Abstract
Background:
The standard of care for unresectable, locally advanced non-small-cell lung cancer (LA-NSCLC) is chemoradiotherapy (CRT) followed by durvalumab, based on the PACIFIC study. Although multiple Japanese phase II studies have shown high efficacy and tolerability of CRT with cisplatin plus S-1 (SP), no prospective study using durvalumab after SP-based CRT has been reported.
Objectives:
We conducted a multicenter phase II study of this approach, the interim analysis of which showed a high transition rate to durvalumab consolidation therapy. Here, we report the primary analysis results.
Design:
In treatment-naïve LA-NSCLC, cisplatin (60 mg/m2, day 1) and S-1 (80–120 mg/body, days 1–14) were administered with two 4-week cycles with concurrent thoracic radiotherapy (60 Gy) followed by durvalumab (10 mg/kg) every 2 weeks for up to 1 year.
Methods:
The primary endpoint was 1-year progression-free survival (PFS). The expected 1-year PFS and its lower limit of the 80% confidence interval (CI) were set as 63% and 47%, respectively, based on the results of TORG1018 study.
Results:
In all, 59 patients were enrolled, with 51 (86.4%) proceeding to durvalumab. The objective response rate throughout the study was 72.9% (95% CI: 59.7–83.6%). After median follow-up of 21.9 months, neither median PFS nor OS was reached. The 1-year PFS was 72.5% (80% CI: 64.2–79.2%, 95% CI: 59.1–82.2%), while the 1-year overall survival was 91.5% (95% CI: 80.8–96.4%). No grade 5 adverse events were observed throughout the study. The most common adverse event during the consolidation phase was pneumonitis (any grade, 78.4%; grade ⩾3, 2.0%). Eventually, 52.5% of patients completed 1-year durvalumab consolidation therapy from CRT initiation.
Conclusion:
This study of durvalumab after SP-based CRT met its primary endpoint and found a 1-year PFS of 73% from CRT initiation. This study provides the first prospective data on the prognosis and tolerability of durvalumab consolidation from the initiation of CRT.
Trial registration:
Japan Registry of Clinical Trials, jRCTs031190127, registered 1 November, 2019, https://jrct.niph.go.jp/latest-detail/jRCTs031190127
Introduction
Lung cancer is the leading cause of death related to cancer worldwide. 1 Non-small-cell lung cancer (NSCLC) accounts for 80% of all lung cancer diagnoses. Approximately 30% of patients with NSCLC present with stage III locally advanced disease, while most patients have unresectable disease. 2 Concurrent chemoradiotherapy (CRT), traditionally consisting of platinum-based doublet chemotherapy and concurrent thoracic radiotherapy (TRT), has been established as the standard of treatment for patients with unresectable, locally advanced NSCLC (LA-NSCLC). 3 However, this combined treatment often results in recurrence, with 5-year survival rates of 15–20%, although CRT is intended to be curative.4,5
A recent randomized phase III study, the PACIFIC study, the results of which were published in 2017, led to a revolution in the treatment of LA-NSCLC. In the PACIFIC study, the co-primary endpoints of progression-free survival (PFS) and overall survival (OS) were both significantly prolonged in the durvalumab group compared with those in the placebo group.6,7 The 5-year OSs were 42.9% and 33.4%, and the 5-year PFSs were 33.1% and 19.0% in the durvalumab and placebo groups, respectively. 8 Thus, platinum-based CRT followed by 1-year durvalumab consolidation therapy has been the standard treatment for unresectable LA-NSCLC. However, PFS and OS from the initiation of CRT have remained unclear because the PACIFIC study included patients who did not have disease progression after CRT.
Furthermore, the optimal chemotherapy regimen during the concurrent CRT phase has not been determined. In Western countries, cisplatin and etoposide or weekly low-dose carboplatin and paclitaxel have been commonly used regimens during CRT.9,10 The Japanese guidelines recommend cisplatin and docetaxel and weekly low-dose carboplatin and paclitaxel as CRT chemotherapies for unresectable LA-NSCLC. These have been established as standard regimens on the basis of their benefits in respective phase III studies (OLCSG0007 and WJTOG0105), using the combination regimen of mitomycin, vindesine, and cisplatin as the control arm.11,12 Meanwhile, a cisplatin and S-1 (SP) regimen has also been shown to have high antitumor efficacy and good tolerability in many phase II studies.13–18 Thus, the SP regimen is considered to be one of the best regimens available in Japan, 19 but no patients treated with it were enrolled in the PACIFIC study.
