Abstract
Sarcomatoid malignant pleural mesothelioma (MPM) is a rare and aggressive cancer with limited therapeutic options. We describe an exceptionally rare case of sarcomatoid MPM in a man in his 50s who developed three severe immune-related adverse events (irAEs)—Grade 3 pneumonitis, Grade 3 hepatitis, and Grade 4 agranulocytosis—within 55 days of initiating nivolumab plus ipilimumab. Corticosteroid treatment and granulocyte colony-stimulating factor resulted in recovery from these toxicities, while two liver biopsies provided essential diagnostic insights, distinguishing drug-induced liver injury from immune-related hepatitis. Despite receiving only a limited number of immune checkpoint inhibitor doses and discontinuing therapy, the patient exhibited rapid pleural tumor regression and sustained clinical benefit. This case highlights the potential association between severe immune-related side effects and favorable treatment response in MPM, and underscores the importance of pathology-supported diagnosis and shared decision-making in managing complex irAEs.
Plain language summary
This case report describes a man in his 50s who was diagnosed with a rare and aggressive type of cancer called sarcomatoid malignant pleural mesothelioma, often linked to asbestos exposure. He was treated with a new type of cancer therapy known as immune checkpoint inhibitors (ICIs), specifically nivolumab and ipilimumab. Although this therapy led to rapid tumor shrinkage, the patient experienced three severe immune-related side effects within six weeks: -Lung inflammation (pneumonitis) -Liver inflammation (hepatitis) -A dangerously low white blood cell count (agranulocytosis) Doctors used liver biopsies and careful diagnostic methods to understand the causes of these side effects. With prompt treatment using steroids and supportive care, the patient recovered from the side effects and still achieved remarkable cancer control, even after stopping immunotherapy. This case shows that: -Severe immune side effects may be linked to a strong anti-cancer response -Quick diagnosis and personalized care can help manage risks -Shared decision-making between patients and doctors is vital It offers valuable insights into how complex side effects can be managed during powerful immunotherapy treatments.
Keywords
Introduction
Malignant pleural mesothelioma (MPM) is an aggressive tumor strongly associated with asbestos exposure, with a particularly poor prognosis. The sarcomatoid subtype is known to be highly resistant to standard platinum-based chemotherapy and generally demonstrates extremely low response rates.1,2
As a novel treatment strategy for such refractory disease, immune checkpoint inhibitors (ICIs) have been introduced. The CheckMate 743 trial demonstrated that first-line nivolumab plus ipilimumab significantly prolonged overall survival compared to standard chemotherapy in patients with unresectable MPM, with efficacy also observed in the sarcomatoid subtype. 3
However, ICIs can induce immune-related adverse events (irAEs), which can unpredictably affect various organs such as the lungs,4,5 liver,6,7 and hematopoietic system,8–11 sometimes presenting as high-grade toxicities. Their onset is often abrupt, and the management remains clinically challenging. Particularly, immune-related agranulocytosis is exceedingly uncommon and sparsely documented in the literature. 12 Moreover, previous clinical and translational studies have suggested that the occurrence of irAEs may correlate with enhanced therapeutic efficacy and improved durability of response to ICIs.13–16 This potential association provides an important biological and clinical context for interpreting the unique clinical course observed in our case.
Here, we report an exceptionally rare case of sarcomatoid MPM that developed severe irAEs, including grade 3 pneumonitis, grade 3 hepatitis, and grade 4 agranulocytosis, following treatment with nivolumab plus ipilimumab. Remarkably, despite these toxicities, the patient demonstrated significant tumor regression, with notable pleural shrinkage and resolution of effusion. This case offers valuable clinical insight into the potential link between irAEs and therapeutic response.
Case presentation
A 59-year-old male, a former smoker (20 cigarettes/day for 8 years between ages 20 and 28), worked as an electrician. In childhood, he lived near an asbestos-related factory and had been previously advised to undergo an asbestos health screening.
In November 2024, routine health screening detected a left-sided pleural effusion, despite the absence of symptoms. Around February 10, 2025, he developed a persistent fever, followed by exertional dyspnea. Chest computed tomography (CT) showed left-sided pleural effusion and diffuse pleural thickening (Figure 1). Although pleural fluid adenosine deaminase was elevated at 65.3 U/L, both polymerase chain reaction and culture for Mycobacterium tuberculosis were negative. Due to a known contact history with tuberculosis patients, anti-tuberculosis therapy with isoniazid, rifampicin, pyrazinamide, and ethambutol was initiated on February 7.

