Abstract
Background:
The benefit of adding immunotherapy to induction chemotherapy before definitive chemoradiotherapy (CRT) in older patients with unresectable stage III non-small-cell lung cancer (NSCLC) remains unclear.
Objectives:
This real-world study aimed to evaluate the efficacy and safety of induction chemoimmunotherapy followed by CRT in this patient population.
Design:
A real-world multicenter study.
Methods:
In this retrospective, multicenter study, we enrolled patients aged ⩾65 years with unresectable stage III NSCLC who received CRT between January 2014 and June 2024. Patients were stratified into two groups: the induction chemoimmunotherapy group (I-CRT), who received PD-1/PD-L1 inhibitors plus chemotherapy before CRT, and the control group (Non-I-CRT), who received CRT without any prior immunotherapy. Propensity score matching (PSM) was used to balance baseline characteristics. The primary endpoints were progression-free survival (PFS) and overall survival (OS).
Results:
Among 260 patients, 141 received I-CRT and 119 constituted the Non-I-CRT control group. After 1:1 PSM (97 patients per group), the I-CRT group showed significantly improved outcomes compared to the Non-I-CRT group: median PFS was 23.4 versus 11.6 months (p < 0.001), and median OS was 46.0 versus 24.4 months (p = 0.009). The incidence of Grade 3/4 adverse events was comparable between the matched groups (20.6% vs 28.9%, p = 0.52).
Conclusion:
Induction chemoimmunotherapy before CRT is feasible in selected older NSCLC patients, offering survival benefits without significant safety concerns.
Keywords
Introduction
The PACIFIC trial established concurrent chemoradiotherapy (cCRT) followed by durvalumab consolidation as the standard of care for unresectable stage III non-small-cell lung cancer (NSCLC), 1 yet long-term outcomes remain suboptimal with a 5-year overall survival (OS) rate of 42.9%. 2 This therapeutic challenge is particularly pronounced in older patients ⩾65 years, who experience higher rates of treatment-related toxicities, poorer functional reserve, and inferior survival outcomes compared to younger counterparts. 3 Notably, this vulnerable population remains understudied as most clinical trials systematically exclude patients with comorbidities or impaired performance status.
While immunotherapy has revolutionized perioperative management of NSCLC, its potential role in the pre-cCRT setting for unresectable disease remains underexplored.4–6 A recent retrospective analysis by Guan et al. 7 suggested that induction chemoimmunotherapy prior to cCRT may improve progression-free survival (PFS) without significantly increasing toxicity in stage III NSCLC. However, more than two-thirds of the patients in this study were non-older patients, leaving critical questions unanswered regarding this strategy’s applicability to geriatric oncology practice. Furthermore, historical data from research demonstrated no survival benefit with induction chemotherapy prior to cCRT, 8 prompting investigation into whether synergistic effects between chemotherapy and immune checkpoint inhibitors could overcome this therapeutic plateau, specifically in older adults.
To address these knowledge gaps, we conducted a multicenter retrospective study focusing on patients ⩾65 years with unresectable stage III NSCLC. Our dual objectives were to (1) compare survival outcomes between patients receiving induction chemoimmunotherapy versus induction chemotherapy alone prior to cCRT and (2) systematically evaluate the safety profile of this therapeutic sequence in a geriatric population. This investigation provides critical real-world evidence to guide treatment decisions for a demographic that is frequently excluded from prospective clinical trials.
Materials and methods
The reporting of this retrospective study adheres to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 9 The completed STROBE checklist is provided as Supplemental File 1.
Study population
This retrospective analysis enrolled consecutive patients aged ⩾65 years diagnosed with unresectable stage III NSCLC who received CRT with or without induction chemoimmunotherapy at Beijing Chest Hospital Affiliated to Capital Medical University and Hebei PetroChina Central Hospital between January 2014 and June 2024. The inclusion criteria were as follows: (1) histological/cytological confirmation of stage III NSCLC; (2) completion of CRT combined with/without PD-1/PD-L1 inhibitor-based induction therapy. Exclusion criteria included the following: (1) prior malignancy-specific treatment; (2) targeted therapy administration; (3) exclusive induction immunotherapy without CRT; and (4) concurrent/post-radiotherapy immunotherapy. Driver gene status was assessed for all patients as part of the standard diagnostic workup. Universal testing was performed for the primary actionable drivers, EGFR and ALK, in the entire cohort. Any patient with an identified sensitizing mutation in these genes was excluded from the study cohort.
Participants were stratified by treatment modality: the induction chemoimmunotherapy before CRT (I-CRT) group versus those who did not receive induction immunotherapy (Non-I-CRT). The Non-I-CRT group represented the heterogeneous standard-of-care, encompassing patients treated with induction chemotherapy plus CRT (C-CRT) or CRT. For exploratory analysis of cCRT recipients, subgroups were defined as induction chemotherapy followed by cCRT (C-cCRT) and induction chemoimmunotherapy followed by cCRT (I-cCRT). Clinicopathological characteristics, including histopathological subtypes (WHO classification) and TNM staging (8th edition), were extracted from medical records.
Drug treatment
Immunotherapy regimens utilized PD-1/PD-L1 inhibitors approved for NSCLC in China: camrelizumab, nivolumab, pembrolizumab, sintilimab, or tislelizumab. Chemotherapy regimens (platinum doublet-based) were individualized according to histology, performance status, and organ function.