Therefore, we conducted a multicenter, single-arm phase II study of durvalumab consolidation therapy following SP-based CRT for patients with LA-NSCLC. We previously reported an interim analysis indicating that SP-based CRT showed a high transition rate (86.4%) to durvalumab consolidation therapy. 20 Here, we report the results of the primary analysis.
This study is abbreviated as the SAMURAI study (A phase II study of
Materials and methods
Study design
The SAMURAI study was designed as a single-arm phase II study to evaluate the efficacy and safety of durvalumab consolidation therapy following SP-based CRT for treatment-naïve unresectable LA-NSCLC. The study protocol was reported previously. 19
The study protocol was approved by the Certified Institutional Review Board of Yokohama City University on September 17, 2019 (CRB19-002), and was registered in the Japan Registry on Clinical Trials on November 1, 2019 (registration number: jRCTs031190127). All patients provided written informed consent before study entry. The study was funded by AstraZeneca Co., Ltd. The study design, subjects, treatment plan, and evaluation methods were in line with those of the PACIFIC study.
Patient eligibility
This study involved a two-step registration procedure: the first registration was performed before the initiation of CRT, and the second registration was performed after the completion of CRT and before the initiation of durvalumab consolidation therapy.
The main inclusion criteria and exclusion criteria were reported previously. 19 The inclusion criteria for the first registration were as follows: aged 20 years or older; histological or cytological diagnosis of NSCLC; unresectable LA-NSCLC; presence of measurable lesion(s) defined in the Response Evaluation Criteria in Solid Tumor (RECIST) version 1.1; Eastern Cooperative Oncology Group performance status (ECOG PS) of 0 or 1; and preserved bone marrow and organ functions. The inclusion criteria for the second registration were as follows: stable disease (SD), partial response (PR), or complete response (CR) to SP-based CRT; ECOG PS of 0 or 1; preserved bone marrow and organ functions (same as the criteria for the first registration); and SpO2 ⩾ 92% or PaO2 ⩾ 70 Torr (room air).
The main exclusion criteria for the first registration were as follows: interstitial pneumonia or pulmonary fibrosis; active or previous autoimmune disease; active inflammatory disease; active infection or uncontrolled disease; and immunosuppressant medication within 28 days prior to the initiation of study treatment (excluding nasal or inhaled steroids and systemic steroid therapy ⩽10 mg/day prednisolone). The main exclusion criteria for the second registration were as follows: grade ⩾2 pneumonitis prior to registration and uncontrolled disease or active infection.
Study treatment
Chemotherapy with intravenous cisplatin (60 mg/m2, day 1) and oral S-1 (80–120 mg/body, days 1–14) was initiated within 14 days after the first registration. It was specified that two cycles of chemotherapy with a duration of 4 weeks per cycle should be administered before the completion of radiotherapy. The dose of S-1 was determined on the basis of the body surface area as follows: 80 mg/day for <1.25 m2; 100 mg/day for 1.25–1.49 m2; and 120 mg/day for ⩾1.5 m2. TRT was initiated on day 1 of chemotherapy and administered at a dose of 2 Gy once daily for 5 days per week for a total dose of 60 Gy using an X-ray generator (6–10 MV). The extent and method of radiation have been detailed in a previous report. 20 Chemotherapy was terminated when TRT was completed, as in the PACIFIC study.
In patients with SD, PR, or CR to SP-based CRT, durvalumab (10 mg/kg) via intravenous infusion was initiated within 14 days after the second registration. Durvalumab was administered every 2 weeks for up to 1 year. It was specified that durvalumab should be initiated within 42 days after completion of CRT.
Assessment
To evaluate the response, thoracoabdominal computed tomography (CT) and head CT or magnetic resonance imaging were performed before the second registration, and thoracoabdominal CT was performed every four cycles after the initiation of durvalumab. Patients who completed 1 year of durvalumab were required to be monitored for disease progression by thoracoabdominal CT every 3 months until progressive disease (PD). The assessment methods were also reported previously. 20 The response was evaluated according to RECIST version 1.1. All adverse events were graded according to the Common Terminology Criteria for Adverse Events, version 5.0.
We measured tumor programmed death ligand 1 (PD-L1) expression levels, using 22C3 immunohistochemistry assay (Dako 22C3 PharmDx), in tissue samples collected prior to the initiation of CRT in some cases. We also assessed the presence of EGFR mutation and ALK rearrangement.