Radiographic and CT findings before and after immunotherapy. Chest radiographs and CT images at two time points. Top row: March 10, 2025 (Day −29) before initiation of ICI therapy, demonstrating left-sided pleural thickening and massive pleural effusion with collapsed left lung. Bottom row: June 20, 2025 (Day 73) after two doses of nivolumab and one dose of ipilimumab, showing marked tumor regression, pleural retraction, and near-complete re-expansion of the left lung.
Despite therapy, the fever persisted, prompting referral to our hospital for further evaluation. On admission (March 10), marked elevation of hepatic enzymes was noted (aspartate aminotransferase (AST) 3061 U/L, alanine aminotransferase (ALT) 2547 U/L), prompting discontinuation of all anti-tuberculosis medications due to suspected drug-induced liver injury (DILI). Concurrent abdominal CT revealed a solitary hypodense lesion in segment 5 of the liver. A liver biopsy performed 7 days later (Day −22) revealed both tumor cell infiltration and features consistent with acute drug-induced hepatitis. Liver enzymes improved thereafter.
Thoracoscopic pleural biopsy was performed on March 31. Intraoperative images revealed diffusely thickened and nodular parietal pleura (Figure 2). Histopathological evaluation confirmed sarcomatoid MPM on April 4, based on immunohistochemistry showing cytokeratin AE1/AE3 (CK AE1/AE3) (+), WT-1 (+), and calretinin (−) (Figure 3).

Thoracoscopic findings of the pleural cavity. Image taken on March 31, 2025 (Day −8) showing diffusely thickened parietal pleura with multiple nodular and fibrous lesions. These gross findings prompted pleural biopsy under direct vision.

Histopathological and immunohistochemical findings of pleural biopsy. Obtained on April 8, 2025 (Day 0), confirming sarcomatoid malignant pleural mesothelioma. (a) HE staining reveals proliferation of spindle-shaped tumor cells. (b) Immunohistochemistry for AE1/AE3 shows diffuse cytoplasmic positivity. (c) D2–40 demonstrates strong membranous positivity. (d) WT-1 shows scattered nuclear positivity. (e) Calretinin is mostly negative. (f) A few cells show weak calretinin positivity. All images captured at ×200 magnification with scale bars indicating 60 μm.
On April 8 (Day 0), nivolumab (360 mg) plus ipilimumab (1 mg/kg) was initiated as first-line therapy under the CheckMate 743 protocol. On Day 8 (April 16), the patient developed fatigue and oxygen desaturation. Chest CT showed bilateral ground-glass opacities in the lower lobes, consistent with grade 3 immune-related pneumonitis (Figure 4). Treatment with methylprednisolone 60 mg/day was initiated, with rapid clinical improvement. The corticosteroid dose was tapered weekly (60–40–20–10 mg/day). Adverse events were assessed according to the Common Terminology Criteria for Adverse Events version 5.0.

Chest CT on April 16, 2025 (Day 8) showing immune-related pneumonitis. Ground-glass opacities and patchy consolidations are seen in both lower lobes, consistent with Grade 3 immune-related pneumonitis (per CTCAE v5.0).
Following shared decision-making (SDM), nivolumab monotherapy was administered as the second dose on Day 38 (May 16). On Day 45 (May 23), transaminase levels rose to AST 208 and ALT 326 U/L, peaking the next day at 295 and 511 U/L, respectively, indicating grade 3 immune-related hepatitis. Viral, autoimmune, and drug-induced causes were excluded. A second liver biopsy performed the same day (Day 45) revealed CD8-positive T-cell infiltration consistent with immune-mediated hepatitis (Figure 5).
