medicaid Services; HIGH, PD-L1 TPS ⩾ 50% population; HR, hazard ratio; ITT, intention-to-treat population; LOW, PD-L1 TPS 1–49% population; NEG, PD-L1 TPS < 1% population; ND, not determined; NivoIpiC, nivolumab + ipilimumab + chemotherapy; NMA, network meta-analysis; OS, overall survival; PembroC, pembrolizumab + chemotherapy; PFS, progression-free survival; SAE, severe adverse event.
Sensitivity analysis
The model robustness was assessed using one-way sensitivity analyses and probabilistic sensitivity analyses (PSAs). In the one-way sensitivity analyses, the ranges of different variables were either based on the 95% confidence intervals (CIs) reported in the published articles or estimated by assuming ±25% from the baseline values (Table 1). PSAs were also performed through 10,000 Monte Carlo repetitions. The probability distribution of HRs (log-normal distribution), costs (gamma distribution), proportions (beta distribution), incidence rates (beta distribution), and utility values (beta distribution) were included. The PD-L1 subgroup sensitivity analyses were also conducted.
Results
Study characteristics and the risk of bias
We identified a total of 3791 studies through database searching (Figure 1) and finally included three studies (CheckMate 9LA, Keynote-189, and Impower132) (Supplemental Table S4), involving 1686 patients with advanced non-squamous NSCLC, according to the inclusion and exclusion criteria (Figure 1), and we summarized their characteristics in Supplemental Table S2. The patients with advanced non-squamous NSCLC were treated with nivolumab + ipilimumab + chemotherapy (NivoIpiC) (n = 248), pembrolizumab + chemotherapy (PembroC) (n = 410), atezolizumab + chemotherapy (AteC) (n = 292), or CT alone (n = 738).
The risks of the study bias were summarized in Supplemental Figure S1. Even though two studies had high risks of performance bias and one study had high risks of reporting bias, selection bias, detection bias, attrition bias, and other biases remained low or unknown among the included studies.
Efficacy and safety results
The NMAs for efficacy included four treatments for PFS and OS in the overall patients with non-squamous NSCLC and three treatments for the patients with negative (TPS < 1%), low (TPS 1–49%), or high (TPS ⩾ 50%) tumor PD-L1 expression (Supplemental Figure S2). Due to a lack of data on non-squamous PD-L1 subgroups in CheckMate 9LA, nivolumab + ipilimumab + CT was excluded in the PD-L1 subgroup analyses.
As for OS [Figure 3(a)], all the immunotherapy-based treatments, except atezolizumab + CT in the patients with high PD-L1 expression (HR 0.73, 95% CI 0.31–1.72) and atezolizumab + CT in the patients with low PD-L1 expression (HR 1.18, 95% CI 0.8–1.75), achieved significant OS prolongation in the total population and across subgroups with various PD-L1 expressions. Pembrolizumab + CT was most likely to be ranked first for extending OS in the total population (90.73%), patients with high PD-L1 expression (67.29%), patient with low PD-L1 expression (97.36%), and patients with negative PD-L1 expression (85.73%) [Supplemental Figure S3(A), (C), (E), and (G)]. Pembrolizumab + CT, as for the pooled HR profiles, significantly prolonged OS in the total population (HR 0.65, 95% CI 0.49–0.87) and in patients with low PD-L1 expression (HR 0.56, 95% CI 0.33–0.96) compared with atezolizumab + CT.

The presented league table displays the pairwise comparisons of overall survival or progression-free survival, denoted as (a) and (b), respectively. The results are presented as HR along with their corresponding 95% CI. These ratios represent the treatment defined by the columns compared to the treatment defined by the rows.
As for PFS [Figure 3(b)], all the immunotherapy-based treatments, except atezolizumab + CT in the low PD-L1 expression group (HR 0.8, 95% CI 0.56–1.15; Figure 3), led to significant PFS prolongation compared with CT alone in the total population and across all the subgroups with various PD-L1 expressions. Among these immunotherapy treatments, pembrolizumab + CT was most likely to be ranked first for extending PFS in the total population (93.16%), patients with high PD-L1 expression (74.32%), and patients with low PD-L1 expression (94.94%) [Supplemental Figure S3(B), (D), and (F)]. However, atezolizumab + CT had the highest probability (94.55%) to be ranked first for extending PFS in paitents with negative PD-L1 expression. In line with the ranking profile, pembrolizumab + CT significantly prolonged PFS compared with nivolumab + ipilimumab + CT in the total population (HR 0.66, 95% 0.50–0.88).
