Abstract
Objectives:
Blocking the programmed cell death-1 (PD-1)/programmed cell death-ligand 1 (PD-L1) axis could reverse the immune tolerance in the liver microenvironment of hepatocellular carcinoma (HCC). We investigated the efficacy and the safety of PD-1/PD-L1 inhibitors and the possibility of hepatitis B surface antigen (HBsAg) or hepatitis B virus (HBV) DNA levels as prognostic biomarkers in patients with HBV-related HCC.
Design:
A retrospective study.
Methods:
We retrospectively analyzed patients with HBV-related HCC and positive quantitative HBsAg (qHBsAg) who received PD-1/PD-L1 inhibitor therapy at least once. The primary endpoints were overall survival (OS) and objective response rate (ORR), with the secondary endpoint being disease control rate (DCR). Cox regression models were used to illustrate the association of patient characteristics with survival.
Results:
A total of 235 patients with HBV-related HCC were included in this study. The median OS for all patients was 20.9 months. The ORR and DCR were 15.7% and 70.6%, respectively. Baseline HBV DNA levels were associated with DCR (p = 0.004). Patients in the qHBsAg-response group in the fourth week had a longer OS after PD-1/PD-L1 inhibitor treatment compared to those in the qHBsAg nonresponse group (29.1 months vs 14.9 months, p = 0.04). Multivariate Cox regression analysis suggested that positive baseline HBV DNA (adjusted hazard ratio (aHR) = 2.6, p = 0.04) and qHBsAg nonresponse at week 4 after PD-1/PD-L1inhibitor therapy (aHR = 2.2, p = 0.04) were independent risk factors for survival.
Conclusion:
PD-1/PD-L1 inhibitor therapy in patients with HBV-related HCC showed better efficacy and safety. Negative HBV DNA and a short-term decline in qHBsAg from baseline were associated with superior survival prognosis.
Keywords
Introduction
Hepatocellular carcinoma (HCC), which accounts for 90% of liver cancers, is the third leading cause of cancer-related deaths worldwide.1,2 Globally, liver cancer fatalities due to hepatitis B virus (HBV) and hepatitis C virus (HCV) infection are estimated at 265,000 (33%), with the Asia–Pacific region bearing the highest HBV prevalence, accounting for three-quarters of all chronic HBV-positive subjects worldwide.3–5 In China, a country with a high risk of HCC, it accounts for 45.3% of global liver cancer incidence and 47.1% of mortality. Notably, most HCC cases in China are related to HBV carrier status.6,7
Immune checkpoint inhibitors (ICIs) have recently become a promising option to improve the prognosis of advanced HCC. 8 Programmed cell death-1 (PD-1), a major inhibitory immune checkpoint receptor, is involved in regulating the intensity of tumor response. 9 Although PD-1/programmed cell death-ligand 1 (PD-L1) inhibitors in phase I/II clinical trials have shown improved efficacy and safety, 10 their efficacy and safety in patients with HBV-related HCC need further real-world studies. Such studies are required particularly in cases with high levels of hepatitis B surface antigen (HBsAg) and viral loads, since they were rarely enrolled in clinical trials.
HBsAg plays a vital role in the development and recurrence of HCC.11–14 Furthermore, several studies have shown that patients with HCC with a negative HBsAg expression have better prognostic outcomes than those with a positive HBsAg expression.15–19 Notably, PD-1/PD-L1 blockers may affect the tumor response and play a role in anti-HBV therapy.20–22 Studies have also demonstrated that inhibiting the PD-1/PD-L1 pathway can reverse T-cell depletion and enhance the specific anti-HBV T-cell response in human liver T cells and HBV models, such as mice and marmots.23,24 In the Checkmate-040 cohort, nivolumab was used for the first time in patients with advanced HCC; 3 (6%) of 51 patients with chronic hepatitis B (CHB) experienced a 1 log decrease in HBsAg during nivolumab treatment. 10 Nivolumab, with or without the GS-4774 vaccine, has been effective in restoring the HBV-specific immune response in those with chronic HBV infection. 25 These studies suggest that PD-1/PD-L1 inhibitors may treat CHB. However, it remains unclear whether PD-1/PD-L1 inhibitors can decrease HBsAg levels and if baseline HBsAg or HBV DNA levels, or their variations, affect the survival of patients with HCC. This study investigates the antiviral effect of PD-1/PD-L1 inhibitor treatment and the possibility of using HBsAg or HBV DNA as prognostic biomarkers for HCC clinical outcomes.