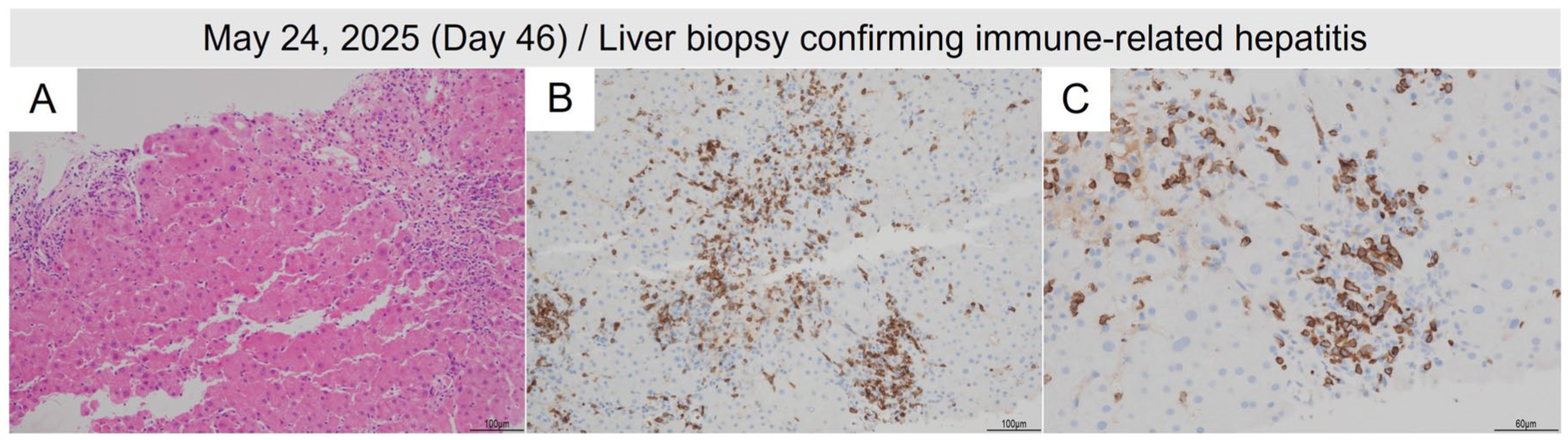
Histopathological findings of liver biopsy on May 28, 2025 (Day 56). Findings are consistent with immune-related hepatitis. (a) HE staining shows interface hepatitis with lymphocytic infiltration at the limiting plate. (b) CD3 immunostaining highlights dense T-cell infiltration. (c) CD8 immunostaining shows predominant cytotoxic T-cell infiltration. Scale bars indicate 100 μm for (a) and (b), and 60 μm for (c).
Detailed serial laboratory data, including AST, ALT, alkaline phosphatase, prothrombin time/international normalized ratio, and fibrinogen, are provided in Table S1 and Figure S1. 17
On Day 46, the patient developed grade 4 agranulocytosis, with a neutrophil count of 5/μL. Anti-neutrophil antibodies were negative. Although bone marrow examination was not performed, the patient’s neutrophil count recovered with 7-day granulocyte colony-stimulating factor (G-CSF) therapy and increased corticosteroids (methylprednisolone 110 mg/day), supporting a diagnosis of immune-related agranulocytosis.
Regarding treatment efficacy, CT imaging on Day 37 (May 15) showed a reduction of pleural thickening and hepatic metastasis. Response evaluation CT on Day 73 (June 20) revealed marked tumor shrinkage, re-expansion of the left lung, and resolution of pleural effusion (Figure 1).
The overall treatment course, including irAEs and corresponding laboratory changes, is summarized in Figure 6.

Clinical timeline of treatment, irAEs, and biochemical parameters. The upper panel depicts the dosing schedule of mPSL (mg/day). The lower panel shows longitudinal changes in AST, ALT, and ANC (logarithmic scale). Key clinical events are annotated with corresponding day values, where Day 0 indicates the initial administration of nivolumab plus ipilimumab. irAEs included pneumonitis (Grade 3, Day 8), hepatitis (Grade 3, Day 45), and agranulocytosis (Grade 4, Day 45), all of which were managed with corticosteroid titration and G-CSF support. Pleural biopsy (Day −8) and second liver biopsy (Day 45) were pivotal in guiding clinical decision-making.
As of August 2025, the patient remains alive, with his general condition maintained compared with the time of diagnosis. He continues to be followed closely in the outpatient clinic with ICI therapy suspended.
This case report was prepared in accordance with the CARE guidelines, 18 and the completed checklist is provided as Table S2.
