Abstract
Background:
Locoregionally advanced nasopharyngeal carcinoma (LANPC) is the predominant stage at diagnosis and is commonly treated with cisplatin-based chemoradiotherapy (CRT). Although this approach achieves excellent locoregional control, distant metastasis remains the leading cause of treatment failure. Immunotherapy with programmed cell death protein 1 (PD-1) inhibitors, supported by the immunogenicity of Epstein–Barr virus-associated disease, has emerged as a promising strategy.
Objectives:
To synthesize evidence from phase III trials on the efficacy and safety of PD-1 blockade combined with definitive chemoradiotherapy in LANPC.
Design:
Systematic review and meta-analysis of randomized controlled trials.
Data sources and methods:
We systematically searched for phase III randomized controlled trials evaluating PD-1 blockade plus chemoradiotherapy versus chemoradiotherapy (with or without placebo) in previously untreated LANPC. Hazard ratios (HRs) with 95% confidence intervals (CIs) were extracted for event-/failure-free survival (EFS/FFS), overall survival (OS), distant metastasis-free survival (DMFS), and locoregional relapse-free survival (LRFS). Pooled effects were estimated with random-effects models, with heterogeneity assessed using I² and prediction intervals. Safety outcomes were summarized descriptively.
Results:
Three trials including 1237 patients were identified. PD-1 blockade significantly improved EFS/FFS (HR 0.66, 95% CI: 0.49–0.89; I² = 18.6%; prediction interval 0.46–0.96), with the greatest effect on DMFS (HR 0.61, 95% CI: 0.43–0.85; I² = 0%). LRFS showed a nonsignificant trend toward benefit (HR 0.67, 95% CI: 0.41–1.11; I² = 43.6%), though a clear advantage was evident in cisplatin-based regimens (HR 0.53, 95% CI: 0.34–0.82). OS data were immature (HR 0.94, 95% CI: 0.60–1.48; I² = 0%). Rates of severe and fatal adverse events were comparable between groups.
Conclusion:
This first meta-analysis of phase III evidence demonstrates that PD-1 blockade combined with cisplatin-based chemoradiotherapy improves disease control in LANPC without added toxicity. Longer follow-up from ongoing studies is needed to establish the OS benefit and optimize treatment strategies.
Trial registration:
PROSPERO CRD-420251147756.
Plain language summary
Nasopharyngeal carcinoma is a type of cancer that starts in the upper part of the throat behind the nose. Many people are diagnosed when the cancer is already advanced but still treatable. The usual treatment is chemotherapy with cisplatin combined with radiotherapy, which controls cancer in the throat and nearby areas. However, many patients still develop cancer spread (metastasis) to distant parts of the body. In recent years, immunotherapy has shown promise for this cancer. Drugs called PD-1 inhibitors work by helping the immune system recognize and attack cancer cells. Since nasopharyngeal carcinoma is often linked to the Epstein–Barr virus, which stimulates the immune system, researchers believed that adding PD-1 inhibitors to standard treatment might improve outcomes. We reviewed and combined the results of three large clinical trials involving 1,237 people with advanced nasopharyngeal carcinoma. Some patients received standard chemoradiotherapy, while others received chemoradiotherapy plus PD-1 inhibitors. We looked at cancer control, survival, and side effects. The results showed that adding PD-1 inhibitors significantly reduced the risk of the cancer returning or spreading. The greatest improvement was seen in preventing distant metastasis. The treatment also seemed more effective when combined with cisplatin chemotherapy. Data on overall survival are not yet mature, meaning it is too early to know if patients live longer. Importantly, adding PD-1 inhibitors did not increase the risk of severe or life-threatening side effects. In summary, this study shows that combining PD-1 immunotherapy with standard treatment improves disease control in advanced nasopharyngeal carcinoma without extra toxicity. Longer follow-up is needed to confirm survival benefits and to refine the best treatment approach.
Keywords
Introduction
Nasopharyngeal carcinoma (NPC) is a malignancy with distinct epidemiological and biological features that set it apart from other head and neck cancers. 1 It arises from the epithelial lining of the nasopharynx and is strongly associated with Epstein–Barr virus (EBV) infection, which plays a central role in pathogenesis across histological subtypes.2,3 NPC is most prevalent in East and Southeast Asia, where it represents a major public health burden. 4 Due to its deep anatomical location and nonspecific early symptoms, diagnosis is often delayed, and the majority of patients present with locoregionally advanced disease (stage III–IVB). 5
For locoregionally advanced NPC (LANPC), concurrent chemoradiotherapy (CCRT) with cisplatin has long been the standard of care, supported by multiple phase III trials and reflected in international guidelines. 6 The introduction of induction chemotherapy followed by chemoradiotherapy (CRT), or CCRT followed by adjuvant chemotherapy, has further improved disease control, particularly in high-risk patients. 7 Despite these advances, however, outcomes remain suboptimal: approximately 20%–30% of patients with LANPC experience disease recurrence or distant metastasis within 3–5 years, underscoring the need for more effective treatment strategies. 8
Immunotherapy offers a biologically compelling approach in this setting. The tumor microenvironment of NPC is characterized by dense lymphocytic infiltration, EBV-driven antigen expression, and high levels of immune checkpoint activation, suggesting susceptibility to programmed cell death protein 1 (PD-1) blockade.9,10 Importantly, CRT itself may enhance antitumor immunity by increasing tumor antigen release, promoting epitope spreading, and reversing local immune suppression—raising the possibility of synergy with checkpoint inhibitors.11,12 Several phase III randomized trials (e.g., CONTINUUM, DIAMOND, DIPPER) have recently evaluated PD-1 inhibitors given in combination with, or following, standard CRT in LANPC, testing strategies such as peri-CRT, adjuvant consolidation, or cisplatin-sparing regimens.13–15 In parallel, several other studies presented findings on recent advances that have transformed the management of NPC, with immunotherapy emerging as a key adjunct to standard cisplatin-based CRT, highlighting the growing role of PD-1/PD-L1 inhibitors in enhancing systemic disease control and improving outcomes in both metastatic and LANCP settings.16,17 Moreover, imaging biomarkers such as [¹8F]FDG PET/CT are gaining relevance in predicting treatment response and guiding patient selection. 18 Together, these developments underscore a shift toward precision immuno-oncology and provide the rationale for our synthesis of phase III evidence on PD-1 blockade in curative-intent NPC therapy.
