Abstract
Objective:
Cemiplimab combined with chemotherapy has emerged as a promising treatment option for advanced non-small cell lung cancer (NSCLC). Accordingly, this study has been conducted to evaluate the cost-effectiveness of this combination therapy in comparison to chemotherapy alone from the perspective of the United States healthcare system.
Methods:
The present study is based on a partitioned survival model developed from clinical data obtained during the 2-year follow-up of the phase III EMPOWER-Lung 3 part 2 trial. The purpose of this investigation is to estimate the 10-year life expectancy and total healthcare costs of patients with advanced NSCLC by leveraging primary outcomes that evaluated costs, quality-adjusted life years (QALYs), and incremental cost-effectiveness ratio (ICER).To establish the willingness-to-pay (WTP) threshold for the analysis, a value of $150,000/QALY was adopted. Sensitivity analysis was performed to determine the impact of varying levels of uncertainty on the results of this study.
Results:
When compared to chemotherapy alone, the addition of cemiplimab to chemotherapy has been demonstrated to result in an incremental gain of 1.593 QALY at an additional cost of $109351.298. This equates to an incremental cost-effectiveness ratio (ICER) of $68644.883/QALY. One-way sensitivity analyses were conducted on the model, which acknowledged the influence of several parameters, such as subsequent costs, the utility of progressive disease, the cost of best supportive care, the cost of cemiplimab per mg, and the utility of progression-free survival on the outcomes. Nonetheless, none of these parameters yielded an ICER lower than the WTP threshold.
Conclusions:
From the perspective of the United States healthcare system, the utilization of cemiplimab in combination with chemotherapy as a first-line treatment option for NSCLC appears to be a cost-effective approach as compared to using chemotherapy as a standalone therapy.
Keywords
Introduction
Cancer is a pervasive public health concern with global implications and represents the second most common cause of death in the United States. Recent projections indicate that by the year 2023, there will be an estimated 1,958,310 new cancer cases and 609,820 cancer-related deaths in the United States. 1 Lung cancer continues to hold the top spot as the leading contributor to cancer-related mortality, causing an estimated 1.8 million deaths globally, representing approximately 18% of all cancer-related deaths. 2 Non-small cell lung cancer (NSCLC) is the prevailing form of lung cancer, accounting for over 80% of lung cancer cases. However, the 5-year survival rate remains disappointingly low at 15%. 3 Development of novel interventions for prevention and treatment of NSCLC represents an important avenue for improvement of survival outcomes in this deadly disease.
The treatment of NSCLC has witnessed significant advancements in recent years, predominantly due to the availability of immune checkpoint inhibitors (ICIs) such as programmed cell death-1 (PD-1) and programmed death ligand 1 (PD-L1) inhibitors. 4 These inhibitors have revolutionized the management of NSCLC by modulating the body’s immune response against cancer cells and have shown remarkable success in improving patient outcomes. The ability of these inhibitors to enhance the immune response selectively has promising implications for patients suffering from NSCLC and provides a viable alternative to traditional chemotherapy. These recent advancements herald an era of precision medicine in oncology, with immunotherapy emerging as a promising approach toward achieving cancer control. 5 Cemiplimab represents a promising anti-PD-L1 inhibitor that has demonstrated efficacy in advanced NSCLC when used as monotherapy or in conjunction with chemotherapy for both squamous and nonsquamous histologies. 6 The phase III EMPOWER-Lung 3 recently released its 2-year follow-up results, providing critical insights into the efficacy of cemiplimab plus chemotherapy in treating advanced squamous or nonsquamous NSCLC. With a median follow-up period of 28.4 months, the study showed continued improvements in patient outcomes when treated with cemiplimab plus chemotherapy compared to chemotherapy alone. 7
The advent of ICIs has revolutionized the field of oncology, offering a promising opportunity for cancer treatment. However, their high costs have sparked concerns about affordability and the economic feasibility of utilizing these novel medications. 8 Hence, it is crucial to examine the therapeutic efficacy alongside cost-effectiveness when making treatment decisions, and formulating healthcare policies. 9 The aim of the present study is to assess the cost-effectiveness of cemiplimab, when used in combination with chemotherapy, in comparison to chemotherapy alone, as a first-line treatment for patients diagnosed with metastatic NSCLC, from the perspective of the United States healthcare system. This research seeks to provide an in-depth analysis of the economic implications associated with the use of cemiplimab in the management of metastatic NSCLC, through the application of rigorous analytical methods and evidence-based assessments.