Radiotherapy
All patients underwent computed-tomography-based simulation for radiotherapy planning. The techniques employed included intensity-modulated radiotherapy (IMRT), or volumetric modulated arc therapy (VMAT), at the discretion of the treating radiation oncologist. The clinical target volume (CTV) encompassed the primary tumor and involved regional lymph nodes. An appropriate margin (5–10 mm) was added to the CTV to generate the planning target volume. Radiotherapy was delivered using conventional fractionation (1.8–2.0 Gy per fraction, once daily, five fractions per week). The definitive radiation dose typically ranged from 54 to 66 Gy.
Study outcomes
Primary endpoints were PFS and OS. PFS encompassed disease progression (PD), death, or last follow-up (whichever occurred first); OS was defined from treatment initiation to death/last follow-up. Secondary endpoints included objective response rate (ORR), defined as complete or partial response per RECIST v1.1; locoregional recurrence-free survival (LRFS), measured to first locoregional progression or death; and distant metastasis-free survival (DMFS), measured to first distant metastasis or death. Tumor response was assessed by the treating clinicians and radiologists per RECIST v1.1. Treatment-related adverse events (TRAEs) were graded using CTCAE v5.0. Follow-up protocol included quarterly clinical/radiographic evaluations (CT/PET-CT) for 24 months, transitioning to biannual assessments thereafter.
Statistical analysis
Categorical variables were compared with χ2/Fisher’s exact tests. Survival analyses employed Kaplan–Meier curves with log-rank testing. To further reduce confounding factors, propensity score matching (PSM, 1:1) balanced baseline characteristics between groups using logistic regression. The hazard ratios (HRs) and the corresponding 95% confidence intervals (CIs) were calculated with the Cox proportional hazards model. Sensitivity analyses, including redefining time-zero as the start of CRT and a Cox model with induction immunotherapy as a time-varying exposure, were performed to validate the primary findings and are detailed in the Supplemental Materials. Statistical significance was set at two-sided p < 0.05. Analyses were conducted using R (version 4.0.3; R Core Team, 2020), accessible via the official website: https://www.R-project.org/
