Abstract
Background:
Pembrolizumab plus chemotherapy (Pembrolizumab-CT) has been suggested as first-line treatment over pembrolizumab alone in aggressive non-small-cell lung cancer (NSCLC) with ⩾50% PDL1, but studies comparing these two strategies are lacking.
Objectives:
To compare overall survival under pembrolizumab and Pembrolizumab-CT depending on tumor aggressiveness in PDL1 ⩾50% advanced NSCLC patients.
Design:
A multicenter retrospective study was conducted of all patients with advanced NSCLC, PDL1 ⩾50% and ECOG 0–1, who received pembrolizumab or Pembrolizumab-CT as first-line treatment.
Methods:
Tumor aggressiveness was defined as a sum of longest diameters (SLD) ⩾ 100 mm, a largest lesion diameter (LLD) >60 mm, ECOG 1, or need for corticosteroid therapy. Overall survival was analyzed in the whole population and in subgroups as restricted mean survival time (RMST) adjusted for the main prognostic variables.
Results:
Ninety-six of the 164 included patients (58.5%) received pembrolizumab, and 68 (41.5%) received Pembrolizumab-CT. In the study group overall, the RMST was significantly shorter under Pembrolizumab-CT than under pembrolizumab (−7.9 months; p = 0.03). RMSTs were significantly shorter in the Pembrolizumab-CT group among patients with LLD <60 mm (−8.6 months, p = 0.04) and among ECOG-0 patients (−12.3 months, p < 0.001). RMSTs did not differ significantly between groups in patients with SLD ⩾ 100 mm (−1.1 months; p = 0.82), in patients with SLD <100 mm (−3.1 months, p = 0.54), in patients with LLD ⩾ 60 mm (0.9 months; p = 0.75), in patients with need for corticosteroid therapy (−2.9 months, p = 0.62), or in ECOG-1 patients (−6.1 months, p = 0.12). Toxicity-related hospitalizations appeared more frequent under Pembrolizumab-CT (20.5%) than under pembrolizumab (12.5%).
Conclusion:
Pembrolizumab-CT was not associated with improved survival compared with pembrolizumab alone, in PDL1 ⩾ 50% advanced NSCLC patients, even in cases of aggressive disease. Chemotherapy-related toxicities may have had a negative effect on survival.
Keywords
Introduction
The standard first-line treatment for patients with advanced (stage IIIC/IV) non-small-cell lung carcinoma (NSCLC) in the absence of a targetable driver alteration includes anti-PD1 immune checkpoint inhibitors such as pembrolizumab. Pembrolizumab alone is associated with longer survival than chemotherapy alone in patients with PDL1 tumor proportion scores ⩾50%, 1 and pembrolizumab plus chemotherapy (Pembrolizumab-CT) is associated with better survival than chemotherapy alone regardless of PDL1 expression status.2,3
Therefore, for patients with PDL1 tumor proportion scores ⩾50%, pembrolizumab alone or in combination with chemotherapy is an option. The rationale for adding chemotherapy to pembrolizumab mainly relies on immunogenic tumor microenvironment and the down-regulation of regulatory T lymphocytes induced by chemotherapy exposure,4–6 as well as an acceptable safety profile since the rates of adverse events are similar for chemotherapy alone and Pembrolizumab-CT (except for febrile neutropenia). 2 However, the only available results comparing pembrolizumab alone and Pembrolizumab-CT in patients with PDL1 ⩾50% come from a retrospective study 7 and a pooled analysis of randomized trials 8 and are inconclusive as to the superiority of either strategy.