Methods
Model structure
A partition survival model has been developed in order to estimate the costs and clinical outcomes for patients with metastatic NSCLC. The model has incorporated three mutually exclusive health states: death, progression-free disease, and progressed disease (PD; Figure 1). The simulation cycle was set at 3 weeks, aligning with the EMPOWER-Lung 3 part 2 clinical trial, and the horizon time was set at 10 years. The decision to set the temporal horizon of the model at 10 years was based upon the poor 5-year overall survival rate for patients with metastatic NSCLC, and the fact that the long-term survival outcomes under current medical practices remain uncertain. We accounted for the potential overestimation of costs and outcomes in the model by implementing a half-cycle correction.
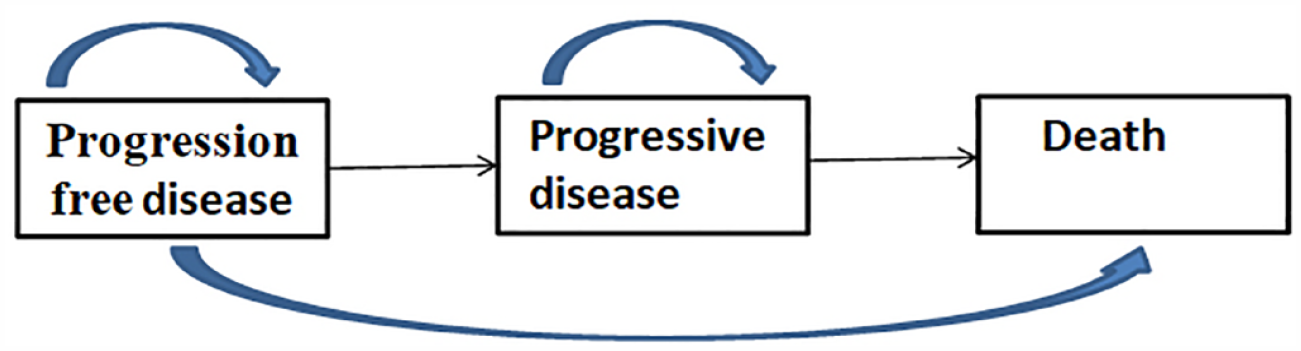
The model structure.
The study utilized a discount rate of 3% for both health utility and costs, and considered only direct medical expenses. 10 Clinical outcomes were measured in terms of total cost, quality-adjusted life years (QALYs), and incremental cost-effectiveness ratio (ICER). To determine the cost-effectiveness of a treatment regimen, a willingness-to-pay (WTP) threshold of $150,000 per QALY was established, with a regimen deemed cost-effective if its ICER fell below this threshold. 11 The analysis was conducted using the TreeAge Pro 2011 software (Williamstown, MA, USA).
Population and treatment
The present study focused on patients who are consistent with those who participated in the phase III EMPOWER-Lung 3 Part 2 Trial. The study recruited patients with advanced NSCLC who lacked tumor genomic aberrations in EGFR, ALK, or ROS1.The study employed a 2:1 randomization and stratified patients by histology to receive either cemiplimab 350 mg or placebo every 3 weeks, in combination with four cycles of chemotherapy. The choice of histology-specific chemotherapy was determined by the investigator and included pemetrexed plus carboplatin, pemetrexed plus cisplatin, paclitaxel plus carboplatin, and paclitaxel plus cisplatin. Patients were treated for up to 108 weeks or until disease progression or unacceptable toxicity occurred. Patients with nonsquamous NSCLC who were assigned a pemetrexed-containing regimen were required to undergo maintenance therapy with pemetrexed.
