Abstract
Background:
Multiple anti-programmed cell death-1/programmed cell death-ligand 1 (PD-1/PD-L1) inhibitors and zolbetuximab, an anti-claudin 18.2 antibody, have shown efficacy in the first-line treatment of HER2-negative gastric cancers. How to choose the best regimen remains an unsolved question.
Objectives:
We aimed to conduct a comparative analysis of the therapeutic advantages between immunotherapy and anti-claudin-18.2-targeted therapies in the first-line treatment of HER2-negative, unresectable, or metastatic gastric cancers.
Design:
Network meta-analysis was employed to systematically compare efficacy and safety data derived from various clinical trials.
Data sources and methods:
We included phase III randomized controlled trials in PubMed, Embase, Web of Science, Cochrane Library, and major conference abstracts. Network meta-analysis was used to compare the efficacy of each first-line therapeutic agent and to indirectly compare immunotherapy with anti-claudin-18.2-targeted therapy.
Results:
Eight trials comprising a total of 6455 patients were included. For the overall survival (OS) analysis, no statistically significant differences were observed between pembrolizumab [hazard ratios (HR) = 1.00, 95% CI: 0.94–1.07], sintilimab (HR = 0.99, 95% CI: 0.89–1.09), sugemalimab (HR = 0.98, 95% CI: 0.87–1.10), tislelizumab (HR = 0.97, 95% CI: 0.87–1.09), zolbetuximab (HR = 0.98, 95% CI: 0.91–1.07), and nivolumab (HR = 1.00). For the progression-free survival (PFS) analysis, no statistically significant differences were observed between pembrolizumab (HR = 1.00, 95% CI: 0.93–1.06), sintilimab (HR = 0.91, 95% CI: 0.83–1.00), sugemalimab (HR = 0.92, 95% CI: 0.84–1.02), tislelizumab (HR = 0.93, 95% CI: 0.84–1.03), zolbetuximab (HR = 0.96, 95% CI: 0.88–1.05), and nivolumab (HR = 1.00). For the overall response rate analysis, all regimens presented similar effects on ORR. In addition, anti-claudin-18.2-targeted therapies presented similar OS (HR = 0.99, 95% CI: 0.95–1.04) and PFS (HR = 1.01, 95% CI: 0.91–1.12) compared to immunotherapy, although their toxicity profiles were distinct.
Conclusions:
Our network meta-analysis showed no significant difference in PFS, OS, or ORR between different checkpoint inhibitors or between immunotherapy and anti-claudin-18.2-targeted therapies in the first-line treatment of HER2-negative, unresectable, or metastatic gastric cancers.
Introduction
Gastric cancer (GC) is the fifth most commonly diagnosed malignancy and the fourth leading cause of cancer mortality worldwide, resulting in more than 768,000 deaths in 2020. 1 The incidence of GCs varies across regions, with the highest estimated rate in Asia. 1 Despite a decline in global incidence within the past few decades, unresectable or metastatic GCs remain incurable and portend dismal prognosis. In the past few decades, the mainstay of first-line regimens for unresectable GCs encompasses platinum-based chemotherapy.2,3 Nevertheless, treatment modalities for unresectable or metastatic GCs have expanded greatly in recent years. With the revolution of precision medicine, novel medications such as immune checkpoint inhibitors (ICIs) and targeted therapies are emerging, enabling oncologists to treat late-stage GCs with further choices. 4 For the first-line treatment of HER2-negative advanced GCs, ICIs and anti-claudin-18.2-targeted therapies are both optional.
