Abstract
Background:
Once recurrent or persistent locoregional tumour after radical chemoradiotherapy (CRT) for non-small cell lung cancer (NSCLC) is identified, few curative-intent treatment options are available. Selected patients might benefit from surgical salvage. We performed a systematic review of the available literature for this emerging treatment option.
Methods:
A systematic literature search was performed based on the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) statement. Publications about persistent or (locoregional) recurrent disease after radical/definitive CRT for locally advanced non-small cell lung cancer were identified.
Results:
Eight full papers were found, representing 158 patients. All were retrospective series and data were heterogeneous: definition and indication for salvage surgery varied and the median time from radiotherapy to surgery was 4.1–33
Conclusion:
There are limited, low-level, heterogeneous data in support of salvage surgery after radical CRT. Based on this, perioperative mortality appears acceptable and long-term survival is possible in (highly) selected patients. In suitable patients (fit, no distant metastases, tumour appears completely resectable and preferably with confirmed viable tumour), this treatment option should be discussed in an experienced multidisciplinary lung cancer team.
Introduction
The guideline-recommended, curative-intent (radical) treatment for fit patients with locally advanced (stage III) non-small cell lung cancer (NSCLC) is high-dose (typically equivalent to ⩾60 Gy in 2 Gy/fraction) concurrent chemoradiotherapy (CRT). 1 Although this is increasingly being used, 2 failure of this strategy is not uncommon, with a 2-year reported incidence of locoregional and distant failure after 60 Gy of 30.7% and 46.6%, respectively. 3 Once recurrent or persistent locoregional tumour after radical CRT is identified, a second attempt at radical treatment may be possible in selected patients. For example, fit patients with isolated local failure in the previous irradiated high-dose volume, might be candidates for surgical ‘salvage’ and removal of vital tumour. However, such surgery can be technically more challenging and associated with increased risks, particularly when central/hilar structures have been irradiated and centrally located surgery is needed. Not all surgical teams may have experience of operating in these situations. For such reasons, we suspect that salvage surgery is not always considered in all multidisciplinary tumour boards. In this report, we have systematically collected and reviewed the available literature regarding salvage surgery for the treatment of locoregional failure after curative-intent (or definitive, or radical) CRT. These data could be used to aid decision making during multidisciplinary tumour board discussions and to inform shared decision making with patients. Greater awareness of salvage options is likely to increase demand for this treatment.
Methods
Study selection
A literature search was performed based on the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) statement. 4 Publications about persistent or (locoregional) recurrent NSCLC following curative-intent CRT were identified in the bibliographic databases PubMed (Appendix 1, Supplementary Table 1), Embase (Appendix 1, Supplementary Table 2), and the Cochrane Library (via Wiley) (Appendix 1, Supplementary Table 3), from inception to 21 February 2018. Search terms included controlled terms (MeSH in PubMed and Emtree in Embase) as well as free text terms. We used free text terms only in the Cochrane Library. Search terms expressing ‘non-small cell lung cancer’ were used in combination with ‘chemoradiotherapy’ and ‘recurrence’. Only full-length English-language papers were included in the review.
All articles were initially screened by title and then by abstract to progressively exclude nonrelevant reports. The articles selected on the basis of their abstract were accessed in full and further screened. Finally, the reference lists of relevant articles, reviews, and editorials were searched. Articles meeting the following criteria were included in this systematic review: any type of original full-length English-language report concerning surgery for locoregional recurrence or persistent tumour after any form of radical CRT for NSCLC. Debate over article selection was resolved by consensus. All final studies were independently reviewed by two authors to extract relevant information, including article type, number of patients, radiotherapy details, time between radiotherapy and surgery, type of surgery, morbidity, mortality, follow up and survival.
Data analysis
A quantitative data analysis (meta-analysis) was planned; however, the reported data were not sufficiently homogeneous for this (with respect to parameters such as time of follow up, morbidity and overall survival). In addition, due to the small sample sizes and heterogeneity in the aforementioned study characteristics, pooling of data was not appropriate. Therefore, we performed a qualitative overview of the current publications and summarized the most important outcome measures.