The results of the clinical trial indicate that the median duration of treatment exposure was 38.8 weeks (interquartile range: 20.7–92.5) with the administration of cemiplimab in conjunction with chemotherapy, while chemotherapy alone resulted in a median duration of 21.3 weeks (interquartile range: 12.0–38.4). In our analysis, adverse events (AEs) classified as grade 3–4 were identified if their incidence rate exceeded 3% in both the cemiplimab and chemotherapy treatment arms. It is essential to highlight that a notable percentage of patients in the cemiplimab plus chemotherapy cohort (51.3%) and the chemotherapy-only cohort (68.2%) received subsequent anticancer treatments due to treatment progression. In consultation with medical professionals, we assumed a chemotherapy protocol, utilizing platinum and docetaxel, as the primary modality for subsequent cancer therapy. 12 It is pertinent to note that the clinical trial did not establish the optimal third-line treatment or demonstrate the efficacy of individual therapeutic strategies. As a result, for our study, we assumed the best supportive care approach after the failure of second-line treatment.
Model survival and transition probabilities
The survival data for each treatment group in the phase III EMPOWER-Lung 3 Part 2 Trial were extracted by utilizing the GetData Graph Digitizer (version 2.25, http://www.getdata-graph-digitizer.com/) to analyze the survival curves. The extracted data were then reconstructed using the R software. To ensure accuracy, multiple distribution options, such as the log-logistic, log-normal, weibull, gompertz, exponential, and gamma distributions, were considered. 13 After rigorous statistical analysis and visual inspection, the log-logistic distribution was found to have the lowest Akaike information criterion and Bayesian information criterion values. 14 Therefore, it was selected as the optimal distribution for predicting the long-term survival status of patients (Supplemental Table 1, Supplemental Figure 1). This analytical approach provides a more reliable and evidence-based way of obtaining survival data, ultimately enhancing the robustness of the study’s findings.
The survival function was computed by calculating the time transition probability, yielding the Log-logistic distribution expression S(t) = 1/(1 + λtγ). 15 The shape parameter (γ) and scale parameter (λ) were estimated with R software, and their corresponding values can be found in Table 1.
