Abstract
Objectives
To analyze differences in programmed cell death protein 1/ligand 1 (PD-1/PD-L1) expression, as well as the relationships between ultrasound/contrast-enhanced ultrasound characteristics and PD-1/PD-L1 expression, among invasive breast cancer molecular subtypes.
Methods
The study included 172 invasive breast cancer patients with surgical resection and pathological confirmation at the First Affiliated Hospital of Xinjiang Medical University from June 2016 to April 2022. PD-1/PD-L1 expression was detected by immunohistochemistry. All patients underwent conventional ultrasound and some underwent contrast-enhanced ultrasound examination before resection.
Results
PD-1 and PD-L1 were expressed in 112 and 121 cases, respectively. The luminal B and HER-2 subtypes had the lowest and highest PD-1 expression rates, respectively. The luminal B and triple-negative subtypes had the lowest and highest PD-L1 expression rates, respectively. Among 112 PD-1-positive cases, most luminal B cases exhibited ill-defined margins, while distant metastasis was more common in triple-negative cases. Among 121 PD-L1-positive cases, many HER-2-positive and triple-negative cases presented as large masses (diameter ≥ 2 cm), while luminal B cases were more likely to show calcification. Most luminal B PD-L1-positive cases displayed indistinct margins on contrast-enhanced ultrasound.
Conclusions
PD-1 expression differed among molecular subtypes of invasive breast cancer. Ultrasound/contrast-enhanced ultrasound features correlated with PD-1/PD-L1 expression in different breast cancer subtypes.
Keywords
Introduction
Differences in molecular subtypes of breast cancer are of great significance for judging the prognosis and selecting treatment options. In recent years, in addition to traditional breast cancer treatment options such as surgery, endocrine therapy, chemoradiotherapy, and targeted therapy, emerging immunotherapy methods have gradually attracted attention. Programmed cell death protein 1/programmed death ligand 1 (PD-1/PD-L1) inhibitors are a focus of immunotherapy research. Activation of PD-1/PD-L1 signaling can mediate immune suppression in the breast cancer microenvironment and play an important role in the occurrence, progression, invasion, and metastasis of breast cancer. Therefore, blockade of the PD-1/PD-L1 signaling pathway can provide a new immunotherapy target for breast cancer patients. Ultrasound and ultrasound imaging play important roles in the diagnosis and immunotherapy of breast cancer. The correlation between PD-1/PD-L1 expression in invasive breast cancer and ultrasound and contrast-enhanced ultrasound indicators has been reported in the literature, but the correlation between PD-1/PD-L1 in different molecular subtypes of invasive breast cancer has not been reported in detail. The present study aimed to assess PD-1 and PD-L1 expression in different molecular subtypes of invasive breast cancer using immunohistochemistry while also observing the two-dimensional and contrast-enhanced ultrasound characteristics of breast cancer. Furthermore, this study aimed to analyze the variations in immune factor expression (PD-1/PD-L1) among different molecular subtypes of invasive breast cancer and explore the relationship between ultrasound/contrast-enhanced ultrasound features and PD-1/PD-L1 expression across these subtypes. Ultimately, this research provides a relatively accurate and objective imaging foundation for predicting PD-1/PD-L1 expression in breast cancer and for guiding targeted drug therapy utilizing PD-1 inhibitors.
Methods
Study object
The study included patients with invasive breast cancer confirmed by surgical pathology from the First Affiliated Hospital of Xinjiang Medical University from June 2016 to April 2022. The inclusion criteria were as follows: first-time female patients diagnosed with pathologically confirmed invasive breast cancer who had not received any anticancer treatment, had complete preoperative ultrasonography and pathology reports, and had signed the informed consent form. The study was approved by the Ethics Committee of the First Affiliated Hospital of Xinjiang Medical University.
Ultrasound examination methods
The LOGIQ E9 ultrasound diagnostic instrument from GE Company (Wauwatosa, WI, USA) was used for the examination. A high-frequency probe (ML6-15-D) operating at 6 to 15 MHz facilitated the two-dimensional ultrasound examination. The patient was in a supine position, with both breasts and armpits fully exposed, and adjustments were made as needed for optimal imaging. After identifying the tumor, the examiner rotated the probe to perform a comprehensive scan of the lesion, noting its location, size, shape, echo characteristics, and boundaries with surrounding tissue. A color Doppler examination was then conducted to assess the blood flow distribution and supply, following the Adler semi-quantitative grading standard. 1 The largest lesion or area with the most blood flow was selected, and the ultrasound examination was converted to breast contrast-enhanced imaging mode. Patients were instructed to breathe quietly, and those with an available left peripheral elbow vein received an injection of 4.8 mL of contrast agent (SonoVue, Bracco Suisse S.A., Plan-Les-Ouates, Switzerland) followed by 5.0 mL of 0.9% saline. Dynamic images were collected for 3 minutes and stored. Observations were performed based on the Chinese Guidelines for Clinical Application of CEUS 2 and included indicators such as blood vessel nourishment, enhancement degree, perfusion mode, enhancement uniformity, boundary definition, and any enlargement of the lesion after angiography (longitudinal or transverse diameter enlarged by > 3 mm compared with that on conventional ultrasound). Measurements for each diameter were performed three times. All ultrasonography images were interpreted by two experienced physicians, with a third physician consulted in case of disagreement.