However, differences in survival may have been masked by the proportional hazards (Cox) models used in these studies, because immunotherapy carries a risk of hyperprogression,1,9 estimated at 4%–30% according to the study,10,11 which could invalidate the proportional hazards hypothesis. As a result, Cox models may yield false negatives and lead to an erroneous conclusion, as has been shown in immunotherapy studies.12,13 Alternative approaches, such as restricted mean survival time (RMST) analysis, do not have this limitation. 14
Furthermore, the relative efficacy of pembrolizumab alone and Pembrolizumab-CT may depend on other criteria. For example, American Society of Clinical Oncology (ASCO) guidelines 15 recommend pembrolizumab monotherapy in general because of its lower toxicity and lower cost, but suggest (expert opinion) that Pembrolizumab-CT might be preferred in patients with high tumor burden, a voluminous visceral lesion, or severe symptoms. Data supporting this recommendation are limited. Tumor burden measured by imaging according to RECIST 1.1 criteria seems to be a prognostic factor and to be predictive of response to pembrolizumab. Among patients with high tumor burden, regardless of PDL1 status, two studies16,17 found worse overall survival and more frequent progression in patients treated with Pembrolizumab-CT than in those treated with pembrolizumab alone.
The objective of the present study was to use RMST analysis to compare the effectiveness of pembrolizumab alone and Pembrolizumab-CT in NSCLC patients with PDL1 tumor proportion scores ⩾50%, depending on the aggressiveness of the tumor (defined based on tumor burden, greatest tumor volume, altered performance status, or need for corticosteroid therapy). Secondary objectives were to evaluate the effectiveness of these treatments by RMST analysis and traditional Cox regression, and to perform exploratory RMST analyses in subgroups stratified by other prognostic factors, as well as to compare progression-free survival, response rate, and toxicity.
Methods
Design and patients
This was a retrospective study of all patients treated with pembrolizumab or Pembrolizumab-CT for NSCLC in three French hospitals: the university hospital (CHU) of Lille, the Teissier Clinic of Valenciennes, and the general hospital of Dunkerque. Inclusions took place between September 2017 and April 2022.
The inclusion criteria were age ⩾18 years, treatment with pembrolizumab or Pembrolizumab-CT for NSCLC, PDL1 expression in more than 50% of tumor cells, stage III (not amenable to radiotherapy) or stage IV disease, no prior NSCLC treatment, and ECOG-0 or ECOG-1 performance status. Patients treated with pembrolizumab alone after December 2019 (the date at which chemotherapy plus pembrolizumab was approved in France) were included only if it could be determined, from medical records, that the patient had been offered both treatment options and had chosen pembrolizumab monotherapy. This criterion also limited group imbalance by excluding patients treated with pembrolizumab alone for medical reasons such as frailty. In keeping with approved indications in France and international recommendations,15,18,19 patients with oncogene (EGFR, ALK, and ROS1) addicted tumors and patients with ECOG ⩾2 performance status were excluded. Eligible patients were identified from prescription records. All data were obtained retrospectively from patient records.
Treatment modalities and follow-up
Pembrolizumab was administered at 200 mg intravenously every 3 weeks in both groups. Patients in the Pembrolizumab-CT group also received four cycles of doublet chemotherapy. This consisted, for non-squamous NSCLC, of four 3-week cycles of cisplatin (75 mg/m2) or carboplatin (AUC 5) according to medical assessment, associated with pemetrexed (500 mg/m2). For squamous NSCLC, the treatment consisted of four 3-week cycles of carboplatin (AUC 6) with paclitaxel (200 mg/m2), in accordance with the previously published pivotal trials.2,3 Clinical and laboratory parameters were monitored between cycles. Follow-up included contrast-enhanced chest CT and upper abdominal and cerebral CT or MRI.
In the Pembrolizumab-CT group, in the absence of disease progression after the first four induction cycles, maintenance therapy consisted of 200 mg pembrolizumab every 3 s weeks for both squamous and non-squamous NSCLC, in addition to pemetrexed (500 mg/m2) for non-squamous histology. In the pembrolizumab monotherapy group, patients continued to receive 200 mg pembrolizumab every 3 weeks. Treatment was continued in both groups for a maximum of 2 years or until unacceptable toxicity or disease progression.