Results
Search results
Figure 1 depicts the results of our search strategy (see also Appendix 1). The initial search resulted in 2456 relevant records: 747 from PubMed, 1643 extracted from Embase and 67 from the Cochrane Library. After screening of all titles, 53 abstracts were identified for further screening. An additional 22 records were excluded during this phase, resulting in 31 reports for full text evaluation. Conference abstracts or poster presentations (n = 15), duplicate papers (n = 7), papers with duplicate patients (same author and inclusion period) (n = 1), and non-English-language papers (n = 1), were excluded. Cross checking references from selected papers identified two additional references, of which one was included in the final analysis. At the end of this process, a total of eight full papers were available for this systematic review (Table 1), representing 158 patients.
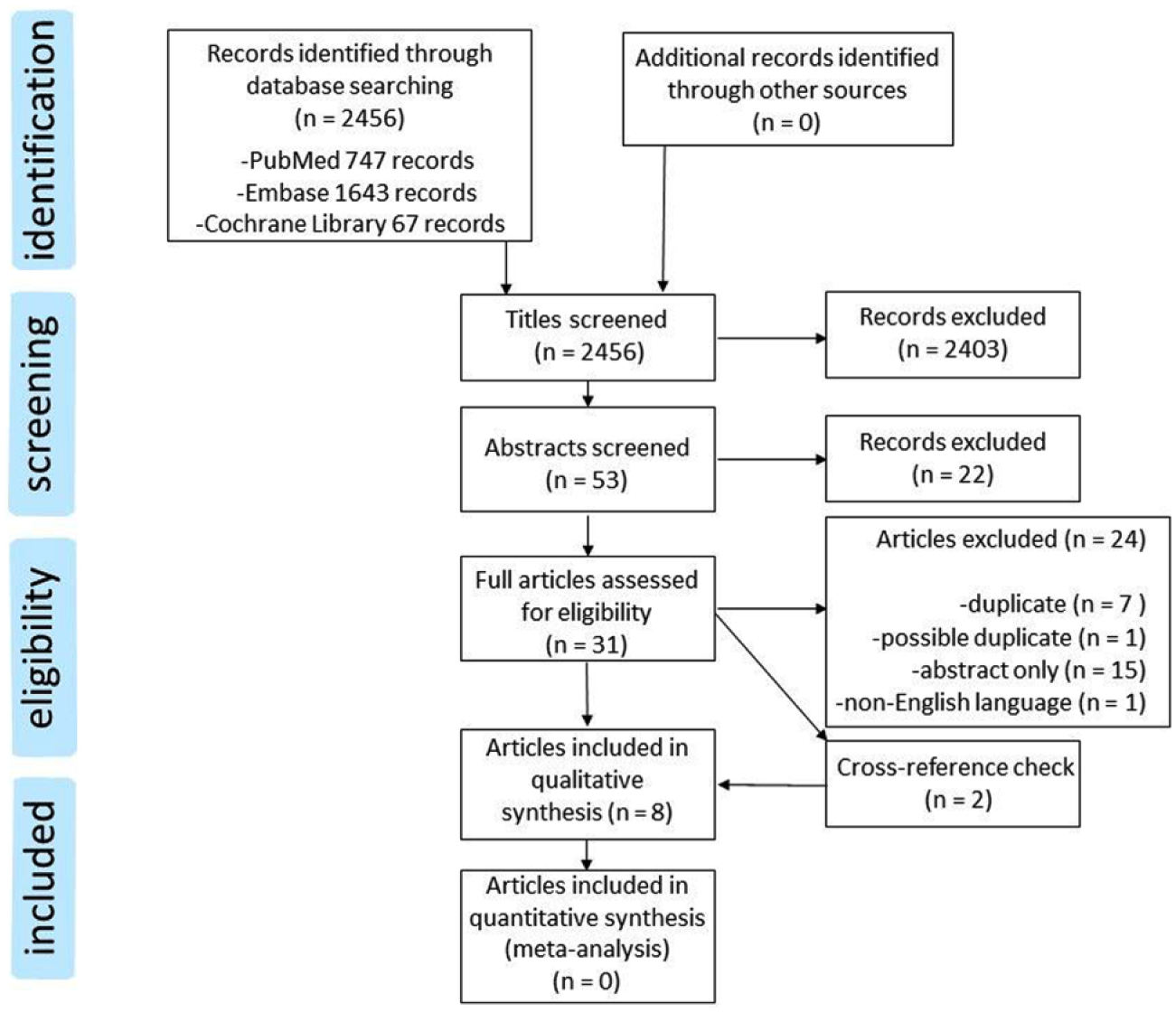
Flowchart depicting study selection according to the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA) statement.