CheckMate-649, a global, large-scale, phase III clinical trial, has taken the lead to confirm the superiority of nivolumab in combination with chemotherapy versus chemotherapy alone in the treatment of patients with advanced GCs, regardless of the programmed cell death-ligand 1 (PD-L1) combined positive score (CPS). 5 Based on this landmark study, the U.S. Food and Drug Administration approved nivolumab plus chemotherapy for the first-line treatment of patients with advanced GCs independent of PD-L1 CPS. Since then, other clinical trials have also reported positive results indicating the efficacy of combinational immunotherapy in the first-line treatment of advanced GCs, such as ORIENT-16, RATIONALE-305, Keynote-859, etc.6–8
Claudin 18.2 is a tight junction molecule that is involved in multiple functions of epithelial layers, including permeability regulation and intercellular communication. 9 In healthy tissue, claudin 18.2 is predominantly located within normal gastric mucosa and is inaccessible to circulating antibodies, while its epitopes become exposed during malignant transformation. 9 Zolbetuximab is a chimeric monoclonal antibody that binds to claudin 18.2 and induces tumor cell death through antibody-dependent cellular cytotoxicity and complement-dependent cytotoxicity.10,11 Recently, the results of two key studies, SPOTLIGHT and GLOW, were announced, which are global, phase III clinical trials comparing the efficacy of chemotherapy plus zolbetuximab versus chemotherapy plus placebo for claudin-18.2-positive (defined as moderate-to-strong expression in ⩾75% tumor cells) advanced GCs.12,13 These pilot studies confirmed that the addition of zolbetuximab to chemotherapy presented a manageable safety profile and could prolong the progression-free survival (PFS) and overall survival (OS) of patients with claudin-18.2-positive tumors.
Despite this promising therapeutic progress, new challenges also arise. How can the best first-line regimen for HER2-negative advanced GCs be chosen if both zolbetuximab and ICIs are selectable? In particular, how can the optimized treatment strategy be determined if the PD-L1 and claudin 18.2 of the tumor are both highly expressed? Numerically, the median OS brought by zolbetuximab was longer than that by nivolumab (18.23 months versus 13.83 months, for the total patients in the SPOTLIGHT and the CheckMate-649 studies).5,13 However, the distinction between populations and cytotoxic regimens might account for the difference, making it difficult for clinicians to compare the survival data directly. In addition, despite multiple high-quality clinical trials on ICIs, head-to-head comparisons of these two drugs for treating unresectable or metastatic GCs are lacking.
Therefore, in the present study, we performed a network meta-analysis to assess the effectiveness of multiple ICIs and zolbetuximab in the first-line treatment of unresectable or metastatic HER2-negative GCs.
Materials and methods
Literature search strategy
We searched for phase III randomized controlled trials (RCTs) that compared the efficacy between claudin-18.2-targeted therapy or immunotherapy plus chemotherapy with placebo plus chemotherapy in the first-line treatment of unresectable or metastatic HER2-negative GCs by utilizing the following electronic databases: PubMed, Embase, Web of Science, and Scopus Cochrane Database. Conference abstracts presented at the annual meetings of the European Society for Medical Oncology, American Society for Clinical Oncology (ASCO), and ASCO GI during the last 10 years (2013–2023) were also reviewed. The publication time of the literature was restricted to March 2015 to March 2023. The search terms ‘gastric cancer’, ‘first-line’, and ‘randomized controlled trial’ were used with language limited to English. Our meta-analysis followed the Preferred Reporting Items for Systematic Reviews and Meta-analyses extension for Network Meta-analyses (PRISMA-NMA) guidelines. 14 The full search terms and the PRISMA-NMA Checklist have been included in the supplementary materials.
Study selection criteria
The screened clinical trials were deemed eligible if they met the following inclusion criteria: (1) all patients were pathologically diagnosed with HER2-negative gastric or gastroesophageal adenocarcinoma and received first-line palliative therapy; (2) fluorouracil and platin-based doublets should be included in the chemotherapy regimens in each arm; (3) only single-agent target therapy or immunotherapy was investigated in the experimental arm; and (4) studies should report at least one of the outcomes of interest, including OS, PFS, and ORR. Noninterventional studies were excluded.
Outcomes
The primary endpoint was OS, which was defined as the time from randomization to death from any cause. The secondary endpoints were PFS (defined as the time from randomization to disease progression or any cause of death, whichever occurred first), ORR (defined as the percentage of patients experiencing an objective response as the best response to treatments), and the safety data (incidence of grade ⩾3 treatment-related adverse events).
Data extraction and synthesis
Data were independently extracted by two investigators (XT and WY) and cross-checked. In the case of disagreement, the original documents were checked, and decisions were made under negotiation. Trial name, year of data publication, treatment regimens, and the number of participants in each cohort were recorded. Median OS, median PFS, ORR, hazard ratios (HRs) for OS and PFS, and their 95% confidential intervals (CIs) were extracted.