Immunohistochemical detection methods
All postoperative specimens of invasive breast cancer were examined by immunohistochemical methods.
Reagents and instruments
The tissue paraffin microtome, thermostatic tissue section incubator, 100-µL pipettor, 100- to 1000-µL pipettor, optical microscope, image acquisition and collection system, rabbit anti-human PD-1 monoclonal antibody, and rabbit anti-human PD-L1 monoclonal antibody were purchased from AbM (Shanghai) Trading Co. LTD. (Shanghai, China). The Ready-to-use SABC-POD kit (rabbit IgG) was purchased from Wuhan Boster Biological Technology Co. LTD. (Wuhan, China). Goat serum, a DAB chromogenic kit, 3% hydrogen peroxide solution, pH 9.0 antigen repair buffer, phosphate-buffered saline, hematoxylin staining solution, and neutral gum were purchased from Beijing Zhongshan Jinqiao Co. LTD. (Beijing, China).
Immunohistochemical staining
Breast cancer tissue samples were fixed with 10% formaldehyde and embedded in paraffin, then sectioned (thickness of 3 to 4 mm) and baked at 70°C for 2 hours. After deparaffinization and rehydration, the sections were rinsed with distilled water. Next, 3% hydrogen peroxide was used to block endogenous peroxidase activity, followed by washing with PBS. Then, an EDTA repair solution was applied with heat for antigen retrieval. After cooling, the samples were washed again. Goat serum was added to block non-specific binding, followed by incubation with a mouse anti-human PD-L1 monoclonal antibody at 37°C. Finally, DAB was used for color development. The samples were counterstained with hematoxylin, then dehydrated and mounted. Staining results were observed under a microscope. The procedure was performed strictly in accordance with the instructions of the immunostaining kit provided by Wuhan Boster Biotechnology Co. LTD.
PD-1/PD-L1 interpretation standard
The results were based on the interpretation standards of Hasan 3 and Dill. 4 Positive expression of PD-L1 in cells was defined as a brown-yellow reaction of the cell membrane or cytoplasm in ≥20% of tumor stromal cells. Positive PD-1 cell expression was defined as a brown reaction of the cell membrane or cytoplasm in ≥5% of tumor stromal cells.
Statistical analysis
IBM SPSS 27.0 statistical software (IBM Corp., Armonk, NY, USA) was used for the statistical analysis. Count data are expressed as proportions (%), and the chi-square or Fisher exact probability method was used for analysis. Spearman correlation analysis was used to analyze the relationship between positive PD-1/PD-L1 expression and the ultrasound and contrast-enhanced ultrasound characteristics of different molecular subtypes of invasive breast cancer. A value of P < 0.05 was considered statistically significant.
Results
Among the 172 cases of invasive breast cancer, 169 were invasive ductal carcinoma and 3 were solid papillary carcinoma with invasion. PD-1 expression was positive in 112 cases and negative in 60 cases. There were 121 cases of positive PD-L1 expression and 51 negative PD-L1 cases. Among the 172 patients, 36 were human epidermal growth factor receptor 2 (HER-2) positive, 32 were the luminal A subtype, 35 were triple-negative, and 69 were the luminal B subtype.
Immunohistochemical results of different molecular subtypes of invasive breast cancer
PD-1 and PD-L1 expression across various molecular subtypes of invasive breast carcinoma is summarized in Table 1. The positive expression rates for PD-1 were as follows: 83.3% among HER-2 positive (30/36; Figure 1), 62.5% among luminal A (20/32; Figure 2), 77.1% among triple-negative (27/35; Figure 3), and 50.7% among luminal B (35/69; Figure 4) cases. The HER-2 positive subtype showed the highest rate of PD-1 expression, while the luminal B subtype had the lowest rate, with significant differences between the HER-2-positive and luminal B subtypes and between the triple-negative and luminal B subtypes (P < 0.05). The positive PD-L1 expression rates were as follows: 72.2% in the HER-2 positive type (26/36; Figure 5), 68.8% in the luminal A subtype (22/32; Figure 6), 74.3% in the triple-negative type (26/35; Figure 7), and 68.1% in the luminal B subtype (47/69; Figure 8). The positive PD-L1 expression rate was lowest in the luminal B subtype and highest in the triple-negative subtype, but there was no significant difference among the four subtypes (P > 0.05).
Expression of PD-1/PD-L1 in different molecular subtypes of invasive breast cancer [n (%)].