The NMAs for safety included three treatments, excluding nivolumab + ipilimumab + CT due to a lack of the safety profile in patients with non-squamous NSCLC reported by CheckMate 9LA (Supplemental Figure S4) for all TRAEs [Supplemental Figure S4(A)] and TRAEs of grade ⩾ 3 [Supplemental Figure S4(B)]. The ranking profiles showed that CT alone was most likely to be ranked first in engendering the fewest AEs (77.41%) and AEs of grade ⩾ 3 (99.33%), while atezolizumab + CT had a high probability to be ranked first in causing the most grade ⩾ 3 AEs (77.86%). Based on the ORs, both immunotherapy-based treatments, including atezolizumab + CT (OR 1.86, 95% CI 1.33–2.6) and pembrolizumab + CT (OR 1.54, 95% CI 1.1–2.18), increased the probabilities of TRAEs of grade ⩾ 3 compared with CT alone.
Cost-effectiveness results
We summarized the base-case mean cost, LYs, QALYs, and ICERs of each treatment in Table 2. Since the median OS of patients with PD-L1 TPS ⩾50% treated with atezolizumab + CT was not reached, we did not include the data of this group in the cost-effectiveness analysis.
Baseline results.

AteT, atezolizumab + chemotherapy; ICER, incremental cost-effectiveness ratio; ITT, intention-to-treat population; LYs, life-years; NA, not applicable; NivoIpiC, nivolumab + ipilimumab + chemotherapy; PembroC, pembrolizumab + chemotherapy; QALYs, quality-adjusted life-years.
From the perspective of the US health care payers, in terms of the total population, the mean costs of atezolizumab + CT, nivolumab + ipilimumab + CT, and pembrolizumab + CT were $271,718.22, $284,176.50, and $349,088.46, respectively, and the QALYs were 1.01, 1.19, and 1.44, respectively. Compared with atezolizumab + CT, the ICERs of nivolumab + ipilimumab + CT and pembrolizumab plus CT were $68,963.1 and $179,355.6 per QALY gained, respectively, and the ICER of pembrolizumab + CT compared with nivolumab + ipilimumab + CT was $258,894.4 per QALY gained. In patients with PD-L1 TPS <1%, the mean costs of combining CT with atezolizumab and pembrolizumab were $282,689.19 and $272,344.16, respectively, and the QALYs were 1.00 and 1.25, respectively. The cost-effectiveness of pembrolizumab + CT dominated that of atezolizumab + CT in this setting. In patients with PD-L1 TPS 1–49%, the mean costs of atezolizumab + CT and pembrolizumab + CT were $225,231.95 and $336,030.44, respectively. The QALYs were 1.32 and 0.82, respectively. Compared with atezolizumab + CT, the ICER for pembrolizumab plus CT was $222,605.8 per QALY gained. The one-way sensitivity analysis (Supplemental Figure S7) revealed that the ICER of pembrolizumab + CT versus atezolizumab + CT in the total population was mostly sensitive to cost of pembrolizumab [Supplemental Figure S7(A)], while the ICER of pembrolizumab + CT versus nivolumab + ipilimumab + CT in the total population was most sensitive to the HR for OS [Supplemental Figure S7(B)], and the ICER of atezolizumab + CT versus nivolumab + ipilimumab + CT was most sensitive to body weight [Supplemental Figure S6(C)]. In terms of patients with PD-L1 TPS 1–49%, the ICER of pembrolizumab + CT versus atezolizumab + CT was primarily sensitive to the HR for OS [Supplemental Figure S7(D)]. As for patients with PD-L1 TPS <1%, the ICER of pembrolizumab + CT versus atezolizumab + CT was primarily sensitive to HR for PFS [Supplemental Figure S7(E)]. At a WTP threshold of $150,000 per QALY, nivolumab + ipilimumab + CT had the highest probability of cost-effectiveness in the total population [63%; Figure 4(a)], while pembrolizumab + CT had the highest probability of being cost-effective in patients with PD-L1 1–49% [93%; Figure 4(b)]. In contrast, pembrolizumab + CT had the highest probability [96%; Figure 4(c)] of being cost-effective at a WTP threshold of $150,000 per QALY in patients with PD-L1 TPS <1%.

The cost-effectiveness acceptability curves for pembrolizumab plus chemotherapy, atezolizumab plus chemotherapy, and nivolumab plus ipilimumab plus chemotherapy in (a–c) the United Sates and (d–f) China. (a and d) The intend-to-treatment population. (b and e) patients with PD-L1 TPS 1–49%. (c and f) Patients with PD-L1 TPS <1%.
