Abstract
The general immune landscape of nasopharyngeal carcinoma (NPC) renders immunotherapy suitable for patients with NPC. Immune checkpoint inhibitors (ICIs) based on programmed death-1/programmed death ligand-1 (PD-1/PD-L1) blockade have made a breakthrough with the approval of PD-1 inhibitor for refractory recurrence and/or metastatic (R/M NPC) and the approval of PD-1 inhibitor in combination with gemcitabine and cisplatin as first line for R/M NPC in 2021 in China. The incorporation of ICIs into the treatment paradigms of NPC has become a clinical hot spot and many prospective clinical studies are ongoing. In this review, we provide a comprehensive overview of the rationale for immunotherapy in NPC and current status, advances and challenges of immunotherapy in NPC based on published clinical data, and ongoing trials. We focus on the clinical application and advances of PD-1 inhibitor monotherapy and its combination with chemotherapy and summarize the clinical explorations of other immunotherapy approaches, for example, combination of PD-1/PD-L1 inhibitors with antiangiogenic inhibitor with molecular targeted agents, cancer vaccines, adaptive immunotherapy, and new ICI agents beyond PD-1/PD-L1 inhibitors in R/M NPC. We also describe the clinical studies’ status and challenges of ICIs-based immunomodulatory strategies in local advanced NPC and pay attention to the biomarker application for personalized immunotherapy of NPC in the hope to provide insights for clinical practice and future clinical studies.
Introduction
Nasopharyngeal carcinoma (NPC) has a unique geographical and ethical distribution, which is attributed to both genetic and environmental factors. 1 Based on GLOBOCAN estimates, in 2018, there were 129,079 new cases and 72,987 deaths due to NPC. 2 NPC occurs more frequently in Southeast Asia than in the rest of the world and is particularly observed in Southern China, which has incidence of >20 cases per 100,000 people and a mortality risk of 6 cases per 100,000 people. 3 The non-keratinizing subtype is the most common pathological subtype of NPC and accounts for >95% of cases in endemic areas, which is predominantly associated with Epstein–Barr virus (EBV) infection, greater response to radiation therapy, and better survival than the keratinizing NPC subtype.4–7
For non-metastatic NPC, radiotherapy is the primary curative treatment. The strategy of combining chemotherapy with radiotherapy is a pivotal advancement for locally advanced NPC (LANPC). Approximately 5–11% of patients present with de novo metastatic disease, whereas 15–30% of patients who are treated for locally advanced disease develop local recurrence and/or metastatic (R/M) NPC.8,9 Platinum-based doublet chemotherapeutic regimens, preferentially gemcitabine plus cisplatin (GP), are generally considered the standard first line of care for patients with R/M NPC. 10 For patients with R/M NPC receiving first-line chemotherapy, their estimated median overall survival (OS) is 15.7 months (95% CI, 12.3–19.1) and median progression-free survival (PFS) is 7.6 months (95% CI, 6.2–9.0). 11 For the patients undergoing second- or later-line therapies, the estimated median OS is approximately 11.5 months and median PFS is 5.4 months (95% CI, 3.8–7.0). 11 No consensus has been reached regarding the treatment following disease progression after the first-line therapy. Current conventional treatments, including radiotherapy, chemotherapy, and surgical resection, are often associated with significant adverse effects and limited efficacy. 12 Thus, there is an urgent need for novel treatment strategies to further improve the outcomes of patients with NPC.
In recent years, immunotherapy has prompted a revolution in the clinical management of many cancers. Immune checkpoint inhibitors (ICIs) based on programmed death-1/programmed death ligand-1 (PD-1/PD-L1) blockade have made a breakthrough and were approved for the treatment of R/M NPC in 2021 in China. The incorporation of ICIs into the current treatment paradigms of LANPC is a clinical hot spot, and many prospective phase II or III clinical studies are ongoing. This review provides a comprehensive overview of the current landscape of immunotherapy in NPC by presenting the rationale of immunotherapy, updated status and challenges of immunotherapy in R/M NPC and LANPC based on the published clinical trial data and ongoing trials data. We also described and analyzed the current status and controversies of different combinatorial strategies and biomarkers application for personalized NPC immunotherapy in the hope to provide insights for clinical practice and future clinical studies.
Rationale for immunotherapy in NPC
NPC is regarded as a highly immune-inflamed tumor due to its unique immune landscape. Close associations with chronic EBV infection, abundant lymphocytic infiltrations, and high expression of PD-L1, and several key immune molecular regulating the activation of T-cells (CD40, CD70, CD80, and CD86) are often observed in EBV-induced NPC. 10 We profiled the rationale supporting immunotherapy in NPC by analyzing the basic immunobiological foundations, including tumor antigenicity, tumor microenvironment (TME), and tumor immunogenicity. The biological steps required to achieve an effective immune response in NPC is depicted Figure 1. 13

The biological steps to achieve an effective immune response in NPC. This figure is based on the original cancer–immunity cycle proposed by Chen and Mellman. 13 The steps and factors affecting the response of ICIs are edited to reflect the specific immunobiology of NPC. The release of NPC-specific antigens (1) and their subsequent presentation by dendritic cells (DCs) and other antigen-presenting cells (APCs) (2), is followed by T-cell priming and activation (3), trafficking into tumors (4), tumor infiltration (5), and recognition and killing of tumor cells (6 and 7). Notable NPC-specific characteristics of this cycle favoring antitumor immunity include the release of NPC-specific antigens (including EBV-derived antigens and a large number of neoantigens), activated monocytes which could also be considered as a kind of APCs, promoting the trafficking of immune cells into tumors [via tumor inflammasomes, increased chemoattraction, notable interferon-α (IFN-α) and IFN-γ], dense infiltration of immune cells into tumors, high expression of inhibitory receptors, elevated cytotoxicity genes, and chemokine receptors. Conversely, antitumor immunity could be impeded by the loss of NPC-specific neoantigens, major histocompatibility complex protein (MHC) aberration, defective DCs or antigen-processing machinery components (APM) deficiency, high epithelial indoleamine 2,3-dioxygenase, abundant myeloid-derived suppressor cells (MDSC) or regulatory T-cells (Tregs), rich M2-type tumor-associated macrophages (TAMs), and several features typically associated with T-cell exhaustion.
Antigenicity is the initial stimulus that activates the tumor immune response and depends on the quality and quantity of tumor antigens, and the antigen’s successful presentation. Neoantigens are the main antigens that induce antitumor activity.14,15 Compared with other cancers in the cancer genome atlas, a large number of neoantigens have been detected in NPC. 14 Since EBV infection is a driving factor for NPC, EBV antigens, such as latent membrane protein (LMP-1, LMP-2) and nuclear antigen-1 (EBNA1), are regarded as surrogate neoantigens.4,16,17 In addition to having efficient antigens, the interferon response is elevated in NPC and is related to a higher proportion of antigen-presenting cells. 18 However, the aberration of major histocompatibility complex protein (MHC) or APM components impede the successful antigen presentation in NPC and leads to the invasion of immune surveillance.19–24 Subsequently, immune cell trafficking and infiltration of the TME play an essential role in activating an immune response.25,26
Distinct features of tumor immunogenicity to overcome an immunosuppressive microenvironment and eventually kill tumor cells provide more potential for efficient immunotherapy in NPC. Infiltrating lymphocytes (TILs), including CD4 + T, CD8 + T, and CD57 + NK cells, are abundant in tumor tissues.27–30 However, upregulated MDSC,31–34 overexpression of PD-L1, and various co-stimulators associated with T-cell exhaustion and dysfunction in NPC include cytotoxic T-lymphocyte-associated antigen 4 (CTLA-4), lymphocyte activation gene 3 (LAG-3), T-cell immunoglobulin and ITIM domain (TIGIT), T-cell immunoglobulin and mucin domain 3 (TIM3), and CD276, which participate in the blockade of immune response and could be considered as targets for immune therapy.18,29,30,35–41 The general immune texture of NPC renders the patients potentially appropriate for immunotherapy, especially for ICIs, although some immunosuppressive elements warrant further study (i.e. loss of neoantigen, MHC protein aberration, defective DCs, and abundance of Tregs). When ICIs are used to stop an immune response cascade, a strong antitumor immune activity would be elicited to kill tumor cells, which could pave a novel and promising treatment for NPC. The approval of toripalimab and camrelizumab supported the establishment of a standard treatment setting for patients with refractory R/M NPC.