Results
Patient allocation and group definitions
A total of 260 consecutive older patients with unresectable stage III NSCLC were included in this analysis and stratified into two primary groups based on the treatment received: 141 patients constituted the I-CRT, and 119 patients constituted the control group Non-I-CRT. Within the Non-I-CRT group, 88 patients received induction chemotherapy followed by CRT (C-CRT), forming the primary comparator for the I-CRT group. The remaining 31 patients in the Non-I-CRT group started with CRT. To minimize confounding, a PSM cohort of 97 patient pairs was generated.
Baseline characteristics
We analyzed 260 consecutive older patients (median age: 69, range: 65–80) with wild-type driver genes who did not receive consolidation immunotherapy. This predominantly male cohort had a median ECOG PS of 1. To enhance the robustness of our comparisons, we performed 1:1 PSM based on the following covariates: sex, age, smoking status, ECOG performance status, WHO histology, disease stage, CRT modality, and radiation dose. After PSM, patient characteristics were well-balanced across all matched variables (Table 1). The balance of these covariates between the I-CRT and Non-I-CRT groups after PSM is visually summarized in Supplemental Figure 1 (Love plot), demonstrating adequate balance across all included variables. Furthermore, we conducted inverse probability of treatment weighting (IPTW) as a sensitivity analysis, which corroborated the primary findings (Supplemental Figure 2). The consistency of survival benefits was also observed in the IPTW-adjusted Kaplan–Meier curves for both PFS and OS (Supplemental Figure 3), reinforcing the validity of our results. A detailed comparison of radiotherapy-specific parameters, including techniques (IMRT, VMAT) and fractionation, is provided in Supplemental Table 1, which confirms their balanced distribution between the groups.
Baseline demographic and clinical characteristics of the patients.
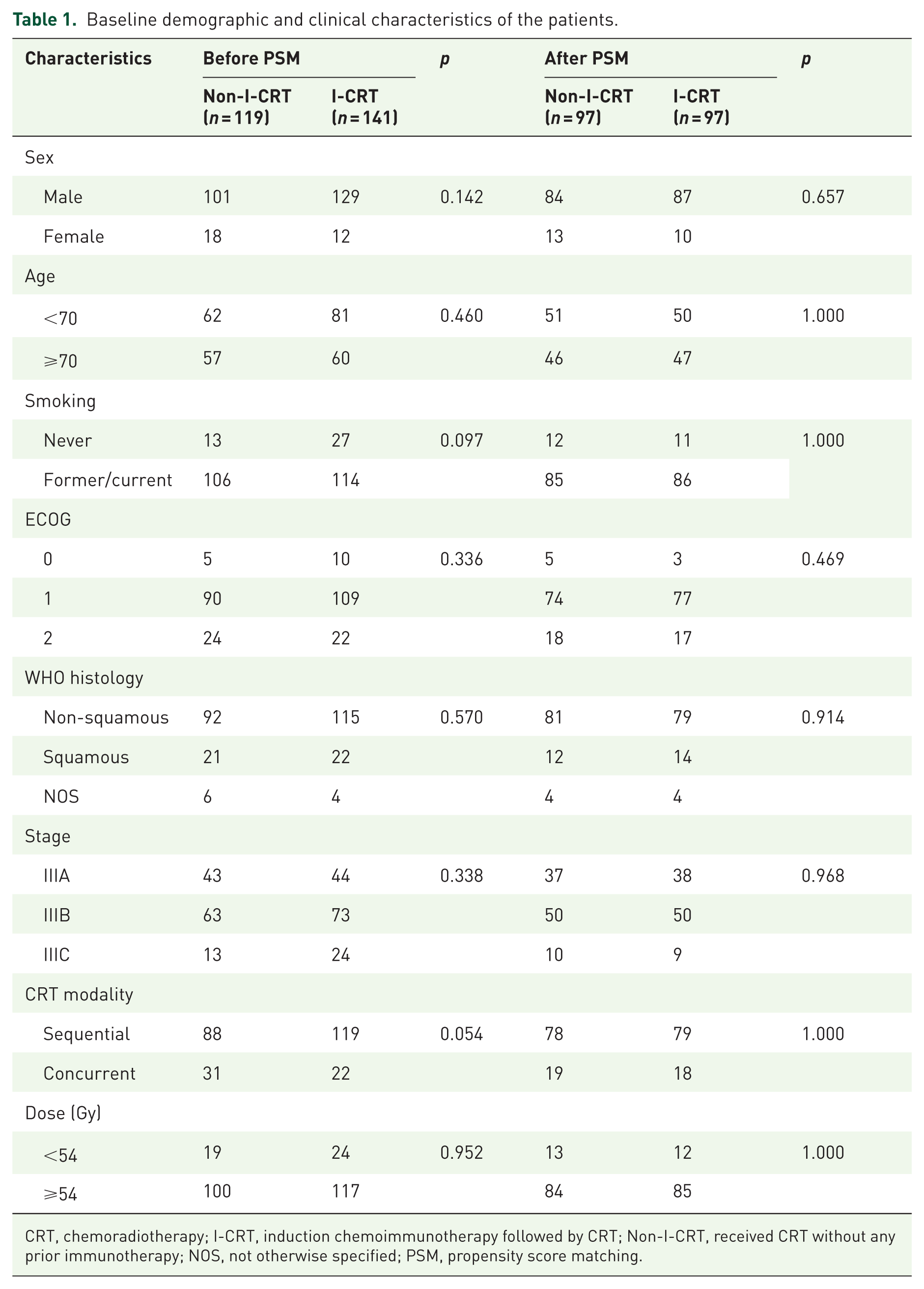
CRT, chemoradiotherapy; I-CRT, induction chemoimmunotherapy followed by CRT; Non-I-CRT, received CRT without any prior immunotherapy; NOS, not otherwise specified; PSM, propensity score matching.