Statistical analysis
The primary endpoint was 1-year PFS from the first registration. The secondary endpoints were PFS from the first registration; PFS from the second registration; the 18-month OS from the first registration; objective response rate (ORR) throughout the study and in the consolidation phase; the proportions of patients who proceeded to durvalumab, patients who completed TRT, and patients who completed CRT; and adverse events not only in the concurrent CRT phase but also in the consolidation phase. In addition, we evaluated whether the PD-L1 status and the timing of durvalumab initiation after CRT could be predictors of PFS and OS in an exploratory data analysis.
PFS was defined as the time from each registration to confirmation of disease progression or any cause of death. OS was defined as the time from the first registration to any cause of death. Patients who completed TRT were defined as those who completed TRT at the protocol-specified dose of 60 Gy, and patients who completed CRT were defined as those who completed TRT and had two cycles of chemotherapy during TRT.
The sample size design and statistical methods were described previously. 20 Assuming that the duration of CRT was 6 weeks and that a period of up to 6 weeks was required from the end of CRT to the initiation of durvalumab consolidation therapy, it was estimated that a period of up to 3 months was required from the initiation of CRT to that of durvalumab consolidation therapy. We accordingly used the 9-month PFS (64%) in the PACIFIC study as a reference to calculate the expected 1-year PFS in this SAMURAI study. However, the PFS of 64% was based on the assumption that all patients treated with CRT could proceed to durvalumab. A randomized phase II study of SP-based CRT versus cisplatin and docetaxel-based CRT in Japan (TORG1018) found PD and mortality rates during SP-based CRT of 2% and 0%, respectively. 18 Accordingly, the expected 1-year PFS in this SAMURAI study was calculated as 64% of 98% of all patients after excluding patients with PD (2% of all patients), that is, approximately 63% (0.98 × 0.64 = 0.63). Patients who failed to proceed to durvalumab because of adverse events were included to calculate the expected 1-year PFS based on the assumption that they would not experience disease progression or die of their disease thereafter. The threshold 1-year PFS [the lower limit of the 80% confidence interval (CI)] was assumed to be 47% based on the results of TORG1018 study. 18 The necessary sample size was calculated to be 52 patients at α = 0.10 (one-sided) and β = 0.8, and the planned sample size of 58 patients was determined to allow for a dropout rate of approximately 10% (withdrawal of consent and ineligibility). Medians for PFS and OS were estimated by the Kaplan–Meier method. Between-group comparisons were performed by the log-rank test. The 95% CIs for the proportions of patients who proceeded to durvalumab, the ORR, and the disease control rate were calculated using the Clopper–Pearson method. The analysis was carried out using SAS ver. 9.4 (SAS Institute Inc., Cary, NC, USA).
Results
Patient characteristics
A total of 59 patients were enrolled in this study between 16 December 2019 and 18 August 2020, from 22 institutions in Japan. The patient characteristics are shown in Table 1. The median age was 68 years, 86.4% were men and 91.5% were current or former smokers. Two-thirds of patients (61.0%) had a non-squamous histology. The tumor PD-L1 expression level was ⩾50% in 33.9%, 1–49% in 28.8%, and <1% or unknown in 37.3%. Three patients (5.1%) had EGFR mutation and four patients (6.8%) had ALK rearrangement.
Patient characteristics of the intention-to-treat population (n = 59).