From the China perspective, the mean costs of atezolizumab + CT, nivolumab + ipilimumab + CT, and pembrolizumab + CT were $85,686.76, $110,386.59, and $164,769.46, and the QALYs were 0.98, 1.15, and 1.39, respectively, in the total population. Compared with atezolizumab + CT, the ICERs of nivolumab + ipilimumab and pembrolizumab + CT were $145,983.4 and $195,863.3, respectively, in the total population. In patients with PD-L1 <1%, the costs of atezolizumab + CT and pembrolizumab + CT were $89,856.14 and $139,757.56, while the QALYs were, respectively, 0.97 and 1.20, yielding an ICER of $215,057.7. Similarly, in patients with PD-L1 1–49%, the costs of the two treatments were $73,557.28 and $155,523.98, while the QALYs were 0.81 and 1.28, leading to an ICER of $173,939.0 per QALY gained. The one-way sensitivity analysis (Supplemental Figure S8) identified that all the cost-effectiveness results in China were mostly sensitive to HR for OS. As for the PSA, atezolizumab + CT had the highest probability to be cost-effective in the total population [94%; Figure 4(d)] at the WTP threshold of $38,017 per QALY gained. Unlike the results from the US perspective, atezolizumab + CT also had higher probabilities of cost-effectiveness compared with pembrolizumab + CT in both patients with PD-L1 1–49% [100%; Figure 4(e)] and patients with PD-L1 <1% [100%; Figure 4(f)].
Discussion
The combination of ICIs with CT has been proven to be a promising first-line strategy in treating patients with advanced non-squamous NSCLC, the efficacy of which, however, varies with tumor PD-L1 expression. To date, several ICI + CT combination strategies have been recommended for the treatment of advanced or metastatic non-squamous NSCLC in the NCCN guideline. 19 However, the cost-effectiveness of these therapies remains to be explored.
The results from the NMAs indicated that pembrolizumab + CT resulted in better survival benefits compared with the other two ICI-based treatments in the total population and in most PD-L1 expression subgroups, which was consistent with a former NMAs. 34 The evaluation of safety profiles demonstrated that all three ICI + CT combination treatments increased the incidences of TRAEs of grade ⩾3 compared with CT alone but had similar chances of causing TRAEs of any grades or TRAEs of grade ⩾3. Nevertheless, the cost-effectiveness analysis revealed that each strategy had distinct rankings in different PD-L1 expression subgroups from the perspective of the US healthcare payers. Specifically, nivolumab + ipilimumab + CT was the most favorable strategy in the total population, while atezolizumab + CT was more favorable in patients with PD-L1 1–49%, and pembrolizumab + CT was preferable in patients with PD-L1 <1%. The sensitivity analyses revealed that the results were generally sensitive to the HRs for OS or PFS of ICI + CT combinations, suggesting that the efficacies of the treatments were the most definitive factor in influencing the cost-effectiveness. Also, the costs of the ICIs account for one of the most significant factors, suggesting that the high price of ICIs is also an unneglectable factor. In contrast, from the Chinese perspective, atezolizumab + CT was the most cost-effective strategy in the total population, patients with PD-L1 1–49%, and patients with PD-L1 <1%, due to distinct cost inputs and the much lower WTP threshold. These results provide significant insights for patients and clinicians to choose which ICI to use. To be more specific, in the United States, nivolumab + ipilimumab + CT is more favorable for patients with non-squamous NSCLC, while atezolizumab + CT and pembrolizumab + CT should be recommended for patients with PD-L1 1–49% and patients with PD-L1 <1%, respectively, to meet the optimal cost-effectiveness. In China, atezolizumab + CT is an economic option despite PD-L1 expression.