Treatment delivery
The I-CRT group completed a median of four cycles (range: 1–6) of induction immunotherapy and four cycles (range: 2–6) of chemotherapy. Immunotherapy regimens comprised sintilimab (34.0%, n = 48), camrelizumab (24.8%, n = 35), tislelizumab (18.4%, n = 26), pembrolizumab (17.0%, n = 24), and nivolumab (5.8%, n = 8). In the Non-I-CRT group, 88 patients received a median of 4 cycles (range: 2–6) of induction chemotherapy.
Treatment completion rates and chemotherapy backbone regimens were well-balanced between the two groups. The completion rate of definitive CRT was similar (I-CRT: 83% vs Non-I-CRT: 84%, p = 0.82), as was the tolerance to induction therapy without dose reduction (78.0% vs 80.7%, p = 0.60). Carboplatin-based doublets were the predominant chemotherapy backbone in both groups (87.9% vs 86.6%), while the remainder received cisplatin-based regimens (13.5% vs 16.0%, p = 0.57).
Survival outcomes
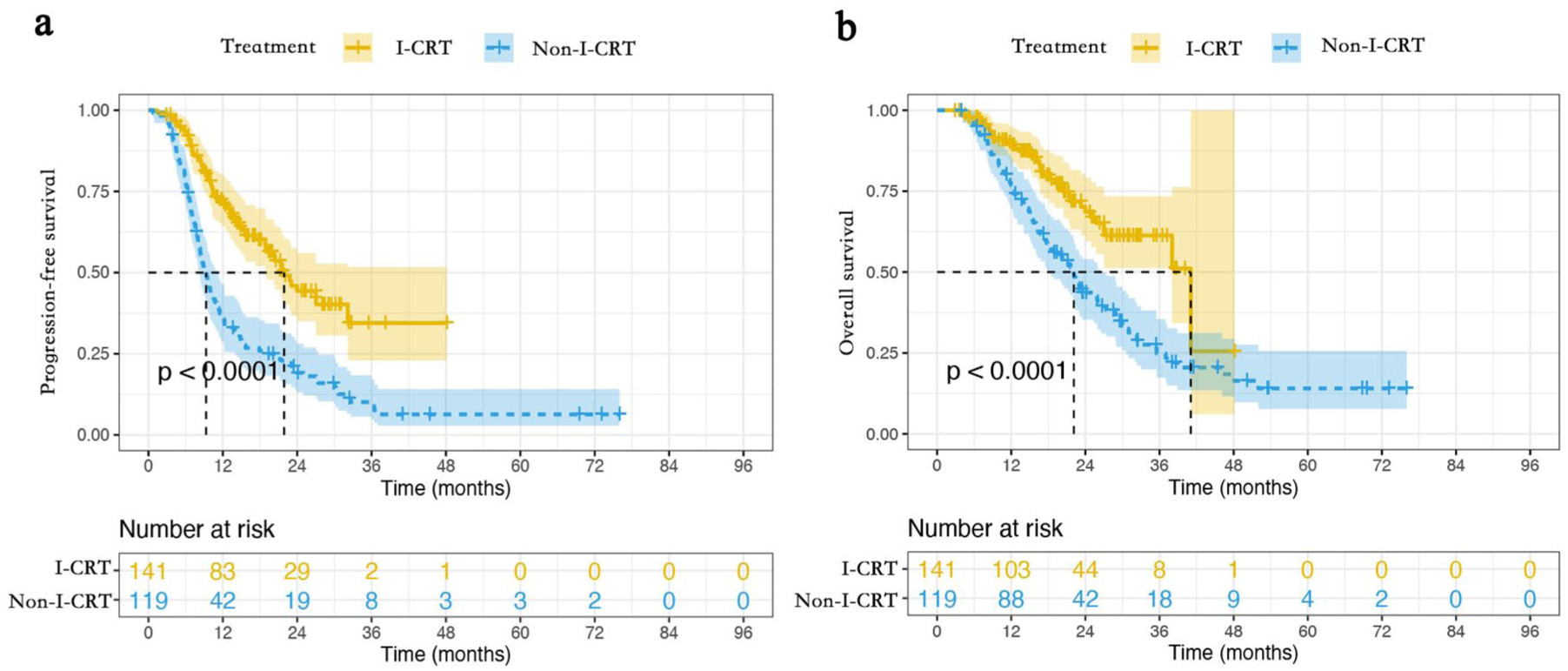
The median follow-up for the I-CRT and Non-I-CRT groups was 19.2 (4.9–54.4) and 22.0 months (5.9–86.5 months), respectively. PD occurred in 167 patients (64.2%), including 63 (44.7%) in the I-CRT group and 104 (87.4%) in the Non-I-CRT group. Mortality events totaled 122 (46.9%), with 37 deaths (26.2%) in the I-CRT group versus 85 (71.4%) in the Non-I-CRT group. Stratified analysis revealed superior outcomes for I-CRT. The median PFS was 23.9 months in the I-CRT group compared to 11.4 months in the Non-I-CRT group. The 1-year PFS rate was 78.2% versus 46.1%, and the 2-year PFS rate was 49.1% versus 21.2% (p < 0.001, Figure 1(a)). The median OS was 46.0 months in the I-CRT group compared to 24.1 months in the Non-I-CRT group. The 1-year OS rate was 90.9% versus 83.7%, and the 2-year OS rate was 71.8% versus 50.4% (p < 0.001, Figure 1(b)). Subgroup analyses demonstrated consistent PFS and OS benefits across most predefined populations (Figures 2 and 3).
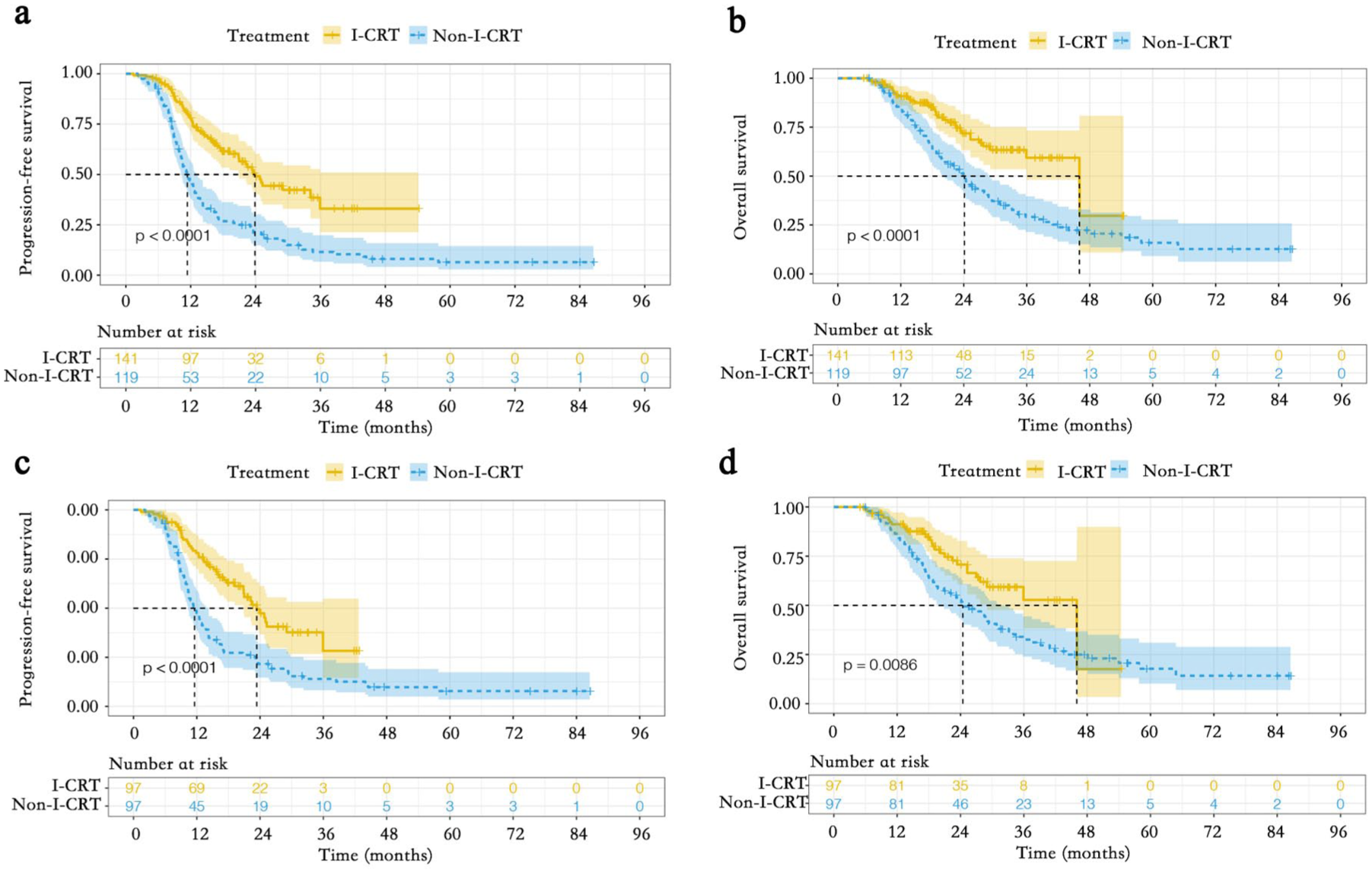
PFS and OS between the I-CRT group and the Non-I-CRT group before and after PSM. (a) PFS from the initiation of treatment before PSM. (b) OS from the initiation of treatment before PSM. (c) PFS from the initiation of treatment after PSM. (d) OS from the initiation of treatment after PSM.