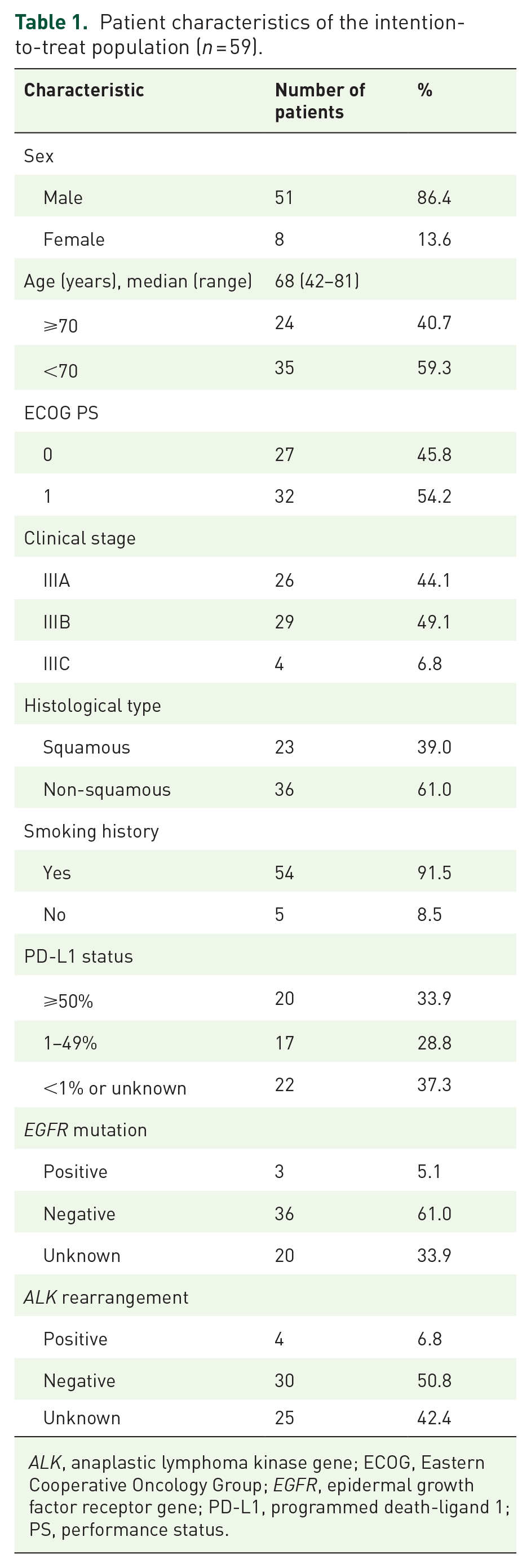
ALK, anaplastic lymphoma kinase gene; ECOG, Eastern Cooperative Oncology Group; EGFR, epidermal growth factor receptor gene; PD-L1, programmed death-ligand 1; PS, performance status.
Patient flow
Patient flow is summarized in Figure 1. Among all 59 enrolled patients, 8 could not fulfill the eligibility criteria for the second registration. Two patients experienced PD and six patients could not proceed to consolidation therapy because of adverse events. Among 51 patients who were able to proceed to consolidation therapy, 20 (39.2%) could not complete 1-year durvalumab consolidation therapy. The reasons for discontinuing consolidation therapy were PD in 12 patients (23.5%); adverse events in 7 patients (13.7%), including grade 2 pneumonitis in 5 patients, grade 3 pneumonitis in 1 patient, and grade 2 thrombocytopenia in 1 patient; and patient’s request in 1 patient. Finally, 31 patients, equivalent to 52.5% from the first registration and 60.8% from the second registration, completed the protocol treatment.

Patient flow in this phase II study of S-1 and cisplatin with concurrent TRT followed by durvalumab for unresectable, LA-NSCLC.
Treatment delivery
TRT at 60 Gy was completed in 57 of 59 patients (96.6%), with a median total radiation dose of 60 Gy (range: 12–60 Gy). In all, 54 patients (91.5%) completed CRT. Median time from the end of CRT to the initiation of durvalumab was 18 days (range: 3–38). In all, 14 patients (27.5%) were administered durvalumab within 14 days.
Efficacy
ORR
The antitumor efficacy is summarized in Table 2. The ORR throughout the study was 72.9% (95% CI: 59.7–83.6%), whereas that in the consolidation phase was 45.1% (95% CI: 31.1–59.7%). The ORR during the CRT phase was 62.7% (95% CI: 49.1–75.0%), as we reported previously. 20
Antitumor efficacy.

CI, confidence interval; CR, complete response; ORR, objective response rate; PD, progressive disease; PR, partial response; SD, stable disease.
PFS and OS from the first registration
After a median follow-up time of 21.9 months (range: 2.2–27.0), median PFS from the first registration was not reached with a 1-year PFS of 72.5% (80% CI: 64.2–79.2%, 95% CI: 59.1–82.2%) (Figure 2). Median OS from the first registration was also not reached with 1-year OS and 18-month OS of 91.5% (95% CI: 80.8–96.4%) and 86.4% (95% CI: 74.7–93.0%), respectively (Figure 3).