Discussion
This case represents a clinically significant and extremely rare example of sarcomatoid MPM in which nivolumab plus ipilimumab therapy triggered a rapid onset of multiorgan irAEs within only 45 days, followed by a marked clinical response within 73 days. Similar early-onset multiorgan irAEs have been rarely reported in the literature, particularly in sarcomatoid MPM, underscoring the uniqueness of this clinical course.1,2
First, the timing and extent of tumor response are noteworthy. Within 11 weeks of initiating ICI therapy, the patient demonstrated dramatic regression of pleural thickening, resolution of pleural effusion, and re-expansion of the lung. Such an early and robust response is rarely documented in sarcomatoid MPM, a notoriously refractory disease. These findings are in line with previous studies suggesting that early-onset irAEs may correlate with favorable ICI responses. 14 These findings may suggest a possible association between early severe irAEs and tumor response; however, causality cannot be confirmed from a single case and should be considered hypothesis-generating rather than definitive. Emerging evidence suggests that severe irAEs may reflect robust T-cell activation and epitope spreading, which can enhance antitumor immunity. Dual blockade of CTLA-4 and PD-1 may further amplify tumor-specific immune responses, potentially explaining the parallel occurrence of both toxicity and tumor regression observed in this case.6,7
Second, the use of serial liver biopsies was critical in guiding diagnosis and management. The first biopsy clarified the etiology of the initial liver injury as drug-induced and partially tumor-associated. The second biopsy, following the onset of hepatitis after ICI rechallenge, demonstrated CD8+ T-cell infiltration consistent with immune-mediated hepatitis, excluding viral, autoimmune, and residual anti-tuberculosis drug causes. These findings support prior evidence emphasizing the diagnostic value of liver biopsy in immune-related hepatitis.6,7
Differentiating immune-related hepatitis from DILI was a major diagnostic challenge in this case. The initial severe hepatotoxicity following anti-tuberculosis therapy improved promptly after drug discontinuation, consistent with DILI. In contrast, the second episode of hepatotoxicity occurred approximately 7 weeks after initiating ICIs, aligning with the typical timeframe of immune-related hepatitis. 13 Clinically, the second episode required high-dose corticosteroids for resolution, whereas the first episode improved with withdrawal of anti-tuberculosis drugs alone, supporting an immune-mediated mechanism. 13 Laboratory findings further supported this distinction, with a hepatocellular pattern of enzyme elevation, negative viral and autoimmune serologies, and normal IgG levels. Histopathological evaluation was decisive: the second biopsy demonstrated interface hepatitis with prominent CD3+ and CD8+ T-cell infiltration, accompanied by lobular necrosis, but without eosinophils, cholestasis, or granulomas. These features are typical of immune-related hepatitis and differ from patterns observed in idiosyncratic DILI.11,13 Taken together, the temporal profile, clinical course, laboratory data, and pathological findings collectively supported the diagnosis of immune-related hepatitis rather than DILI in this patient.
Furthermore, the development of grade 4 agranulocytosis following ICI administration is exceedingly rare. Though bone marrow biopsy was not performed, negative antineutrophil antibodies and recovery with G-CSF and corticosteroids support the diagnosis of ICI-induced agranulocytosis, consistent with mechanisms reported by Delanoy et al. 8 and Michot et al. 11 However, the absence of a bone marrow examination limits the ability to definitively classify the agranulocytosis subtype, such as distinguishing between marrow suppression and peripheral destruction. 13
Although bone marrow aspiration is generally considered a relatively low-risk procedure, we judged that performing it during profound neutropenia would pose a non-negligible risk of infection and clinical deterioration, and therefore, it was not performed. 19 As a result, acute hematologic disorders such as acute leukemia could not be completely excluded, which we acknowledge as an important limitation of this report. Nevertheless, the temporal association with ICI administration, the absence of alternative causes, and the prompt recovery after G-CSF and corticosteroid therapy strongly support an immune-related etiology.
Rechallenge with ICIs after grade 3 pneumonitis deviated from current international guidelines, which generally recommend permanent discontinuation of ICIs in such circumstances.20,21 In this case, the decision was made through a multidisciplinary discussion and SDM with the patient. The lack of effective alternative treatments for sarcomatoid MPM, the remarkable initial tumor regression, and the patient’s strong desire to continue immunotherapy—after being fully informed of the potential risks—all contributed to this individualized decision. We emphasize that this approach should be interpreted with caution and cannot be generalized. Importantly, the patient’s perspective was also considered: he expressed a strong desire to continue immunotherapy despite the risks, and this preference was carefully integrated into the SDM process.
To further contextualize our case, we have incorporated references to relevant reports and registry data describing early-onset irAEs in thoracic malignancies, 17 and we have discussed similarities and differences with our patient’s course. This highlights both the rarity of early severe irAEs in MPM and the potential clinical implications of their possible association with antitumor benefit.