Methods
Study design and patients
We conducted a retrospective cohort study involving patients diagnosed with HCC through histological or imaging examination. These patients were seropositive for quantitative HBsAg (qHBsAg) and had received at least one cycle of PD-1/PD-L1 inhibitor therapy at Southern Medical University Nanfang Hospital from July 2018 to December 2022. Recorded data included patient demographics, Child-Pugh stage, Barcelona Clinic Liver Cancer (BCLC) stage, Eastern Cooperative Oncology Group performance status (ECOG PS), type of nucleos(t)ide analogs (NAs) for HBV, antitumor therapy before and/or during PD-1/PD-L1 inhibitor treatment, the levels of serological markers for HBV infection, alanine aminotransferase (ALT), aspartate aminotransferase (AST), and alpha-fetoprotein (AFP) levels at baseline.
To be eligible for this study, patients had to meet the following inclusion criteria: (1) diagnosis of HCC through histological or imaging examination; (2) age ⩾ 18 years; (3) seropositive qHBsAg at least 6 months before PD-1/PD-L1 inhibitor therapy; (4) no previous antiviral therapy with interferon; (5) at least one imaging evaluation before and after PD-1/PD-L1 inhibitor therapy; (6) and at least one test result for qHBsAg and HBV DNA before and after PD-1/PD-L1 inhibitor treatment. Patients were excluded if they met any of the following criteria: (1) the presence of other positive viral markers, including immunoglobulin M antibodies to the hepatitis A virus, HCV, or hepatitis E virus or immunoglobulin G antibodies to the hepatitis D virus or human immunodeficiency virus (HIV); (2) a current or past history of another malignant tumor or autoimmune-related diseases; (3) the presence of spontaneous bacterial peritonitis with liver cancer; (4) or current or previous treatment with corticosteroids or immunosuppressants. Ultimately, 235 patients with complete data were enrolled in the study.
For PD-1/PD-L1 inhibitors, camrelizumab, pembrolizumab, toripalimab, sintilimab, tislelizumab, atezolizumab, or nivolumab were allowed. The dosages were 200 mg (atezolizumab: 1200 mg; toripalimab: 240 mg; nivolumab: 240 mg per 2 weeks), administered intravenously every 3 weeks. The choice of PD-1/PD-L1 inhibitors and treatment duration were jointly decided by physicians and patients in real-world practice. This included information on medical history, lab results, radiological findings, and treatment regimens both pre- and post-PD-1/PD-L1 inhibitor therapy. All follow-ups were conducted from the start of PD-1/PD-L1 inhibitor treatment until May 31, 2023.
HBV reactivation in patients with positive HBsAg was defined according to the American Association for the Study of Liver Diseases 2018 hepatitis B guidance. 26 Radiological response was recorded using computed tomography or magnetic resonance imaging initially at baseline, 6–12 weeks after PD-1/PD-L1 inhibitor therapy initiation, and then at 3-month intervals. Tumor response was evaluated according to the modified Response Evaluation Criteria in Solid Tumors. Serological markers for HBV infection, including qHBsAg and HBV DNA, were assessed at least every 4 weeks after starting PD-1/PD-L1 inhibitor therapy.
Patients were divided into two groups based on the detectability of the baseline HBV DNA: the HBV DNA-positive group, with a baseline HBV DNA quantification >20 IU/mL before PD-1/PD-L1 inhibitor treatment; and the HBV DNA negative, with a baseline HBV DNA quantification ⩽20 IU/mL before PD-1/PD-L1 inhibitor treatment. Patients were also divided into two groups according to the degree of decline in qHBsAg levels after PD-1/PD-L1 inhibitors therapy: (1) the qHBsAg-response group, where qHBsAg decreased by more than 0.1 log10 IU/mL compared with baseline; and (2) the qHBsAg nonresponse group, where qHBsAg did not decrease by more than 0.1 log10 IU/mL from baseline or increased from baseline.
Statistical analysis
The primary endpoints of this study were overall survival (OS) and objective response rate (ORR). OS was defined as the time between the start of the first PD-1/PD-L1 treatment and the last follow-up visit or death, whichever occurred first. ORR was defined as the proportion of patients whose tumor size was reduced to a predetermined value and maintained this reduction for a minimum duration, typically encompassing the sum of the proportion of complete response (CR) to partial response (PR). Disease control rate (DCR) was a secondary endpoint, defined as the percentage of evaluable cases achieving remission (PR + CR) and disease stabilization (stable disease (SD)) with treatment. Quantitative data were expressed as mean ± standard deviation for normally distributed values and as median (range) for non-normally distributed variables. Categorical variables are expressed as counts (percentages). The two-sampled t test or Mann–Whitney U test was used to compare continuous variables. The Pearson χ2 analysis or Fisher’s exact test was used to compare categorical variables. The cut-off value for baseline qHBsAg level (best cut-off value = 500 IU/mL) was determined using the restrictive cubic spline (RCS) method (Figure 1). Survival time was estimated using the Kaplan–Meier method; any differences in survival time were evaluated with a stratified log-rank test. Independent predictive variables for OS were determined with univariate and multivariate Cox regression analyses. Statistical analyses were performed using IBM SPSS Statistics 25.0 (Armonk, NY: IBM Corp). Statistical significance was set at p < 0.05. All statistical tests were two-sided.