Summary of studies included in the qualitative analysis.

Where reported in weeks, it was calculated in months: months = weeks/4.33.
Values from Results section text, different data presented in Table 2.
Bi-S, bisegmentectomy; BL, bilobectomy; Br-plasty, bronchoplasty; Br Sl, bronchial sleeve; Cc, concurrent; CRT, chemoradiotherapy; CTx, chemotherapy; CW, chest wall; DFS, disease-free survival; L, lobectomy; LN, lymph node; n.r., not reported; P, pneumonectomy; PA, pulmonary artery; Pl Br, plexus brachialis; Pt, (carbo-/cis-/neda-) platinum-based CTx; PV, pulmonary vein; Retro, retrospective series; RT, radiotherapy; S, segmentectomy; SA, subclavian artery; Sq, sequential; SVC, superior vena cava; Sx, surgery; Va Sl, vascular sleeve; VATS, video-assisted thoracic surgery; Vert, vertebral resection; W, wedge.
Study characteristics
All selected articles were retrospective series, published during the previous decade (2008–2018). The articles included 158 patients,5–12 and the first reported patient was operated on in 1995.
7
However, not all of these patients had full-dose (C)RT6,7,11 or local tumour recurrence within the previous radiotherapy volume,5,6 and some reports included patients that were operated within 3
Definitions and indications for salvage surgery.

CT, computed tomography; CRTx, chemoradiotherapy; CTx, chemotherapy; FDG, fluorodeoxyglucose; MRI, magnetic resonance imaging; NSCLC, non-small cell lung cancer; PET, positron-emission tomography; RTx, radiotherapy.
Postoperative survival and recurrence data.
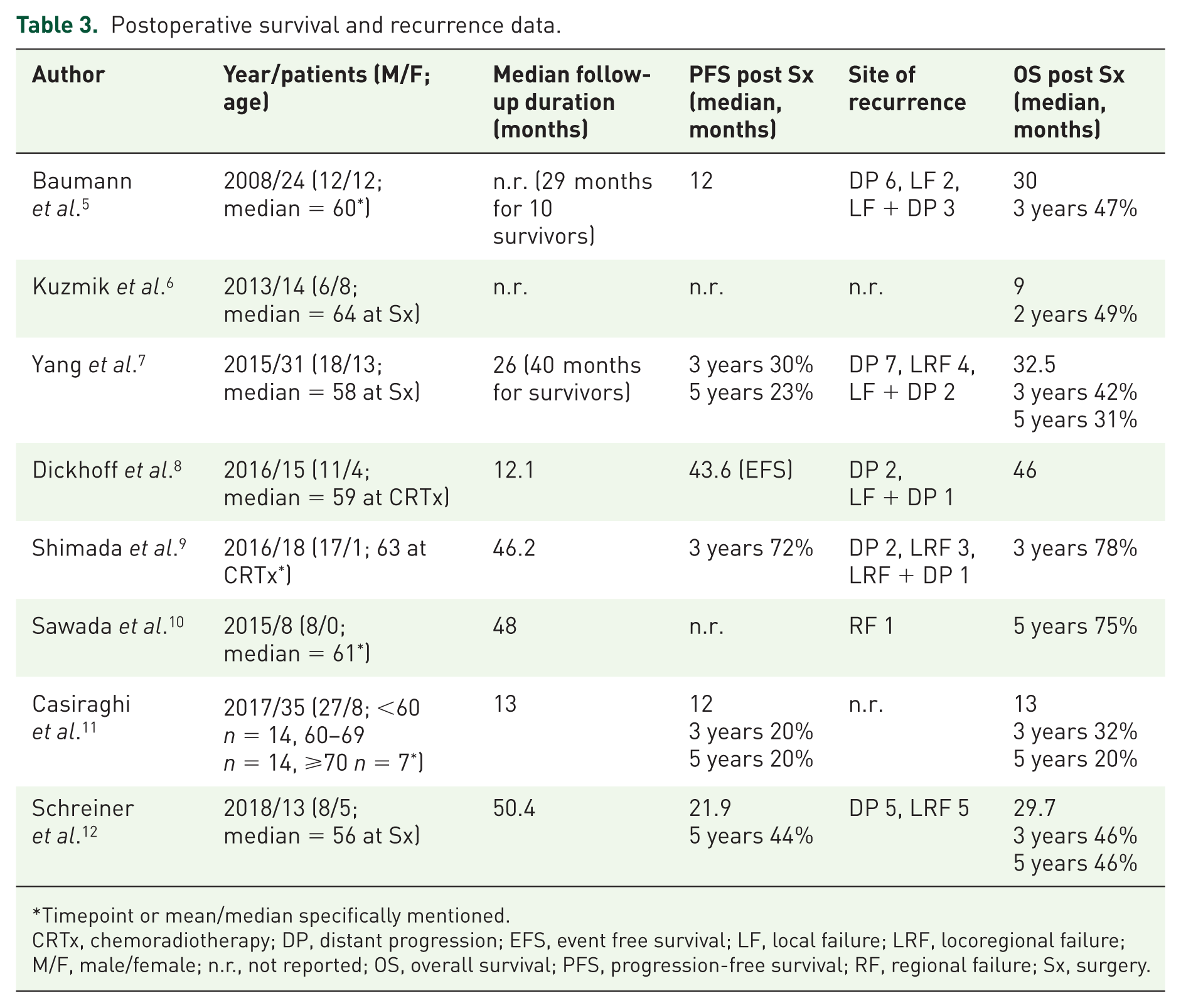
Timepoint or mean/median specifically mentioned.
CRTx, chemoradiotherapy; DP, distant progression; EFS, event free survival; LF, local failure; LRF, locoregional failure; M/F, male/female; n.r., not reported; OS, overall survival; PFS, progression-free survival; RF, regional failure; Sx, surgery.
Primary treatment
The median radiotherapy dose during primary treatment varied from 57 to 66
The median time from radiotherapy to surgery varied between 4.1 and 33
Preoperative mediastinal nodal involvement
In 7/8 of the studies, suspected mediastinal lymph involvement was not considered a reason to exclude patients from salvage surgery.5,7–12 Several authors reported that they performed mediastinoscopy and provided some numerical data: Casiraghi and colleagues performed the procedure in 20/35 (57%) patients and did not specifically describe any related complications; 11 Yang and colleagues performed mediastinoscopy ‘along with lobectomy’ in 15/31 patients (48%), with no specific mention of complications. 7 They had one patient with mediastinal nodal involvement (N2) in their series; this was station five, which had not been evaluated by preoperative mediastinoscopy; Baumann and colleagues performed mediastinoscopy in 19/24 (79%) patients (including one repeat) and described a patient who needed aortic arch replacement following injury during mediastinoscopy. 5
Salvage surgery