Quality assessment of included studies
The quality of the studies included in this network meta-analysis was assessed using the revised Cochrane risk-of-bias tool: RoB2. 15
Statistical analysis
First, we performed indirect comparisons of HRs for OS, PFS, and ORR with each first-line therapeutic agent. Then, we compared the efficacy of first-line immunotherapy to first-line anti-claudin-18.2-targeted therapy by integrating individual efficacy data. Common effects models and random effects models were used to estimate the pooled effects, and forest plots were generated. A high risk of heterogeneity was considered when I 2 ⩾ 50%. In the indirect analysis, a high risk of heterogeneity within designs was considered when the p value for the Q value < 0.05 (via chi-square test). The network meta-analyses were conducted by R 3.6.3 (R Software, Austria) with R package netmeta. All reported p values were two-sided, and statistical significance was considered when p < 0.05.
Results
Literature review results
A total of 2911 potentially relevant publications were identified in the literature search. After initial abstract review and duplicate removal, eight original studies met all the inclusion criteria and thus were considered eligible, comprising 6455 patients for final analysis (Figure 1).
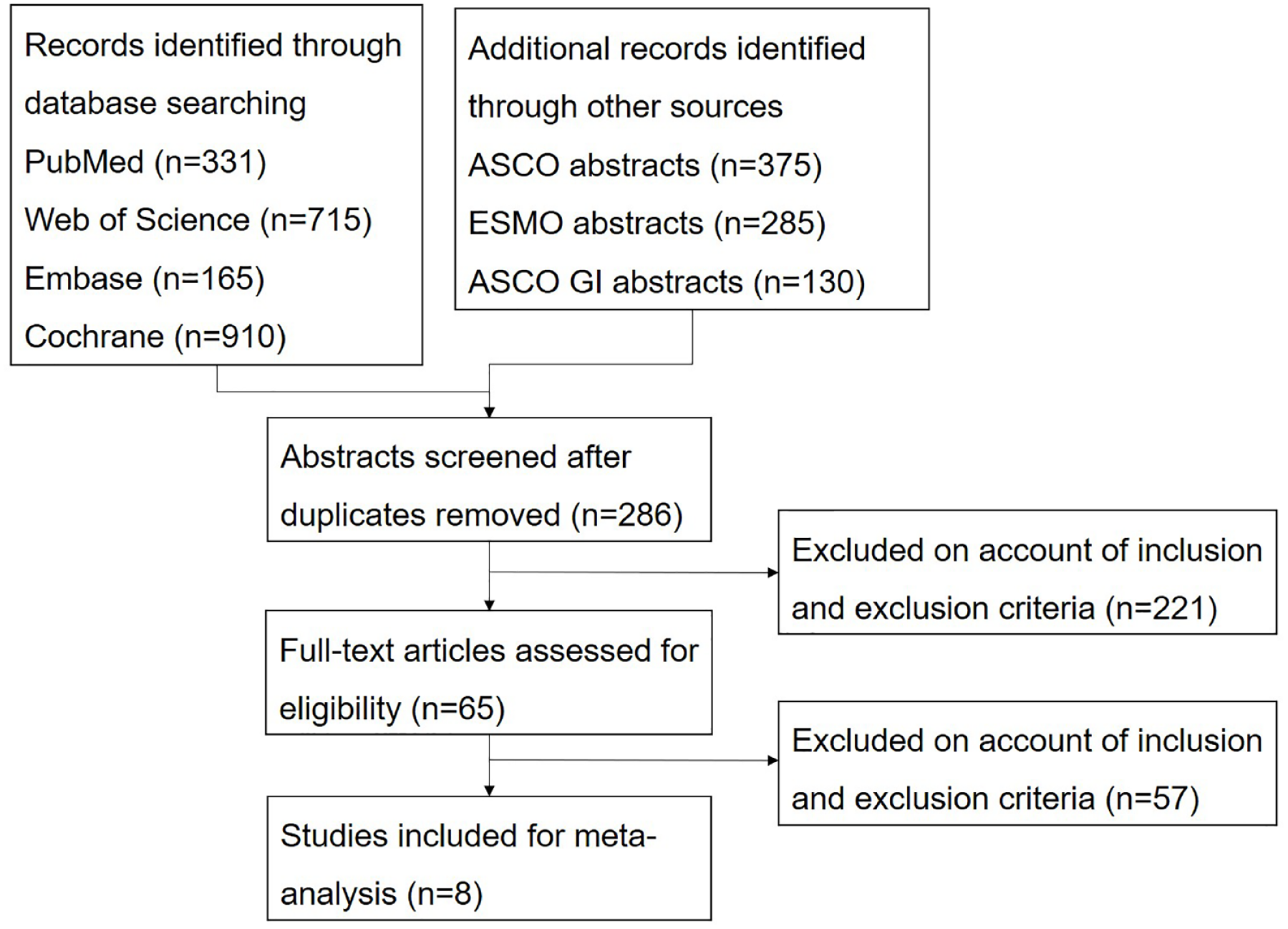
Flow chart of the study.