Therefore, in this context, immunotherapy is increasingly being explored not only in the metastatic setting but also in the curative management of LANPC. Several phase III randomized clinical trials have recently evaluated the integration of PD-1 inhibitors with standard chemoradiotherapy, either during induction, concurrent, or adjuvant phases, with promising improvements in event-free survival (EFS).13–15 These studies address a critical question in clinical practice: whether the addition of immune checkpoint blockade to definitive CCRT can enhance long-term disease control and survival in patients with LANPC. Accordingly, the present meta-analysis aims to systematically review and synthesize the evidence from phase III randomized controlled trials to assess the efficacy and safety of PD-1 blockade in combination with CCRT for the treatment of LANPC.
Materials and methods
This systematic review and meta-analysis was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 statement (Supplemental File 1). The study protocol was prospectively registered with PROSPERO (registration number: CRD 420251147756). A comprehensive literature search was performed in PubMed, SCOPUS, and the Web of Science for studies published up to September 1, 2025. The search strategy combined controlled vocabulary (MeSH terms) and free-text keywords to maximize sensitivity. The following terms were used: (“nasopharyngeal carcinoma” OR “nasopharyngeal cancer”) AND (“immunotherapy” OR “immune checkpoint inhibitors” OR “nivolumab” OR “pembrolizumab” OR “atezolizumab” OR “avelumab” OR “durvalumab” OR “camrelizumab” OR “cemiplimab” OR “tislelizumab” OR “sintilimab” OR “spartalizumab” OR “toripalimab”, “randomized” OR “phase III” OR “trial”; Supplemental File 2). This strategy was designed to ensure inclusion of all relevant randomized clinical trials assessing immune checkpoint inhibitors in NPC.
Inclusion and exclusion criteria
Studies were considered eligible if they were randomized phase III clinical trials published as full peer-reviewed articles in English, enrolling adults (⩾18 years) with histologically confirmed, treatment-naïve, LANPC. Interventions had to involve an anti-PD-1 antibody administered in combination with, or following, definitive CRT, including regimens where PD-1 blockade was given during induction, concurrent, adjuvant, or peri-CRT phases, provided CRT was part of curative-intent therapy. Comparators were standard CRT without PD-1 blockade or placebo plus CRT, as defined by the trial protocol. Eligible studies were required to report hazard ratios (HRs) with 95% confidence intervals (CIs) for EFS, failure-free survival (FFS), progression-free survival (PFS), and/or overall survival (OS), with secondary outcomes such as distant metastasis-free survival (DMFS), locoregional relapse-free survival (LRFS), and safety considered when available. We excluded phase I/II trials, non-randomized or single-arm studies, retrospective analyses, case series, case reports, reviews, editorials, commentaries, and conference abstracts without a peer-reviewed publication; studies of recurrent or metastatic NPC, pediatric (<18 years), non-cancer, or animal populations; trials of PD-1/PD-L1 monotherapy in later-line recurrent/metastatic settings; interventions not based on PD-1 blockade (e.g., EBV-cytotoxic T lymphocytes, vaccines, cytotoxic T-lymphocyte antigen 4 inhibitors); studies lacking extractable HRs and CIs for time-to-event outcomes; publications in languages other than English; and duplicate reports, in which case the most complete or most recent version was retained.
Data collection process
Extracted information included key trial characteristics (name, year of publication, phase, study location, and sample size), patient population, treatment regimens, and follow-up duration. The primary outcome of interest was EFS/FFS, defined as the time from randomization to disease progression, locoregional or distant relapse, initiation of non-protocol therapy, or death from any cause. Secondary outcomes included OS, DMFS, and LRFS, each defined according to the original trial protocols. Safety outcomes included grade ⩾3 treatment-related adverse events (TRAEs), serious adverse events (AEs), fatal events, and immune-related toxicities, when available. For time-to-event outcomes, HRs with 95% CIs were extracted, and for safety outcomes, risk estimates were summarized descriptively. When multiple reports of the same trial were available, the most recent or most mature dataset was used. Methodological quality and risk of bias were assessed using the Cochrane Risk of Bias 2.0 tool (Cochrane, London, United Kingdom), which evaluates five domains: randomization process, deviations from intended interventions, missing outcome data, measurement of outcomes, and selective reporting. Each domain was judged as low risk, some concerns, or high risk of bias. The certainty of the evidence for each outcome was further appraised using the GRADE approach, considering risk of bias, inconsistency, indirectness, imprecision, and publication bias.
Risk of bias assessment
The risk of bias for each included trial was independently assessed by two reviewers (K.S. and K.B.-P.) using the Cochrane Risk-of-Bias Tool for Randomized Trials (RoB 2) tool (Figure 1). Each domain was rated as low risk, high risk, or some concerns according to Cochrane criteria. Any discrepancies in assessment were adjudicated by a third reviewer (A.Go.).

Risk of bias assessment of included trials. Risk of Bias 2.0 evaluation for the three randomized controlled trials (CONTINUUM, DIPPER, DIAMOND) across key domains. All studies were judged as low risk in randomization, deviations, missing data, measurement, and reporting. Overall risk of bias was rated as low for EFS/FFS but some concerns for OS outcomes due to immature data.
Statistical analysis
HRs with corresponding 95% CIs for EFS/FFS, OS, DMFS, and LRFS were extracted from each eligible trial. When multiple analyses were available, the most mature dataset was used. HRs were log-transformed, and standard errors were derived from reported 95% CIs. Pooled estimates were generated using the inverse-variance method under both fixed- and random-effects models (DerSimonian–Laird), with the random-effects model considered primary. Between-study heterogeneity was assessed using Cochran’s Q test (p < 0.10 indicating significance) and quantified with the I² statistic. Prediction intervals were calculated to evaluate the expected range of effects in future studies. Forest plots were used to display individual and pooled HRs on a logarithmic scale. Sensitivity analyses were performed with a leave-one-out approach, sequentially excluding each study to test the robustness of results. Because fewer than 10 trials were included, publication bias was not formally assessed using funnel plots or Egger’s test. All analyses were conducted in R (version 2025.05.1; R Foundation for Statistical Computing, Vienna, Austria) using the meta and metafor packages. A two-sided p < 0.05 was considered statistically significant. AE data were extracted from the full analysis sets of the CONTINUUM, DIPPER, and DIAMOND trials. Safety endpoints included the incidence of grade ⩾3 TRAEs, fatal AEs, and selected high-grade toxicities (e.g., hematologic and hepatic). For each trial, risk ratios (RRs) with 95% CIs were calculated by comparing event rates between the PD-1 blockade plus CRT arm and the CRT-only arm, with CIs estimated using the Wald method. Results with zero events in one or more cells were reported as not estimable without continuity correction. Between-study heterogeneity was assessed descriptively and was expected to be minimal given the uniform treatment setting. Safety outcomes were pooled using the Mantel–Haenszel fixed-effect model, yielding RRs with 95% CIs. Forest plots were generated to display trial-level and pooled estimates.