PD-1, programmed cell death protein 1; PD-L1, programmed death ligand 1; HER-2, human epidermal growth factor receptor 2.

Positive expression of programmed cell death protein 1 (PD-1) in human epidermal growth factor receptor 2 (HER-2) positive invasive breast cancer.

Positive expression of programmed cell death protein 1 (PD-1) in Luminal A invasive breast cancer.

Positive expression of programmed cell death protein 1 (PD-1) in triple-negative invasive breast cancer.

Positive expression of programmed cell death protein 1 (PD-1) in Luminal B invasive breast cancer.

Positive expression of programmed cell death ligand 1 (PD-L1) in human epidermal growth factor receptor 2 (HER-2) positive invasive breast cancer.

Positive expression of programmed cell death ligand 1 (PD-L1) in Luminal A invasive breast cancer.
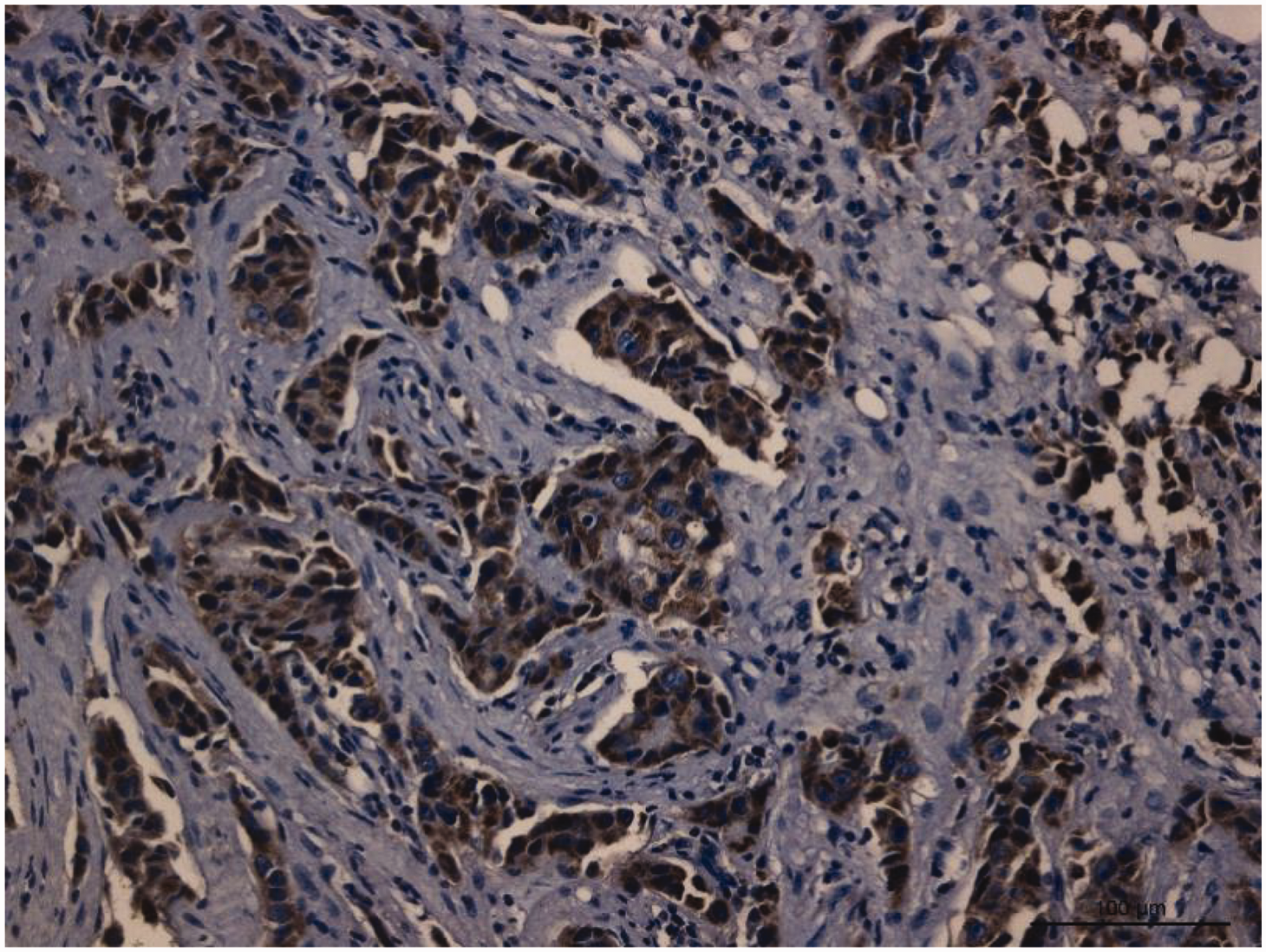
Positive expression of programmed cell death ligand 1 (PD-L1) in triple-negative invasive breast cancer.