PD-1/PD-L1 inhibitors in R/M NPC
Many PD-1/PD-L1 inhibitors have been evaluated in R/M NPC. Toripalimab and camrelizumab monotherapy were approved for refractory R/M NPC by National Medical Products Administration (NMPA) in 2021. The promising antitumor activity and manageable safety profile of PD-1/PD-L1 inhibitors have promoted the investigation of various combination approaches, including chemotherapy, targeted therapy, other immunotherapeutic agents, or radiotherapy. Camrelizumab combined with GP regimen as first-line setting was approved for R/M NPC by NMPA in June 2021 based on the results of CAPTAIN study. 42 Toripalimab combined with GP regimen as first-line setting was approved for R/M NPC by NMPA in November 2021 based on the results of JUPITER-02 study. Here, we focus on the clinical application and advance of PD-1 inhibitor monotherapy and its combination with chemotherapy and summarize the clinical explorations of other combination strategies, for example, combination of PD-1/PD-L1 inhibitors with antiangiogenic inhibitor, radiotherapy, or new ICI agents in R/M NPC
PD-1/PD-L1 inhibitor monotherapy in R/M NPC
For the second- or later-line setting, 10 clinical trials with 1163 R/M NPC patients were included. All patients progressed during or after platinum-based chemotherapy and then received PD-1/PD-L1 inhibitor monotherapy. The pooled objective response rate (ORR) was 22.5% (95% CI 18.3–26.7%) [Figure 2(a)] and disease control rate (DCR) was 52.1% (95% CI 43.4–60.9%) [Figure 2(b)]. The forest plots of grade ⩾3 treatment-related adverse effects (TRAEs) incidence was 14.9% (95% CI 10.8–19.1%) [Figure 2(c)]. Both NCT02605967 study and KEYNOTE-122 study were randomized clinical trials which compared the efficacy and safety between ICIs monotherapy and chemotherapy based on physician’s choice. The pooled ORR and DCR had no significant difference [Figure 3(a) and (b)], the incidence of grade ⩾3 TRAEs was significantly lower in ICIs group [Figure 3(c)]. For unselected refractory R/M NPC patients, the antitumor activity of PD-1 antibody monotherapy was moderate, which was similar to chemotherapy.

The forest plot of ORR, DCR, and grade ⩾3 TRAEs incidence in ICI monotherapy for R/M NPC: (a) the forest plot of ORR in ICI monotherapy for R/M NPC. (b) The forest plot of DCR in ICI monotherapy for R/M NPC. (c) The forest plot of grade ⩾3 TRAEs incidence in ICI monotherapy for R/M NPC.
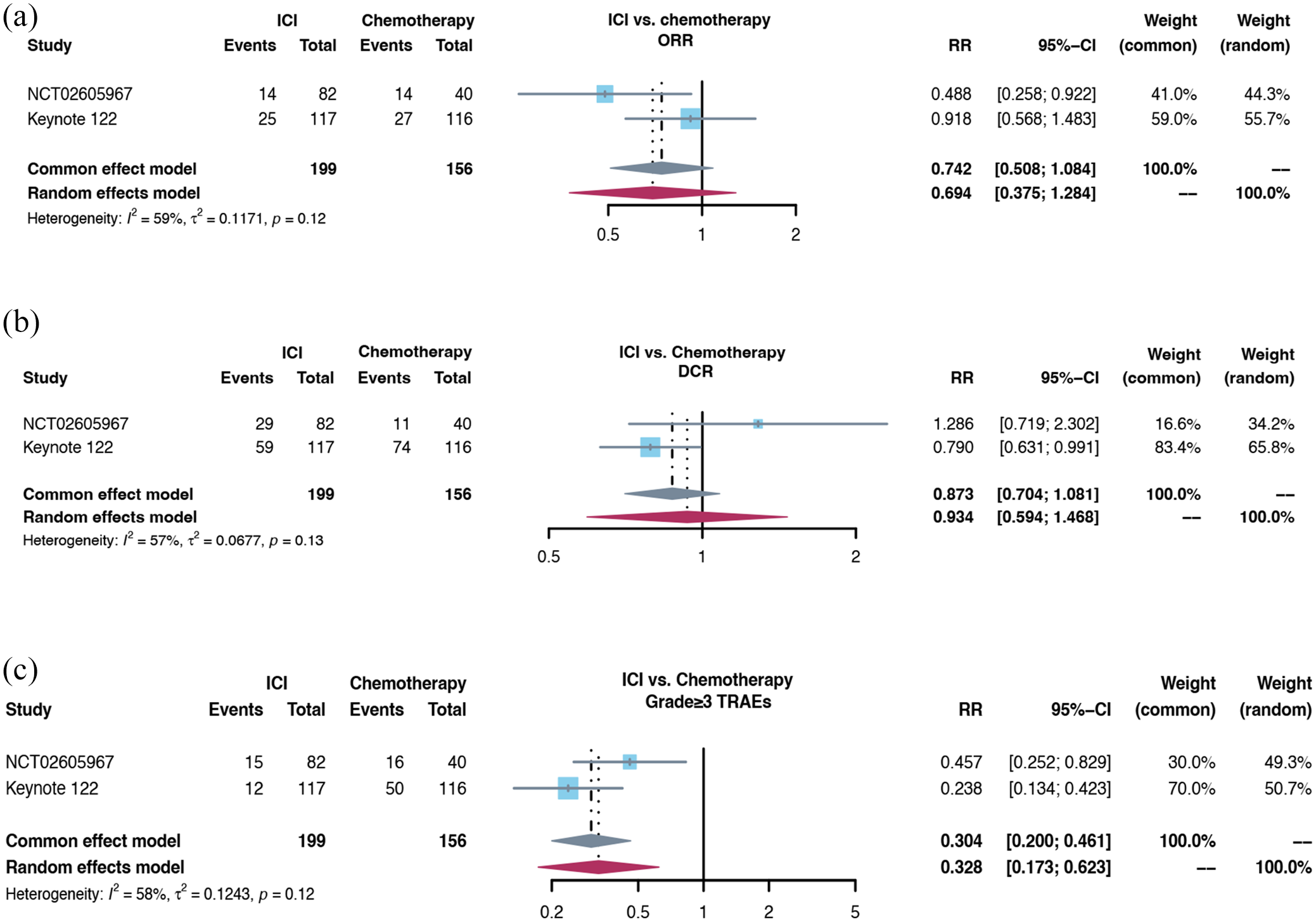
The forest plot of ORR, DCR, and grade ⩾3 TRAEs incidence in comparison between ICI monotherapy and chemotherapy for R/M NPC: (a) the forest plot of ORR in comparison between ICI monotherapy and chemotherapy for R/M NPC. (b) The forest plot of DCR in comparison between ICI monotherapy and chemotherapy for R/M NPC. (c) The forest plot of grade ⩾3 TRAEs incidence in the comparison between ICI monotherapy and chemotherapy for R/M NPC.