Results
Literature selection
Our systematic search retrieved a total of 381 records. After removing the duplications (n = 41), we screened the abstracts of the remaining 340 records and excluded 228 records because of the topic irrelevance. We also excluded reviews (86), case reports (n = 3), commentaries (n = 6), editorials (n = 5), studies not in English (n = 3), pediatric population (n = 2), and animal studies (n = 4). We screened the titles and abstracts of the remaining three articles then we evaluated the full texts of the remaining three articles and included all the three randomized clinical trial studies in the meta-analyses. The PRISMA diagram for the article selection process is presented in Figure 2.

PRISMA flow diagram of study selection. Flow of information through the systematic review and meta-analysis. Of 381 records identified, 340 abstracts were screened after removal of duplicates. Following exclusion of ineligible articles, three phase III randomized clinical trials were included in the final analysis.
Study characteristics
Three phase III randomized controlled trials (CONTINUUM, DIAMOND, and DIPPER), enrolling a total of 1237 patients, were included in this meta-analysis. All trials employed 1:1 randomization and were conducted in multicenter settings across China. The median age across studies was approximately 45 years, and over 90% of participants were male, consistent with the epidemiology of NPC in endemic regions. The vast majority of patients had non-keratinizing histology, with baseline EBV DNA positivity reported in most cases. Eligible patients had stage III–IVB locoregionally advanced disease without distant metastases at diagnosis, and randomization was stratified by stage or EBV DNA status to ensure balanced arms, though stratification factors and EBV cutoff values varied across studies. Baseline demographic and disease characteristics were generally well matched between treatment and control groups. The primary endpoint in all trials was EFS or FFS, as defined by each protocol and assessed by independent review. Since all three studies evaluated PD-1 blockade in the curative-intent, non-metastatic setting, consolidation radiotherapy after systemic treatment was not relevant, as definitive chemoradiotherapy formed the treatment backbone in both experimental and control arms. Adequate EFS/FFS events accrued to allow primary endpoint analyses, whereas OS remained a secondary endpoint and was immature in all reports at the time of publication. All three studies were rated as low risk of bias when assessed using the Cochrane RoB 2.0 tool. Details on trial characteristics are presented in Table 1.