Positive expression of programmed cell death ligand 1 (PD-L1) in Luminal B invasive breast cancer.
The correlations between PD-1-positive expression and ultrasound and contrast-enhanced ultrasound features in different molecular subtypes of invasive breast cancer are shown in Table 2 and Table 3.
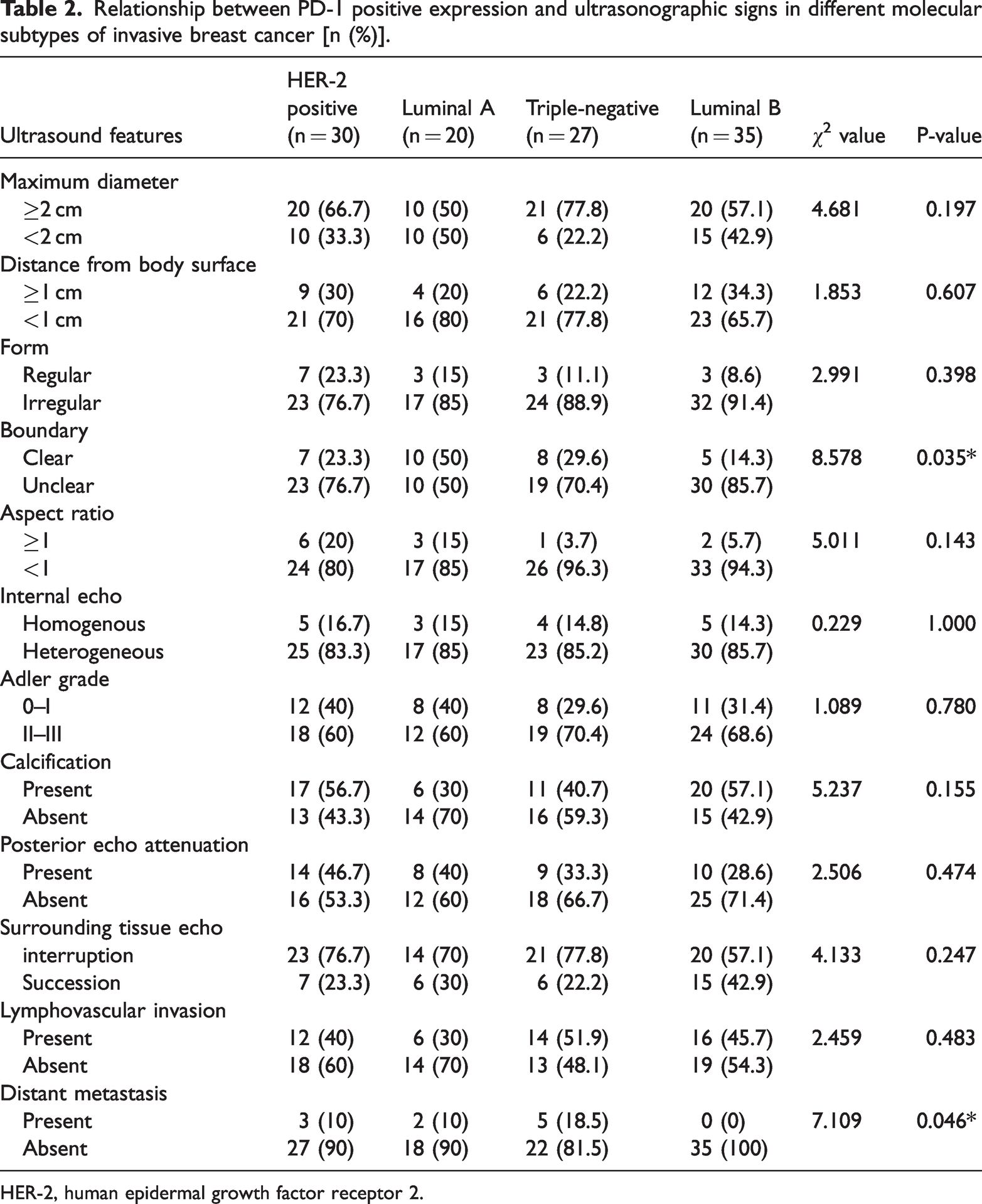
Relationship between PD-1 positive expression and ultrasonographic signs in different molecular subtypes of invasive breast cancer [n (%)].
HER-2, human epidermal growth factor receptor 2.
Relationship between PD-1 positive expression and contrast-enhanced ultrasound characteristics in different molecular subtypes of invasive breast cancer [n (%)].