POLARIS-02 study is a single-arm and multicenter phase II study which evaluated the efficacy, safety, and correlative biomarkers of toripalimab in previously treated R/M NPC and enrolled 190 patients. The ORR was 20.5% (39/190), median duration of response (DOR) was 12.8 months, and median OS was 17.4 months. The ORR and DOR of 92 patients who failed at least two lines of systemic chemotherapy were 23.9% and 21.5 months, respectively. However, 27 (14.2%) patients experienced grades 3–5 TRAEs. Hypothyroidism, hyperthyroidism, and abnormal liver function were the main immune-related adverse events. 43
CAPTAIN study is also a single-arm and multicenter phase II study which evaluated the efficacy and safety of camrelizumab in previously treated R/M NPC and enrolled 93 patients. The ORR was 34.0% (31/91) and DCR was 59.0% (54/91). The median PFS was 5.6 months with a median follow-up time of 9.9 months. The incidence of grade 3 or 4 TRAEs was 16% (15/91), and the most common adverse events were stomatitis, anemia, and abnormal liver function. 43
The NCT02605967 study is the first randomized phase II study to compare spartalizumab (PD-1 monoclonal antibody) with chemotherapy in R/M NPC patients who progressed on or after platinum-based chemotherapy. The patients were randomized to receive either spartalizumab (n = 82) or chemotherapy (n = 40). Chemotherapy was administered according to the investigator’s choice and one-third of the patients received chemotherapy combinations with two or more drugs. Spartalizumab showed a safety profile consistent with that of other anti-PD-1 antibodies. The most common TRAEs were fatigue (10.3%) and pruritus (9.3%). The primary endpoint of median PFS was not met. The median PFS in spartalizumab group was 1.9 versus 6.6 months in chemotherapy group (p = 0.915), and the ORR in spartalizumab group was lower (17.1% versus 35.0%). However, the median OS (25.2 versus 15.5 months) and median DOR (10.2 versus 5.7 months) were longer in spartalizumab group than in chemotherapy group. 44
The KEYNOTE-122 study (NCT02611960) was the first randomized phase III study comparing pembrolizumab with single-agent standard chemotherapy based on the investigator’s choice (capecitabine, gemcitabine, or docetaxel) in patients with platinum-pretreated R/M NPC. The results were first reported in 2021 European Society of Medical Oncology (ESMO). There were 117 patients in pembrolizumab group and 116 patients in chemotherapy arm. The study failed to meet the primary endpoint of OS. The median OS in pembrolizumab arm was 17.2 and 15.3 months in chemotherapy group (p = 0.226). The median PFS in pembrolizumab group was 4.1 versus 5.5 months in chemotherapy group. The ORR was similar (21.4% versus 23.3%). The incidence of grades 3–5 TRAEs was 10.3% in pembrolizumab group versus 43.8% in chemotherapy group. Although the study did not meet the primary endpoint, pembrolizumab showed a trend of better OS and a lower incidence of treatment-related adverse events. Examination of Kaplan–Meier OS curves for pembrolizumab versus chemotherapy showed an early favorable trend toward chemotherapy. Meanwhile, the separation in favor of pembrolizumab was sustained after 13 months, indicating a long-term survival benefit with pembrolizumab.
In these two randomized controlled studies, a crossover of the survival curves was observed. With the extension of follow-up time, we can see further improvements of OS. This might reflect the time taken by ICI to induce an effective antitumor immune response and greater durability of benefit from immunotherapy in patients who achieved response or prolonged stable disease.
These phenomena have been observed in studies of PD-1 or PD-L1 inhibitors in unselected patients with other refractory metastatic solid tumor types, such as non-small cell lung cancer (KEYNOTE-010), 45 head and neck squamous cell carcinoma (KEYNOTE-040), 46 esophageal cancer (KEYNOTE-181), 47 and gastric cancer (KEYNOTE-061 or KEYNOTE-062).48,49 This violation of the proportional hazard assumption supports the idea that alternative statistical approaches for addressing changes in risk over time will be necessary for future clinical studies evaluating the delayed onset of benefit typically observed with immunotherapies.
Coupled with these results, it could be inferred that both chemotherapy and PD-1 inhibitor monotherapy could be choices for R/M NPC patients who progressed during or after platinum-based chemotherapy. However, compared with chemotherapy, the TRAEs incidence is significantly lower in ICIs group with good tolerance. A subset of patients responding to anti-PD-1 antibody could benefit from long-term survival. The clinically meaningful survival prolongation and favorable safety profile of ICIs supported the prioritized recommendations of ICIs, especially for those patients with heavy-treated R/M NPC who had limited treatment options and poorer tolerance. Given to only a small subset of patients benefiting from PD-1 immunotherapy, molecular biomarkers will further help to identify the patients who benefit more from PD-1 inhibitors. The novel ICIs is a hot spot in the future research. Further studies regarding ICI as monotherapy or as part of combination therapy in earlier stage settings should be considered.
Combination of PD-1/PD-L1 inhibitor and chemotherapy in R/M NPC
The combination of ICIs and chemotherapy, which can eliminate or modulate immune suppressive cells in the TME, has shown promising synergy.50–52 The immunosuppression states could be unleashed by chemotherapy (including gemcitabine, platinum, and 5-fluorouracil) by promoting antigen presentation, enhancing T-cell responses, and relieving immunosuppression in TME.41,50,53–59 Given the promising results of ICI monotherapy and preclinical evidence of the synergy between chemotherapy and ICIs, clinical exploration of the combination of ICIs with chemotherapy is required to promote the management of R/M NPC (Table 1).
The combination of camrelizumab with the GP regimen has shown promising antitumor activity as first-line setting for R/M NPC in a phase I trial. 42 The CAPTAIN-1st study is a randomized, double-blind, phase III trial that explored the addition of camrelizumab to the GP regimen as a first-line therapy. In total, 263 eligible patients were randomly assigned to the camrelizumab group (n = 134) or placebo group (n = 129). At the prespecified interim analysis (15 June 2020), the independent review committee-assessed median PFS was significantly longer in the camrelizumab group [HR = 0.54, 9.7 versus 6.9 months, HR = 0.54, p = 0.0002)]. In the camrelizumab group, 94% (126/134) and 91% (118/129) of patients in the placebo group had grade ⩾3 TRAEs, of which a decrease in white blood cell count, decreased neutrophil count and anemia, and decreased platelet count were the most common (Table 2). 60
Completed clinical trial of PD-1/PD-L1 inhibitors monotherapy in recurrent and/or metastatic (R/M) NPC.

ALT, alanine aminotransferase; AST, aspartate aminotransferase; CI, confidence interval; CT, chemotherapy; DCR, disease control rate; DOR, duration of response; GGT, Gamma-glutamyl transferase; mAb, monoclonal antibody; N/A, not available; NPC, nasopharyngeal carcinoma; NR, not reached; ORR, objective response rate; OS, overall survival; PD-1, programmed death-1; PD-L1, programmed death ligand-1; PFS, progression-free survival; R/M, recurrence and/or metastatic; TRAE, treatment-related adverse effect; FAS, full analysis set.
In the study of KEYNOTE-028, all R/M NPC patients had PD-L1 expression in 1% or more of tumor cells or tumor-infiltrating lymphocytes.
In the study of spartalizumab and KEYNOTE-122, the data of efficacy and safety are shown as anti-PD-1 antibody arm versus CT arm.
Completed clinical trial of PD-1/PD-L1 inhibitors combination in R/M NPC.