Baseline characteristics and clinical outcomes of randomized trials evaluating immunotherapy-based strategies in locoregionally advanced nasopharyngeal carcinoma.

CCRT, concurrent chemoradiotherapy; CRT, chemoradiotherapy; DMFS, distant metastasis-free survival; EBV, Epstein–Barr virus; EFS, event-free survival; FFS, failure-free survival; LANPC, Locoregionally advanced nasopharyngeal carcinoma; LRFS, locoregional relapse–free survival; OS, overall survival; TRAE, treatment-related adverse events.
Treatment efficacy
All three phase III trials (CONTINUUM, DIPPER, DIAMOND) used EFS or FFS as the primary endpoint, analyzed after the prespecified number of events had accrued. In the pooled analysis of 1237 patients, the addition of PD-1 blockade to CRT significantly improved EFS/FFS (HR 0.66, 95% CI: 0.49–0.89, p = 0.006), with low between-study heterogeneity (I2 = 18.6%, Q = 2.46, p > 0.05), indicating consistent treatment effects (Table 2-(A), Figure 3(a)). OS was a secondary endpoint in all trials and remained immature at the time of reporting. In the pooled analysis, PD-1 blockade did not significantly reduce the risk of death (HR 0.94, 95% CI: 0.60–1.48, p = 0.79), with no heterogeneity (I2 = 0%, Q = 0.29, df = 2; Table 2-(B), Figure 3(b)).
Pooled HRs for EFS/FFS and OS in LANPC.
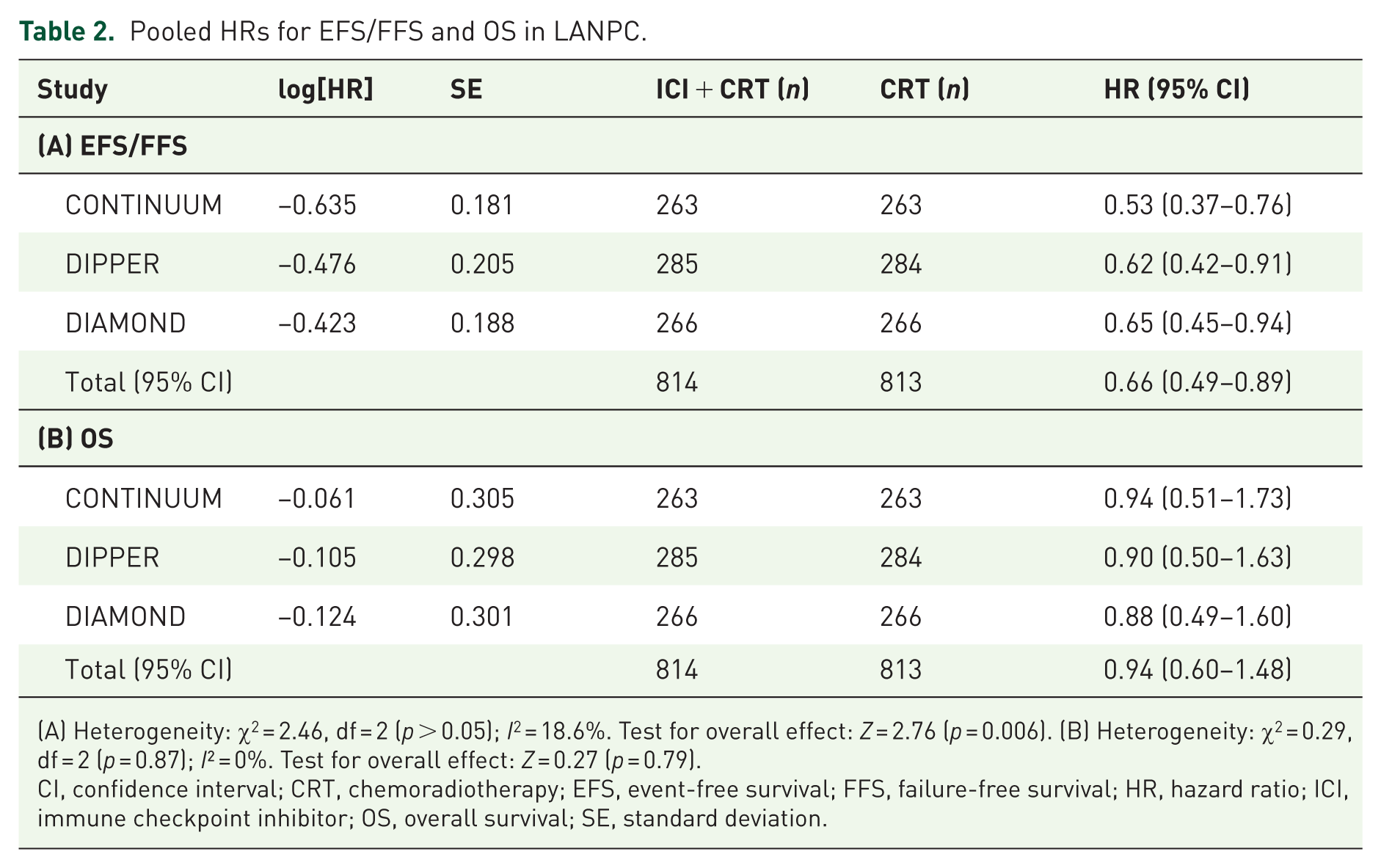
(A) Heterogeneity: χ2 = 2.46, df = 2 (p > 0.05); I2 = 18.6%. Test for overall effect: Z = 2.76 (p = 0.006). (B) Heterogeneity: χ2 = 0.29, df = 2 (p = 0.87); I² = 0%. Test for overall effect: Z = 0.27 (p = 0.79).
CI, confidence interval; CRT, chemoradiotherapy; EFS, event-free survival; FFS, failure-free survival; HR, hazard ratio; ICI, immune checkpoint inhibitor; OS, overall survival; SE, standard deviation.

Pooled effect of PD-1 blockade on EFS/FFS and OS. Forest plots of HRs for (a) EFS/FFS and (b) OS, comparing PD-1 blockade plus chemoradiotherapy versus chemoradiotherapy alone. PD-1 blockade significantly improved EFS/FFS, while OS data remained immature with no statistically significant difference.
Subgroup analyses
Distant metastasis-free survival
The addition of PD-1 blockade improved DMFS in the CONTINUUM and DIPPER trials, while the DIAMOND trial showed no significant difference. Pooled analysis confirmed a significant 39% reduction in the risk of distant relapse (HR 0.61, 95% CI: 0.43–0.85, p = 0.0039), with no evidence of heterogeneity (I² = 0%). These findings support the efficacy of integrating PD-1 blockade in reducing metastatic spread (Table 3-(A), Figure 4).
Pooled HRs for DMFS and LRFS in LANPC.
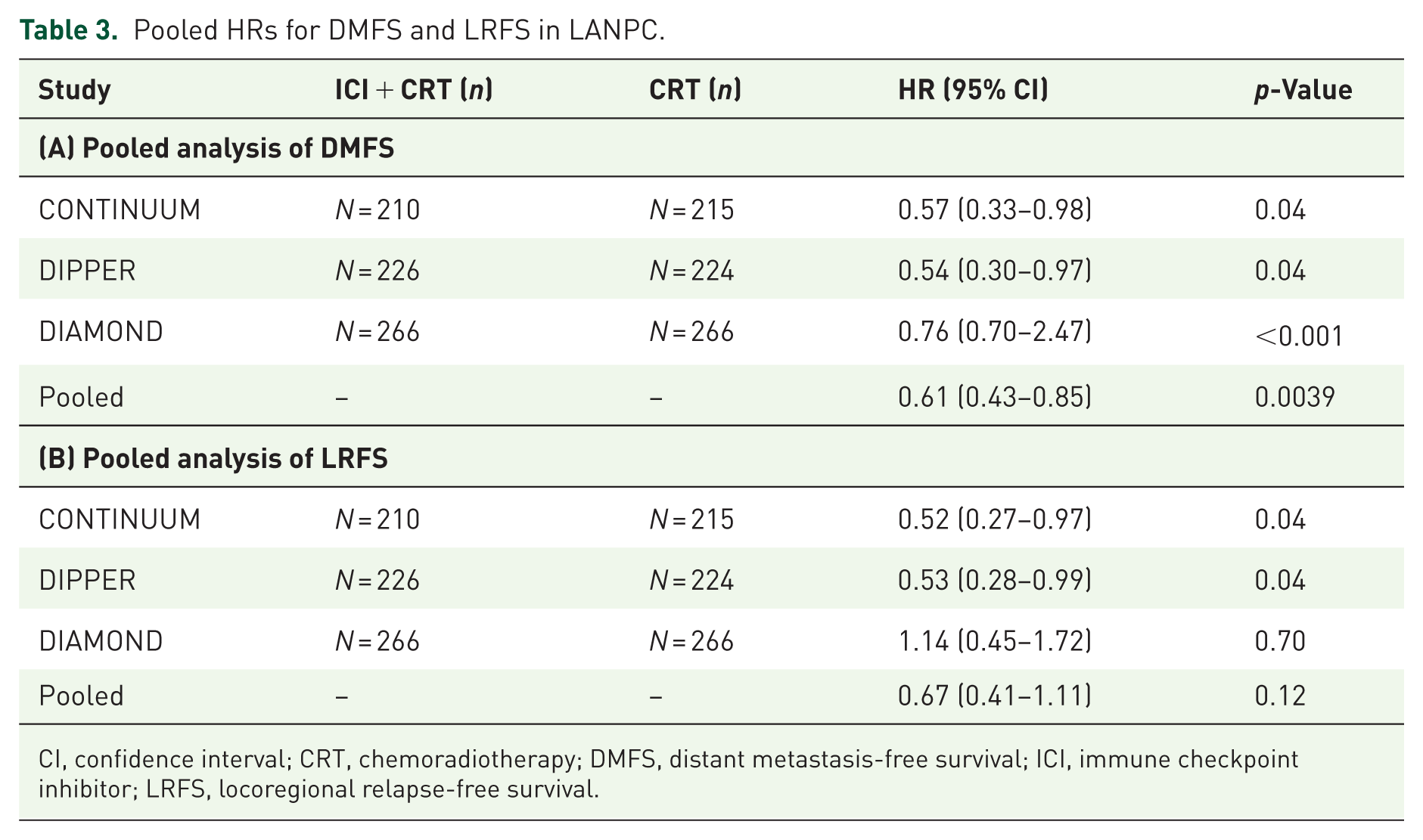
CI, confidence interval; CRT, chemoradiotherapy; DMFS, distant metastasis-free survival; ICI, immune checkpoint inhibitor; LRFS, locoregional relapse-free survival.