Characteristics of the included studies
Eight incorporated RCTs involved patients with treatment-naïve, unresectable or metastatic, HER2-negative GCs receiving chemotherapy regimens containing fluoropyrimidine and platinum, as well as biological therapies including anti-PD-1 monoclonal antibodies (nivolumab, pembrolizumab, sintilimab, sugemalimab, and tislelizumab) and anti-claudin 18.2 monoclonal antibodies (zolbetuximab). The results of the risk-of-bias assessment for all included studies are presented in Figure 2. Among 6455 patients, 2721 received chemotherapy and ICIs, 537 received chemotherapy and claudin-targeted therapies, and 3197 patients were treated with chemotherapy and matching placebo. The baseline characteristics among the included RCTs are indicated in Table 1.
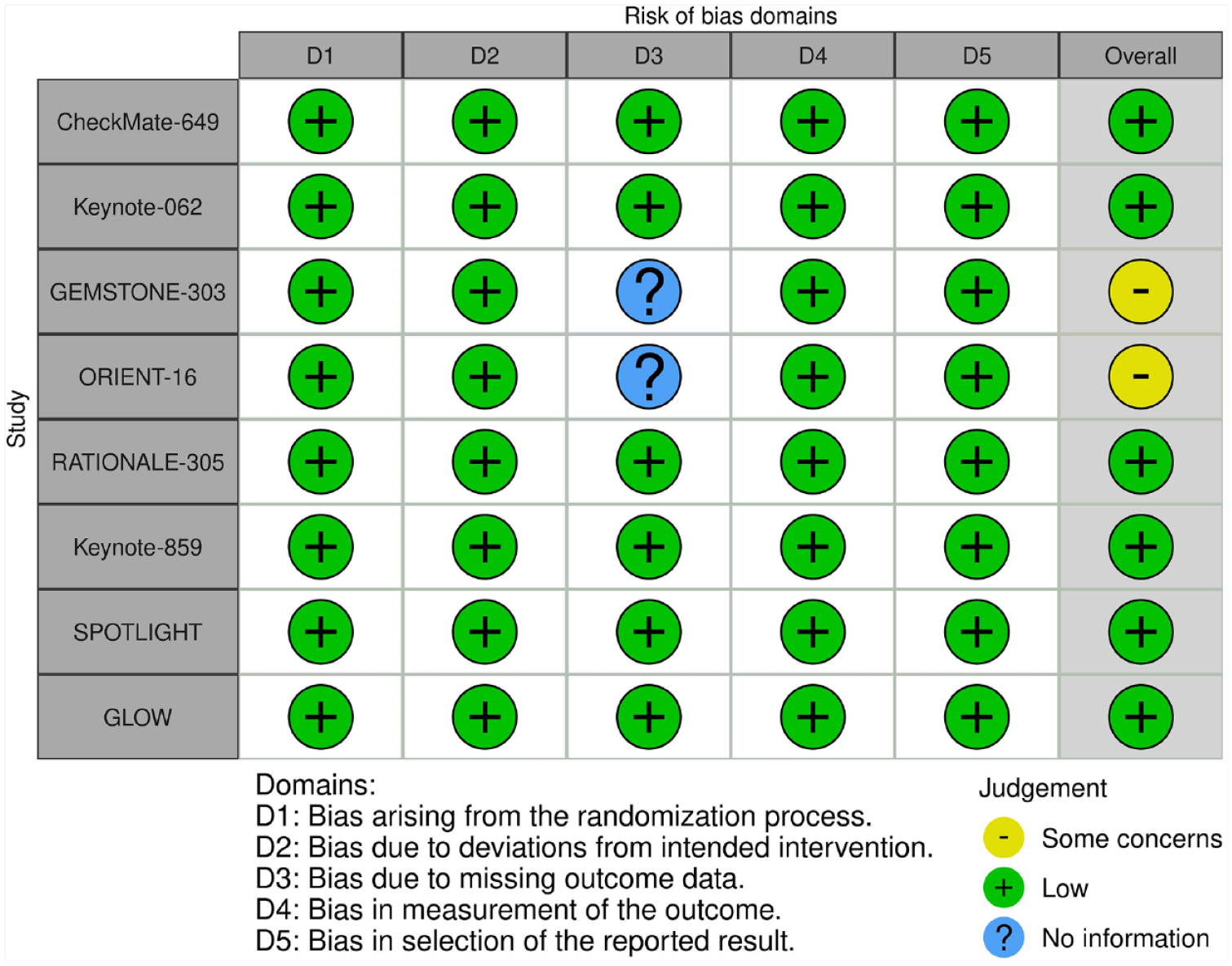
Risk-of-bias assessments for included studies.
