Abstract
Background:
The effect of vulnerability on the outcomes of patients with locally advanced non-small cell lung cancer (NSCLC) undergoing concurrent chemoradiotherapy (CCRT) remains unclear.
Objectives:
To examine the impact of vulnerability on the outcomes in patients with locally advanced NSCLC undergoing CCRT.
Design:
We analyzed data from the Japan Lung Cancer Society Integrated Clinical Trial Database, which included 1288 patients with locally advanced NSCLC treated with CCRT.
Methods:
Vulnerability was defined as meeting one or more of the following: age ⩾75 years; history of chronic obstructive pulmonary disease (COPD) or emphysema; chronic kidney disease with at least one other comorbidity; or cancer cachexia (C-reactive protein >1.0 mg/dL and albumin <3.5 g/dL).
Results:
Among 741 eligible patients, 283 (38.2%) were classified as vulnerable. Vulnerable patients had significantly shorter overall survival (OS; 19.7 vs 27.4 months, p = 0.003), whereas progression-free survival (PFS) did not differ significantly (8.8 vs 10.4 months, p = 0.151). In multivariate analysis adjusted for factors including cisplatin (CDDP) use, the association between vulnerability and OS was attenuated (hazard ratio = 1.221, 95% confidence interval: 0.959–1.555, p = 0.105). The vulnerable patients were significantly less likely to undergo subsequent therapies (60.4% vs 71.6%, p = 0.003). Among the vulnerability components, cachexia showed the strongest association with shorter PFS (8.0 vs 10.5 months, p = 0.009) and OS (16.5 vs 27.4 months, p < 0.001).
Conclusion:
Clinical vulnerability was associated with poorer OS after CCRT, mediated by multiple factors, including reduced CDDP use, lower subsequent therapy rates, and cachexia. Individualized strategies that balance treatment intensity, supportive care, and access to post-CCRT are essential for improving outcomes.
Introduction
Lung cancer is the leading cause of cancer-related deaths in Japan, with over 70,000 deaths annually. 1 Approximately one-third of non-small cell lung cancer (NSCLC) cases are locally advanced, and the standard treatment involves concurrent chemoradiotherapy (CCRT) followed by 1 year of durvalumab administration.2,3 However, some patients are intolerant to CCRT. This vulnerable patient population includes elderly individuals, those with comorbidities, poor performance status, and patients with cancer cachexia, all of whom have low treatment completion rates and a high incidence of severe adverse events that contribute to poor treatment outcomes. 4 Although such patients are occasionally included in clinical trials, the extent of their inclusion and influence on treatment outcomes remains uncertain.
To address this gap, we analyzed data from previous clinical trial databases to assess the effects of vulnerable patient characteristics on treatment outcomes during CCRT.
Patients and methods
Inclusion and exclusion criteria
We used data from the Database Committee of the Japan Lung Cancer Society’s integrated clinical trial database, which contains data on clinical trials on curative CCRT for locally advanced NSCLC. The database comprised 1288 patients. The enrollment periods varied among the included clinical trials, ranging from November 1999–February 2001 to January 2013–October 2016. The details of the included clinical trials have been presented previously. 5
We defined vulnerability as meeting one of the following three criteria: patients aged ⩾75 years or those with a database record of chronic obstructive pulmonary disease (COPD) or emphysema as a comorbidity; patients with chronic kidney disease (CKD; serum creatinine ⩾1.2 mg/dL) 6 and at least one other comorbidity record, including diabetes mellitus, hypertension, arrhythmia, COPD, pulmonary emphysema, or asthma, and patients who had cancer cachexia. Because sufficient data on body weight loss were not available in our database, we used C-reactive protein levels >1.0 mg/dL and serum albumin levels <3.5 g/dL as indicators of cachexia, based on a previous report. 7 We classified a case as vulnerable if it fulfilled at least one of three predefined criteria. In contrast, cases that did not meet any of these criteria were classified as nonvulnerable.
As no standardized definition of vulnerability exists in oncology, and previous literature, including our narrative review, has employed different clinical factors to characterize vulnerable NSCLC populations, we adopted this pragmatic definition suitable for the context of curative CCRT. 4 This study was reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 8 The completed STROBE checklist is available in the Supplemental File 1.
Ethics approval
This study was approved by the Ethics Review Boards of Kyoto Prefectural University of Medicine and each hospital (approval no. ERB-C-3132), and conformed to the principles of the Declaration of Helsinki (2024). Since this was a retrospective study, the need for informed consent was waived by the ethics committee. Patients were given the opportunity to opt out of the official study website.