Pooled absolute benefits of PD-1 blockade. Absolute risk differences at 3 years for LRFS, DMFS, and EFS/FFS. NNT are indicated for each endpoint. The most pronounced benefit was observed for DMFS, reflecting reduced distant relapse.
Locoregional relapse-free survival
For LRFS, the pooled analysis demonstrated only a non-significant trend toward benefit with PD-1 blockade (HR 0.67, 95% CI: 0.41–1.11, p = 0.12), with moderate heterogeneity (I2 ≈ 44%). While CONTINUUM and DIPPER each showed significant improvements, the neutral results from DIAMOND attenuated the pooled effect, leaving uncertainty about the role of PD-1 blockade in locoregional disease control (Table 3-(B), Figure 4(b)).
Absolute benefits in EFS/FFS
Across the three included trials, the addition of PD-1 blockade to definitive CRT improved absolute EFS or FFS rates at 3 years. In the CONTINUUM trial, 3-year FFS was 86.0% with sintilimab versus 76.0% with CRT alone, corresponding to an absolute benefit of +10.0% (95% CI: +2.6% to +17.4%) and a number needed to treat (NNT) of 10.0. Similarly, in the DIPPER trial, 3-year EFS was 86.9% with adjuvant camrelizumab compared with 77.3% in the control arm, yielding an absolute benefit of +9.6% (95% CI: +2.6% to +16.6%; NNT 10.4). By contrast, the DIAMOND trial showed comparable 3-year FFS rates between the cisplatin-sparing toripalimab regimen (88.3%) and standard chemoradiotherapy (87.6%), with a nonsignificant absolute difference of +0.7% (95% CI: −6.0% to +7.4%; NNT 142.9). When pooled across all three trials (N = 1237), PD-1 blockade provided a consistent absolute improvement in 3-year EFS/FFS of +6.5% (95% CI: +2.4% to +10.5%), translating into a clinically meaningful NNT of 15.5 patients treated to prevent one additional progression, relapse, or death event within 3 years (Table 4, Figure 5).
Absolute benefits in EFS/FFS, DMFS, and LRFS at 3 years.

Absolute differences in 3-year outcomes with the addition of PD-1 blockade to definitive chemoradiotherapy across individual trials and pooled analysis. Absolute benefit is expressed as percentage points with 95% CIs. NNT is calculated as the reciprocal of the absolute risk reduction.
CI, confidence interval; CRT, chemoradiotherapy; DMFS, distant metastasis-free survival; EFS, event-free survival; FFS, failure-free survival; LRFS, locoregional relapse-free survival; NNT, number needed to treat; PD-1, programmed cell death protein 1.

Forest plots of DMFS and LRFS by trial. Forest plots showing HRs for (a) DMFS and (b) LRFS. PD-1 blockade significantly improved DMFS, while LRFS showed a favorable but less consistent trend, influenced by cisplatin use.
Absolute benefits in DMFS
At 3 years, the addition of PD-1 blockade to chemoradiotherapy consistently reduced the risk of distant relapse in two of the three trials. In CONTINUUM, 3-year DMFS was 91.0% with sintilimab versus 84.0% with chemoradiotherapy alone, yielding an absolute benefit of +7.0% (95% CI: +0.8% to +13.2%) and an NNT of 14.3. In DIPPER, 3-year DMFS was 89.0% versus 82.0% (absolute benefit +7.0%, 95% CI: +0.5% to +13.5%; NNT 14.3). In contrast, the DIAMOND trial showed comparable 3-year DMFS rates (93.2% vs 91.6%), with a nonsignificant absolute difference of +1.6% (95% CI: −3.9% to +7.1%). When pooled across all trials, PD-1 blockade conferred a significant absolute improvement in 3-year DMFS of +4.8% (95% CI: +1.3% to +8.3%), corresponding to an NNT of 20.8 (Table 4, Figure 5).
Absolute benefits in LRFS
For locoregional control, the benefits were more modest. In CONTINUUM, 3-year LRFS was 95.0% with sintilimab compared to 91.0% with control (absolute benefit +4.0%, 95% CI: −0.8% to +8.8%; NNT 25). DIPPER showed similar results (94.0% vs 90.0%, absolute benefit +4.0%, 95% CI: −1.0% to +9.0%; NNT 25). By contrast, DIAMOND demonstrated similar 3-year LRFS rates between treatment groups (92.9% vs 93.6%, absolute difference −0.7%, 95% CI: −5.9% to +4.5%). The pooled analysis revealed a nonsignificant overall trend toward improved locoregional control with PD-1 blockade, with an absolute benefit of +2.5% (95% CI: −0.3% to +5.4%) and an estimated NNT of 39.4 (Table 4, Figure 5).