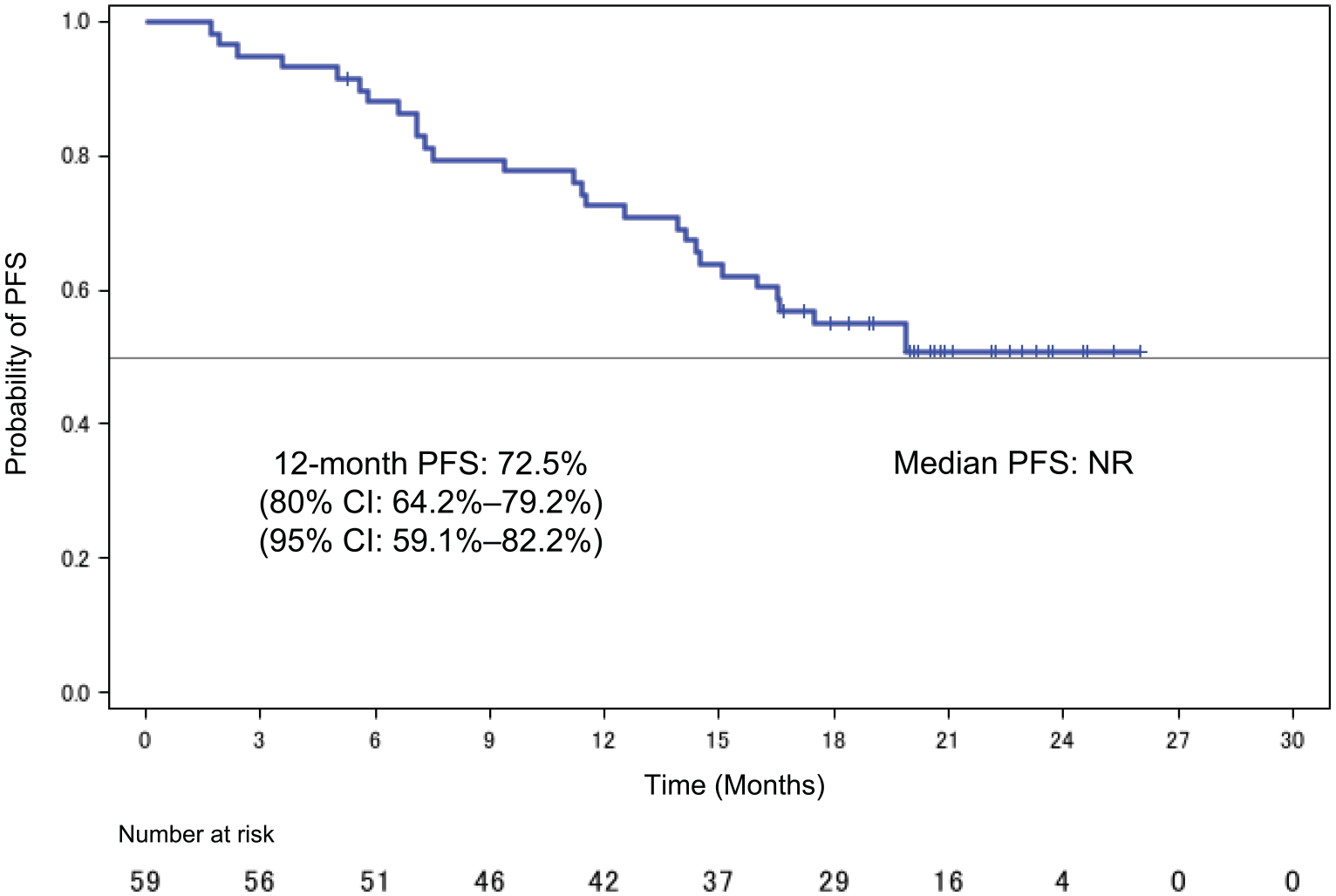
Kaplan–Meier curve for PFS from the first registration in the intention-to-treat population.

Kaplan–Meier curve for OS from the first registration in the intention-to-treat population.
In an exploratory data analysis, PD-L1 status was not a predictor of PFS and OS in the current follow-up period despite adjustment for other clinical factors, including sex, age, PS, smoking history, stage, and presence of driver mutation, by multivariate analysis.
PFS and OS from the second registration
After a median follow-up time of 19.8 months (range: 3.5–25.1), median PFS from the second registration was not reached with a 1-year PFS of 70.1% (95% CI: 55.3–80.7%) (Supplemental Figure S1). Median OS from the second registration was also not reached with a 1-year OS of 96.1% (95% CI: 85.2–99.0%) (Supplemental Figure S2).
As an exploratory data analysis, PFS and OS from the second registration in patients who received durvalumab for <14 days or ⩾14 days after CRT were evaluated, and no statistically significant difference was found between the groups (Supplemental Figure S3). Furthermore, this timing of durvalumab initiation after CRT was not a predictor of PFS and OS in the current follow-up period, despite adjustment for other clinical factors, including sex, age, PS, smoking history, stage, and presence of driver mutation, by multivariate analysis.