Baseline characteristics of the included studies.

CPS, combined positive score; FOLFOX, 5-FU + oxaliplatin + leucovorin; FP, fluorouracil + cisplatin; ITT, intention-to-treat; mFOLFOX6, 5-FU + oxaliplatin + leucovorin; OS, overall survival; PFS, progression-free survival; XELOX, capecitabine + oxaliplatin; XP, capecitabine + cisplatin.
Comparisons of the efficacy between different first-line therapeutic agents
In terms of the indirect comparison of OS, pembrolizumab (HR = 1.00, 95% CI: 0.94–1.07) (p = 0.457), sintilimab (HR = 0.99, 95% CI: 0.89–1.09; p = 0.595), sugemalimab (HR = 0.98, 95% CI: 0.87–1.10; p = 0.645), tislelizumab (HR = 0.97, 95% CI: 0.87–1.09) (p = 0.683), and zolbetuximab (HR = 0.98, 95% CI: 0.91–1.07; p = 0.626) all presented similar effects on OS in contrast to nivolumab (Figure 3). Compared to the combinations of chemotherapy and nivolumab, chemotherapy alone resulted in an inferior OS (HR = 1.11, 95% CI: 1.06–1.16; p = 0.004; Figure 3). The Q value was 0.61 (p = 0.74), indicating a low risk of heterogeneity within designs.
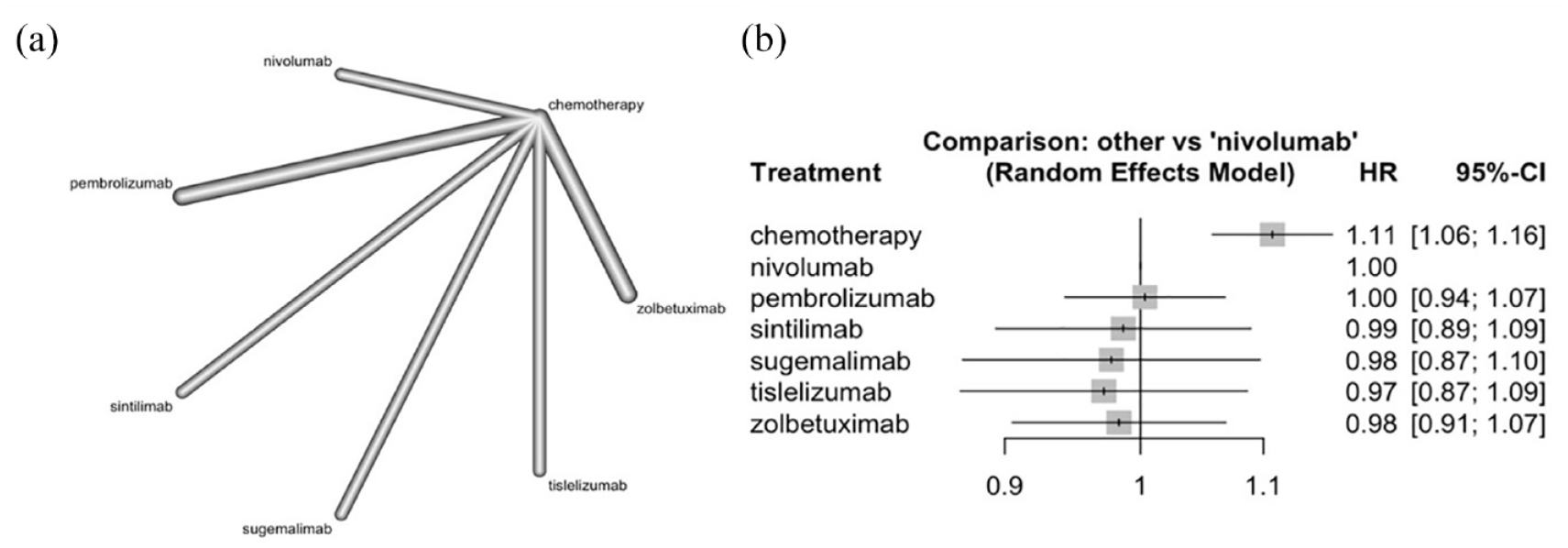
Indirect analysis of OS among patients receiving first-line therapeutic agents. (a) The network map, with the nodes representing different treatments and the connecting lines representing the comparisons; (b) the forest plot.
In terms of the indirect comparison of PFS, pembrolizumab (HR = 1.00, 95% CI: 0.93–1.06; p = 0.323), sintilimab (HR = 0.91, 95% CI: 0.83–1.00; p = 0.835), sugemalimab (HR = 0.92, 95% CI: 0.84–1.02; p = 0.757), tislelizumab (HR = 0.93, 95% CI: 0.84–1.03) (p = 0.723), and zolbetuximab (HR = 0.96, 95% CI: 0.88–1.05; p = 0.562) all presented similar effects on PFS in contrast to nivolumab (Figure 4). Likewise, compared to the combinations of chemotherapy and nivolumab, chemotherapy alone resulted in inferior PFS (HR = 1.11, 95% CI: 1.05–1.16; p < 0.001; Figure 4). The Q value was 1.06 (p = 0.59), indicating a low risk of heterogeneity within designs.