For patients undergoing resection (n = 152), a total of 44 pneumonectomies, 11 bilobectomies, 89 lobectomies, 6 segmentectomies and 3 wedge resections were performed. In one study,
5
the same patient was operated on twice (initial lobectomy and then completion pneumonectomy 20
One study reported six patients in whom intraoperative decision making resulted in no resection, 11 and one study did not mention resection margin. 6 Leaving these patients out, the proportion of R0 (complete microscopic) resections was 85–100%. Vital tumour was reported in all studies and ranged between 61% and 100% of patients. Where reported, mediastinal lymph node dissection was performed in a varying number of patients; 14/24 (58%); 5 ‘routinely performed’; 7 all patients had lymph nodes resected but number of stations varied between one and eight (median four); 8 18/18 (100%) with varying extent; 9 8/8 (100%) 10 based on pN staging reported for all patients; ‘all patients’ underwent radical lymphadenectomy with systematic node dissection; 11 ‘all lung resections were followed by systematic hilar and mediastinal lymph node dissection’. 12 Where reported, the incidence of pathologically confirmed N2 disease in the resection specimen was 3, 7 8, 12 13, 8 21 5 and 50%. 10 In addition, some patients who had stage IV disease prior to CRT or salvage surgery were included: the incidence was 3, 11 8, 5 8, 12 13 7 and 14%. 6 There were two patients in one study who had adjuvant chemotherapy for N2 disease found on pathological examination of the resected specimen. 10
Morbidity, mortality and survival
All studies reported perioperative morbidity and mortality in some way (Table 4): the median reported morbidity ranged from 28% to 58% and perioperative mortality varied between 0% in four studies,6,7,9,10 up to 11.4% (4/35 patients) at 90 days. 11 In each of the remaining three studies, one patient died perioperatively.5,8,12 Overall survival (OS) and the recurrence pattern are presented in Table 3. OS was reported in all studies and progression-free survival (PFS) in 6/8 studies. As previously mentioned in the Methods section, pooling OS and PFS data was not appropriate. Except for one study, 10 distant metastases were the most frequent primary cause of progression. None of the articles included quality of life/patient-reported outcome measures.
Details of perioperative morbidity and mortality after salvage surgery.

Requiring rethoracotomy.
This patient subsequently died.
One treated with direct suture and flap cover, one with thoracostomy after 2
ARDS, adult respiratory distress syndrome; CTCAE, common terminology criteria for adverse event; G, grade.
Discussion
To the best of our knowledge, this is the first systematic review of the literature concerning salvage surgery for recurrent or persistent NSCLC after high-dose (C)RT. We identified eight eligible full-length English-language papers, all of which described retrospective series, representing a total of 158 patients. They show that salvage surgery consisting of anatomical lung resection and mediastinal lymph node dissection is feasible, that it can be performed with an acceptable mortality rate (comparable with planned trimodality protocols) despite frequent major resections involving additional organs, and that it can deliver promising clinical outcomes.13,14 However, the 158 patients were accumulated over a combined total of 77.6