In addition, we cited further case-based reports describing concurrent or sequential severe irAEs associated with favorable outcomes in other cancers, thereby providing a broader context to support the hypothesis that irAEs may serve as clinical correlates of treatment benefit. 22 Despite the severity of irAEs, the patient exhibited remarkable tumor regression, consistent with prior literature suggesting an association between irAEs and both response rate and durable clinical benefit.15,23
Limitations
This report has several limitations. First, as a single case, the findings cannot be generalized regarding irAE patterns or ICI efficacy in MPM. Second, although longer-term follow-up is ongoing, the patient remains alive as of August 2025, which is encouraging but insufficient to draw conclusions about durability. Third, bone marrow aspiration was not performed; while generally considered low risk, it was avoided due to the high risk of infection during profound neutropenia. As a result, acute hematologic disorders such as acute leukemia could not be definitively excluded, underscoring the diagnostic limitation in accurately confirming immune-related agranulocytosis. Fourth, although serial liver biopsies were informative, they showed both tumor infiltration and irAE-related changes, making quantitative attribution difficult. Fifth, biomarker analyses, including driver oncogenic alterations, PD-L1 expression, and tumor mutational burden, were not performed, precluding deeper mechanistic insights into the relationship between irAEs and treatment response. Finally, the patient’s recovery was multifactorial (G-CSF plus corticosteroids), and the decision to rechallenge ICIs was based on SDM without formal assessment of psychological factors, both of which limit the certainty and generalizability of our interpretations.
Conclusion
This case highlights a highly aggressive form of immune activation with sequential onset of grade 3 pneumonitis, grade 3 hepatitis, and grade 4 agranulocytosis—all within 7 weeks of initiating combination ICI therapy for sarcomatoid MPM.
Despite the severity of irAEs, the patient exhibited remarkable tumor regression within 11 weeks. This underscores the potential for strong therapeutic efficacy, even in rapidly evolving and high-toxicity clinical contexts. It also highlights the critical importance of careful monitoring and SDM in managing immunotherapy.
Furthermore, this observation raises the possibility that severe irAEs may serve as predictive markers of treatment efficacy in MPM, underscoring the need for further investigation in larger cohorts.
Supplemental Material
sj-docx-3-tam-10.1177_17588359251397331 – Supplemental material for Early severe immune-related pneumonitis, hepatitis, and agranulocytosis with radiographic response in sarcomatoid malignant pleural mesothelioma treated with nivolumab and ipilimumab: a case report highlighting dual liver biopsies and rechallenge decision-making
Supplemental material, sj-docx-3-tam-10.1177_17588359251397331 for Early severe immune-related pneumonitis, hepatitis, and agranulocytosis with radiographic response in sarcomatoid malignant pleural mesothelioma treated with nivolumab and ipilimumab: a case report highlighting dual liver biopsies and rechallenge decision-making by Tomohiro Oba, Katsuhiro Itogawa, Yoko Machida, Yuji Ono, Yuho Morita, Daisuke Nakatani, Keiichi Akasaka, Akiko Adachi and Hidekazu Matsushima in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-2-tam-10.1177_17588359251397331 – Supplemental material for Early severe immune-related pneumonitis, hepatitis, and agranulocytosis with radiographic response in sarcomatoid malignant pleural mesothelioma treated with nivolumab and ipilimumab: a case report highlighting dual liver biopsies and rechallenge decision-making
Supplemental material, sj-jpg-2-tam-10.1177_17588359251397331 for Early severe immune-related pneumonitis, hepatitis, and agranulocytosis with radiographic response in sarcomatoid malignant pleural mesothelioma treated with nivolumab and ipilimumab: a case report highlighting dual liver biopsies and rechallenge decision-making by Tomohiro Oba, Katsuhiro Itogawa, Yoko Machida, Yuji Ono, Yuho Morita, Daisuke Nakatani, Keiichi Akasaka, Akiko Adachi and Hidekazu Matsushima in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-xlsx-1-tam-10.1177_17588359251397331 – Supplemental material for Early severe immune-related pneumonitis, hepatitis, and agranulocytosis with radiographic response in sarcomatoid malignant pleural mesothelioma treated with nivolumab and ipilimumab: a case report highlighting dual liver biopsies and rechallenge decision-making
Supplemental material, sj-xlsx-1-tam-10.1177_17588359251397331 for Early severe immune-related pneumonitis, hepatitis, and agranulocytosis with radiographic response in sarcomatoid malignant pleural mesothelioma treated with nivolumab and ipilimumab: a case report highlighting dual liver biopsies and rechallenge decision-making by Tomohiro Oba, Katsuhiro Itogawa, Yoko Machida, Yuji Ono, Yuho Morita, Daisuke Nakatani, Keiichi Akasaka, Akiko Adachi and Hidekazu Matsushima in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We are deeply grateful to the patient and his family for their understanding and cooperation in the publication of this case. We also thank all medical professionals involved in the diagnosis, treatment, and follow-up care.
Declarations
Availability of data and materials
Not applicable.
Grant number
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