Tumor aggressiveness
Tumor aggressiveness was defined according to ASCO guidelines on the basis of overall tumor burden, largest lesion diameter (LLD), performance status, and need for corticosteroid therapy as a proxy for symptom severity. 15
Tumor burden was measured according to iRECIST criteria, 20 as the sum of longest measurable diameters (SLD), with a maximum of five lesions in all and two lesions per organ, on the most recent images (brain MRI, PET scan, chest CT) prior to treatment initiation. Tumor lesions were measured along the long axis, and ganglionic lesions along the short axis. As interpretations of tumor burden tend to vary between investigators,21–25 measurements were independently performed in a blinded manner by two experienced clinicians (M.L. and C.G.), and the mean of the two measurements was retained. When measurements disagreed by more than 10%, a third measurement was performed jointly by both clinicians. In the absence of previously defined thresholds, the SLD was defined as high if it exceeded the median sum of the largest lesions in the study population, which was about 100 mm, a value previously used by Katsurada et al. 17 Similarly, for the LLD, the largest lesion was defined as large if it exceeded the median LLD in the study group (60 mm). These thresholds were selected based on the study data prior to any further analysis and were therefore not guided by the efficacy data of the corresponding subgroups.
Tumor symptom severity was evaluated using ECOG performance status scores26,27 and need for corticosteroid therapy.1,28,29 A high ECOG performance status and prescription of corticosteroids are indicative of symptomatic disease and unfavorable prognosis. Corticosteroid therapy has also been associated with reduced pembrolizumab efficacy when administered alone. 30
Studied parameters
The main endpoint was overall survival, defined as the time between treatment initiation and death. The primary analysis assessed overall survival depending on treatment received and tumor aggressiveness: SLD ⩾100 mm, LLD ⩾60 mm, ECOG-1, and need for corticosteroid therapy. Secondary analyses assessed overall survival depending on treatment received in the entire study group and in subgroups stratified by number of metastatic sites, 31 PDL1 tumor proportion scores (⩾90% vs <90% 32 ), and age.
In addition, we described real-world progression-free survival (PFS), which was defined as the time elapsed between treatment initiation and occurrence of the first among the following events: clinically evaluated tumor progression or death; toxicity evaluated using the common terminology criteria for adverse events; imaging progression according to iRECIST criteria 20 as assessed by attending clinicians. Due to the lack of a consensus definition for hyperprogressive disease and the absence of repeated imaging prior to the start of pembrolizumab, which is necessary to assess both tumor growth rate and tumor growth kinetics, 33 we used a pragmatic approach and retained the occurrence of hyperprogressive disease according to the attending clinicians’ conclusions in the presence of major progressive disease at the first disease evaluation along with clinical deterioration.
Statistical analysis
Qualitative variables were summarized as counts and percentages. Quantitative variables were described as mean and standard deviation when normally distributed, and as median and interquartile range (IQR) otherwise. Ordinal data were handled as continuous variables in multivariate analyses, assuming proportional scaling. SLD, LLD, and PDL1 were dichotomized based on the above-mentioned thresholds to allow subgroup analyses.
Overall survival outcomes were evaluated using the Kaplan–Meier method, with RMST analysis 34 to account for the non-proportionality of hazards in immunotherapy studies.1,35 Results were expressed as differences in RMST with 95% confidence intervals (95% CIs). The cutoff time point tau was defined as the shortest of the two groups’ maximum observation times.
Overall survival differences between treatment arms in the study group were also analyzed using a Cox proportional hazard model to assess the impact of model choice on results. The results of the Cox model were expressed as hazard ratios (HRs) and corresponding 95% CIs, with a log-rank test for statistical significance.
Analyses were adjusted for ECOG performance status, patient age, histology, PDL1 tumor proportion score, presence or not of brain metastases, and number of metastatic sites. Analyses of secondary endpoints were also adjusted for tumor burden. The α value was set at 0.05 for all analyses. Missing data were handled by available-case analysis.
We estimated that at least 80 survival events would be required to achieve sufficient statistical power for the primary outcome, corresponding to 160 patients based on an estimated survival rate of 50% for the first 2 years of follow-up.
All analyses were performed in R version 4.1.3. (R Foundation for Statistical computing).