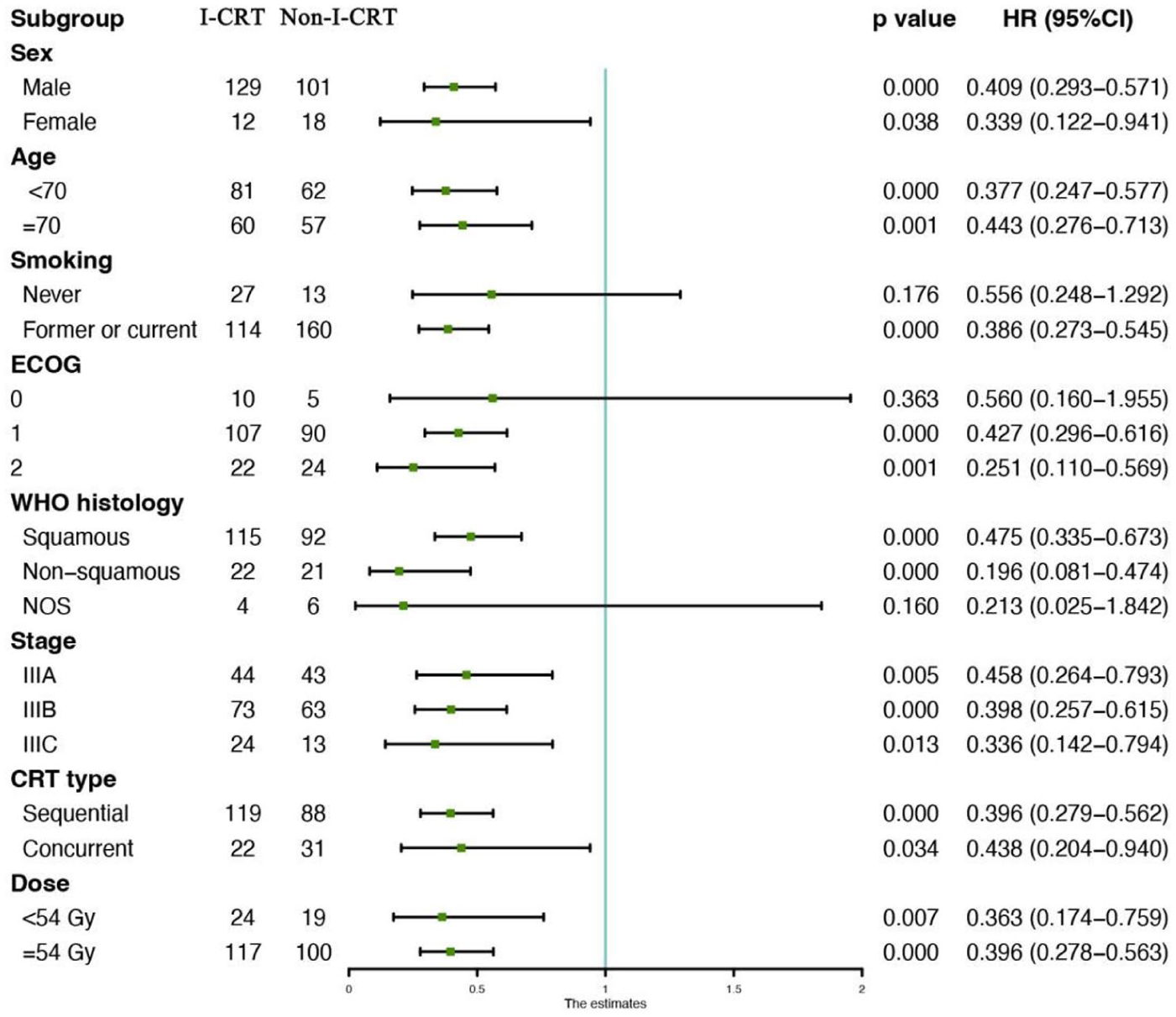
Subgroup analysis of prognostic factors for PFS in the whole population.