PD-1, programmed cell death protein 1; HER-2, human epidermal growth factor receptor 2.
Table 2 illustrates significant differences in distant metastasis and tumor boundary characteristics among various molecular subtypes of invasive breast cancer with positive PD-1 expression in a cohort of 112 patients (P < 0.05). Notably, 50% of the luminal A subtype exhibited unclear boundaries, while 85.7% of the luminal B subtype demonstrated similar ambiguity (Figures 9–10), resulting in a statistically significant difference between these subtypes (P < 0.05). The incidence of distant metastasis in the PD-1 positive triple-negative group was 18.5%, in contrast to 0% in the luminal B group, resulting in a statistically significant difference (P < 0.05). No significant differences were identified in other ultrasound features, such as lesion size, among the four molecular subtypes with positive PD-1 expression. Table 3 indicates that there were no significant differences in contrast-enhanced ultrasound characteristics, including the enhancement pattern, enhancement amplitude, boundary clarity post-enhancement, or presence of filling defects.

Programmed cell death protein 1 (PD-1) positive Luminal B breast cancer mass with an unclear boundary.

Programmed cell death protein 1 (PD-1) positive Luminal A breast cancer mass with an unclear boundary.
Different molecular subtypes of invasive breast cancer with PD-L1-positive expression and their correlations with ultrasound and ultrasound imaging characteristics are shown in Table 4 and Table 5.
Relationship between PD-L1 positive expression and ultrasonographic signs in different molecular subtypes of invasive breast cancer [n (%)].

PD-L1, programmed death ligand 1; HER-2, human epidermal growth factor receptor 2.
Relationship between PD-L1 positive expression and contrast-enhanced ultrasound characteristics in different molecular subtypes of invasive breast cancer [n (%)].

PD-L1, programmed death ligand 1; HER-2, human epidermal growth factor receptor 2.
As shown in Table 4, statistically significant differences were observed in the presence or absence of calcification and lesion size among the different invasive breast cancer molecular subtypes in 121 PD-L1-positive cases (P < 0.05). Triple-negative and HER-2 positive masses (≥2 cm in diameter) with PD-L1 positivity accounted for 80.8% and 69.2% of cases, respectively, while luminal A and B type tumors accounted for 45.5% and 44.7% of cases, respectively, as shown in Figures 11–14. Statistically significant differences were observed between the HER-2 positive type and luminal B type, the luminal A type and triple-negative type, and the luminal B type and triple-negative type (P < 0.05). Calcification was present in 63.8% of PD-L1 positive luminal B lumps, 31.8% of luminal A lumps, and 38.6% of triple-negative lumps, as shown in Figures 15–17. The differences between luminal A lumps and luminal B lumps and between triple-negative lumps and luminal B lumps were statistically significant (P < 0.05). No significant differences were found for the remaining ultrasound characteristics (such as aspect ratio, Adler grade, posterior echo). Table 5 shows that among the contrast-enhanced ultrasound features, there was a statistically significant difference in whether the boundary was clear after contrast enhancement. Moreover, 63.2% of the luminal B subtype with positive PD-L1 expression had an unclear boundary while only 14.3% of the HER-2 positive subtype had an unclear boundary after contrast enhancement, as shown in Figures 18–19, resulting in a significant difference between the two groups (P < 0.05). There were no significant differences in other aspects such as the contrast enhancement mode, contrast enhancement amplitude, and internal filling defects.

The sizes of programmed cell death ligand 1 (PD-L1) positive triple-negative invasive breast cancer mass is approximately 3.5 cm × 2.8 cm.