Indirect analysis of PFS among patients receiving first-line therapeutic agents. (a) The network map, with the nodes representing different treatments and the connecting lines representing the comparisons; (b) the forest plot.
ORR data were available for pembrolizumab, nivolumab, sintilimab, tislelizumab, and zolbetuximab. In terms of the indirect comparison of ORR, the combinations of chemotherapy and nivolumab (OR = 1.5, 95% CI: 0.92–2.4), pembrolizumab (OR = 1.5, 95% CI: 1.1–2.2), sintilimab (OR = 1.5, 95% CI: 0.88–2.5), tislelizumab (OR = 1.3, 95% CI: 0.79–2.3), and zolbetuximab (OR = 1.1, 95% CI: 0.72–1.6) all presented similar effects on ORR in contrast to chemotherapy alone (Figure 5).

Indirect analysis of ORR among patients receiving first-line therapeutic agents. (a) The network map, with the nodes representing different treatments and the connecting lines representing the comparisons; (b) the forest plot.
Comparisons of the efficacy between immunotherapy and anti-claudin-18.2-targeted therapy
We pooled the OS and PFS results of six trials that investigated the superiority of chemotherapy combined with anti-PD-1 antibodies versus chemotherapy combined with placebo in the treatment of unresectable or metastatic HER2-negative GCs. When compared to immunotherapy, anti-claudin-18.2-targeted therapies appeared to result in similar OS (HR = 0.99, 95% CI: 0.95–1.04) (p = 0.802) and PFS (HR = 1.01, 95% CI: 0.91–1.12; p = 0.810; Figure 6).

Indirect comparisons of OS (a) and PFS (b) among patients receiving chemotherapy, immunotherapy, and zolbetuximab.