Pooled efficacy outcomes and sensitivity analyses
In the pooled analysis of three phase III trials (CONTINUUM, DIPPER, and DIAMOND), PD-1 blockade combined with definitive CRT significantly improved EFS/FFS, with a HR of 0.66 (95% CI: 0.49–0.89). The prediction interval (0.46–0.96) indicated that future studies are likely to confirm this benefit, and leave-one-out sensitivity analyses demonstrated stable results across trials, with HRs ranging from 0.58 to 0.72. For DMFS, the pooled HR was 0.61 (95% CI: 0.43–0.85), with a narrow prediction interval (0.43–0.85), highlighting robust and consistent reductions in metastatic risk. Sensitivity analyses confirmed that no single study disproportionately influenced the results. LRFS showed a nonsignificant trend favoring immunotherapy (HR 0.67, 95% CI: 0.41–1.11), though the wide prediction interval (0.32–1.43) suggests potential variability in future trials. Notably, removing the DIAMOND study yielded a statistically significant effect (HR 0.53, 95% CI: 0.29–0.96), underscoring its impact on between-study heterogeneity. OS remained immature at the time of reporting, with a pooled HR of 0.94 (95% CI: 0.60–1.48) and no evidence of survival benefit to date. Collectively, these results confirm that PD-1 blockade primarily improves disease control by reducing progression and metastatic relapse, while effects on locoregional relapse and OS require longer follow-up and additional studies (Table 5).
Pooled efficacy results.

CI, confidence interval; DMFS, distant metastasis-free survival; EFS, event-free survival; FFS, failure-free survival; HR, hazard ratio; LRFS, locoregional relapse-free survival; OS, overall survival.
Subgroup analyses by treatment strategy
Subgroup analyses were performed according to PD-1 timing strategy and concurrent cisplatin use. Both peri-CRT (induction, concurrent, and adjuvant; CONTINUUM) and adjuvant-only (DIPPER) approaches yielded consistent and statistically significant improvements in EFS/FFS and DMFS, with HRs ranging from 0.52 to 0.59 across endpoints. LRFS was also favorably impacted in these cisplatin-containing regimens (HR 0.52–0.53), whereas OS data remained immature (Table 6). By contrast, the cisplatin-sparing strategy (DIAMOND) demonstrated no significant advantage across efficacy endpoints, with HRs approximating unity for EFS/FFS (HR 0.92, 95% CI: 0.66–1.79), DMFS (HR 0.76, 95% CI: 0.40–1.43), and LRFS (HR 1.14, 95% CI: 0.45–1.72). Pooled analysis by cisplatin use confirmed that retaining cisplatin conferred robust benefits, reducing the risk of progression or failure by 43% (HR 0.57, 95% CI: 0.42–0.79; I2 = 0%) and significantly improving DMFS (HR 0.56, 95% CI: 0.37–0.83) and LRFS (HR 0.53, 95% CI: 0.34–0.82). In contrast, cisplatin omission did not improve outcomes across any survival endpoint (Table 7).
Timing strategies for PD-1 blockade: subgroup analysis.

CI, confidence interval; DMFS, distant metastasis-free survival; EFS, event-free survival; FFS, failure-free survival; HR, hazard ratio; LRFS, locoregional relapse-free survival; OS, overall survival; PD-1, programmed cell death protein 1.
Subgroup analysis by cisplatin use.
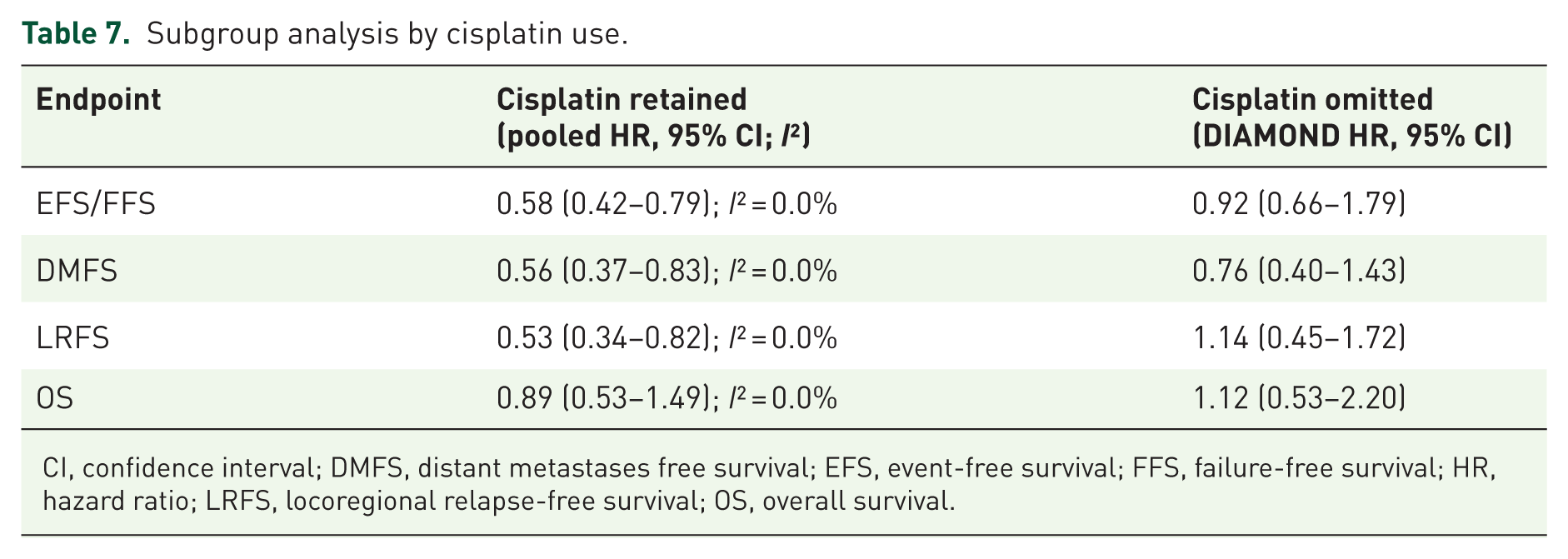
CI, confidence interval; DMFS, distant metastases free survival; EFS, event-free survival; FFS, failure-free survival; HR, hazard ratio; LRFS, locoregional relapse-free survival; OS, overall survival.