Cost and health utility
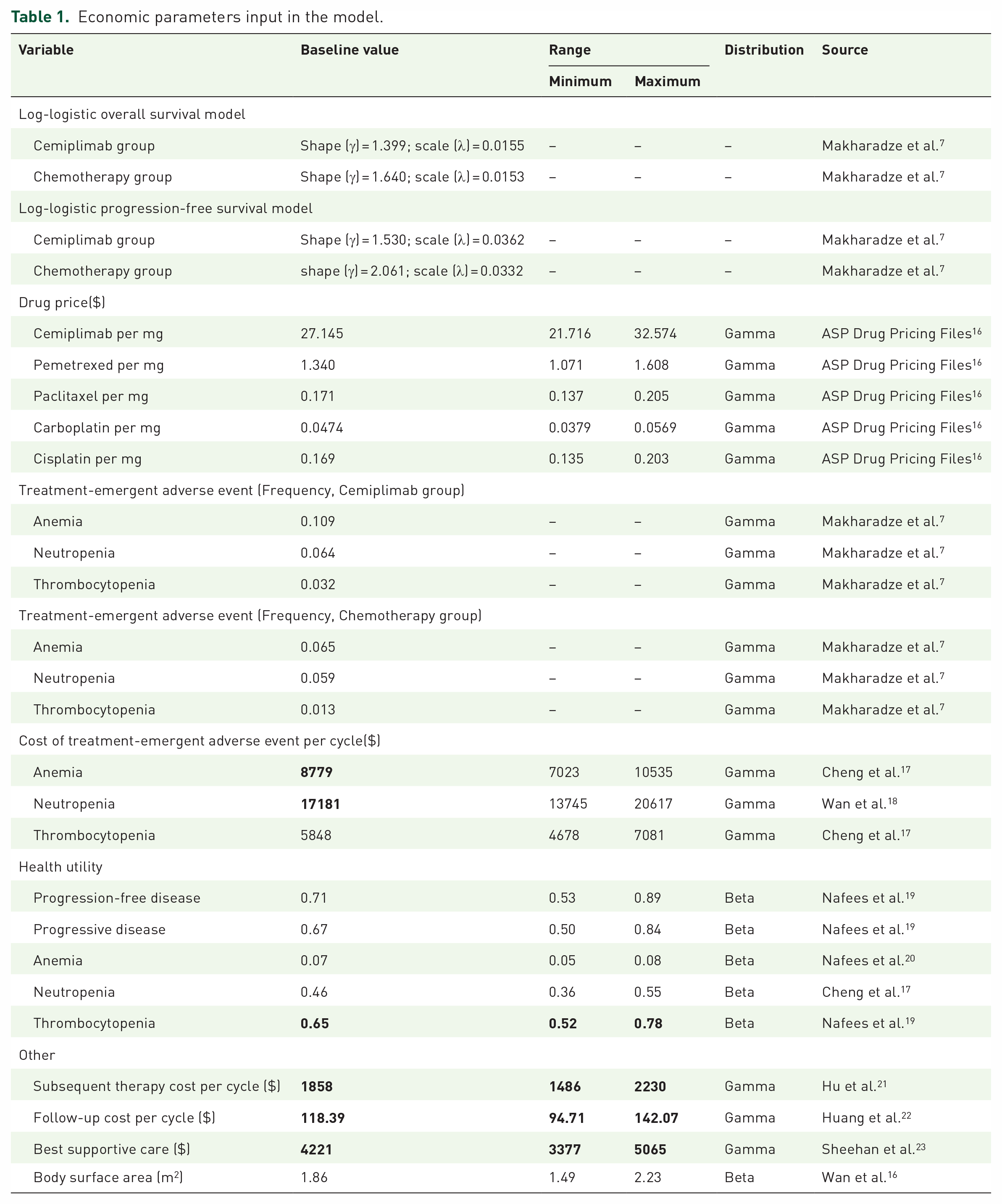
Our primary focus was on direct costs associated with medical care. The total cost for both treatment arms was calculated by taking into account various clinical drugs, subsequent therapy, best supportive care, and costs incurred in managing treatment-emergent grade 3 and 4 adverse events (TEAEs). To estimate the costs of drugs, we utilized the Medicare Part B drug average sales price data from the US Centers for Medicare & Medicaid Services for May 2023. The chemotherapy dose was derived by considering multiple factors, including a body weight of 70 kg, a body surface area of 1.86 m2, and a creatinine clearance of 70 mL/min. 18
In accordance with expert medical advice, we have employed a chemotherapy protocol consisting of platinum and docetaxel as the principal modality for subsequent cancer therapy. It is noteworthy to highlight that the clinical trial undertaken did not establish the definitive third-line treatment or conclusively demonstrate the efficacy of individual therapeutic strategies. Consequently, in our study, we have assumed the utilization of a best supportive care approach following the failure of second-line treatment. This therapeutic paradigm primarily encompasses a diverse array of interventions aimed at ameliorating symptoms and furnishing comprehensive support to patients during and after unsuccessful cancer treatments. The scope of this supportive approach encompasses various aspects, such as implementing nutritional support interventions, providing interventions for symptomatic pain relief, offering psychological counseling, and implementing other related strategies.
In order to assess the health-related quality of life for each health condition, utility values between 0 and 1 were incorporated into the model. The phase III EMPOWER-Lung 3 Part 2 trial clinical investigation did not provide health-related data, thus published literature was referenced in order to determine the utility values for progression-free survival (PFS) and progressive disease (PD) in the two treatment arms. Moreover, the model accounted for the disutility associated with TEAEs. A detailed summary of the cost and utility values can be found in Table 1.
Economic parameters input in the model.