Safety
Detailed safety data could be generated from the CheckMate-649, Keynote-062, Keynote-859, SPOTLIGHT, and GLOW trials. Grade ⩾3 treatment-related adverse events occurred in 59.0–73.2% of patients treated with the combination of chemotherapy and ICIs, with neutropenia and neutrophil count decrease being the two most common grade ⩾3 events. Grade ⩾3 treatment-related adverse events occurred in 72.8–86.7% of patients treated with the combination of chemotherapy and zolbetuximab, with nausea and vomiting being the two most common grade ⩾3 events.
Discussion
Herein, we presented the first and the latest comparative study that provided the reference value for choosing the optimized first-line therapy of HER2-negative GCs. The results of our network meta-analysis showed no significant difference in PFS, OS, or ORR between different ICI agents in the first-line treatment of HER2-negative, unresectable, or metastatic GCs. In addition, immunotherapy was also demonstrated to be comparable to anti-claudin-18.2-targeted therapy in terms of PFS, OS, and ORR. Therefore, either of the two options is feasible. However, the toxicity profiles of these two treatments are distinct: ICIs typically cause immune-related adverse events, which are multiorgan-affected toxicities triggered by overactivated immune responses, while anti-claudin 18.2 antibodies predominantly lead to gastrointestinal adverse events (e.g. vomiting, nausea) mediated by the ‘on-target off-tumor’ effect.10,16 Accordingly, in the real world, patient tolerance should also be considered when choosing the appropriate regimens.
Although different ICI agents presented similar treatment effects according to our analysis, we noticed that the absolute survival benefits patients generated varied across studies. For example, the combinational treatment of chemotherapy and ICIs resulted in an OS of 17.2 months in the RATIONALE-305 trial but only 13.8 months in the CheckMate-649 trial.5,6 In addition, although pembrolizumab was used in both the Keynote-062 and Keynote-859 trials, the results of these studies were distinct.8,17 To be more specific, the Keynote-062 trial incorporated cisplatin into the chemotherapy regimen, whereas other trials utilized oxaliplatin, an alkylate known for inducing more potent immunogenic cell death. Furthermore, in comparison to the CheckMate-649 trial, the RATIONALE-305 study enrolled a greater percentage of Asian patients (74% versus 25%) and patients with PD-L1 CPS ⩾ 1 (100% versus 60%). This indicates that the RATIONALE-305 study had a higher enrichment of patients sensitive to ICIs. Therefore, it is obvious that different populations, structures of ICIs, and chemotherapy regimens might have influenced the overall treatment efficacy. In clinical practice, the regimen used in the study with numerically the best survival data should be recommended preferentially. It is also noteworthy that sugemalimab was the first anti-PD-L1 antibody that showed efficacy in the first-line treatment of advanced GCs. Previous data indicated that anti-PD-L1 antibodies resulted in fewer adverse events than anti-PD-1 antibodies. 18 Whether this regimen is safer among ICIs still needs to be examined in further studies.
To optimize the first-line treatment regimen, a better understanding of the interactions between claudin 18.2 expression and the tumor immune microenvironment is essential. According to a meta-analysis conducted in 2021, no significant correlation was found between claudin 18.2 expression and major clinicopathological parameters, including tumor, nodes, metastases (TNM) stages, Lauren classification, HER2 status, and grading. 19 Recently, Shitara et al. conducted a comprehensive characterization of claudin 18.2 expressions in 408 advanced GCs, which also suggested no evidence of a correlation between claudin 18.2 positivity and clinicopathological factors, except for dominance of Bormann type IV. 20 In addition, this study also revealed that molecular subtyping and PD-L1 CPS had little impact on claudin 18.2 expression. 20 Neither of the aforementioned two studies found any difference in the prognosis or chemotherapeutic/immunotherapeutic efficacy between patients with claudin-18.2-positive and claudin-18.2-negative GCs.19,20 However, other studies of the same type revealed that claudin-18.2-positive GCs are more advanced and harbor a more diffuse/poorly differentiated pathological appearance, which usually heralds poor response to immunotherapy.21,22 Of note, different detection antibodies and cutoff values were adopted in different studies. According to the multidimensional analysis of the immune microenvironment of claudin-18.2-positive GCs, claudin 18.2 positivity is correlated with a higher infiltration of non-exhausted CD8+ and CD4+ T cells (due to the lack of immune checkpoint molecules on their surface) within the immune microenvironment and shows a poor response to ICIs. 23 Therefore, the prognostic significance of claudin 18.2 positivity still needs further investigation, considering the conflicting results at present and the heterogeneity of the study designs.