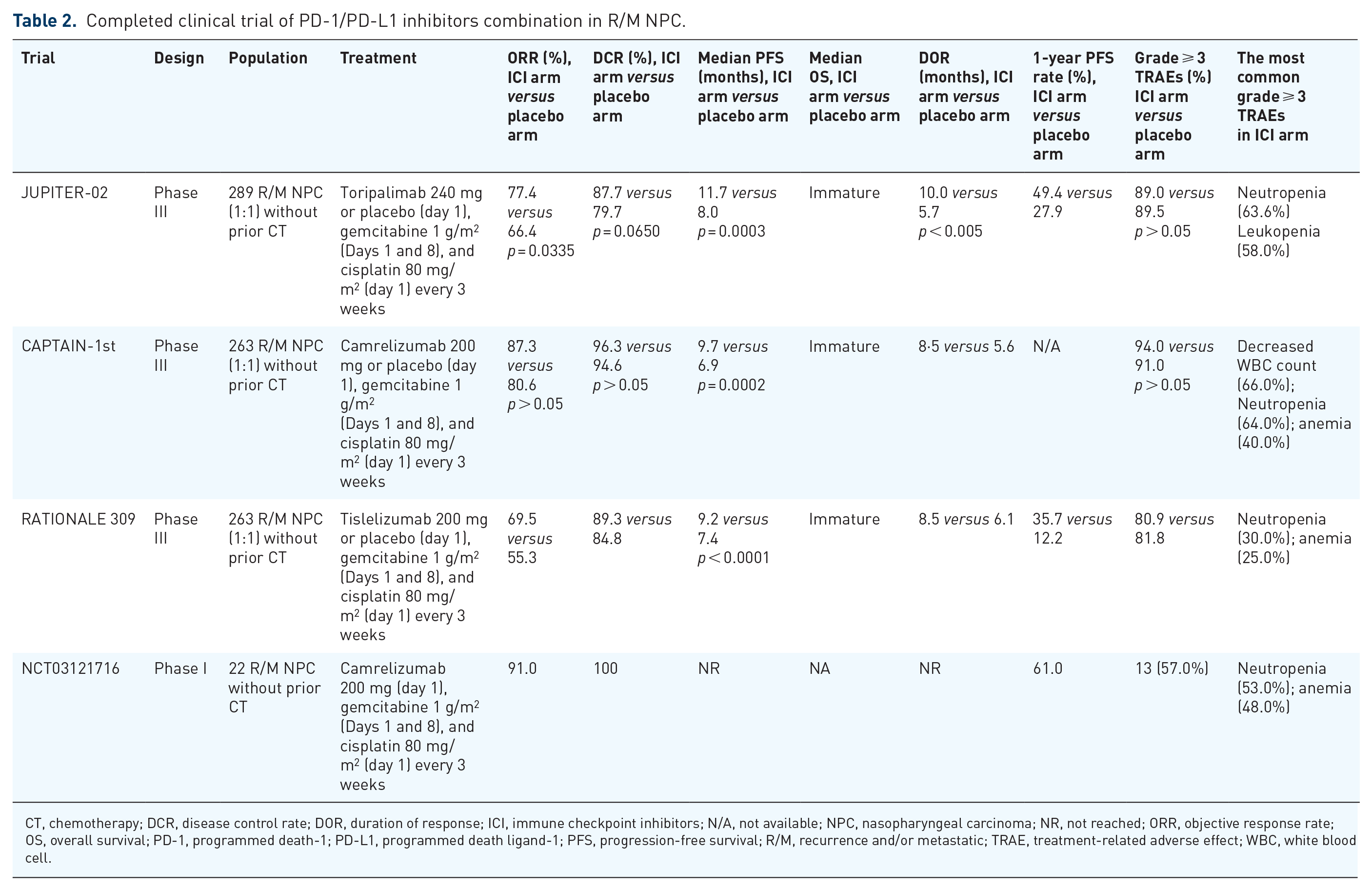
CT, chemotherapy; DCR, disease control rate; DOR, duration of response; ICI, immune checkpoint inhibitors; N/A, not available; NPC, nasopharyngeal carcinoma; NR, not reached; ORR, objective response rate; OS, overall survival; PD-1, programmed death-1; PD-L1, programmed death ligand-1; PFS, progression-free survival; R/M, recurrence and/or metastatic; TRAE, treatment-related adverse effect; WBC, white blood cell.
The JUPITER-02 study was a multinational, randomized, double-blind, phase III study conducted in mainland China, Taiwan, and Singapore that compared the GP regimen in combination with either toripalimab or placebo in patients with R/M NPC as the first-line setting. 61 A total of 289 patients were randomized to either toripalimab or placebo in combination with GP every 3 weeks for up to six cycles, followed by maintenance therapy with toripalimab or placebo. The interim analysis showed that median PFS in toripalimab group was significantly superior to that in placebo group (HR = 0.52, 11.7 versus 8.0 months). Significant improvements of 1-year PFS rates and ORR were observed in toripalimab group. In toripalimab group, 89.0% developed grade 3 or worse adverse events, similar to those in the placebo arm (89.5%) (Table 2).
The RATIONALE 309 study, a phase III randomized clinical trial, explored the value of adding tislelizumab, an anti-human PD-1 monoclonal IgG4 antibody, or placebo to GP regimen for R/M NPC as first-line setting. 62 A total of 263 patients from Asia were enrolled. The interim analysis showed that tislelizumab in combination with chemotherapy demonstrated a statistically significant improvement in PFS compared with chemotherapy alone (9.2 versus 7.4 months). Clinical trials using other anti-PD-1 inhibitors, including nivolumab, penpulimab, and SHR-1701, in combination with chemotherapy are ongoing (Table 3).
Ongoing clinical trials of ICIs in R/M NPC.

AC, adjuvant chemotherapy; AE, adverse event; CCRT, concurrent chemoradiothrapy; CT, chemotherapy; DLTS, incidence of dose-limiting toxicities; DNA, deoxyribonucleic acid; EBV, Epstein–Barr virus; GCP, Cisplatin + Gemcitabine + Carboplatin; GFP, Cisplatin + Gemcitabine + 5-Fluorouracil; GP, Cisplatin + Gemcitabine; GTX, Capecitabine + Gemcitabine + Docetaxel; IC, induction chemotherapy; ICIs, immune checkpoint inhibitors; NPC, nasopharyngeal carcinoma; ORR, objective response rate; OS, overall survival; PD-1, programmed death-1; PD-L1, programmed death ligand-1; PFS, progression-free survival; R/M, recurrence and/or metastatic; RT, radiotherapy; SAE, serious adverse event; SBRT, stereotactic body radiation therapy.
Nanatinostat: a selective class I HDAC inhibitor which induces EBV early lytic phase protein generation, activating (val) ganciclovir to its cytotoxic form.
Represents anti-PD-1 antibody; avelumab: anti-PD-L1 antibody; INCB099318: an oral, small molecule that targets PD-L1; XmAb22841: CTLA-4/LAG-3 bispecific antibody; IBI310: anti-CTLA-4 antibody; AK-104: PD-1/CTLA-4 bispecific antibody; ipilimumab: anti-CTLA-4 antibody; SHR-1701: a bifunctional fusion protein targeting PD-L1 and TGF-β; TQB2858 injection: a bifunctional fusion protein against PD-L1 and transforming growth factor-β; XmAb23104: a bispecific antibody targeting PD-1 and ICOS; XmAb20717: a bispecific antibody targeting PD-1 and CTLA-4 bispecific antibody; BOR: best overall response rate.
The published data from three phase III studies in the first-line setting reported positive results of primary endpoint PFS with a manageable safety profile, which supported that PD-1 inhibitors plus GP regimen become a new standard of care for patients with R/M NPC in the first-line setting. However, OS events were not mature; longer follow-up is needed to assess whether or not the PFS benefit could translate into OS prolongation and long-term adverse effects. All patients enrolled were from the endemic region where the predominant histology was undifferentiated non-keratinizing carcinoma. Whether or not the combined regimen has the same clinical efficacy in regions where keratinizing carcinoma dominance is yet to be elucidated. Comprehensive analysis of potential predictive biomarkers is needed to determine the tailor treatments for individual patients.