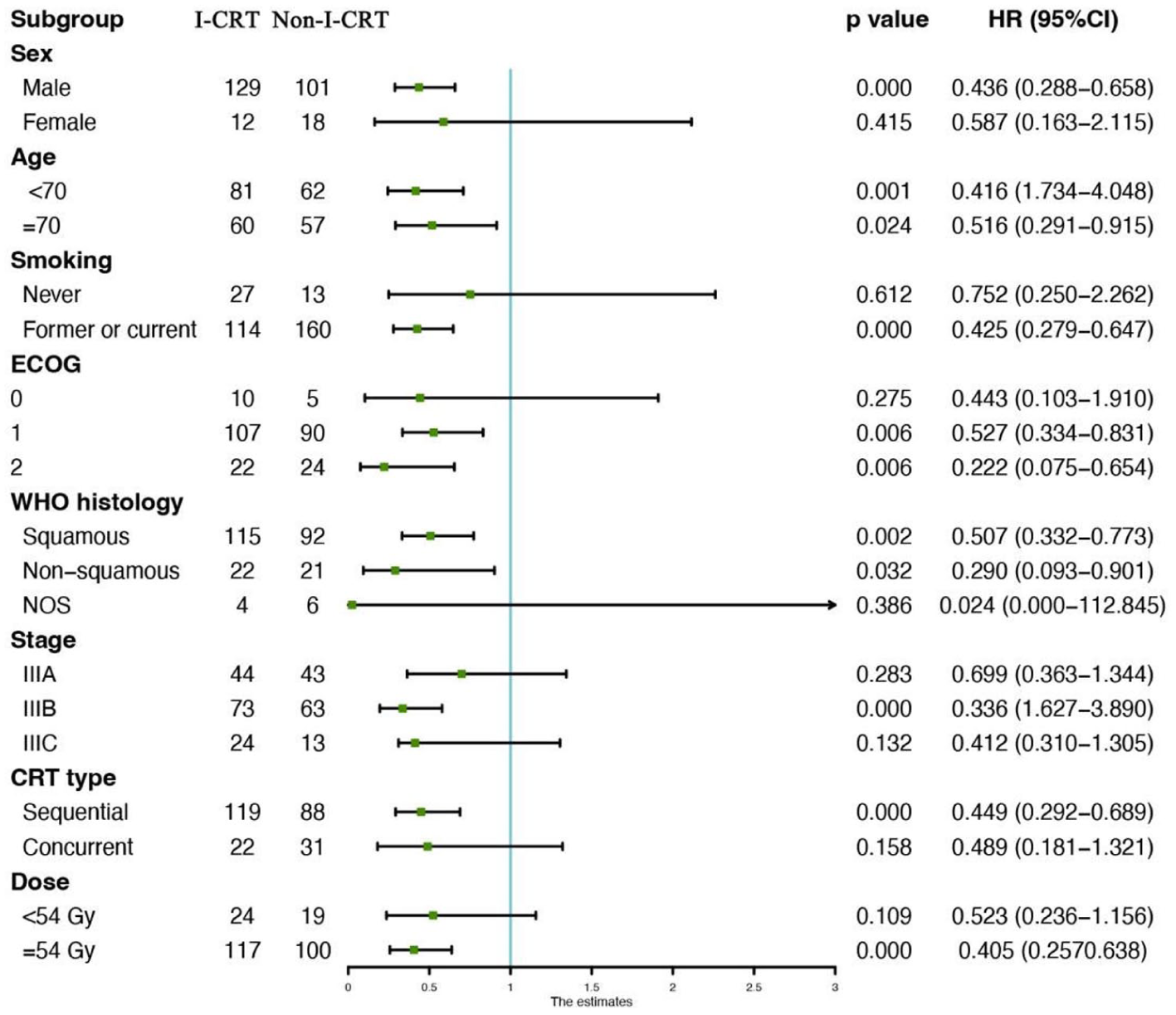
Subgroup analysis of prognostic factors for OS in the whole population.
The survival benefit of the I-CRT strategy was reaffirmed through comprehensive sensitivity analyses. Redefining time-zero as the start of CRT, the I-CRT group maintained significantly improved PFS (HR, 0.51; 95% CI: 0.45–0.68; p < 0.001) and OS (HR, 0.60; 95% CI: 0.41–0.75; p < 0.001), a finding corroborated by Kaplan–Meier analysis with this revised time-zero (Figure 4). Evaluated via a Cox model with induction as a time-varying exposure (Figure 5), the results were equally robust. Consistency was maintained in all comparisons with the entire Non-I-CRT cohort. Furthermore, an era-restricted analysis of patients diagnosed from 2018 onward, which better reflects the modern treatment era, yielded consistent results (Supplemental Figure 4).
PFS and OS between the I-CRT group and the Non-I-CRT group. (a) PFS from the initiation of CRT. (b) OS from the initiation of CRT.

Sensitivity analysis of survival outcomes using a Cox model with induction immunotherapy as a time-varying exposure.
PSM analysis confirmed these trends (I-CRT vs Non-I-CRT). The median PFS was 23.4 months in the I-CRT group compared to 11.6 months in the Non-I-CRT group. The 1-year PFS rate was 78.1% versus 47.5%, and the 2-year PFS rate was 47.4% versus 21.6% (p < 0.001, Figure 1(c)). The median OS was 46.0 months in the I-CRT group compared to 24.4 months in the Non-I-CRT group. The 1-year OS rate was 91.2% versus 85.3%, and the 2-year OS rate was 70.7% versus 52.9% (p = 0.009, Figure 1(d)).