Restricted cubic spline analyses for Baseline qHBsAg level.
Reporting guideline
The reporting of this study conforms to Strengthening the Reporting of Observational Studies in Epidemiology (STROBE; Supplemental Table 1). 27
Results
Study cohort
This study enrolled 235 patients with HBV-related HCC who were treated with PD-1/PD-L1 inhibitors. Among them, 171 (72.8%) patients were ⩽60 years old, and 215 (91.5%) patients were male. Prior to PD-1/PD-L1 inhibitor therapy, 155 (66.0%) patients received antiviral therapy. All patients received antiviral therapy at the initiation of PD-1/PD-L1 inhibitor therapy. Of the patients, 160 (68.1%) were seropositive for baseline HBV DNA (baseline viral load >20 IU/mL), while 75 (31.9%) were seronegative (baseline viral load ⩽20 IU/mL). Other details are listed in Table 1.
Baseline characteristics and clinical outcomes of patients enrolled in the study.

AFP, alpha-fetoprotein; BCLC, Barcelona Clinic Liver Cancer; CI, confidence interval; CR, complete response; DCR, disease control rate; ECOG PS, Eastern Cooperative Oncology Group performance status; ETV, entecavir; HAIC, hepatic artery infusion chemotherapy; HBV, hepatitis B virus; ORR, objective response rate; PD, progressive disease; PD-1/PD-L1, programmed cell death-1/programmed cell death-ligand 1; PR, partial response; qHBsAg, quantitative hepatitis B surface antigen; SD, stable disease; TACE, transcatheter arterial chemoembolization; TAF, tenofovir alafenamide; TDF, tenofovir disoproxil.
Overall tumor response and survival time profiles
The median OS for all patients was 20.9 months (95% confidence interval (CI): 17.2–24.6). Of all the included patients, 8 (3.4%) achieved a CR, 29 (12.3%) achieved a PR, and 129 (54.9%) had SD, yielding an ORR of 15.7% and a DCR of 70.6% (Table 1).
Antiviral efficacy of PD-1/PD-L1 inhibitor therapy
At week 4 post-treatment, 63.2% (72/114) of patients experienced a decrease in qHBsAg levels, with 51.4% (37/72) exhibiting a reduction of more than 0.1 log10 IU/mL. At week 12 post-treatment, qHBsAg data were available in 121 cases. Approximately 71.9% (87/121) of patients had a decrease in qHBsAg levels, of which 62.1% had a decrease of more than 0.1 log10 IU/mL. After 24 weeks of treatment, a decrease in qHBsAg levels was observed in 64.3% (63/98) of patients; a decrease of more than 0.1 log10 IU/mL was detected in 77.8% (49/63). Among the 62 patients with available qHBsAg data at week 48, qHBsAg levels decreased in 77.4% (48/62); a decrease of more than 0.1 log10 IU/mL was observed in 77.1% (37/48) (Figure 2(a)–(d)).

qHBsAg dynamics at weeks (a) 4, (b) 12, (c) 24, and (d) 48 after PD-1/PD-L1 inhibitors therapy. Bars are expressed as qHBsAg change from baseline.
Observation of HBV reactivation
Among the 235 patients with HBV-associated HCC treated with PD-1/PD-L1 inhibitors, one patient with positive baseline HBV DNA who started antiviral therapy simultaneously with PD-1/PD-L1 inhibitors had an increase in HBV DNA levels of 3.35 log10 IU/mL (from 2.57 to 5.92 log10 IU/mL) at week 12, meeting the criteria for HBV reactivation. However, no significant changes in ALT levels were observed in this patient, indicating that the patient did not develop HBV reaction-associated hepatitis. No other patients experienced HBV reactivation during PD-1/PD-L1 inhibitor treatment.