Combination of PD-1/PD-L1 inhibitors and antiangiogenic inhibitors in R/M NPC
Angiogenesis inhibitors reshape the TME and normalize the vascularity to enhance cancer immunotherapy using four methods: depletion of the hypoxic TME, improvement of tumor perfusion, facilitation of T-effector cell infiltration, especially CD4(+) and CD8(+) T lymphocytes, CD4(+)FOXP3(+) Tregs with higher expression of PD-L1, and enhancing the differentiation toward M1-like macrophage.63–67 Combination strategy of PD-1/PD-L1 inhibitors and antiangiogenic inhibitors has been successful and obtained approval of indications in various tumors, such as pembrolizumab plus axitinib for advanced renal cell carcinoma, pembrolizumab plus lenvatinib for advanced endometrial carcinoma, atezolizumab plus bevacizumab for hepatocellular carcinoma, and so on. The combination strategies of ICIs plus angiogenesis inhibitors became attractive and receive key attentions.
Several clinical trials have tested antiangiogenic inhibitors in heavily pretreated R/M NPC patients, with variable objective response rates (ORRs) of 2.7–31.3%68–71 (Table 3). The NPC-AXEL study is a single-arm, phase II study evaluating the activity and safety of the combination of axitinib and avelumab in patients with refractory R/M NPC (NCT04562441). Phase II studies regarding the combination of apatinib and camrelizumab, or the combination of bevacizumab and pembrolizumab for patients failing to first-line therapy are currently recruiting (NCT04586088, NCT03813394).
PD-1/PD-L1 inhibitors in LANPC
PD-1/PD-L1 inhibitors in recurrent LANPC
As a consequence of the current excellent loco-regional control rates attained using the generally accepted treatment paradigms involving intensity-modulated radiotherapy for NPC, only 10–20% of patients will suffer from local and/or nodal recurrence after primary treatment. Due to the possibility of radio-resistance of tumor and the limited tolerance of adjacent normal tissues to sustain further additional treatment, such as re-irradiation or chemotherapy, the clinical management of local recurrent NPC remains great challenges. Although systemic chemotherapy is often given, the high-level evidence was lack and the potential aggravating effect of chemotherapy related to increased late toxicities should also be addressed. Ongoing studies focus on the emerging role of immunotherapy in locally recurrent NPC.
Promising results of ICIs in the second-line setting have been reported. However, all of these studies comprise small series of mixed groups with distant metastases and/or local-regional recurrence. Therefore, the exact role of immunotherapy in the management of locally recurrent NPC remains yet to be evaluated. A phase II trial using toripalimab plus intensity-modulated radiotherapy showed promising antitumor activity and satisfactory tolerance in LANPC failing to previous treatment. However, 25 patients unsuitable for surgery were included. ORR and DCR were 79.2% (19/25) and 95.8% (23/25), respectively. The 1-year PFS rate was 91.8%. The acute and late adverse events were tolerable and included blood triglyceride elevation, creatine phosphokinase elevation, skin reaction, mucositis, nasopharyngeal wall necrosis, nasal bleeding, and trismus. 72
PD-1/PD-L1 inhibitors in LANPC at initial diagnosis
About 70% of NPC patients have locally advanced disease at initial diagnosis. Concomitant radio-chemotherapy is the cornerstone of the treatment of locally advanced forms. For the locally advanced stage, the addition of induction chemotherapy has become the new standard care according to the latest international recommendations to reduce tumor volumes and act early on micro-metastases. Despite these therapeutic advances, the local and especially distant failure rate remains high. Promising efficacy results and manageable safety profile of ICIs monotherapy or in combination with chemotherapy in R/M NPC have been reported. The addition of immunotherapy in the multidisciplinary management of LANPC, specifically anti-PD1or PD-L1 antibody, needs to be evaluated.
Radiotherapy not only directly kills tumor cells but also has a synergistic effect with ICIs by improving tumor antigenicity, enhancing tumor immunogenicity, and reshaping the TME to enhance immune surveillance.73–78 Furthermore, radiotherapy in combination with ICIs could not only upregulate the expression of PD-L1 but also decrease the accumulation of MDSC to enhance antitumor activity. 79
Radiotherapy is considered as the main curative treatment for early-stage NPC. Radiotherapy not only directly kills tumor cells but also has a synergistic effect with ICIs by improving tumor antigenicity, enhancing tumor immunogenicity, and reshaping the TME to enhance immune surveillance.73–78 Furthermore, radiotherapy in combination with ICIs could not only upregulate the expression of PD-L1 but also decrease the accumulation of MDSC to enhance antitumor activity. 79 Based on preclinical evidence and promising results of ICIs in R/M NPC, clinical studies on the addition of ICIs to radiotherapy are being conducted in different settings of LANPC; specific information about ongoing clinical trials is shown in Table 4.
Ongoing clinical trials of ICIs in LANPC.
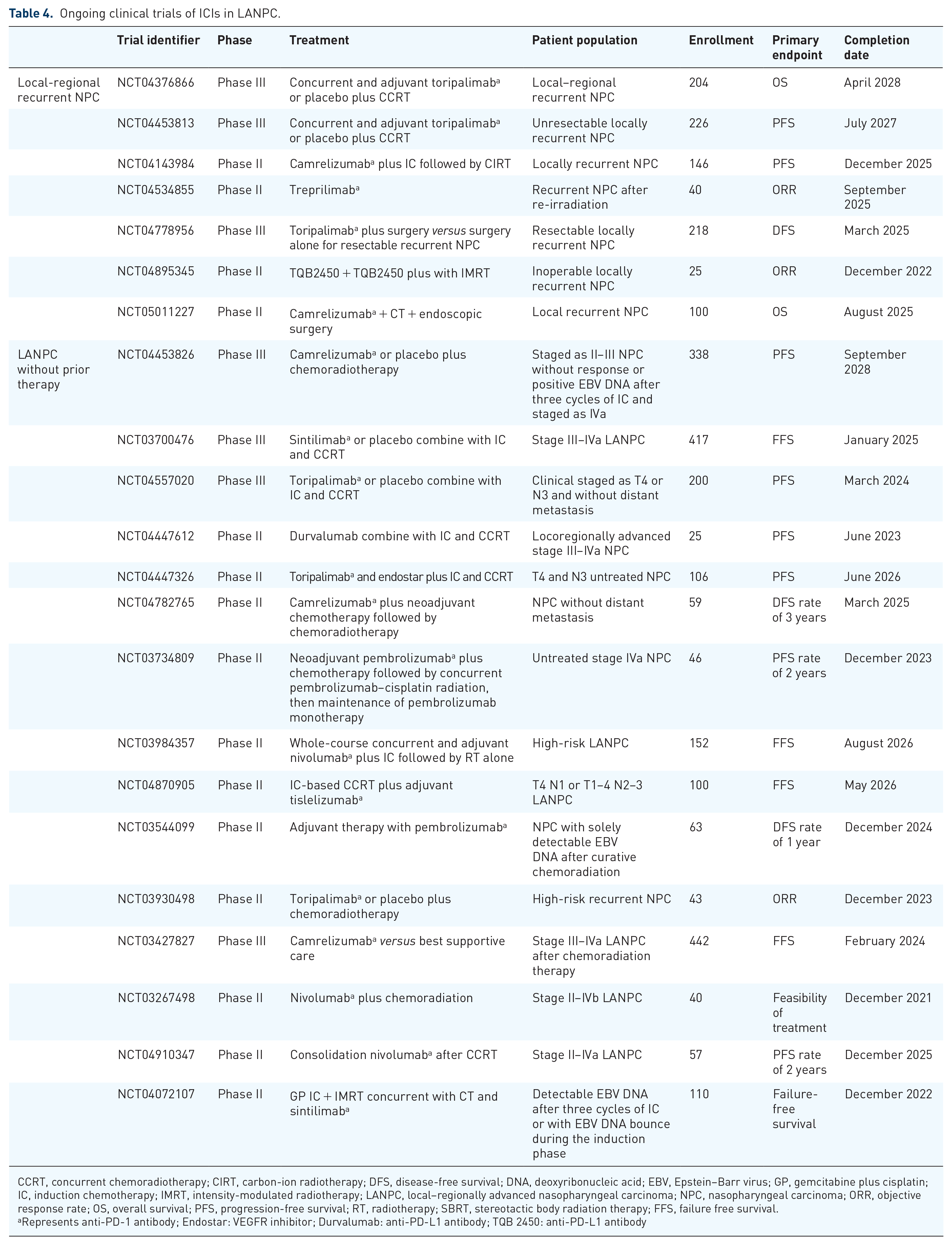
CCRT, concurrent chemoradiotherapy; CIRT, carbon-ion radiotherapy; DFS, disease-free survival; DNA, deoxyribonucleic acid; EBV, Epstein–Barr virus; GP, gemcitabine plus cisplatin; IC, induction chemotherapy; IMRT, intensity-modulated radiotherapy; LANPC, local–regionally advanced nasopharyngeal carcinoma; NPC, nasopharyngeal carcinoma; ORR, objective response rate; OS, overall survival; PFS, progression-free survival; RT, radiotherapy; SBRT, stereotactic body radiation therapy; FFS, failure free survival.