The sizes of programmed cell death ligand 1 (PD-L1) positive human epidermal growth factor receptor 2 (HER-2) positive invasive breast cancer mass is approximately 5.0 cm × 1.8 cm.

The sizes of programmed cell death ligand 1 (PD-L1) positive Luminal A invasive breast cancer mass is approximately 1.2 cm × 1.0 cm.

The sizes of programmed cell death ligand 1 (PD-L1) positive Luminal B invasive breast cancer mass is approximately 1.3 cm × 1.5 cm.

Calcification was common in Luminal B breast cancer with positive programmed cell death ligand 1 (PD-L1) expression.

Programmed cell death ligand 1 (PD-L1) positive triple-negative invasive breast cancer exhibited less calcification in the mass.
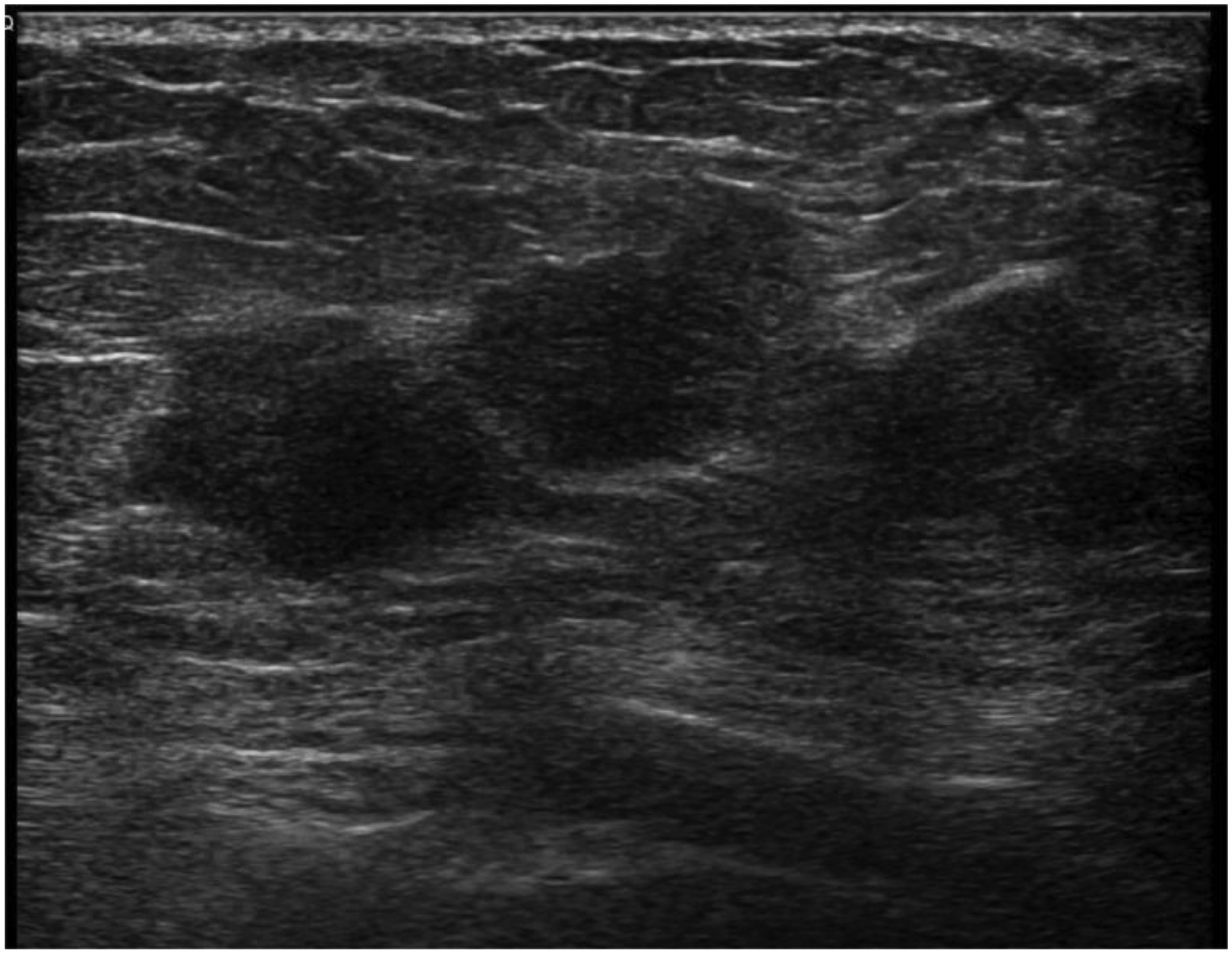
Programmed cell death ligand 1 (PD-L1) positive Luminal A invasive breast cancer exhibited less calcification in the mass.