Endpoints and assessments
Tumor responses were assessed according to the Response Evaluation Criteria in Solid Tumors (RECIST) guidelines. Overall survival (OS) was defined as the period from the date of enrollment to the date of the last observation or death, and progression-free survival (PFS) was defined as the period from the date of enrollment to the date of the last observation or the date of confirmed disease progression or death. The primary endpoint was OS. The secondary endpoint was objective response rate (ORR).
Statistical analyses
Some variables were structurally missing across protocols; therefore, a complete-case analysis was performed. Categorical variables were summarized as frequencies and proportions, and continuous variables were summarized as medians and ranges. Fisher’s exact test and the Mann–Whitney U test were used to assess the differences between groups. Each curve was calculated using the Kaplan–Meier method. OS and PFS were estimated using the Kaplan–Meier method and compared using the log-rank test, and the Cox proportional-hazards model was applied to estimate hazard ratios (HRs) with corresponding 95% confidence intervals (CIs). The proportional hazards assumptions for the presence or absence of vulnerability were evaluated using a complementary log–log plot. Multivariate analysis was performed using a Cox proportional hazards model. The covariates of the multivariate analysis were sex, smoking history, histology, stage, Eastern Cooperative Oncology Group performance status (ECOG-PS), and cisplatin (CDDP) regimen use. Age, commonly used as a covariate, was excluded because it was incorporated into the definition of vulnerability. All statistical tests were two-sided, and statistical significance was set at p < 0.05. Statistical analyses were performed using R version 4.4.3 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Patient characteristics
A total of 1288 patients with NSCLC who received CCRT were assessed. A flow diagram of the study is shown in Figure 1. Of these, 741 were included in the final analysis. A total of 283 patients (38.2%) were classified as vulnerable, whereas 458 (61.8%) were not. Table 1 shows the characteristics of the patients included in this study. In the vulnerable patient group, 228 patients (80.6%) were males, with a median age of 70 years (range: 36–89 years); 249 (88.0%) had a history of smoking; 173 (61.1%) had stage IIIB disease; 107 (37.8%) had ECOG-PS 0; and 92 (32.5%) received a cisplatin-containing regimen. In the nonvulnerable patient group, 372 patients (81.2%) were males, with a median age of 62 years (range: 30–74 years); 394 (86.0%) had a history of smoking; 269 (58.2%) had stage IIIB disease; 249 (54.4%) had ECOG-PS 0; and 252 (55.0%) received a cisplatin-containing regimen. The vulnerable patient group had a significantly higher proportion of older patients (p < 0.001), poor performance status (p < 0.001), squamous cell carcinoma histology (p = 0.009), and non-CDDP regimens (p < 0.001) than the nonvulnerable patient group. Details of the treatment regimens in the vulnerable and nonvulnerable groups are shown in Supplemental Table 1.

Flow diagram of this study.
Patient characteristics.

Alb, albumin; BMI, body mass index; CDDP, cisplatin; COPD, chronic obstructive pulmonary disease; Cre, creatinine; CRP, C-reactive protein; ECOG-PS, Eastern Cooperative Oncology Group performance status; EGFR, epidermal growth factor receptor; Hb, hemoglobin; IQR, interquartile range; LDH, lactate dehydrogenase; Neu, neutrophils; PLT, platelet; SD, standard deviation; WBC, white blood cell.
Treatment response and survival outcomes
The median follow-up duration was 24.4 months (interquartile range, 12.9–41.9), and the longest follow-up time was 200.2 months. In the vulnerable-patient group, 250 PFS events (88.3%) and 226 OS events (79.9%) occurred, whereas in the nonvulnerable-patient group, 385 PFS events (84.1%) and 329 OS events (71.8%) occurred. Vulnerable patients had a lower ORR than non-vulnerable patients who received CCRT (57.6% vs 69.2%, p = 0.002; Supplemental Figure 1).
There were no significant differences in PFS between the vulnerable and nonvulnerable groups (8.8 vs 10.4 months, log-rank test; p = 0.151; Figure 2(a)). On the other hand, the vulnerable group had a significantly shorter OS compared to the nonvulnerable group (19.7 vs 27.4 months, log-rank test; p = 0.003; Figure 2(b)). For both PFS and OS, the complementary log–log plots indicated no evidence of violation of the proportional hazards assumption (Supplemental Figure 2). In the multivariate analysis excluding CDDP regimen use, vulnerable patients had a significantly shorter OS than nonvulnerable patients (HR = 1.423, 95% CI: 1.130–1.792, p = 0.002; Supplemental Table 2). However, when the CDDP regimen was added to the model, vulnerable patients no longer had a significantly shorter OS than nonvulnerable patients, suggesting that treatment selection may mediate this relationship (HR = 1.221, 95% CI: 0.959–1.555, p = 0.105; Table 2). Multivariate analysis also identified stage IIIB (vs IIIA) as an independent predictor of shorter PFS and OS (PFS: HR = 1.255, 95% CI: 1.005–1.567, p = 0.045; OS: HR = 1.377, 95% CI: 1.083–1.750, p = 0.008), whereas the use of CDDP regimens was independently associated with longer PFS and OS (PFS: HR = 0.688, 95% CI: 0.550–0.861, p = 0.001; OS: HR = 0.612, 95% CI: 0.482–0.777, p < 0.001). Sensitivity analyses using mixed-effects Cox models accounting for trial-level clustering showed results consistent with the primary analysis (Supplemental Tables 3 and 4).