When locoregional failure after radical-intent CRT for locally advanced NSCLC occurs, treatment frequently focuses on palliative or supportive care. However, chemotherapy in patients previously treated with platinum-based CRT has been reported to have a response rate as low as 10%,
16
and the role of immunotherapy or targeted therapy in this clinical setting still requires investigation. In a small number of patients, high-dose re-irradiation is an option, and in highly selected patients (in particular those with smaller, noncentral tumours), reported survival (median approximately 13–15
The role of mediastinal lymph node involvement in this clinical scenario is unclear. Five of the eight studies had patients with postoperative N2 disease. This was as high as 50% (4/8) patients in the study of Sawada and colleagues,
10
in which long-term survival was still observed. The data suggest that at the present time, N2 disease is not an absolute contraindication to salvage surgery. However, the disease should be technically completely resectable with acceptable risks. It is important to consider how aggressively the mediastinum should be staged prior to salvage surgery. There is evidence for the safety of mediastinoscopy after induction treatment in therapy-naïve patients;
19
however, it cannot be assumed that this is also true for invasive diagnostics (mediastinoscopy/mediastinotomy) performed ⩾3
Although there have been several reports on the feasibility and safety of planned surgery (trimodality therapy), including pneumonectomy after high-dose CRT in locally advanced NSCLC,21,22 salvage surgery performed a considerable period of time after CRT is a relatively new treatment strategy for patients with recurrent or persistent tumour: all the papers that we identified were published during the last decade and only contain a total of 158 patients. If salvage surgery is being considered, patients should be referred to a sufficiently experienced team that is achieving acceptable outcomes and capable of managing the morbidity that can occur after salvage surgery (although we acknowledge that ‘sufficiently experienced’ and ‘acceptable outcomes’ have yet to be defined). If surgery after neoadjuvant chemotherapy or CRT is used as a benchmark for perioperative mortality, then the data reported here compare reasonably favourably with the 30- and 90-day mortality of 3% and 6.5%, respectively, reported in a National Cancer Database analysis. 23 Specialist teams performing salvage surgery are encouraged to report their results and national lung cancer surgery registries should also capture salvage activity and outcomes.