Sensitivity analysis
To evaluate the resilience of the constructed model, a comprehensive deterministic sensitivity analysis (DSA) was undertaken in this study. In this regard, a one-way sensitivity analysis was conducted by individually manipulating each input parameter by a range of ±20%, coupled with a dynamic variation of the discount rate from 0% to 8%. The resulting outcomes were visually represented through the depiction of a tornado diagram, effectively encapsulating the influence of each parameter on the ICER.
A probabilistic sensitivity analysis (PSA) utilizing 1000 Monte Carlo simulations was conducted to evaluate the robustness of the research findings. The PSA relied on model runs using input parameters modeled as random variables with specific distributions, with the cost parameter modeled using the gamma distribution, and the utility and transition probability parameters considered appropriate for the beta distribution. The outcomes were presented utilizing scatter plot.
This cost-effectiveness analysis conforms to the to the Consolidated Health Economic Evaluation Reporting Standards 2022 (CHEERS 2022), ensuring methodological rigor and scholarly integrity in its presentation. 24
Results
Base-case results
The combination therapy of cemiplimab and chemotherapy has been demonstrated to result in a notable incremental gain of 1.599 QALYs, at an accompanying incremental cost of $109351.298. This leads to an ICER value of $68644.883/QALY (Table 2). These findings suggest that the use of cemiplimab in conjunction with chemotherapy is a cost-effective alternative versus chemotherapy alone, as the ICER falls comfortably below the widely recognized WTP benchmark of $150,000/QALY in the United States.
The results of base-case analysis.

ICER, incremental cost–effectiveness ratio; LYG, life year gained; NA, not applicable; QALY, quality-adjusted life year.
Sensitivity analyses
The findings from the one-way DSA have been visually represented using a tornado diagram, as depicted in Figures 2. The results indicate that subsequent costs exerted the greatest influence on the ICER. Other noteworthy factors that significantly influenced ICER include the utility of PD, the cost of best supportive care, the cost of cemiplimab per mg, and the utility of PFS. However, while these factors did impact the model’s outcomes, none of them displayed a substantial effect as the ICER value consistently remained below the WTP threshold of $150000/QALY when all levels of uncertainty were taken into account.

The result of the one-way sensitivity analysis.
The presented scatter plot in Figure 3 depicts the results of a Monte Carlo simulation, which demonstrates that the cemiplimab group is of superior cost-effectiveness compared to the chemotherapy group. The diagonal line depicted in the plot represents the WTP value, which serves as a threshold for determining the point at which the benefits of cemiplimab surpass its costs. Notably, this study found that when the WTP threshold is set at $150,000/QALY, the cemiplimab group has a 98.10% probability of being considered the more cost-effective treatment option.