Correlation between baseline HBV DNA levels, tumor response, and survival time
Of the 235 patients, 75 (31.9%) were HBV DNA negative, and 160 (68.1%) were HBV DNA positive. ORRs in patients with seronegative and seropositive baseline HBV DNA levels were 10.6% and 18.1%, respectively (p = 0.16), while the DCRs were 82.7% and 65.0%, respectively (p < 0.05). Baseline HBV DNA levels were associated with a better DCR (Table 2). The median OS was 21.9 months (95% CI: 10.7–33.2) for HBV DNA-negative patients and 18.1 months (95% CI: 11.1–25.1) for HBV DNA-positive patients, with significant differences between the two groups (p < 0.05, Figure 3(a) and Table 2).
Efficacy according to different baseline HBV DNA levels.

CI, confidence interval; CR, complete response; DCR, disease control rate; HBV, hepatitis B virus; ORR, objective response rate; PD, progressive disease; PR, partial response; SD, stable disease.
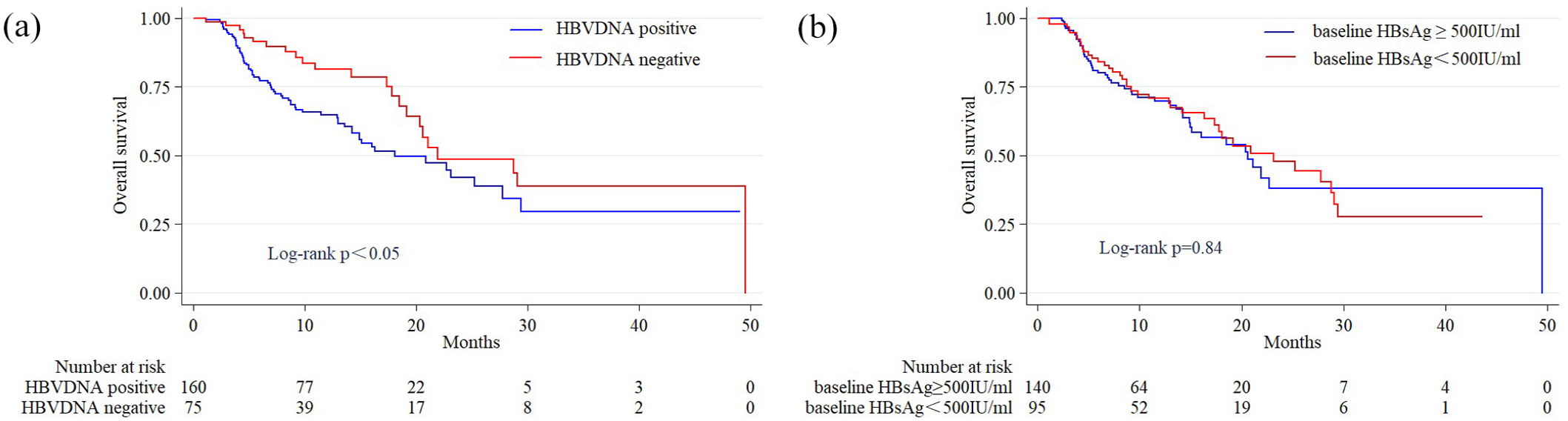
Correlation analysis between baseline qHBsAg and HBV DNA level with survival time. (a) Comparison of survival curves of patients with different baseline HBV DNA levels. (b) Comparison of survival curves of patients with different baseline qHBsAg levels. Log-rank test for group comparisons.
Correlation between baseline qHBsAg levels and survival time
To investigate the correlation between baseline qHBsAg levels and patient mortality, we used the RCS method to simulate and visualize the relationship. Despite the absence of significant difference in the RCS curve results (Figure 1), we selected the corresponding qHBsAg level for hazard ratio = 1 as the cut-off value and divided all patients into two groups: patients with baseline qHBsAg levels ⩽500 IU/mL and those with baseline qHBsAg levels >500 IU/mL. The survival time between the two groups was not statistically significant (p = 0.84, Figure 3(b)).
Correlation between qHBsAg dynamics after PD-1/PD-L1 inhibitor therapy and survival time
Patients with available data were categorized into two groups according to whether the decrease in qHBsAg titer was greater than 0.1 log10 after PD-1/PD-L1 inhibitor treatment: the qHBsAg-response group, where qHBsAg decreased by more than 0.1 log10 IU/mL compared with baseline; and the qHBsAg nonresponse group, where qHBsAg did not decrease by more than 0.1 log10 IU/mL from baseline or increased from baseline. We compared the OS between qHBsAg-response and qHBsAg nonresponse groups at weeks 4, 12, 24, and 48, receiving PD-1/PD-L1 inhibitor therapy. The median OS for the qHBsAg-response group in the fourth week was significantly longer (29.1 months vs 14.9 months, p = 0.04, Figure 4(a)), while the median OS was comparable between qHBsAg-response and qHBsAg nonresponse groups at weeks 12, 24, and 48 (p > 0.05, Figure 4(b)–(d)).