Comparison of the Kaplan–Meier curves for (a) PFS and (b) OS between vulnerable and nonvulnerable patients.
Multivariate analysis of PFS and OS.
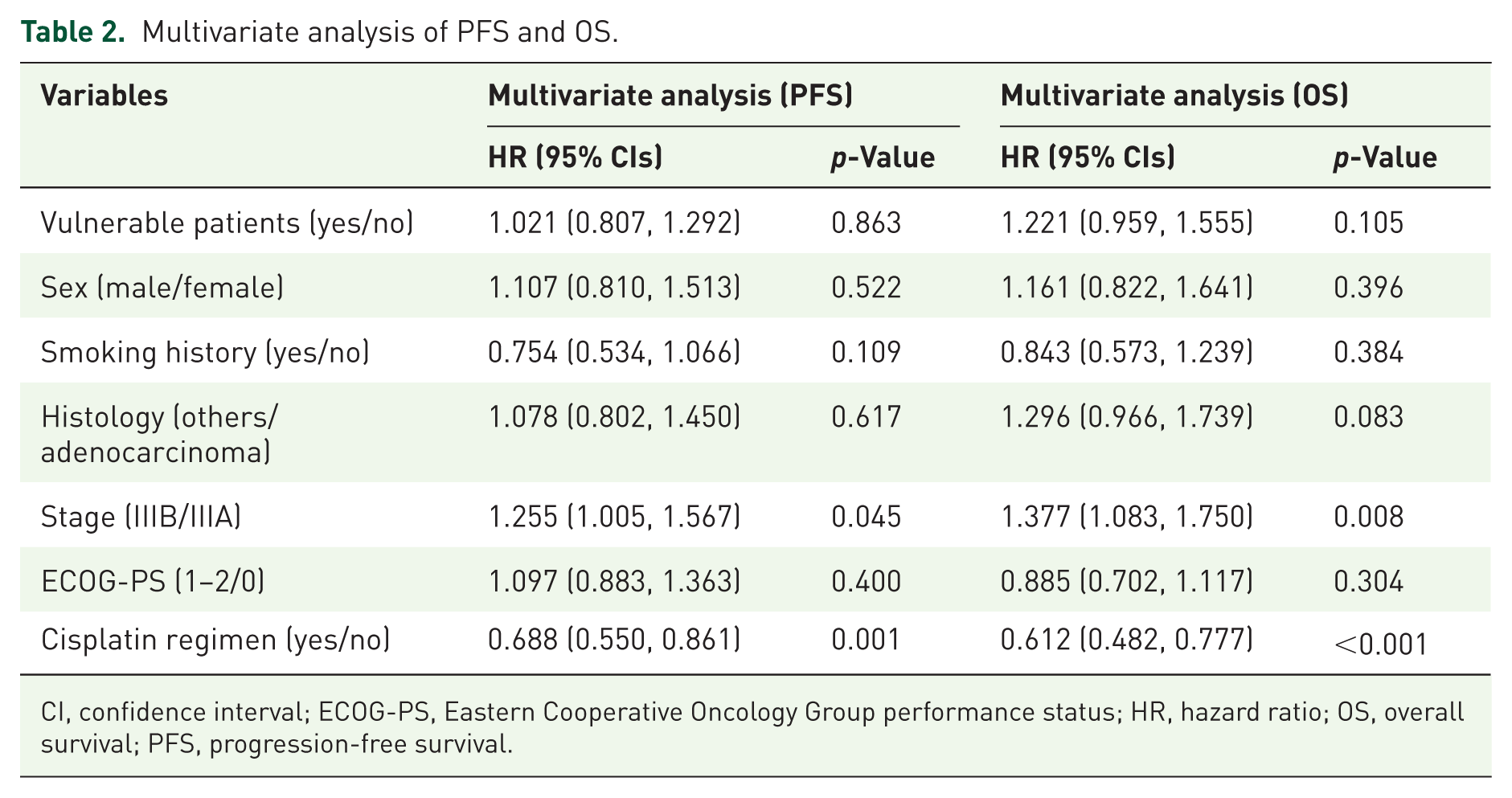
CI, confidence interval; ECOG-PS, Eastern Cooperative Oncology Group performance status; HR, hazard ratio; OS, overall survival; PFS, progression-free survival.