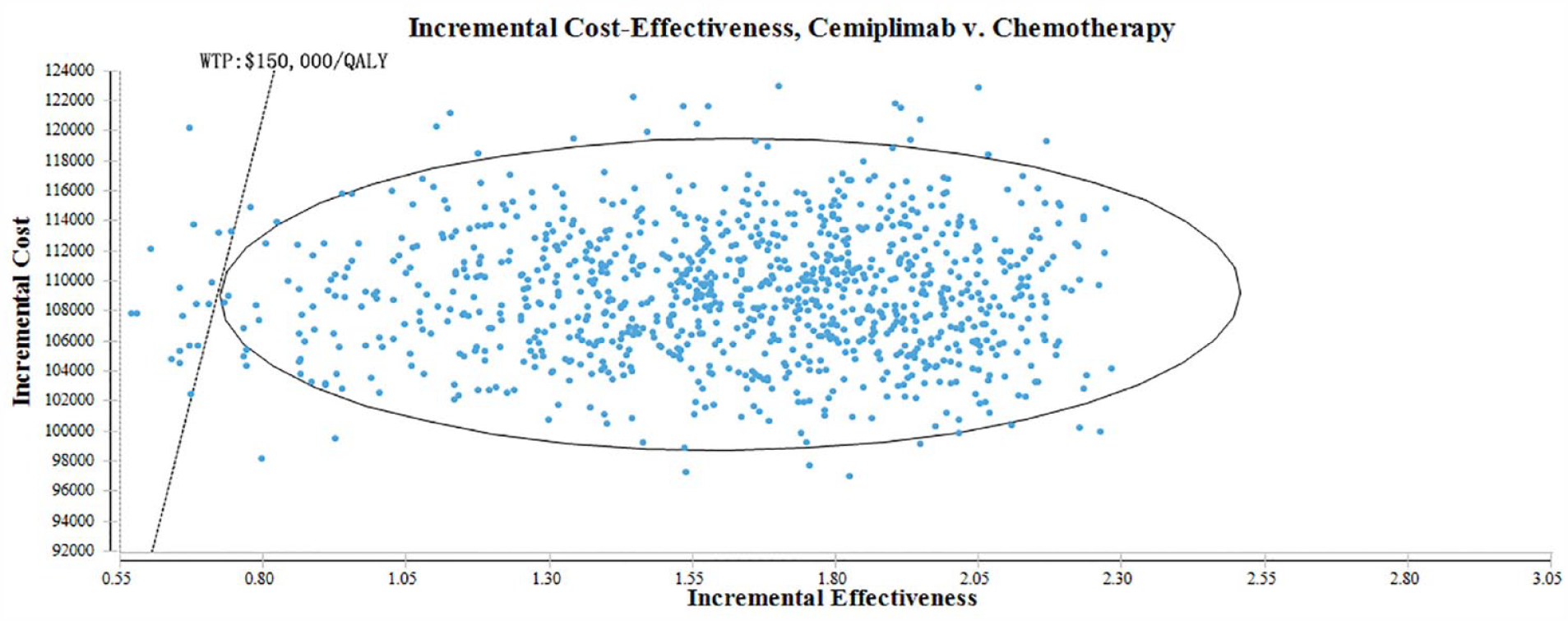
The probabilistic sensitivity analysis scatter plot.
Discussion
Cancer is a significant contributor to global mortality, resulting in approximately 10 million deaths annually across the world. 25 Lung cancer currently ranks as the leading cause of cancer-related mortality worldwide, contributing significantly to the escalating cost of cancer treatment. 26 Immunotherapy, specifically ICIs, has emerged as a promising therapeutic strategy for the treatment of various tumors.27,28 Given the rising incidence of lung cancer, there is growing interest among clinicians in the potential therapeutic benefits of combining cemiplimab with chemotherapy, as demonstrated in the EMPOWER-Lung 3 clinical trial. A recent 2-year follow-up analysis from part 2 of the EMPOWER-Lung 3 trial demonstrated sustained benefits of cemiplimab plus chemotherapy versus chemotherapy alone in patients with advanced NSCLC after 28.4 months of follow-up. However, the high cost of cemiplimab may limit its widespread adoption. 29 As such, this study aims to evaluate the cost-effectiveness of using cemiplimab in combination with chemotherapy as a first-line treatment for metastatic NSCLC patients compared to chemotherapy alone, from the perspective of the United States healthcare system.
The combination therapy of cemiplimab plus chemotherapy has been demonstrated to yield promising results in terms of both cost and QALYs. The total cost incurred for this treatment option is reported to be $268,615.733, resulting in a QALY gain of 3.192. In comparison, the chemotherapy group incurred a cost of $159,264.435, with a corresponding QALY gain of 1.599.A noteworthy finding of this study is the incremental gain of 1.593 QALYs achieved through the use of cemiplimab plus chemotherapy. Although this gain comes at an additional cost of $109,351.298, it signifies a substantial improvement in terms of patient outcomes. To assess the value of this incremental gain, an ICER was calculated, which stands at $68,644.883 per QALY. The results of this study indicate that cemiplimab plus chemotherapy is a cost-effective treatment option for advanced NSCLC patients. This combination therapy provides notable improvements in overall survival, PFS, and quality of life, whereas the associated costs are acceptable within the context of commonly accepted cost-effectiveness thresholds.
This analysis is particularly important as healthcare decisions, especially regarding cancer treatment, are often influenced by several factors that introduce uncertainty. Our findings were further supported by the results of the one-way analysis and PSA, which demonstrated the robustness of this conclusion. Therefore, the study’s results under different scenarios were evaluated to provide decision-makers with a more comprehensive understanding of the benefits and costs associated with the treatment of advanced NSCLC.