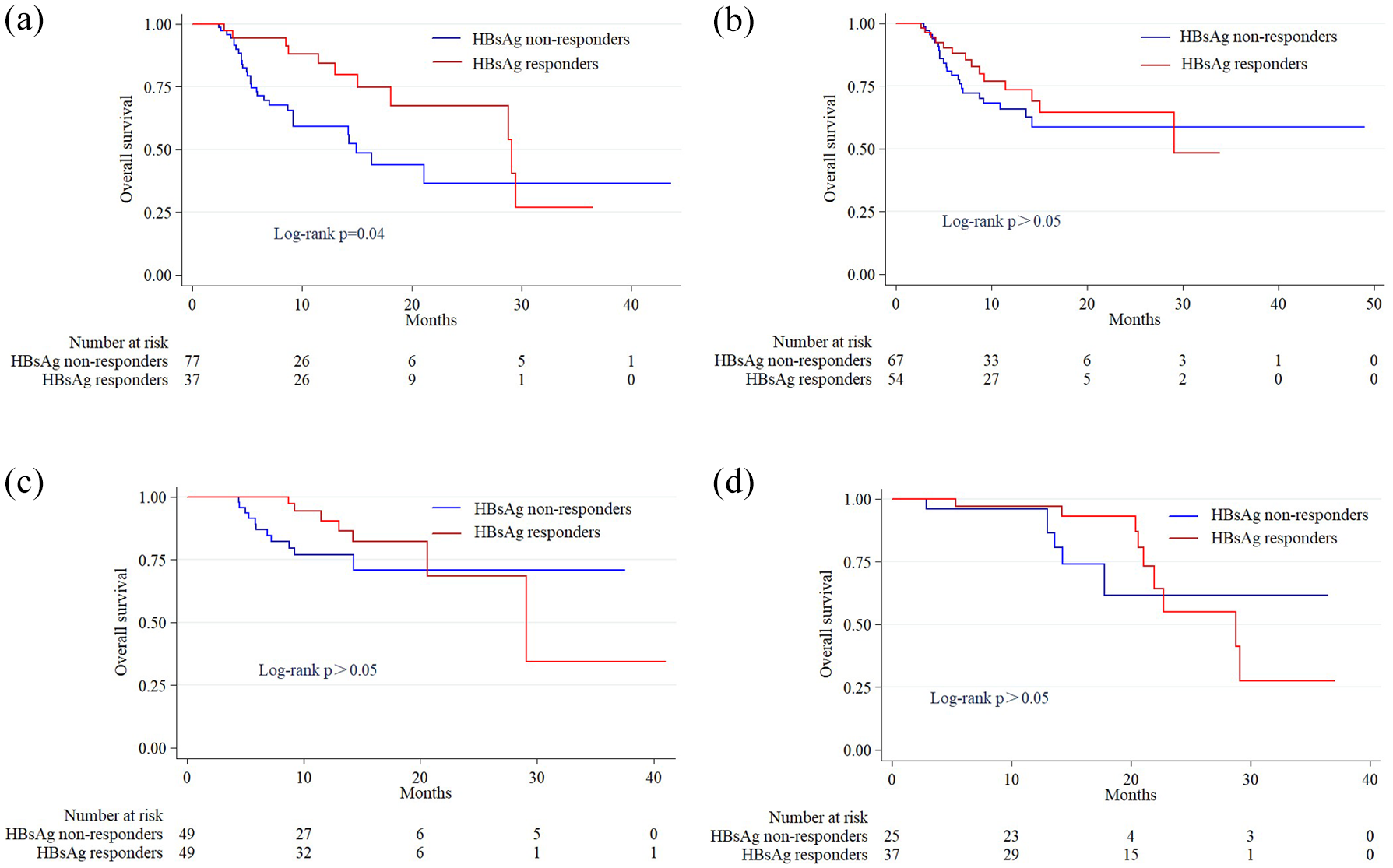
Survival analysis of qHBsAg dynamics at weeks (a) 4, (b) 12, (c) 24, and (d) 48 after PD-1/PD-L1 inhibitors therapy. Log-rank test for group comparisons.
Factors associated with OS time
Univariate Cox survival analysis revealed that baseline HBV DNA levels and qHBsAg kinetics at week 4 after PD-1/PD-L1 inhibitor therapy were significantly associated with OS. Multivariate Cox survival analysis suggested that baseline HBV DNA positivity (adjusted hazard ratio (aHR) = 2.6, 95% CI: 1.1–6.3, p = 0.04) and qHBsAg nonresponse at week 4 (aHR = 2.2, 95% CI: 1.1–4.5, p = 0.04) post-therapy were independent risk factors for survival (Table 3).