Treatment completion and subsequent therapy
The treatment completion rates for CCRT were not significantly different between vulnerable and nonvulnerable patients (61.2% vs 54.3%, p = 0.075; Supplemental Figure 3(A)). However, vulnerable patients were significantly less likely to receive subsequent therapy (60.4% vs 71.6%, p = 0.003; Supplemental Figure 4(A)). Details of the subsequent treatments are shown in Supplemental Table 5.
Impact of individual vulnerability components
When individual components of vulnerability were analyzed separately, cancer cachexia exhibited the strongest negative prognostic impact, significantly reducing both PFS (8.0 vs 10.5 months; log-rank test, p = 0.009) and OS (16.5 vs 27.4 months; log-rank test, p < 0.001; Figure 3(a) and (b)). Patients aged ⩾75 years or with COPD/emphysema had significantly shorter OS compared with their counterparts (22.5 vs 26.6 months; log-rank test, p = 0.009) but showed no difference in PFS (9.4 vs 10.3 months; log-rank test, p = 0.91; Figure 3(c) and (d)). In contrast, CKD with other comorbidities did not significantly affect either PFS or OS (PFS: 14.2 vs 9.8 months; log-rank test, p = 0.445; OS: 26.1 vs 25.5 months; log-rank test, p = 0.7069; Figure 3(e) and (f)).

Kaplan–Meier curves for PFS and OS stratified by clinical factors: (a, b) cancer cachexia; (c, d) age ⩾75 years or history of COPD/emphysema; and (e, f) CKD and other comorbidities.
Patients with cancer cachexia had the lowest CCRT completion rate (43.8% vs 55.8%, p = 0.007; Supplemental Figure 3(B)). Conversely, patients aged ⩾75 years or with COPD/emphysema paradoxically achieved the highest completion rates (88.2% vs 54.3%, p < 0.001; Supplemental Figure 3(C)). Among patients aged ⩾75 years or with a history of COPD/emphysema, 87 patients (76.3%) received carboplatin monotherapy and 97 patients (85.1%) received carboplatin-based chemotherapy (Supplemental Table 6).
Discussion
In real-world practice, more than half of the patients with stage III NSCLC are ineligible for CCRT due to advanced age, comorbidities, or poor performance status. 9 In contrast, clinical trials generally enroll patients with preserved organ function and appreciable performance status. 10 However, our analysis revealed that, even among trial-eligible patients, approximately 40% met the criteria for clinical vulnerability (age ⩾75 years, COPD/emphysema, CKD with other comorbidities, or cancer cachexia), underscoring the substantial presence of frailty, even within controlled trial populations.
The integrated database included the JCOG0301 trial that specifically targeted elderly patients who received carboplatin-based CCRT. 11 The inclusion of the JCOG0301 trial partially contributed to the higher proportion of elderly and clinically vulnerable patients, thereby enhancing the diversity of our cohort and making it representative of real-world clinical practice. Notably, even after excluding patients enrolled in JCOG0301, 28.1% of the remaining patients were classified as vulnerable, indicating that a substantial degree of clinical vulnerability exists even within trial-eligible populations (data not shown). Vulnerable patients demonstrated significantly shorter OS but comparable PFS to nonvulnerable patients. This discrepancy may reflect treatment-related factors: a higher proportion of lower-intensity regimens, reduced receipt of subsequent therapy (60.4% vs 71.6%), systemic impact of cancer cachexia, and increased noncancer mortality related to frailty and comorbidities. The attenuation of the association between vulnerability and OS after adjusting for CDDP use suggested that treatment intensity and selection, rather than vulnerability itself, primarily mediated the observed survival differences.
Among the individual components, cancer cachexia emerged as the strongest predictor of poor prognosis, significantly affecting both PFS and OS. This is consistent with previous studies that have identified cancer cachexia as a key marker of poor tolerance and survival in NSCLC.12–14 Our definition of vulnerability also corresponds to established geriatric assessment tools such as the G8 and Charlson Comorbidity Index, supporting its clinical validity.15–17
These findings highlight the fact that the prognostic impact of vulnerability is largely modified by treatment strategies and supportive interventions. Tailored approaches, including more tolerable chemotherapy regimens and early nutritional and supportive care, may help improve the outcomes in this high-risk population. For instance, the SPIRAL-RT and DUART studies have demonstrated the feasibility and efficacy of durvalumab after radiation therapy alone in patients ineligible for CCRT.18,19 Further prospective studies are required to optimally integrate these alternative regimens into treatment algorithms for this high-risk population.