Teng et al. 35 compared the cost-effectiveness of four ICI regimens, including nivolumab, atezolizumab, pembrolizumab, and durvalumab, as first-line monotherapy for Chinese patients with NSCLC, and the results showed that nivolumab ranked first in terms of cost-effectiveness. This study investigated the cost-effectiveness of several ICIs but could not answer the question of which combination of ICI and CT, which has better efficacy than ICI monotherapy, has the optimal cost-effectiveness. A study by Barbier et al. 36 showed that pembrolizumab + CT was not likely to be cost-effective in Swiss patients with PD-L1 TPS ⩾50%, and Jiang et al. 37 and Cai et al. 10 also revealed that pembrolizumab + CT was not cost-effective for Chinese patients with non-squamous NSCLC. Additionally, from a US healthcare perspective, another study by Liu et al. 9 showed that pembrolizumab + CT was not cost-effective for non-squamous NSCLC patients with PD-L1 TPS ⩾50% compared with pembrolizumab alone. Our acceptability analyses also showed low probabilities of pembrolizumab + CT being cost-effective in all non-squamous NSCLC patients and across subgroups of different PD-L1 expressions at a WTP threshold of $150,000 per QALY. As for atezolizumab + CT, Ding et al. 38 and Yang et al. 11 showed that the combination of atezolizumab and carboplatin plus nab-paclitaxel was not cost-effective for first-line treatment of advanced non-squamous NSCLC from the US payers’ perspective and the Chinese payers’ perspective, respectively. However, our results showed that the combination of atezolizumab and carboplatin/cisplatin plus pemetrexed had a high probability of being cost-effective for US patients (69%), suggesting that the CT regimens can also strongly affect the cost-effectiveness of ICI + CT. In 2023, a cost-effectiveness analysis investigated the cost-effectiveness of pembrolizumab + CT, atezolizumab + CT, nivolumab + ipilimumab + CT, nivolumab + ipilimumab, atezolizumab + bevacizumab + CT, pembrolizumab alone, and CT alone in treating advanced NSCLC in the United States. 39 The results showed that the nivolumab + ipilimumab, pembrolizumab + CT, and pembrolizumab alone were the preferred strategies for patients with PD-L1 <1%, 1–49% and ⩾50%, respectively, which differed from our results. 39 The difference may attribute to different survival estimating references (the study is based on CT, and our analysis is based on pembrolizumab + CT) and a more detailed cost consideration for patients of our study. 39
Based on our results and previous studies, we suggest administrating different ICIs to combine with CT regimens according to patients’ PD-L1 expression, as the cost-effectiveness ranks varied across different PD-L1 expression subgroups. PD-L1 testing is a recommended and routinely applied practice for advanced NSCLC patients, and our results highlight the importance of early detection of PD-L1 expression for non-squamous NSCLC patients with negative driver gene mutations to determine the most cost-effective treatment. Additionally, although pembrolizumab + CT has a favorable efficacy and has been listed as a preferred option in treating advanced NSCLC in the latest NCCN guideline, 19 the cost-effectiveness of pembrolizumab + CT remains inferior. Our sensitivity analysis suggested that the cost of pembrolizumab accounts inserted an essential impact on the comparisons of cost-effectiveness, indicating that a discount on pembrolizumab price may significantly improve the cost-effectiveness of pembrolizumab + CT and benefit more patients with lower costs. In short, our study provides important information for clinicians and policymakers in making treatment decisions for advanced non-squamous NSCLC patients.
This study has the following limitations. First, since CheckMate 9LA enrolled both patients with squamous NSCLC and patients with non-squamous NSCLC, the available data of different PD-L1 expression subgroups in CheckMate 9LA involved patients with both cancer types. Accordingly, we excluded NivoIpiC in the subgroup analyses by PD-L1 expression. Second, although the regimens used in the three RCTs were similar, disparities, such as differences in patients’ baseline characteristics, duration of follow-up, and courses of CT, remain. Consequently, the discrepancies led to mild deviations of predicted OS and PFS curves of IMpower132 and CheckMate 9LA from the original K–M curves when we fitted the curves using NMA-generated HRs. Third, the cost-effectiveness of atezolizumab + CT in patients with PD-L1 TPS ⩾50% was not revealed in our study due to its relatively small sample size and the fact that the median OS of this group was not reached in the original data.
Conclusion
The NMAs revealed that pembrolizumab + CT seemed to have the best efficacy compared with nivolumab + ipilimumab + CT and atezolizumab + CT in the total population with advanced non-squamous NSCLC and most subgroups with different PD-L1 expressions, and the safety profiles of the ICI plus CT combinations were similar. However, the cost-effectiveness analyses showed that from the US perspective, nivolumab + ipilimumab + CT was most favorable in the total population, and pembrolizumab + CT and atezolizumab + CT had the best cost-effectiveness in patients with PD-L1 TPS <1% and 1–49%, respectively. However, from the Chinese perspective, atezolizumab + CT was the most cost-effective strategy in the total population, patients with PD-L1 TPS <1%, and patients with PD-L1 TPS 1–49%.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241255613 – Supplemental material for First-line treatments for advanced non-squamous non-small cell lung cancer with immune checkpoint inhibitors plus chemotherapy: a systematic review, network meta-analysis, and cost-effectiveness analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359241255613 for First-line treatments for advanced non-squamous non-small cell lung cancer with immune checkpoint inhibitors plus chemotherapy: a systematic review, network meta-analysis, and cost-effectiveness analysis by Wentao Tian, Lishui Niu, Yin Shi, Shuishi Li and Rongrong Zhou in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