Univariable and multivariable Cox regression analyses of prognostic factors for overall survival.

AFP, alpha-fetoprotein; BCLC, Barcelona Clinic Liver Cancer; CI, confidence interval; ECOG PS, Eastern Cooperative Oncology Group performance status; HBV, hepatitis B virus; HR, hazard ratio; qHBsAg, quantitative hepatitis B surface antigen.
Correlation between clinical parameters and qHBsAg dynamics at week 4 after PD-1/PD-L1 inhibitor therapy
To identify other factors associated with qHBsAg dynamics at week 4 post-therapy, we analyzed the baseline characteristics of some clinical parameters. There were no statistically significant differences in age, gender, BCLC stage, Child-Pugh class, ECOG PS, baseline HBV DNA levels, baseline qHBsAg levels, baseline AFP levels, PD-1 inhibitors combined with targeted drugs, or PD-1 inhibitors combined with interventional therapy. However, patients in the qHBsAg-response group underwent more cycles and a longer duration of PD-1/PD-L1 inhibitor therapy (p < 0.05, Table 4).
Comparison of baseline variables between qHBsAg responders and qHBsAg nonresponders at week 4 after immunotherapy.
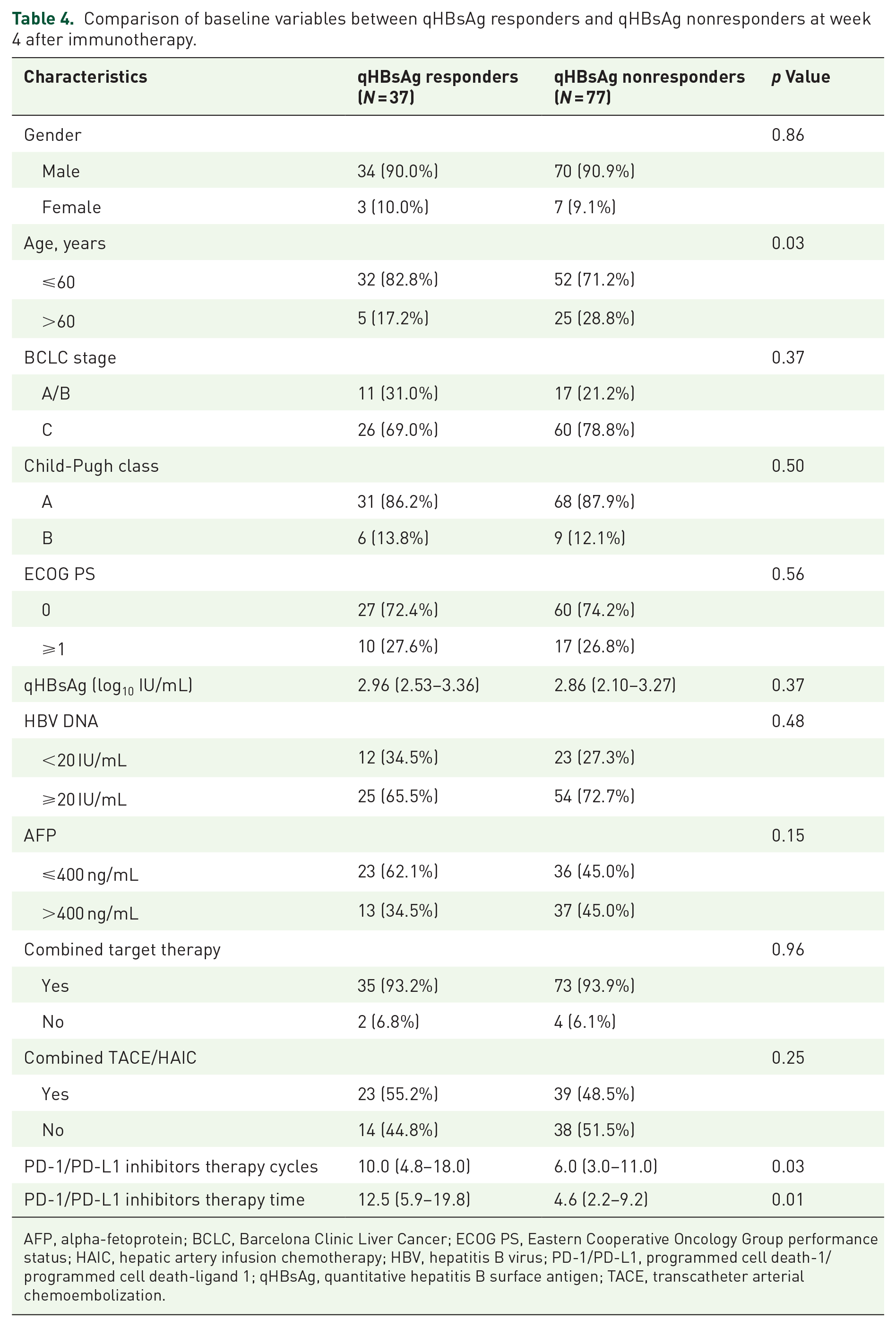
AFP, alpha-fetoprotein; BCLC, Barcelona Clinic Liver Cancer; ECOG PS, Eastern Cooperative Oncology Group performance status; HAIC, hepatic artery infusion chemotherapy; HBV, hepatitis B virus; PD-1/PD-L1, programmed cell death-1/programmed cell death-ligand 1; qHBsAg, quantitative hepatitis B surface antigen; TACE, transcatheter arterial chemoembolization.