Subgroup analyses by PD-L1 expression and baseline EBV DNA levels
In subgroup analyses according to PD-L1 expression, the addition of PD-1 blockade to CRT significantly improved EFS/FFS both in patients with PD-L1-negative tumors (pooled HR = 0.62, 95% CI: 0.44–0.88) and in those with PD-L1-positive tumors (pooled HR = 0.65, 95% CI: 0.47–0.91). There was no statistically significant interaction between PD-L1 status and treatment effect (Q_between = 3.22, p = 0.20), suggesting consistent benefit regardless of PD-L1 expression. When stratified by baseline EBV DNA levels, patients with low EBV showed a pooled HR of 0.58 (95% CI: 0.42–0.81), while those with high EBV had a pooled HR of 0.61 (95% CI: 0.45–0.84). Again, there was no evidence of heterogeneity between EBV strata (Q_between = 8.41, p = 0.14). Collectively, these findings indicate that PD-1 + CRT provides consistent survival benefit irrespective of PD-L1 expression or baseline EBV DNA levels (Table 8, Figure 6(a) and (b)).
Subgroup analyses by PD-L1 expression and baseline EBV DNA levels.

CI, confidence interval; EBV, Epstein–Barr virus; HR, hazard ratio; PD-L1, programmed cell death ligand 1.

Subgroup analyses by PD-L1 expression and EBV DNA levels. Forest plots of HRs for EFS/FFS stratified by (a) PD-L1 CPS subgroups and (b) baseline or post-induction EBV DNA levels. Benefits of PD-1 blockade were observed across most biomarker-defined subgroups, with greater consistency in PD-L1-positive and EBV low patients.
Safety analysis of PD-1 blockade in LANPC
Across the three included trials (CONTINUUM, DIPPER, DIAMOND), grade ⩾3 AEs were reported in 58% of patients treated with PD-1 blockade plus CRT and in 59% of those receiving CRT alone (HR 0.99, 95% CI: 0.77–1.28; p = 0.90). Fatal AEs were rare, occurring in 1.7% of patients in the PD-1 arms and 1.4% in the control arms (HR 1.20, 95% CI: 0.45–3.22; p = 0.70; Table 9). The pooled safety analysis showed that PD-1 blockade did not increase the incidence of severe or fatal toxicities. The RR for TRAEs of grade ⩾3 was 0.99 (95% CI: 0.77–1.28; p = 0.90), suggesting no difference compared with standard therapy. Similarly, the risk of fatal AEs was not significantly different between groups (RR 1.20, 95% CI: 0.45–3.22; p = 0.70; Table 10, Figure 7(a) and (b)).
Per-trial breakdown of AE.

AE, adverse events; CI, confidence interval; CRT, chemoradiotherapy; PD-1, programmed cell death protein 1; TRAE, treatment-related adverse event.
Pooled analysis of adverse events (Mantel–Haenszel fixed-effect).

AE, adverse event; CI, confidence interval; HR, hazard ratio; RR, risk ratio; TRAE, treatment-related adverse event.

Safety outcomes of PD-1 blockade combined with CRT. Forest plots of RRs for (a) grade ⩾3 TRAEs and (b) fatal AEs, stratified by trial. Severe toxicities and fatal events were balanced between treatment arms, indicating no excess safety burden from PD-1 blockade.
Discussion
In this meta-analysis of three phase III randomized trials (CONTINUUM, DIPPER, and DIAMOND) including 1237 patients with LANPC, the addition of PD-1 inhibitors to definitive CRT significantly improved EFS/FFS, corresponding to a relative risk reduction of approximately one-third. This benefit was consistent across studies, with low heterogeneity, underscoring the robustness and generalizability of the effect across different PD-1 agents, treatment strategies, and patient populations. Absolute benefit analyses confirmed clinically meaningful improvements in 3-year EFS/FFS, reinforcing the magnitude of benefit in the curative-intent setting. To our knowledge, this represents the first meta-analysis to synthesize phase III evidence on PD-1 blockade in combination with CRT for LANPC. To our knowledge, this represents the first meta-analysis to systematically evaluate the integration of PD-1 blockade with curative-intent CRT in this population.
Advanced NPC remains a highly lethal disease with limited therapeutic options.19,20 The current first-line standard of care—gemcitabine plus cisplatin—was established by a pivotal phase III trial demonstrating significant improvement in PFS. 21 However, long-term outcomes remain suboptimal, highlighting the urgent need for more effective diagnostic and treatment strategies. 21
In this context, immunotherapy has emerged as a promising approach, particularly in advanced NPC, supported by strong biological rationale. The disease’s close association with EBV infection contributes to a distinct tumor microenvironment characterized by dense lymphocytic infiltration and frequent PD-L1 overexpression on tumor cells.10,22 Furthermore, PD-1 expression on tumor-infiltrating T cells reflects functional exhaustion from chronic viral antigen stimulation.23,24 Collectively, these features suggest that NPC is especially susceptible to immune checkpoint blockade. On this basis, PD-1 inhibitors have been evaluated in early-phase clinical trials, yielding encouraging results and establishing proof of concept for their therapeutic potential in NPC.25–27
Recent evidence also highlights the expanding role of [¹8F]FDG PET/CT as a non-invasive biomarker for predicting and monitoring response to neoadjuvant chemoradiotherapy in NPC. In a study by Quartuccio et al., 18 both pre- and post-treatment metabolic parameters—particularly reductions in SUVmax and total lesion glycolysis—correlated strongly with histopathologic and clinical response following neoadjuvant CRT. 18 These findings suggest that [¹8F]FDG PET/CT may facilitate response-adapted treatment strategies, enabling early identification of non-responders who might benefit from intensified systemic therapy or the incorporation of immunotherapeutic approaches.
These biological insights provide a mechanistic framework for interpreting our findings, as the results of the present meta-analysis further support the clinical efficacy of PD-1 blockade in enhancing systemic disease control in LANPC. In our meta-analysis, the most compelling efficacy signal emerged for DMFS, emphasizing immunotherapy’s unique capacity to prevent systemic relapse—the predominant mode of treatment failure in LANPC despite advances in radiotherapy precision and chemotherapy delivery. This observation is biologically plausible, as checkpoint blockade enhances immune surveillance and facilitates the eradication of micrometastatic disease, an area where chemoradiotherapy alone is insufficient. Preventing distant spread is particularly critical given its strong association with limited curative options and poor long-term survival outcomes.
By contrast, improvements in LRFS were more modest and heterogeneous. Importantly, the benefit in LRFS appeared dependent on the chemotherapy backbone. CONTINUUM and DIPPER, both of which incorporated concurrent cisplatin, demonstrated significant reductions in locoregional recurrence, supporting the hypothesis that cisplatin’s radiosensitizing effects may synergize with immunotherapy-induced immune activation. In contrast, the cisplatin-sparing DIAMOND trial showed no LRFS improvement, underscoring the indispensable role of cisplatin in locoregional control and suggesting that PD-1 inhibition cannot fully substitute for platinum-based therapy. These findings have practical implications, reinforcing that immunotherapy should be integrated with, rather than replace, cisplatin-based CRT in fit patients.