Additional efficacy outcomes
We further assessed the treatment efficacy by analyzing ORR, LRFS, and DMFS. The ORR was significantly higher in the I-CRT group than in the Non-I-CRT group (67.4% (95/141) vs 34.5% (41/119), p < 0.001). The I-CRT regimen was associated with significantly longer LRFS (median, 38.5 vs 25.1 months; HR, 0.69; 95% CI: 0.48–0.98; p = 0.039) and DMFS (median, 31.2 vs 15.8 months; HR, 0.55; 95% CI: 0.40–0.76; p < 0.001) compared to the Non-I-CRT regimen.
Treatment-related adverse events
Comparative analysis revealed a comparable incidence of TRAEs between matched groups, with no statistically significant intergroup differences identified (Table 2). Notably, the survival benefits were attained without additional safety compromises, as reflected in similar rates of severe toxicities (grade 3/4): pneumonitis (11.8% vs 12.1%; p = 1.00), esophagitis (1.7% vs 1.4%; p = 0.86), and hematologic toxicities (16.0% vs 12.1%; p = 0.47). To clarify the timing and etiology of TRAEs, we conducted a stratified analysis. Grade 3/4 pneumonitis occurred predominantly during or after CRT in both the Non-I-CRT (11.8%) and I-CRT (11.7%) groups. In the I-CRT group, 6.4% of these cases exhibited overlapping clinical and imaging features suggestive of both immune-mediated and radiation-induced lung injury. Esophagitis occurred exclusively during or after CRT and was attributed to radiation-induced mucositis. Hematologic toxicities were more frequent during or after CRT, consistent with the cumulative myelosuppressive effects of combined CRT (Supplemental Table 4).
TRAEs between the CRT and I-CRT groups.

I-CRT, induction chemoimmunotherapy followed by CRT; Non-I-CRT, received CRT without any prior immunotherapy; TRAEs, treatment-related adverse events.
Induction chemoimmunotherapy versus induction chemotherapy
Given the limited statistical power of this exploratory subgroup analysis (I-cCRT, n = 22; C-cCRT, n = 31), the following findings should be interpreted with caution. Baseline characteristics were balanced between I-cCRT and C-cCRT cohorts (Supplemental Table 2). Survival outcomes favored I-cCRT, the median follow-up for the I-cCRT and C-cCRT groups was 17.5 months (ranging from 5.7 to 34.6 months) and 19.9 months (ranging from 5.9 to 86.5 months), respectively. As illustrated in Supplemental Figure 5, PFS was significantly superior in the I-cCRT group compared to the C-cCRT group (median NR vs 11.5 months, p = 0.029). The 1-year and 2-year PFS rates were 66.3% versus 48.1% and 54.2% versus 21.2%, respectively. OS was numerically prolonged in the I-cCRT group compared to the C-cCRT group (median NR vs 23.2 months, p = 0.150). The 1-year and 2-year OS rates were 80.8% versus 82.3% and 71.8% versus 48.0%, respectively. The incidence of TRAEs was similar in both groups of older patients, with no statistically significant difference observed (Supplemental Table 3).
Discussion
This multicenter real-world study demonstrates the feasibility, efficacy, and manageable toxicity of I-CRT in older patients (⩾65 years) with unresectable stage III NSCLC—a population underrepresented in clinical trials. Our PSM analysis revealed significant survival benefits for I-CRT over conventional CRT without prior immunotherapy, achieved without a significant increase in severe toxicities. This study specifically investigates I-CRT as an alternative sequence for the many older patients in real-world practice who cannot receive consolidation immunotherapy due to toxicity concerns, financial constraints, or declining performance status.10–12
The exclusion of patients who received consolidation immunotherapy was intentional, allowing us to isolate the effect of the induction strategy. It should be noted that the longer PFS observed in our control group compared to the PACIFIC trial’s placebo arm primarily reflects differing time-zero definitions: our analysis started at first systemic therapy (including induction and CRT phases), whereas PACIFIC began after CRT completion. 13 While this precludes direct cross-trial comparison, it does not affect the internal validity of our cohort analysis, in which both groups were assessed consistently from the same starting point.
The application of cCRT in this demographic is constrained by factors such as extensive tumor volume and reduced treatment tolerance, resulting in CRT remaining the predominant therapeutic approach for most older patients. 14 Notably, even among those receiving consolidation immunotherapy, clinical benefits appear attenuated compared to younger cohorts undergoing sequential CRT. 15 The optimal management strategy for older stage III NSCLC patients remains controversial across clinical studies. While the JCOG 0301 phase III trial and subsequent meta-analyses demonstrated significant survival advantages of cCRT over radiotherapy alone despite increased toxicity, conflicting evidence exists.16–18 A multicenter retrospective analysis from the Netherlands Cancer Registry revealed no survival differences between treatment modalities in patients with severe comorbidities. 19 In addition, Miller et al. 20 proposed potential survival benefits of sequential CRT over alternative approaches. These discrepancies highlight the lack of consensus regarding therapeutic optimization for older NSCLC patients, underscoring the critical need for personalized treatment strategies tailored to geriatric oncology populations.
Building on emerging evidence from neoadjuvant immunotherapy in resectable NSCLC, our study pioneers the investigation of upfront immunotherapy timing in unresectable disease. The induction approach offers distinct mechanistic advantages: (1) early targeting of micrometastatic disease through systemic immune activation; (2) tumor volume reduction, facilitating subsequent radiation planning; (3) biological selection of immunotherapy-responsive patients before definitive local therapy.21,22 Notably, our protocol requiring only four median immunotherapy cycles (vs 12–24 months in consolidation regimens) significantly enhanced treatment feasibility in this vulnerable population.