Discussion
In this study, we first investigated the efficacy and the rate of HBV reactivation during PD-1/PD-L1 inhibitor treatment in HBV-associated HCC in the real world. ICI therapy for HCC is safe, with only 0.4% of patients experiencing HBV reactivation. No hepatitis was observed in this group, likely because all patients received antiviral therapy during their PD-1/PD-L1 inhibitor therapy. A study suggested that antiviral therapy may reduce the risk of HBV reaction or virological breakthrough when using ICIs. 28 Therefore, healthcare workers and patients with HCC should regularly monitor HBV DNA and liver function tests during PD-1/PD-L1 inhibitor treatment.
Further, we explored the correlation between PD-1/PD-L1 inhibitor therapy and HBV serum markers (qHBsAg and HBV DNA) in HCC. By recording qHBsAg levels at weeks 4, 12, 24, and 48 after PD-1/PD-L1 inhibitor treatment. The decrease in qHBsAg levels was observed in more than 50% of patients at all four time points. Patients with a response to qHBsAg (qHBsAg decreased ⩾ 0.1 log10 IU/mL) at week 4 had a better survival rate than those without a response (qHBsAg decreased <0.1 log10 IU/mL or even increased). This implies that these patients are more responsive to PD-1/PD-L1 inhibitors, indicating a heightened sensitivity of the immune system to blocking the PD-1/PD-L1 pathway. Moreover, neither Cox analysis nor log-rank survival analysis showed a statistically significant difference in OS between the two groups at weeks 12, 24, and 48, potentially due to the small sample size. However, the current results support our conclusion that early qHBsAg changes can be used to identify patients with HBV-associated HCC who are more likely to benefit from PD-1/PD-L1 inhibitor therapy. Thus, patients with HBV-associated HCC undergoing PD-1/PD-L1 inhibitor treatment should be regularly monitored for changes in qHBsAg levels to enable clinicians to adjust treatment regimens accordingly.
All enrolled patients were positive for baseline qHBsAg and received concomitant nucleoside analog antiviral therapy at the start of PD-1/PD-L1 inhibitor therapy, regardless of prior anti-HBV therapy. We are concerned about HBsAg for three main reasons. First, past studies have confirmed that a positive HBsAg expression is a high-risk factor for late recurrence post-surgery, a poor prognosis of TACE treatment in HCC, and shorter survival in patients with HCC treated with lenvatinib combined with ICIs.16–18,29 Second, HBsAg plays a role in the occurrence and development of HCC; the pathogenesis of CHB combined with HCC is based on a liver immunosuppressive microenvironment that leads to immune cell dysfunction, including immune checkpoint interactions such as PD-1/PD-L1. Blocking the PD-1/PD-L1 pathway may have a dual effect: it may exert antitumor therapeutic effects while reducing HBsAg. Third, ICIs have achieved good efficacy in the treatment of liver cancer; nevertheless, the lack of prognostic-related biomarkers is an objective fact. Although baseline HBsAg levels have been used in studies to predict clinical outcomes with different grouping criteria, few studies have examined the specific relationship between qHBsAg levels and the mortality risk in patients with HCC. Our data suggest that baseline qHBsAg levels do not affect the prognosis of PD-1/PD-L1 inhibitor therapy in patients with HCC.