Post-protocol treatment
During the follow-up period, 28 patients experienced PD, occurring during the CRT phase in two patients, during the consolidation phase in 13 patients, and after protocol treatment in 13 patients. Regarding the patterns of relapse, 14 patients experienced locoregional relapse only, 11 patients experienced distant relapse only, and 3 patients experienced both locoregional and distant relapse. Overall, 22 of the 28 (78.6%) patients received subsequent chemotherapy after PD.
Toxicity analysis
Major toxicities during the CRT phase and the consolidation phase are presented in Table 3. No grade 5 adverse events were observed throughout the study. During the CRT phase, major adverse events of grade ⩾3 were decreased neutrophil count (20.3%), anorexia (10.2%), and febrile neutropenia (8.5%). Pneumonitis was observed in four patients as grades 1–2 (6.8%) and grade ⩾3 (0%). During the consolidation phase, major adverse events were pneumonitis (78.4%), including 76.4% for grades 1–2 and 2.0% for grade 3, hypothyroidism (15.7%), and rash (15.7%).
Hematological and non-hematological major adverse events.

Included are events that were reported in at least 5% of the patients.
CRT, chemoradiotherapy.
Discussion
The SAMURAI study is, to our knowledge, the first prospective trial to evaluate the efficacy and safety of durvalumab consolidation therapy after SP-based CRT in patients with unresectable LA-NSCLC. The 1-year PFS was 72.5% (80% CI: 64.2–79.2%, 95% CI: 66.6–78.4%), which met the primary endpoint.
Our study is notable in that it identifies two main clinical indicators that would be beneficial when CRT followed by 1-year durvalumab consolidation therapy is selected as the treatment strategy for patients diagnosed with stage III LA-NSCLC. First, the prognosis from the initiation of CRT is now more predictable. Since the results of the PACIFIC study, the standard of care for unresectable LA-NSCLC has been CRT followed by 1-year durvalumab consolidation therapy. However, PFS and OS from the initiation of CRT have remained unclear because the PACIFIC study included patients who did not have disease progression after CRT. Therefore, the results of the PACIFIC study cannot be extrapolated to patients diagnosed with stage III LA-NSCLC and thus cannot be used for explaining prognosis to patients and families at the initiation of CRT.
Meanwhile, the benefits of prognostic predictability are not limited to patients receiving CRT. The treatment of stage III LA-NSCLC is an issue that needs to be considered by a multimodal treatment team that includes respiratory surgeons, pulmonary physicians, and radiologists. 21 Actually, discussions about whether surgery or CRT is better for stage III LA-NSCLC have often been performed at this type of team meeting. However, a problem with CRT followed by 1-year durvalumab consolidation therapy is that only data on the 5-year survival rate from the end of CRT are available; for surgery, data are available on the 5-year survival rate from the initiation of treatment.8,22 For this reason, for convenience, the 5-year survival rate from the end of CRT was often used as a reference value for CRT followed by 1-year durvalumab consolidation therapy when the two treatment strategies are compared. However, it is easy to imagine that this would have made a comparison of the two treatment strategies even more difficult in addition to the difficulty of comparison caused by their heterogeneous backgrounds. In our study, the 1-year OS from the first registration before CRT was 91.5%, while the 1-year OS from the second registration after CRT was 96.1%. The reason for this approximately 5% difference is that the transition rate from CRT to durvalumab was not 100%, and disease progression as well as treatment discontinuation due to adverse events occurred during the CRT phase, which may have affected the OS from the initiation of CRT. Therefore, through the SAMURAI study, we insisted on clarifying PFS and OS from the initiation of CRT.