Programmed cell death ligand 1 (PD-L1) positive Luminal B breast cancer with an unclear border on contrast-enhanced ultrasound.

Programmed cell death ligand 1 (PD-L1) positive human epidermal growth factor receptor 2 (HER-2) positive breast cancer with clear boundaries on contrast-enhanced ultrasound.
Discussion
Breast cancer treatment has entered the era of personalized therapy based on molecular typing. Endocrine therapy is effective for luminal breast cancer, which has high sensitivity to postoperative radiotherapy and chemotherapy. However, endocrine therapy has a poor effect on HER-2 positive and triple-negative breast cancer. 5 At present, molecular immune therapy has shown potential for clinical application as a new type of treatment that can use the body’s own immune system to recognize and destroy cancer cells and prevent recurrence and metastasis of breast cancer. 6 PD-L1 is the main ligand of PD-1, 7 which is highly expressed on the surface of a variety of hematopoietic and non-hematopoietic tumor cells, including lung, kidney, breast, pancreatic, cervical, ovarian, and prostate cancer,8,9 and can be used as a new target for molecular immunotherapy. Induced blockade of the PD-1/PD-L1 signaling pathway can inhibit the immune activity of T cells and limit tumor cell death. 10 Ultrasound and ultrasound imaging are the main diagnosis methods of breast cancer, and breast cancer immunotherapy plays an important role in activating the body’s immune system to fight specific tumor-associated antigens, modulating immunoregulatory signals, and thereby promoting T cell-mediated tumor cell death. 11 Related studies have shown that the sonographic findings of breast cancer and molecular markers have certain relevance. A breast cancer study by Au et al. found significant differences in ultrasound borders and posterior echogenic features between low-grade and high-grade, estrogen receptor positive (ER+) and ER−, and PR+ and PR− lesions. 12
The positive expression rate of PD-1 was 65% (112/172), and the positive expression rate of PD-L1 was 70% (121/172), indicating that PD-1/PD-L1 is highly expressed in invasive breast cancer, and this is because of the interaction between PD-L1 and PD-1 on the lymphocyte surface. Thus, PD-1/PD-L1 continuously interferes with the body’s immune response to the surface of tumor cells in breast cancer patients and promotes tumor immune escape. 13 In this study, the positive expression rate of PD-1 was lowest in the luminal B subtype and highest in the HER-2 positive subtype. There were significant differences in PD-1 expression between the triple-negative subtype and luminal B subtype and between the HER-2 positive subtype and luminal B subtype. The positive expression rate of PD-L1 was lowest in the luminal B subtype and highest in the triple-negative subtype. Muenst et al. found that the positive PD-1/PD-L1 expression in breast cancer was related to ER−, progesterone receptor (PR) negative, and HER-2 positive expression,14–16 and the positivity rate of PD-1/PD-L1 was higher in HER-2 positive and triple-negative breast cancer with ER and PR negativity. This finding also suggests that the PD-1/PD-L1 signaling pathway may play an important role in the occurrence, progression, and metastasis of HER-2 positive and triple-negative breast cancer.
In this study, the proportion of PD-1 positive invasive breast cancer with an unclear boundary was highest in the luminal B group (85.7%), and the difference was statistically significant among different molecular subtypes of invasive breast cancer. An unclear tumor boundary may be caused by the increased stromal reaction and high fibrous connective tissue proliferation caused by tumor cells invading the surrounding tissues at different growth rates, which is reflected by an unclear boundary between the tumor and the surrounding tissues on ultrasound images. According to one study, 17 the tumor edge is divided into a cancer bed zone, an inflammatory cell infiltration zone, and a connective tissue hyperplasia zone. The cancer bed zone is formed by the distribution of cancer cells along the base and invasion into the stroma. The infiltration zone of inflammatory cells is mostly the manifestation of the interstitial reaction and edema. The connective tissue proliferation zone contains dense fibrous tissue and has a hard texture, 18 which is common in luminal breast cancer and is the body’s self-protection mechanism against tumor metastasis. 19 This study showed that in the PD-L1 positive invasive breast cancer group, the luminal B subtype had the highest proportion of calcification (63.8%). ER/PR expression is positive, HER-2 expression can be high or low, and Ki-67 expression is mostly high in luminal B tumors. Gao Junrong 20 confirmed that microcalcification is more likely to occur when Ki-67 or HER-2 is highly expressed. Breast cancer cells have a vigorous metabolism, with active anaerobic glycolysis and aerobic oxidation of glucose. When the cells are necrotic and lysed, calcium salt deposits are formed in the acini and ducts. 21 Rapid tumor growth can easily cause an insufficient oxygen supply, followed by ischemic necrosis, calcium salt deposition, and dystrophic calcification. In this study, the boundary of PD-L1 positive luminal B invasive breast cancer was unclear after contrast-enhanced ultrasound because the degree of malignancy and blood supply of luminal B breast cancer were relatively low. The enhancement amplitude and intensity of the mass and the surrounding tissue were close to or slightly higher than those of the surrounding normal breast tissue after contrast-enhanced ultrasound, resulting in an unclear boundary after contrast-enhanced ultrasound.