It is important to note that the treatment landscape for unresectable stage III NSCLC has substantially evolved with the introduction of consolidation durvalumab following definitive CCRT, as demonstrated in the PACIFIC trial. 3 Given that all clinical trials included in the present analysis were conducted before the approval of durvalumab, our data reflect outcomes after CCRT alone and may not fully correspond to those observed in the contemporary immunotherapy era. However, the concept of patient vulnerability remains clinically important, as vulnerable individuals may also experience challenges in tolerating CCRT–durvalumab treatment sequences. Future studies are warranted to examine whether vulnerability influences treatment tolerance, toxicity, and optimal sequencing strategies under current standards of care.
This study has certain limitations. First, as it used a clinical trial database, patients with severe performance impairment or advanced renal dysfunction were excluded, thereby limiting its generalizability to the most vulnerable populations. Second, several variables were structurally uncollected across the original trial protocols, resulting in missing data that could not be addressed through multiple imputation. Therefore, a complete-case analysis was performed, which may have introduced residual selection bias. Third, detailed physiological and functional parameters (e.g., pulmonary function or physical fitness measures) were not collected, limiting our assessment of their impact on treatment tolerance and prognosis. Similarly, comorbidities such as COPD may have been underdiagnosed or underreported because spirometry was not systematically performed, potentially explaining the low prevalence of COPD in the dataset. 20 Finally, the database included only Japanese patients; therefore, the findings may not be generalizable to other populations. Future studies should validate these findings in large-scale, heterogeneous cohorts and explore strategies to optimize supportive care and post-CCRT treatment sequencing for vulnerable patients.
Conclusion
Clinical vulnerability in patients with stage III NSCLC is associated with poorer OS after CCRT, primarily mediated by reduced treatment intensity, lower rates of subsequent therapy, and the adverse effects of cancer cachexia. Optimizing treatment completion, even with less-intensive regimens, and integrating comprehensive supportive care are crucial for improving outcomes in this high-risk population.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251414535 – Supplemental material for Impact of vulnerability on concurrent chemoradiotherapy outcomes in patients with locally advanced non-small cell lung cancer using an integrated clinical trial database
Supplemental material, sj-docx-1-tam-10.1177_17588359251414535 for Impact of vulnerability on concurrent chemoradiotherapy outcomes in patients with locally advanced non-small cell lung cancer using an integrated clinical trial database by Kenji Morimoto, Tadaaki Yamada, Naoya Nishioka, Nobuyuki Yamamoto, Seiji Niho, Isamu Okamoto, Katsuyuki Hotta, Hiroaki Okamoto, Shunichi Sugawara, Toshio Shimokawa, Yuichi Ozawa, Satoshi Oizumi and Koichi Takayama in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-3-tam-10.1177_17588359251414535 – Supplemental material for Impact of vulnerability on concurrent chemoradiotherapy outcomes in patients with locally advanced non-small cell lung cancer using an integrated clinical trial database
Supplemental material, sj-docx-3-tam-10.1177_17588359251414535 for Impact of vulnerability on concurrent chemoradiotherapy outcomes in patients with locally advanced non-small cell lung cancer using an integrated clinical trial database by Kenji Morimoto, Tadaaki Yamada, Naoya Nishioka, Nobuyuki Yamamoto, Seiji Niho, Isamu Okamoto, Katsuyuki Hotta, Hiroaki Okamoto, Shunichi Sugawara, Toshio Shimokawa, Yuichi Ozawa, Satoshi Oizumi and Koichi Takayama in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-2-tam-10.1177_17588359251414535 – Supplemental material for Impact of vulnerability on concurrent chemoradiotherapy outcomes in patients with locally advanced non-small cell lung cancer using an integrated clinical trial database
Supplemental material, sj-pdf-2-tam-10.1177_17588359251414535 for Impact of vulnerability on concurrent chemoradiotherapy outcomes in patients with locally advanced non-small cell lung cancer using an integrated clinical trial database by Kenji Morimoto, Tadaaki Yamada, Naoya Nishioka, Nobuyuki Yamamoto, Seiji Niho, Isamu Okamoto, Katsuyuki Hotta, Hiroaki Okamoto, Shunichi Sugawara, Toshio Shimokawa, Yuichi Ozawa, Satoshi Oizumi and Koichi Takayama in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