Represents anti-PD-1 antibody; Endostar: VEGFR inhibitor; Durvalumab: anti-PD-L1 antibody; TQB 2450: anti-PD-L1 antibody
For LANPC at initial diagnosis, clinical studies on the addition of ICIs to radiotherapy are being conducted in different settings of LANPC; specific information about ongoing clinical trials is shown in Table 4. The phase III clinical trials regarding the addition of toripalimab, camrelizumab, or sintilimab to induction and concurrent chemotherapy are in the recruitment stage (NCT04557020, NCT04453826, NCT03700476, NCT04557020). Interestingly, a phase III clinical trial (NCT04453826) focused on high-risk LANPC (staged as II–III without response, or EBV DNA >0 copies/ml after three cycles of chemotherapy with GP induction, or staged as IVa) are exploring the role of concurrent and adjuvant camrelizumab combined with chemoradiotherapy. The results of this trial are anticipated due to the prospect of personalized treatment based on the response to induction therapy. Phase II clinical trials using whole-course concurrent and adjuvant nivolumab, pembrolizumab, camrelizumab, toripalimab, and durvalumab (anti-PD-L1 antibody) in LANPC patients are also ongoing (Table 4).
In general, the breakthrough of immunotherapy in LANPC is worth looking forward to. The ongoing clinical trials are focusing on those patients with high-risk [detectable EBV DNA following induction chemotherapy and concurrent chemoradiothrapy (CCRT) or stage of T4 N2-3M0] and explore different schedule of ICIs (e.g. induction chemotherapy to CCRT plus ICI, CCRT plus PD1 or PD-L1 inhibitors followed by 1 year of adjuvant PD1 or PD-L1 inhibitors, induction chemotherapy to CCRT plus ICI, and so on). The multiple variants of radiotherapy synergizing ICIs also need to be considered, including the dose, volume, fractionation, and the schedule of radiotherapy. These results from different clinical trials may answer the optimal multidisciplinary management in the era of immunotherapy.
Other immunotherapy in NPC
Based on the immune landscape of NPC, there are other approaches to enhance the patients’ immunity apart from ICIs. Promoting tumor antigenicity, tumor immunogenicity, and stimulating the killing ability of T-cells are three key elements to activate an immune response. EBV-specific vaccines and autologous DCs are aimed at increasing tumor antigenicity, whereas adoptive T-cell transfer focuses on promoting tumor immunogenicity and T-cell killing ability. In addition, the immune modulator is an interesting topic for releasing the immune suppressive TME. Although challenges exist in the translation from bench to bedside, further exploration appears promising.
Cancer vaccine in NPC
As early as 2002, the first clinical study of autologous immune cell vaccine was conducted by injecting autologous DCs with human leukocyte antigen (HLA)-restricted epitopes from EBV LMP-2 into 16 patients with local R/M NPC. Nine patients demonstrated boosted CD8 + T-cell responses and two patients had partial response (PR). 80 Subsequently, a series of clinical trials of therapeutic EBV vaccine have been launched. In 2011, a Singapore clinical trial designed a new Ad-ΔLMP1-LMP2 DC vaccine without HLA restriction and enrolled 16 participants with advanced NPC. However, limited T-cell response was detected. Only one patient had PR and two SD. 81
Virus-based vaccine seems to be an effective and convenient substitute for traditional cell vaccine. An modified vaccinia Ankara EBNA1/LMP (MVA-EL) virus-based vaccine which encodes inactive EBNA1/LMP2 fusion protein was tested in two phase-I clinical trials. One trial enrolled 18 Hong Kong NPC patients; T-cell responses were detected in 15 subjects and had a positive correlation with vaccine dose. 82 In another UK trial, enhanced immune response to either specific antigen was observed in 8 of 14 patients regardless of ethnicities. 83 In addition, with improved accuracy and specificity for the detection of antigens, immunogenic peptide-based therapeutic vaccines tailored for specific immune profiles may be available soon and become a new trend. 84
Adaptive immunotherapy in NPC
Adaptive immunotherapy is a distinct approach to stimulate immune response by bypassing the antigen presentation step and direct activation of the effector cells. Various preclinical studies have explored the application of CTLs, TILs, natural killer (NK) cells, and DCs in the treatment of R/M NPC with certain clinical benefits. 85 Most clinical trials focused on CTLs, and the CTLs-related information are shown in Table 5. Corey Smith et al. conducted a phase II trial to investigate the autologous EBV-specific T-cells generated with a novel AdE1-LMPpoly vector in R/M NPC. In total, 29 patients completed therapy without significant toxicity. The median PFS was 5.5 (95% CI 2.1–9.0) months and the median OS was 38.1 months (95% CI 17.2 months to not reached). 86
Completed clinical trials of EBV-CTLs in NPC.

CR, complete response; CT, chemotherapy; CTL, cytotoxic T-cell; DCR, disease control rate; DFS, disease-free survival; EBV, Epstein–Barr virus; GC, gemcitabine and carboplatin; LMP, latent membrane protein; N/A, not available; N/MRD, no or minimal residual disease; NPC, nasopharyngeal carcinoma; ORR, objective response rate; OS, overall survival; PD, progressive disease; PFS, progression-free survival; PR, partial response; R/M, recurrent/metastatic; R/R, refractory or relapsed; Ref., reference; SD, stable disease; WHO, World Health Organization; IL-2, interleukin-2.
29a:1 Patient of 30 pts died after single administration due to lung abscess.
38b: 35 of 38 pts received GC and EBV-CTL.
21c: 21 of 28 enrolled pts received EBV-CTL.