Previous research has investigated the efficacy of cemiplimab plus chemotherapy as a primary treatment for advanced NSCLC. Kuznik et al. 30 conducted a cost-effectiveness analysis of cemiplimab compared to other first-line treatments for advanced NSCLC with PD-L1 expression ⩾50% from the perspective of a US commercial payer. The authors suggest that cemiplimab is a cost-effective option for this patient population, as compared to pembrolizumab or chemotherapy. Similarly, Zhang et al. 31 found that cemiplimab is a cost-effective choice for first-line treatment of NSCLC in patients with at least 50% PD-L1 expression from an American perspective. Our study is comparable to previous research in this field. However, it should be noted that the clinical trials cited in prior studies differ from those utilized in our research. Furthermore, we have access to extended follow-up data for part 2 of the EMPOWER-Lung 3 trial, which provides more robust and compelling evidence. Our research findings indicate that the co-administration of cemiplimab, an ICI, alongside chemotherapy could yield a significant cost-effective advantage in patients suffering from advanced stages of nonsquamous or squamous NSCLC. This trend held true across varying levels of PD-L1 expression, indicating a broad applicability of this treatment approach.
This investigation has some limitations. First, we had to make assumptions in relation to subsequent anticancer drugs and best supportive care, given the unavailability of specific drug information in corresponding clinical trials. It is possible that this assumption may lead to bias against cost estimates. However, we ensured that our findings were robust across a wide range of variations in the price of subsequent cost to minimize any potential bias. Second, in order to obtain utility values, we had to rely on published literature. As with any literature review, there may be minor inconsistencies between the simulation results and actual health outcomes. To address this issue, we conducted a sensitivity analysis and found that these utility values did not significantly impact the base-case results. A third limitation of our study is that we included only TEAEs in grades 3–4 when calculating disutility values and costs. However, our sensitivity analysis revealed that TEAE-related factors did not affect the outcome, confirming our assumption that grade 1/2 TEAEs would not substantially impact the final report conclusion. We acknowledge these potential limitations and have taken measures to address them through sensitivity analyses and careful consideration of assumptions.
In summary, the current research utilizes an advanced analytical model to estimate the life expectancy and healthcare costs of patients with advanced NSCLC. In conclusion, the addition of cemiplimab to chemotherapy showed improved clinical outcomes at an acceptable cost-effectiveness threshold versus chemotherapy alone. The study’s findings are expected to inform clinical decision-making by providing healthcare professionals with valuable insights into the costs and benefits of advanced NSCLC treatment options.
Conclusion
The utilization of cemiplimab in combination with chemotherapy as a first-line treatment option for NSCLC appears to be a cost-effective approach as compared to using chemotherapy as a standalone therapy from the perspective of the United States healthcare system.
Supplemental Material
sj-docx-1-tam-10.1177_17588359231213619 – Supplemental material for Cemiplimab combined with chemotherapy versus chemotherapy in advanced non-small cell lung cancer: an updated EMPOWER-Lung 3 trial-based cost-effectiveness analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359231213619 for Cemiplimab combined with chemotherapy versus chemotherapy in advanced non-small cell lung cancer: an updated EMPOWER-Lung 3 trial-based cost-effectiveness analysis by Gaofeng Zhu, Hongfu Cai and Zhiwei Zheng in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-2-tam-10.1177_17588359231213619 – Supplemental material for Cemiplimab combined with chemotherapy versus chemotherapy in advanced non-small cell lung cancer: an updated EMPOWER-Lung 3 trial-based cost-effectiveness analysis
Supplemental material, sj-jpg-2-tam-10.1177_17588359231213619 for Cemiplimab combined with chemotherapy versus chemotherapy in advanced non-small cell lung cancer: an updated EMPOWER-Lung 3 trial-based cost-effectiveness analysis by Gaofeng Zhu, Hongfu Cai and Zhiwei Zheng in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