OS data remain immature, with the pooled HR showing no significant difference. This is not unexpected given the relatively short follow-up, the high efficacy of CRT alone, and the availability of effective salvage therapies in endemic regions. Nevertheless, early and consistent gains in EFS/FFS and DMFS—both established surrogates for OS in head and neck cancer—suggest that longer follow-up is likely to reveal survival advantages.
Our findings align with and extend prior evidence from the metastatic setting, where PD-1 blockade is firmly established as a standard of care.28,29 The landmark JUPITER-02 trial demonstrated that toripalimab combined with gemcitabine and cisplatin significantly improved PFS and OS in recurrent/metastatic NPC, 30 while RATIONALE-309 confirmed similar benefits with tislelizumab. 31 Trials such as KEYNOTE-122 and CAPTAIN showed more modest efficacy of PD-1 monotherapy in previously treated patients, emphasizing the importance of combination strategies.25,32 Together, these data established PD-1 inhibitors as the backbone of systemic therapy for advanced disease. Our analysis now provides parallel evidence in the curative-intent setting, suggesting that immunotherapy not only extends systemic disease control but also prevents micrometastatic dissemination, thereby shifting the natural history of LANPC. These findings also resonate with results in other virally associated malignancies, such as HPV-positive oropharyngeal cancer and EBV-positive gastric carcinoma, where PD-1 inhibitors demonstrate enhanced activity in immune-rich, virally driven tumors.33,34
Safety is a critical consideration when integrating immunotherapy into curative-intent regimens. Across trials, the addition of PD-1 blockade did not increase the incidence of grade ⩾3 AEs or fatal AEs. Severe toxicities occurred in 58% of patients in the PD-1 arms and 59% in controls, indicating no excess burden. Immune-related AEs occurred at expected frequencies and were generally manageable with established protocols. Therefore, these findings support the safety of PD-1 blockade in combination with CRT, an essential factor in the curative setting.
This study has several strengths. It represents the first meta-analysis limited to phase III randomized controlled trials evaluating PD-1 blockade combined with definitive CRT for LANPC. Conducted in accordance with PRISMA 2020 guidelines, the analysis ensured methodological rigor and transparency. The included trials were of high quality, showed low heterogeneity, and yielded consistent results across sensitivity analyses, supporting the robustness and internal validity of the findings.
However, certain limitations should be acknowledged. Only three RCTs—all conducted in China—were available, which may limit generalizability to non-endemic regions where disease biology and EBV prevalence differ.35–37 While this enhances internal consistency, validation in global cohorts remains necessary. Moreover, OS data are still immature, and longer follow-up is required to confirm whether improvements in EFS/FFS and DMFS translate into durable survival benefit. Differences in trial design—peri-CRT in CONTINUUM, adjuvant-only in DIPPER, and cisplatin-sparing concurrent therapy in DIAMOND—introduce clinical heterogeneity that may affect effect size.
Future research should clarify the optimal timing and duration of PD-1 blockade—whether induction, concurrent, adjuvant, or peri-CRT—and evaluate whether cisplatin can be safely omitted in patients with comorbidities or poor tolerance. Biomarker integration will be critical: EBV DNA clearance is a validated prognostic marker and should serve as a stratification tool in upcoming trials. Additional factors such as PD-L1 expression, tumor mutational burden, and immune gene signatures may further refine patient selection. Finally, health economic analyses are needed to determine the cost-effectiveness of PD-1 blockade with CRT, particularly in resource-limited settings.
In summary, this meta-analysis demonstrates that the addition of PD-1 inhibitors to definitive CRT significantly improves disease control in LANPC, primarily by reducing the risk of distant metastasis, without increasing the burden of severe or fatal toxicities. These results provide the first synthesis of phase III evidence supporting immunotherapy in the curative-intent setting and strongly advocate for its integration into standard CRT, particularly with cisplatin-based regimens. While OS data remain immature, the consistency of early benefits in EFS/FFS and DMFS supports immunotherapy as a highly promising adjunct to CRT, laying the groundwork for incorporation into future clinical practice guidelines for LANPC.
Conclusion
This meta-analysis of three phase III trials demonstrates that adding PD-1 inhibitors to definitive CRT significantly improves disease control in LANPC, with consistent benefits across studies and no increase in severe or fatal toxicities. The strongest effect was observed in reducing distant relapse, highlighting immunotherapy’s capacity to address the predominant mode of treatment failure in this disease. Benefits in locoregional control were most evident when cisplatin was retained, underscoring its continued importance as the backbone of CRT. While OS data remain immature, the consistent gains in EFS/FFS and DMFS strongly support the integration of PD-1 blockade into standard CRT, particularly in cisplatin-eligible patients. Future global trials with longer follow-up and biomarker-driven strategies will be essential to validate these findings, optimize treatment sequencing, and ensure equitable implementation in diverse clinical settings.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251412043 – Supplemental material for Immunotherapy for locoregionally advanced nasopharyngeal carcinoma: a meta-analysis of randomized clinical trials
Supplemental material, sj-docx-1-tam-10.1177_17588359251412043 for Immunotherapy for locoregionally advanced nasopharyngeal carcinoma: a meta-analysis of randomized clinical trials by Katarzyna Stawarz, Anna Gorzelnik, Wojciech Klos, Jacek Korzon, Filip Kissin, Karolina Bienkowska-Pluta, Grzegorz Stawarz, Jakub Zwolinski and Dorota Kiprian in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251412043 – Supplemental material for Immunotherapy for locoregionally advanced nasopharyngeal carcinoma: a meta-analysis of randomized clinical trials
Supplemental material, sj-docx-2-tam-10.1177_17588359251412043 for Immunotherapy for locoregionally advanced nasopharyngeal carcinoma: a meta-analysis of randomized clinical trials by Katarzyna Stawarz, Anna Gorzelnik, Wojciech Klos, Jacek Korzon, Filip Kissin, Karolina Bienkowska-Pluta, Grzegorz Stawarz, Jakub Zwolinski and Dorota Kiprian in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