A phase II clinical trial evaluated the efficacy and safety of GC regimen followed by up to six doses of EBV-CTL for R/M NPC. 87 In total, 35 patients were enrolled and achieved ORR of 71.4% (including 3 CR and 22 PR) and 2-year OS rate was 62.9%. Thereafter, a randomized phase III study (VANCE, NCT02578641) is ongoing, which aims to assess whether the GP regimen followed by adoptive T-cell therapy as first-line treatment for R/M NPC could prolong OS, compared with GP. The study is expected to enroll 330 patients from 30 hospital centers across Asia and the United States. Information of other ongoing clinical trials of EBV-CTLs in NPC is showed in Table 6.
Ongoing clinical trials of EBV-CTLs in NPC.

CT, chemotherapy; CTL, cytotoxic T-cell; DNR, dominant negative receptor; EBV, Epstein–Barr virus; GC, gemcitabine and carboplatin; Mab, monoclonal antibody; MABEL, LMP, BARF-1 and EBNA1; N/A, not available; NPC, nasopharyngeal carcinoma; ORR, objective response rate; OS, overall survival; R/M, recurrent/metastatic; R/R, refractory or relapsed; Ref., reference.
Adoptive immunotherapy could be a potential and effective treatment for NPC. Notably, immune-mediated antitumor activities were observed in heavily pre-treated NPC patients. EBV-CTL showed some efficacy, but T-cell amplification technology needs to be further optimized. The results of EBV TCR-T in the treatment of NPC are worthy of expectation. The translation from bench to bedside is challenging and its current status is yet to be completely discovered. The encouraging results in refractory NPC patients promote the exploration of adaptive therapy as the first-line setting.
New ICI agents beyond PD-1/PD-L1 inhibitors in NPC
Apart from PD-1/PD-L1 pathway, there are other co-inhibitory pathways, such as CTLA4, LAG-3, TIGIT, and TIM3, which suppress immune cell activation and avoid immune surveillance in TME, and co-stimulatory pathways including CD27/CD70, CD40/CD40 L, 4-1BB/4-1BBL, and OX40/OX4, which regulate T-cell function.52–55
The dual immune checkpoint blockade strategies, including CTAL4, TIM-3, LAG-3, and TIGIT, synergized with anti-PD-1/PD-L1 antibody in enhancing TIL function and suppressing tumor growth. They are in phase I–III clinical trials, having not been approved by the FDA or NMPA. The results of these clinical trials exhibited encouraging antitumor activity even in some patients with prior ICI treatment and were tolerable. Bispecific antibody targeting two inhibitory immune checkpoints including CTLA-4 plus PD-L1, LAG-3 plus PD-L1, TIM-3 plus PD-L1, TIGIT plus PD-L1, and PD-1 plus inducible T-cell co-stimulator (ICOS) have been developed. Most bispecific antibodies achieved excellent antitumor efficacies in murine tumor models. Some bispecific antibodies have been in clinical trials, and the specific information is shown in Table 3.
Biomarkers for personalized immunotherapy in NPC
Despite the substantial advancements in clinical care, a small subset of patients with R/M NPC can obtain long-lasting clinical benefits and improved survival from immunotherapy. Identifying biomarkers that can predict response and elucidate the biological mechanisms underlying response or non-response to immunotherapy are of paramount importance. Some predictive biomarkers derived from tumor tissues or peripheral blood, such as PD-L1 expression, plasma EBV DNA, tumor mutational burden (TMB), and actionable hot spot mutations, have been investigated.42,43,61,97–99
PD-L1 expression
Tumor PD-L1 expression has been the most commonly explored biomarker for predicting the response to PD-1 and PD-L1 blockade across tumor types. However, a meta-analysis including 1836 NPC patients from 15 studies concerning PD-L1 expression did not observe a significant correlation between PD-L1 expression and OS, which indicated that the expression level of PD-L1 may not act as a useful prognostic biomarker for NPC. 100 Several studies have explored the predictive value of anti-PD-1/PD-L1 in single or combined therapy in NPC, and the specific information is shown in Table 7.
The PD-L1 expression and the predictive value of NPC.
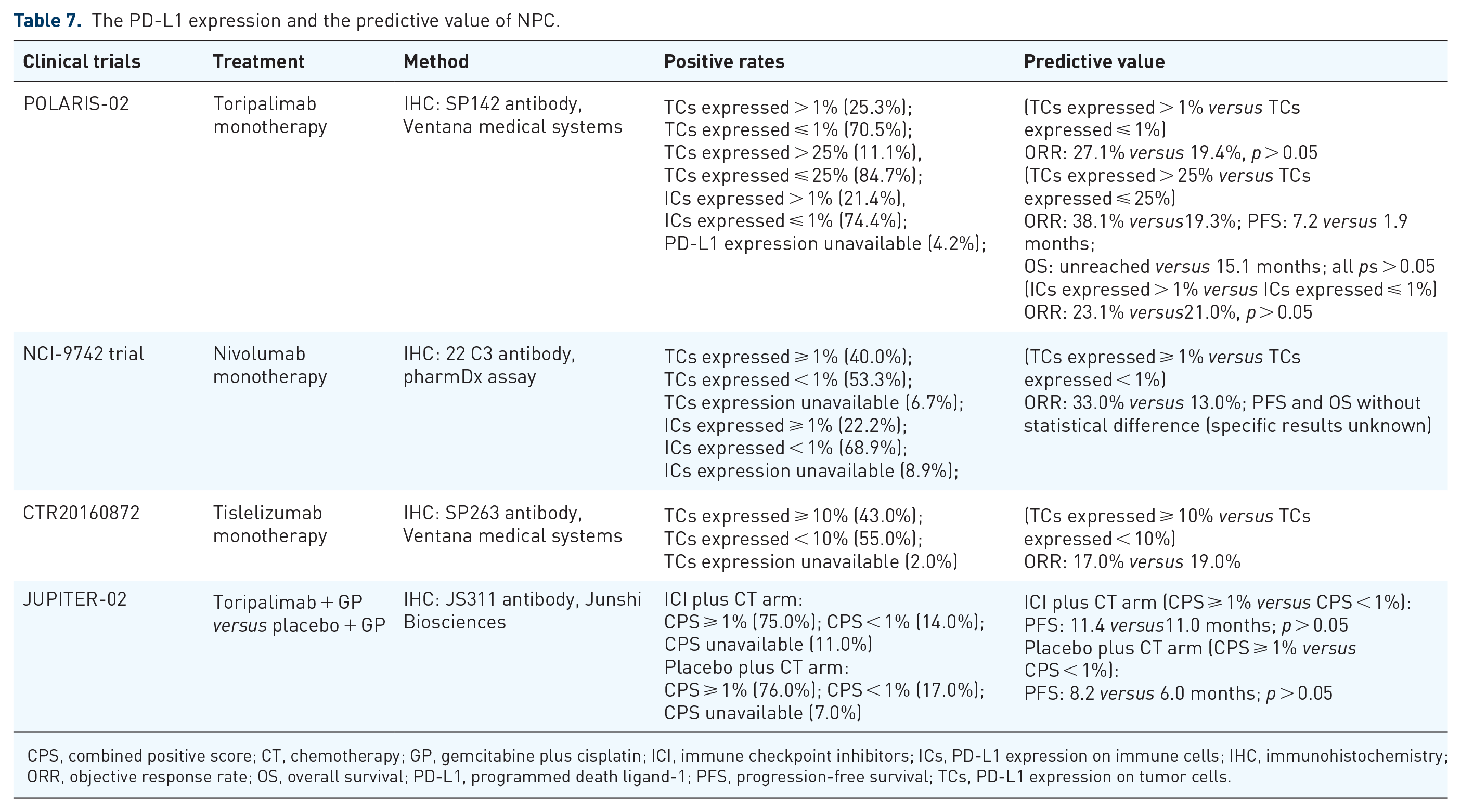
CPS, combined positive score; CT, chemotherapy; GP, gemcitabine plus cisplatin; ICI, immune checkpoint inhibitors; ICs, PD-L1 expression on immune cells; IHC, immunohistochemistry; ORR, objective response rate; OS, overall survival; PD-L1, programmed death ligand-1; PFS, progression-free survival; TCs, PD-L1 expression on tumor cells.