Comprehensively, our meta-analysis indicated that anti-claudin-18.2-targeted therapies and ICIs had similar effects on patients with claudin-18.2-positive, PD-L1 unselected tumors. To further stratify patients based on both PD-L1 CPS and claudin 18.2 expression, different situations should be discussed separately. As suggested in a secondary analysis of four pivotal RCTs, there is a lack of benefit in the addition of ICIs to chemotherapy in PD-L1 low-expressing GCs. 24 Therefore, for PD-L1-positive, claudin-18.2-negative GCs, ICIs should be given top priority, and for PD-L1-negative, claudin-18.2-positive GCs, zolbetuximab is preferred. For PD-L1-positive, claudin-18.2-positive GCs, determining the best regimen is challenging. According to the results of the GLOW study, 22% of claudin-18.2-positive GCs also had CPS ⩾ 5. 12 In addition, we observed that patients with tumors exhibiting very high CPS achieved a numerically prolonged duration of response (10.9 months in the CPS ⩾ 10 subgroup of the Keynote-859 trial, 9.5 months in the CPS ⩾ 5 subgroup of the CheckMate-649 trial) in response to anti-PD-1 therapies. This contrasts with the general patient population that received claudin-targeted therapies, where the duration of response was 6.3 months in the GLOW trial and 9.0 months in the SPOTLIGHT trial. The findings may suggest that for tumors classified as ‘CPS-high, claudin-18.2-positive’, immunotherapy, rather than anti-claudin therapy, is more likely to confer a durable response. A clinical trial with one cohort investigating the efficacy and safety of zolbetuximab combined with chemotherapy and anti-PD-1 antibody is ongoing (ILUSTRO: NCT03505320). Whether the improvement in treatment responses is worth the superposed toxicities is expected to be clarified. For these ‘double-positive’ GCs, sequential therapy is another potential option. First-line immunotherapies exert a positive influence on the efficacy of later therapies in patients with a few cancer types, which is potentially attributed to the long-acting effects of ICIs. 25 Among GC patients, a numerical prolongation of OS could also be observed when ICIs are applied from the later lines to the earlier lines.5,26 A ‘tailing effect’ of the OS curve was observed in the CheckMate-649 trial, suggesting that durable responses could be achieved in a small portion of patients who received ICIs, while this phenomenon was not evident in the SPOTLIGHT and GLOW trials.5,12,13 Inspiringly, whether the efficacy of anti-claudin-18.2-targeted therapies is influenced by treatment lines warrants examination.
Our studies have several limitations, such as the small number of included studies and the different cutoff values for PD-L1 and claudin 18.2 across different studies. Future studies should be conducted to clarify the best cutoff values of these biomarkers. In addition, head-to-head comparative studies and RCTs that investigate combinational or sequential therapies are warranted.
Conclusion
In summary, this network meta-analysis showed no significant difference in PFS, OS, or ORR between different ICI agents or between immunotherapy and anti-claudin-18.2-targeted therapies in the first-line treatment of HER2-negative, unresectable, or metastatic GCs.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241231253 – Supplemental material for Immune checkpoint inhibitors or anti-claudin 18.2 antibodies? A network meta-analysis for the optimized first-line therapy of HER2-negative gastric cancer
Supplemental material, sj-docx-1-tam-10.1177_17588359241231253 for Immune checkpoint inhibitors or anti-claudin 18.2 antibodies? A network meta-analysis for the optimized first-line therapy of HER2-negative gastric cancer by Zhening Zhang, Tong Xie, Yingxuan Wang, Lin Shen, Xiaotian Zhang and Zhi Peng in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359241231253 – Supplemental material for Immune checkpoint inhibitors or anti-claudin 18.2 antibodies? A network meta-analysis for the optimized first-line therapy of HER2-negative gastric cancer
Supplemental material, sj-docx-2-tam-10.1177_17588359241231253 for Immune checkpoint inhibitors or anti-claudin 18.2 antibodies? A network meta-analysis for the optimized first-line therapy of HER2-negative gastric cancer by Zhening Zhang, Tong Xie, Yingxuan Wang, Lin Shen, Xiaotian Zhang and Zhi Peng in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