Several studies have concluded that baseline viral load is not associated with the prognosis of PD-1/PD-L1 inhibitor treatment in patients with HBV-associated HCC.30,31 However, our study used significantly different cut-off values for the grouping, yielding results that differ from those of previous studies but align more closely with common sense. We hypothesize that a lower viral load indicates a less compromised patient’s liver immune system during chronic infection, providing a basis for a better prognosis with subsequent PD-1/PD-L1 ICI therapy. Upregulation of immune checkpoint receptors such as PD-1 on the surface of CD8+ T cells was detected in both CHB and HCC patients. This is the main mechanism involved in T-cell failure and exhaustion. This immunosuppression may be more pronounced in the livers of patients with HCC and concomitant CHB. The overexpression of PD-1 in immune cells inhibits the function of both HBV-specific and tumor-specific CD8+ T cells; the potential collective decay of these two types of cells, leading to more severe immunosuppression, requires further investigation. In most chronic HBV infection cases, the function of immune cells is significantly lower than during the acute infection stage. Long-term antigen stimulation leads to more severe immunosuppression, further inhibiting the liver immune microenvironment in patients with CHB. Consequently, the core substances of viruses, such as HBV DNA and HBsAg, are difficult to clear for a long time. 32 We propose that patients with viral loads below the lower limit of detection before starting PD-1/PD-L1 inhibitor therapy may experience less liver immunosuppression, at least compared to those with positive HBV DNA levels. This renders patients with low viral load more likely to benefit from ICI treatment. It has been found that those with HBV-associated HCC who have high HBV DNA levels after PD-1/PD-L1 inhibitor treatment have a worse prognosis.33,34 Our analysis revealed that pretreatment viral load levels were linked to the prognosis of PD-1/PD-L1 inhibitor therapy in patients with HBV-related HCC. However, this observation is based on a retrospective clinical study; future prospective studies are needed to confirm this hypothesis. In addition, exploring the changes in the number of immune cells within the liver at different stages after PD-1/PD-L1 inhibitor treatment, and how various T-cell function-related factors are expressed may help us better interpret this result. Therefore, more specific and in-depth research and analysis are needed to understand how PD-1/PD-L1 inhibitors affect the changes in hepatitis B serological markers.
Our study has some major limitations. First, the retrospective nature of our analysis introduced unavoidable selection biases and confounders. In addition, due to tumor heterogeneity and individual patient differences, it is difficult to achieve complete randomization and fully consider all factors that may affect patient prognosis. Regarding the antiviral effects of PD-1/PD-L1 inhibitors, we could not confirm whether the decline in HBV DNA was associated with the use of PD-1/PD-L1 inhibitors because the antiviral effects of NAs could not be completely balanced. Third, this was a single-center retrospective study with a relatively small sample size. Our conclusions need to be verified by large-sample clinical studies.
In summary, we validated that dynamic changes in baseline HBV DNA and qHBsAg levels at week 4 after PD-1/PD-L1 inhibitor treatment serve as easily measurable, cost-effective, and noninvasive biomarkers for predicting the prognosis of HBV-related HCC. This finding has significant clinical implications. Early changes in qHBsAg levels can help clinicians identify HBV-related HCC patients who are more likely to benefit from PD-1/PD-L1 inhibitors. Furthermore, for patients receiving PD-1/PD-L1 inhibitors, regular monitoring of qHBsAg levels, particularly at baseline and week 4, is essential for guiding personalized treatment strategies. A lack of qHBsAg response at week 4 may facilitate early disease staging and, in cases of tumor progression, enable timely adjustments to more effective therapies while minimizing the risk of severe immunotherapy-related toxicities. Given its affordability and broad clinical accessibility, assessing qHBsAg dynamics at week 4 is a practical approach that can be seamlessly integrated into routine clinical practice, offering a valuable tool for optimizing immunotherapy monitoring.
Conclusion
PD-1/PD-L1 inhibitors have reliable efficacy and safety in the treatment of patients with HBV-related HCC in the real world. Hepatitis B serum markers such as qHBsAg and HBV DNA can serve as prognostic biomarkers for PD-1/PD-L1 inhibitor treatment.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251347469 – Supplemental material for Relationship between HBsAg/HBV DNA and prognosis in patients with HBV-related hepatocellular carcinoma treated with PD-1/PD-L1 inhibitors
Supplemental material, sj-docx-1-tam-10.1177_17588359251347469 for Relationship between HBsAg/HBV DNA and prognosis in patients with HBV-related hepatocellular carcinoma treated with PD-1/PD-L1 inhibitors by Qiuying Qin, Yuanyuan Zheng, Yiran Wang, Fei Zhou, Dong Li, Linlin Jin, Mujia Zhu, Yabing Guo, Rong Fan, Jinlin Hou, Xiaoyong Zhang and Hongyan Liu in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