The POLARIS-02 trial showed that 21/190 (11.1%) patients treated with toripalimab had high PD-L1 expression (>25%) and were associated with a trend of higher ORR, better median PFS, and median OS than patients with low PD-L1 expression, whereas no significant differences between the two groups were observed. 43 In addition, there was no difference in ORR between PD-L1 positive (>1%) and PD-L1 negative expression on immune cells. Similarly, responses to tislelizumab were observed in R/M NPC, regardless of PD-L1 expression in a phase I/II study of tislelizumab in Chinese patients with advanced solid tumors. 101 The NCI-9742 trial evaluated the antitumor activity of nivolumab in R/M NPC. There was no significant difference between patients with PD-L1-negative and PD-Ll-positive tumors in terms of OS or PFS. However, a descriptive analysis showed that a higher proportion of patients with higher levels of PD-L1 expressing tumors responded to nivolumab than those with PD-L1-negative tumors. 97 The phase III JUPITER-02 study explored the predictive value of PD-L1 expression for PD-1 inhibitor combined chemotherapy in NPC. A clinical benefit from using toripalimab along with the GP regimen was observed regardless of PD-L1 expressions status. 61
There is significant variability in the literature regarding the prevalence and prognostic significance of PD-L1 expression in NPC. The use of PD-L1 as a predictive biomarker in NPC is limited by the lack of consensus in defining a clinically meaningful threshold, the choice of assay, and the optimal analytical method to be used. In summary, current studies did not prove that high PD-L1 expression would be the optimal biomarker to predict efficacy and prognosis in patients with R/M NPC receiving immunotherapy, but its value merits further investigation.
Plasma EBV DNA
Plasma EBV DNA has been considered as a useful biomarker for population screening, disease surveillance, and prognosis or efficacy prediction of chemotherapy and radiotherapy.102–107 The value of plasma EBV DNA as a biomarker in the era of NPC immunotherapy needs to be verified.
The predictive and prognostic effects of baseline plasma EBV DNA in NPC treated with ICIs are under-investigated. In the POLARIS-02 study, patients with baseline EBV DNA titers less than 10,000 IU/mL had numerically higher ORR from toripalimab (26.7% versus 15.4%); however, the difference was not statistically significant. Similarly, a trend toward a lower overall response in the patients with higher EBV DNA levels receiving camrelizumab monotherapy was observed, but the difference was not statistically significant.42,43,60
The dynamic change in plasma EBV DNA after immunotherapy could be a more reliable and promising biomarker for the response. The POLARIS-02 study indicated that an early decrease in plasma EBV titer during the first 4 weeks was significantly correlated with a favorable response and survival benefit to toripalimab monotherapy.43,108 The significant increase in plasma EBV DNA titer could predict the disease progression prior than radiological review. 108 Although the plasma EBV DNA clearance and the increasing or decreasing trend of EBV DNA during the first month with nivolumab monotherapy did not differ significantly in terms of efficacy, more responders showed a decreasing trend of plasma EBV DNA. The failure to demonstrate statistical significance might be due to the small number of patients investigated. 97 The CAPTAIN-1st trial demonstrated that the early clearance of plasma EBV DNA was related to the response rate of the combination of camrelizumab and the GP regimen. 60 However, the predictive value of dynamic plasma EBV DNA for survival is unclear and warrants further exploration. The change in plasma EBV DNA was not significantly related to PFS in patients who received camrelizumab or nivolumab monotherapy, whereas in a phase I study of camrelizumab in combination with GP, the patients with post-baseline undetectable EBV DNA after the first month of camrelizumab had significantly longer PFS than those with detectable post-baseline EBV DNA. 42
TMB
NPC is a tumor with low TMB, as shown by several studies, with a median TMB ranging from 0.9 to 3.3 muts/Mb.42,43,109 Both the POLARIS-02 study of toripalimab monotherapy and the CAPTAIN study of camrelizumab monotherapy had negative results regarding value of TMB.42,43 In the POLARIS-02 study, patients with low TMB had the same median PFS as those with high TMB (1.9 months) and despite having longer median OS (17.4 versus 9.2 months), no statistical difference was observed between the two groups. 43 The clinical response was not significantly related to TMB level, with a cutoff value of 2.9 muts/Mb (top 10% TMB value). In the CAPTAIN study, there was no difference in PFS or ORR between patients with low and high TMB. 42 There are no data regarding the predictive value of TMB in ICI combination treatment in R/M NPC.
Other biomarkers
Recently, researchers have attempted to identify predictors of R/M NPC responses to immunotherapy through whole-exome sequencing (WES). The POLARIS-02 study reported that patients with genomic amplification in the 11q13 region or ETV6 genomic alterations by WES had extremely poor responses to toripalimab. 43 It was also found that copy number loss in either GZMB or GZMH genes was related to the poor survival of R/M NPC patients receiving camrelizumab monotherapy. 110
Immune signatures and subtypes have been attracting attention since the development of single-cell RNA-seq revealed the genomic and immune landscape of NPC.111–113 Chen et al. 112 demonstrated the TME of NPC and identified three immune subtypes (immune-enriched subtype, evaded subtype, and active immune subtype) that could predict the prognosis and response of NPC patients from immunotherapy plus chemotherapy. A prognostic model for immunotherapy was established using the expression of nine immune checkpoints according to five features (B7-H3TAIC, IDO-1TAIC, VISTATAIC, ICOSTAIC, and LAG3TAIC). 114 The integration of four immune markers (PD-L1+, CD163+, CXCR5, and CD117) in the intratumor is useful for predicting distant metastasis in NPC, as this may also be a promising biomarker for immunotherapy. 113 Further studies are required to verify the predictive value of the immune signature in NPC to better understand their potential for suggesting appropriate immunotherapy regimens and estimating the corresponding treatments treatment outcomes in different patient subgroups through immune signature. This might be challenging in clinical practice because of the limited number of patients and hypothesis-generating studies.
Conclusion
The general immune landscape of NPC renders patients suitable for immunotherapy, especially ICIs. Clinical trials regarding R/M NPC have confirmed the safety and antitumor activity of anti-PD-1 monotherapy in late-line settings and the addition of PD-1 inhibitor to chemotherapy in first-line settings. For LANPC, a variety of clinical trials on the addition of PD-1 inhibitor to radio-chemotherapy are ongoing, and their results are worth expecting. In addition, anti-PD-1 treatment strategies, such as its combination with anti-vascular agents and other immunotherapies, are promising and explore broad spheres. EBV vaccine, adoptive T-cell therapy and other adoptive immunotherapy could be a potential and effective treatment for NPC, although their current status is challenging and remains to be completely discovered. Finally, the dynamic changes in plasma EBV DNA could be a useful and practical biomarker for immunotherapy in NPC. However, well-known biomarkers, such as PD-L1 expression and TMB, have not been proven to have a clear clinical predictive value for immunotherapy in NPC. More studies regarding biomarkers that effectively predict the response and prognosis of anti-PD-1 monotherapy or combination therapy are required to maximize the personalized treatment approaches in NPC. Thus, further investigations regarding the molecular and cellular drivers of immune escape to overcome immunotherapy resistance are of great significance and could lead to innovative treatment approaches to improve the treatment outcomes of NPC patients.
Footnotes
Acknowledgements
None
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Author contribution(s)
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflict of interest statement
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Availability of data and materials
Main data are shown in this article and additional data about this study could be obtained from the corresponding author on reasonable request.