In our own practice, we define salvage surgery as resection for, preferably confirmed, recurrent/persistent disease in the irradiated volume, at least 12
Although the limited data presented in this systematic review suggest that salvage surgery can be performed with acceptable risks, the treatment paradigm for locally advanced NSCLC is already changing, with the recent publication of data from the phase III PACIFIC study. 24 As a result of this, eligible patients may receive consolidation durvalumab [antiprogrammed death ligand 1 (PD-L1) antibody] after CRT. The safety of salvage surgery after durvalumab has not been reported and the possibility of increased risks cannot currently be excluded. In a recent report by Forde and colleagues, 25 a short course of neoadjuvant anti-PD-L1 treatment using nivolumab was followed by surgery. Although the number of patients treated and the surgical details are limited, they do not specifically mention increased surgical complications. Nonetheless, vigilance is required when combining new medications of various classes for the first time with radical thoracic surgery in previously irradiated tissues. Caution is also appropriate when operating after radiation doses significantly higher than those reported in these papers, especially in central locations. How much the risk of fistula is reduced by buttressing the bronchial stump with a muscle flap or omentum, and whether this increases the safety of operating after very high doses of radiotherapy, is uncertain. Techniques to assess tissue viability after CRT may merit further investigation for identifying a high risk of complications like fistula and stump dehiscence. 26
Finally, reliable identification of local recurrence on follow-up CT and PET/CT imaging after CRT can be challenging and we therefore believe that an attempt should be made to obtain proof of tumour recurrence prior to exposing a patient to the risks of surgery. This is supported by the viable tumour rate of as low as 61% in the research of Yang and colleagues 7 and 64% of evaluable patients in the study by Shimada and colleagues. 9 In the future, the detection of circulating tumour cells or tumour DNA could facilitate early identification of residual or recurrent disease.27,28 In addition, improved imaging techniques, such as PET/MRI, combined with functional tests, like diffusion-weighted imaging, could further improve detection of recurrent tumour and distinguish it from pseudoprogression or fibrotic changes after CRT. 29
Conclusion
There are limited, low-level, heterogeneous data in support of salvage surgery after radical CRT. Based on this, perioperative mortality appears acceptable, and long-term survival is possible in (highly) selected patients. In suitable patients (fit, no distant metastases, tumour appears completely resectable and preferably with confirmed viable tumour), this treatment option should be discussed within an experienced multidisciplinary lung cancer team.
Supplemental Material
Appendix_1 – Supplemental material for Salvage surgery for recurrent or persistent tumour after radical (chemo)radiotherapy for locally advanced non-small cell lung cancer: a systematic review
Supplemental material, Appendix_1 for Salvage surgery for recurrent or persistent tumour after radical (chemo)radiotherapy for locally advanced non-small cell lung cancer: a systematic review by Chris Dickhoff, Rene H. J. Otten, Martijn W. Heymans and Max Dahele in Therapeutic Advances in Medical Oncology
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest statement
MD has received research funding and honoraria/travel support from Varian Medical Systems (Palo Alto, California, USA).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