Second, the patient flow from the initiation of CRT to the end of 1-year durvalumab consolidation therapy was clarified. Given a predictable patient flow at the initiation of CRT, our study showed that 86.4% of patients were able to actually receive durvalumab and 52.5% of patients completed 1-year durvalumab consolidation therapy. The ability for physicians and patients to have this perspective prior to the initiation of CRT will be important to both as they strive to complete 1-year durvalumab consolidation therapy in daily practice with the shared goal of achieving completion. In addition, we can now use these results as a basis for exploring what to watch out for during the CRT phase to further improve the transition rate to durvalumab. Indeed, the SP-based CRT may be a unique treatment strategy available only in Japan. However, we believe that the obstacles to the transition from CRT to durvalumab that were identified in the SAMURAI study will provide many references for CRT based on other regimens. 20
Regarding the treatment efficacy in the SAMURAI study, it is difficult to make a relative assessment of the results because our study is a single-arm phase II study. However, for reference, the PACIFIC study showed 1-year PFS of 55.9% (95% CI: 51.0–60.4) and 1-year OS of 83.1% (95% CI: 79.4–86.2) from the registration after CRT, while the SAMURAI study showed 1-year PFS of 70.1% (95% CI: 55.3–80.7) and 1-year OS of 96.1% (95% CI: 85.2–99.0) from the registration after CRT.6,7 Focusing on the 95% CIs of 1-year PFS and 1-year OS, we can see that they overlap in the two studies. Therefore, so far, the results suggest that the treatment efficacy of the SAMURAI study may be comparable to that of the PACIFIC study.
Currently, in Japan, the SP regimen is one of the most commonly used regimens in daily clinical practice in administering CRT followed by 1-year durvalumab consolidation therapy. 19 Thus, our current results finally provide us with the desired evidence to support our daily clinical practice. Although our study had a limited sample size, with reference to the list of regimens used in the PACIFIC study, the sample size varied from 1 to 158 patients among 18 regimens. 6 Assuming that the SP regimen was adopted in the PACIFIC study, this regimen, applied to 59 patients in our study, would be considered to be the fourth most-used regimen. From this perspective, our study appears to be worthwhile. However, we must note that the survival data in our study are limited because of the short follow-up period.
Regarding the treatment safety in the SAMURAI study, there were no significant signs of novel toxicity through SP-based CRT followed by 1-year durvalumab consolidation therapy. However, during the consolidation phase, the frequency of pneumonitis was as high as 78.4% for any grade, although it was only 2.0% for grade ⩾3. In this regard, the frequency of pneumonitis in the PACIFIC study was summarized separately for the global and Japanese populations for reference (Supplemental Table S1), where the frequencies of any-grade pneumonitis in both the durvalumab and the placebo groups of the Japanese population are higher than those in the respective groups of the global population.6,23 It can also be seen that the frequency of pneumonitis in the SAMURAI study is almost the same as that in the durvalumab group of the Japanese population in the PACIFIC study. 23 Furthermore, in the DATE study (presented at the ASCO 2022 annual meeting as a single-arm phase II study of CRT followed by 1-year durvalumab consolidation therapy for Japanese patients (n = 47; with a chemotherapy regimen of the physician’s choice) the frequency of pneumonitis was 78.7% for any grade and 4.3% for grade ⩾3. 24 Therefore, we speculate that the higher frequency of pneumonitis observed in our study is more likely based on ethnic differences rather than on the nature of the SP regimen.
Recently, the results of a meta-analysis comparing the frequencies of any-grade pneumonitis during durvalumab consolidation therapy between Western and Asian populations based on the real-world data have been reported, where those in the former and the latter are 22% (95% CI: 17–26) and 62% (95% CI: 29–94), respectively (p = 0.017). 25 The report also discussed the reasons for the higher frequency of pneumonitis in Asians from the perspective of genetic disparities. For example, the single-nucleotide polymorphism at rs1982073, T869C of the TGFb1 gene was demonstrated to be associated with a significantly lower risk of radiation pneumonitis in American patients with NSCLC treated with definitive radiotherapy, while this polymorphism was not detected in Asian patients.26,27 Currently, a phase III study (PACIFIC-5) of the durvalumab consolidation therapy after CRT for mainly Asian patients is underway. 28 An investigation of the pharmacokinetics and immunogenicity of durvalumab is also included in the analysis plan. In the near future, this phase III study is expected to reveal more details about the efficacy and safety of this approach in Asians.