Despite recommending consolidation immunotherapy to all eligible older patients, a substantial proportion declined due to financial constraints, concerns about adverse events, or anticipated challenges with treatment adherence over the prolonged 1- to 2-year duration. 11 Given the limited number of older patients who ultimately received consolidation immunotherapy and to minimize confounding effects from this intervention, we excluded those who underwent consolidation therapy. This approach enabled isolated evaluation of induction chemoimmunotherapy as a standalone therapeutic strategy. Remarkably, even without subsequent consolidation, induction chemoimmunotherapy demonstrated significant clinical benefits. The addition of a median of four induction chemoimmunotherapy cycles doubled both median PFS and 2-year PFS rates compared to CRT in older patients with unresectable stage III NSCLC, while also yielding meaningful OS improvements. These findings suggest that larger-scale studies could potentially validate these survival trends with enhanced statistical power.
The risk associated with TRAEs remains a key area of concern. Existing literature has shown that the concurrent use of immunotherapy and CRT has the potential to elevate the prevalence of pneumonitis among older patients.23,24 In this study, safety analyses revealed three key observations: First, the pneumonitis incidence (12.1% grade 3/4) remained within acceptable ranges despite immunotherapy-radiation overlap. This figure was marginally higher than the 9.3% reported by Wang et al. 25 and the 4.4%–6.0% incidence observed in the real-world application of the PACIFIC regimen.26,27 Notably, in this study, 15.6% of the older patients who received induction chemoimmunotherapy also received cCRT, a factor that might have influenced this outcome. Second, hematologic toxicity profiles aligned with expectations for platinum-based regimens, with no unexpected synergistic toxicity signals. Third, in the I-CRT subgroup proceeding to cCRT (n = 22). These results suggest that the toxicity of induction chemoimmunotherapy in combination with CRT, or even cCRT, is tolerable for older patients.
It is important to note that the term “older” in oncology lacks a universal definition. While we used the ⩾65 years threshold, consistent with many geriatric oncology studies, 28 and our cohort’s median age of 69 years represents a clinically relevant older population, chronological age alone is an imperfect metric. The lack of comprehensive geriatric assessment (CGA) data is a limitation, and future studies should incorporate CGA to better define fitness for treatment. 29
This study has several limitations. First, its retrospective nature introduces potential selection bias. Second, the focus on patients aged ⩾65 years limits generalizability to younger populations. Third, the subgroup analysis of patients receiving cCRT was underpowered due to the small sample size, and those results require validation in larger cohorts. Fourth, unmeasured confounders (e.g., PD-L1 expression, comorbidities) may persist despite PSM. In addition, for patients in the I-CRT group, the overlapping and synergistic toxicities between immunotherapy and radiotherapy often made it difficult to definitively attribute specific adverse events (notably pneumonitis) to a single etiology (immune-mediated vs radiation-induced), which may limit the mechanistic interpretation of the safety profile in this subgroup. Finally, the exclusion of consolidation immunotherapy precludes direct comparison with the PACIFIC regimen. Despite its limitations, this multicenter study with rigorous methodology provides valid evidence. Future randomized trials are needed to confirm whether induction chemoimmunotherapy matches or enhances the efficacy of consolidation therapy, offering a new perspective for treating this growing patient population.
Conclusion
This real-world study suggests that induction chemoimmunotherapy is a potentially safe and effective frontline strategy for selected older patients with unresectable stage III NSCLC. Compared to induction chemotherapy, this approach demonstrated superior tumor response and survival outcomes, supported by rigorous propensity-adjusted and sensitivity analyses. The manageable toxicity profile warrants prospective validation in randomized trials. Future efforts should focus on identifying predictive biomarkers and optimizing toxicity management to personalize this strategy for the vulnerable geriatric population.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251414132 – Supplemental material for Efficacy and safety of induction chemoimmunotherapy in older patients with unresectable stage III NSCLC: a real-world multicenter study
Supplemental material, sj-docx-1-tam-10.1177_17588359251414132 for Efficacy and safety of induction chemoimmunotherapy in older patients with unresectable stage III NSCLC: a real-world multicenter study by Xingyu Du, Huan Li, Song Guan, Hui Wang, Li Wen and Cuimeng Tian in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251414132 – Supplemental material for Efficacy and safety of induction chemoimmunotherapy in older patients with unresectable stage III NSCLC: a real-world multicenter study
Supplemental material, sj-docx-2-tam-10.1177_17588359251414132 for Efficacy and safety of induction chemoimmunotherapy in older patients with unresectable stage III NSCLC: a real-world multicenter study by Xingyu Du, Huan Li, Song Guan, Hui Wang, Li Wen and Cuimeng Tian in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-3-tam-10.1177_17588359251414132 – Supplemental material for Efficacy and safety of induction chemoimmunotherapy in older patients with unresectable stage III NSCLC: a real-world multicenter study
Supplemental material, sj-docx-3-tam-10.1177_17588359251414132 for Efficacy and safety of induction chemoimmunotherapy in older patients with unresectable stage III NSCLC: a real-world multicenter study by Xingyu Du, Huan Li, Song Guan, Hui Wang, Li Wen and Cuimeng Tian in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