In this study, PD-L1 positive luminal A breast cancer exhibited a smaller tumor size and reduced degree of calcification, which aligns with the findings reported by Kurozumi et al. 22 and Gumowska et al. 23 ER and PR expression was positive, while HER-2 expression was negative, and Ki-67 expression was characterized as low in luminal A type neoplasms. Ki-67 is a nuclear protein associated with tumor proliferation, invasion, and metastasis. Its elevated expression correlates with an increased capacity for tumor invasion and metastasis, indicating a higher malignancy grade and a poor treatment response and prognosis.24,25 Conversely, low Ki-67 expression suggests relatively slow tumor growth and a decreased likelihood of dystrophic calcification. Consequently, luminal breast cancer tends to present with a smaller tumor volume 26 and a lower incidence of distant metastasis. 27 Furthermore, the prognosis for this subtype of breast cancer is generally more favorable than that observed in triple-negative and HER-2 positive breast cancer. 28
In this study, PD-1 positive triple-negative breast cancer exhibited a larger mass and the highest rate of distant metastasis, while PD-L1 positive triple-negative breast cancer showed less calcification. Because the growth of triple-negative breast cancer is not controlled by hormones, it grows faster and is more invasive and prone to distant metastasis. 29 Such masses often are large when they are found. Triple-negative breast cancer exhibited the least calcification, while luminal B breast cancer showed the most calcification. Hu Yangling et al. 30 and Zhang et al. 31 found that the microcalcification of breast cancer was closely related to positive ER and HER-2 expression. Gumowska et al. 23 and Ding Tong et al. 32 found that the proportion of calcification in triple-negative breast cancer was lowest among several molecular subtypes, with rates of 21% and 0%, respectively, which is consistent with the results of this study.
The results of this study indicate that PD-L1 positive HER-2 positive tumors typically exhibit a larger volume, which is consistent with the findings of Chen Yan, 33 who reported a positive correlation between HER-2 expression levels and tumor size. Specifically, tumors with high HER-2 expression generally have a larger volume and are accompanied by an elevated degree of malignancy and a poorer prognosis. This phenomenon highlights the distinct biological behavior of HER-2 positive breast cancer, underscoring its significance as a potential prognostic indicator. In the current study, PD-L1 positive, HER-2 positive tumors predominantly displayed well-defined margins on contrast-enhanced ultrasound imaging. This clarity may stem from the high malignancy associated with HER-2 positive breast cancer, as well as its abundant vascular supply. HER-2 positive tumors typically demonstrate enhanced angiogenesis, resulting in a more pronounced enhancement effect during contrast ultrasound imaging compared with that of adjacent normal tissues. 34 This enhancement effect is particularly evident in the early stages of contrast ultrasound, facilitating the recognition of the tumor in clinical diagnostic assessments. There are still some limitations of this study. First, some of the ultrasound data in this study are retrospective, and there may be differences in the retention of standard images, which can impact image interpretation. Second, ultrasonography was only qualitatively studied, and quantitative analysis of ultrasonography was not included; thus, it is hoped that future studies will add quantitative ultrasonography analysis.
Conclusion
The expression of the immune regulatory factor PD-1 varies among distinct molecular subtypes of invasive breast cancer. Furthermore, specific ultrasound and contrast-enhanced ultrasound characteristics are correlated with PD-1/PD-L1 expression across these molecular subtypes. These imaging features may serve as predictive indicators for the expression levels of PD-1/PD-L1 in breast cancer. Consequently, this approach offers precise and objective imaging evidence to support the implementation of targeted pharmacological interventions for breast cancer.
Footnotes
Acknowledgement
We thank Professor Tao Song for her professional advice and assistance in this research.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