Our study has several limitations. First, owing to the short follow-up period, the long-term survival data are limited. Furthermore, probably for the same reason, it was not possible to accurately evaluate whether the PD-L1 status and the timing of durvalumab initiation after CRT could be predictors of PFS and OS. To identify the true efficacy of immune checkpoint inhibitor-based therapy, the OS after a long-term follow-up period, including 3-year OS and 5-year OS, should be important. In the future, it will be necessary to clarify such survival data and predictive factors in the SAMURAI study. Second, it may be difficult to extrapolate the patient flow, as clarified by the SP regimen in the SAMURAI study, to treatment with other standard regimens. It is expected that future studies will clarify the patient flow from the initiation of CRT to the end of 1-year durvalumab consolidation therapy, including the transition rate to durvalumab after CRT, reasons for failure to initiate durvalumab, and completion rate of 1-year durvalumab consolidation therapy from the initiation of CRT, even for the standard treatment regimens. Third, patients with unresectable LA-NSCLC represent a heterogeneous population. Fourth, our study could not reveal the driver gene status in all patients. In our study, 5.1% of patients had EGFR mutations, while 33.9% of cases had an unknown mutation status. In the PACIFIC study, EGFR gene mutation-positive cases accounted for about 5% of all cases, which matches the rate in our study. It is possible that some cases with unknown gene mutation status also included EGFR gene mutation-positive cases; however, the primary endpoint was achieved in the analysis including those cases. Therefore, the interpretation of the study efficacy in this patient population requires caution according to the differences in the distribution of stage IIIA-C, the PD-L1 status, and the driver mutation status, among other factors. In fact, the distribution of subjects among the sub-stages was not balanced in the SAMURAI study, with 44.1% classified as stage IIIA, 49.1% as stage IIIB, and 6.8% as stage IIIC.
Conclusion
The SAMURAI study achieved its primary endpoint. The significance of our study is the identification of two main clinical indicators, including the prognosis from the initiation of CRT and the patient flow from the initiation of CRT to the end of 1-year durvalumab consolidation therapy, which would be beneficial when CRT followed by 1-year durvalumab consolidation therapy is selected as the treatment strategy for patients diagnosed with stage III LA-NSCLC.
Supplemental Material
sj-docx-1-tam-10.1177_17588359221142786 – Supplemental material for A phase II study of S-1 and cisplatin with concurrent thoracic radiotherapy followed by durvalumab for unresectable, locally advanced non-small-cell lung cancer in Japan (SAMURAI study): primary analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359221142786 for A phase II study of S-1 and cisplatin with concurrent thoracic radiotherapy followed by durvalumab for unresectable, locally advanced non-small-cell lung cancer in Japan (SAMURAI study): primary analysis by Hisashi Tanaka, Shigeru Tanzawa, Toshihiro Misumi, Tomonori Makiguchi, Megumi Inaba, Takeshi Honda, Junya Nakamura, Koji Inoue, Takayuki Kishikawa, Masanao Nakashima, Keiichi Fujiwara, Tadashi Kohyama, Hiroo Ishida, Shoichi Kuyama, Naoki Miyazawa, Tomomi Nakamura, Hiroshi Miyawaki, Naohiro Oda, Nobuhisa Ishikawa, Ryotaro Morinaga, Kei Kusaka, Nobukazu Fujimoto, Yasushi Fukuda, Masayuki Yasugi, Takeshi Tsuda, Sunao Ushijima, Kazuhiko Shibata, Takuo Shibayama, Akihiro Bessho, Kyoichi Kaira, Kenshiro Shiraishi, Noriyuki Matsutani and Nobuhiko Seki in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pptx-2-tam-10.1177_17588359221142786 – Supplemental material for A phase II study of S-1 and cisplatin with concurrent thoracic radiotherapy followed by durvalumab for unresectable, locally advanced non-small-cell lung cancer in Japan (SAMURAI study): primary analysis
Supplemental material, sj-pptx-2-tam-10.1177_17588359221142786 for A phase II study of S-1 and cisplatin with concurrent thoracic radiotherapy followed by durvalumab for unresectable, locally advanced non-small-cell lung cancer in Japan (SAMURAI study): primary analysis by Hisashi Tanaka, Shigeru Tanzawa, Toshihiro Misumi, Tomonori Makiguchi, Megumi Inaba, Takeshi Honda, Junya Nakamura, Koji Inoue, Takayuki Kishikawa, Masanao Nakashima, Keiichi Fujiwara, Tadashi Kohyama, Hiroo Ishida, Shoichi Kuyama, Naoki Miyazawa, Tomomi Nakamura, Hiroshi Miyawaki, Naohiro Oda, Nobuhisa Ishikawa, Ryotaro Morinaga, Kei Kusaka, Nobukazu Fujimoto, Yasushi Fukuda, Masayuki Yasugi, Takeshi Tsuda, Sunao Ushijima, Kazuhiko Shibata, Takuo Shibayama, Akihiro Bessho, Kyoichi Kaira, Kenshiro Shiraishi, Noriyuki Matsutani and Nobuhiko Seki in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors would like to thank all of the patients who participated in this study, their families, as well as the investigators. The authors express their gratitude to ASCA Corporation for editing a draft of this article.
Declarations
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