Regulatory framework
Because this study only involved a retrospective analysis of medical records, no ethics committee approval was required under French law. A data protection report was submitted to the Data Protection Delegate of Lille University Hospital under the n°DEC23-056, in agreement with MR004 (CNIL Reference Methodology 004). In keeping with French law, patients were informed by letter that their anonymized data would be used for research purposes unless they objected.
This manuscript was written in accordance with the STROBE guidelines for observational studies (STROBE statement in the Supplemental Material).
Results
Patients and treatments
In all, 1017 patients received either pembrolizumab alone or Pembrolizumab-CT during the study inclusion period, among whom 953 were excluded (746 because they had PDL1 tumor proportion score <50%, 122 because they had an ECOG performance status ⩾2, and 85 because they had received pembrolizumab alone without being offered the choice of Pembrolizumab-CT, which was considered indicative of frailty). Among the 164 included patients, 96 (58.5%) had received pembrolizumab alone and 68 (41.5%) had received Pembrolizumab-CT.
The mean age was 63.7 ± 9.6 years, and 67.7% of patients (n = 111) were men. Most patients had stage IV NSCLC (150 patients, 91.5%) and had an ECOG performance status of 1 (118 patients, 72%). Forty-six patients (28%) had brain metastases: 23 (23.9%) in the pembrolizumab monotherapy group and 23 (33.8%) in the Pembrolizumab-CT group. Ninety-seven patients (59.1%) had more than two metastatic sites (Table 1). Ninety-three patients (56.7%) had PDL1 tumor proportion scores ⩾90%, 56 (58.3%) in the pembrolizumab monotherapy group, and 37 (45.6%) in the Pembrolizumab-CT group.
Baseline characteristics were compared between patients in the pembrolizumab alone and pembrolizumab plus chemotherapy groups.

Results are reported as in n (%), mean ± standard deviation, or median (IQR).
MET, EGFR insertion exon 20, HER2, Nras, STK11.
IQR, interquartile range.
The mean SLD was 107 ± 50 mm, 114 ± 52 mm in the pembrolizumab monotherapy group, and 96 ± 46 mm in the Pembrolizumab-CT group.
The mean LLD was 59 ± 29 mm in the pembrolizumab group and 54 ± 25 mm in the Pembrolizumab-CT group. Corticosteroids (⩾10 mg/day) had been prescribed at the start of treatment in 19 patients (19.8%) in the pembrolizumab group and 16 patients (23.5%) in the Pembrolizumab-CT group.
Efficacy
Overall survival
The mean follow-up time in the study group was 32.9 months (IQR, 18.1–49.8 months). Median overall survival was 20.5 months (95% CI, 12.9–27.6 months) in the pembrolizumab group and 17 months (95% CI, 15.1–not reached) in the Pembrolizumab-CT group. Sixty-two patients in the pembrolizumab monotherapy group (64.5%) and 36 patients in the Pembrolizumab-CT group (52.9%) died during follow-up.
After adjusting for confounders, the RMST was significantly shorter in the Pembrolizumab-CT group than in the pembrolizumab monotherapy group (RMST difference, −7.9 months, 95% CI, −15.1 to −0.7 months; p = 0.03; Figure 1). By contrast, the Cox proportional hazards model adjusted for the same covariates found no significant difference in survival between these two groups (HR, 1.5; 95% CI, 0.9–2.5; p = 0.11; Figure S1).

Adjusted restricted mean overall survival times of patients treated with pembrolizumab alone and pembrolizumab plus chemotherapy (entire study group).
The RMST was non-significantly shorter in the Pembrolizumab-CT group than in the pembrolizumab monotherapy group among patients with SLD ⩾ 100 mm (RMST difference, −1.1 months; 95% CI, −10.5 to 8.3 months; p = 0.82) and among patients with SLD < 100 mm (RMST difference, −3.1 months; 95% CI, −13.0 to 6.9 months; p = 0.54; Figure 2).

Restricted mean overall survival times (RMSTs) compared between patients treated with pembrolizumab alone and pembrolizumab plus chemotherapy in the listed subgroups.
The RMST was significantly shorter in the Pembrolizumab-CT group among patients with LLD <60 mm (RMST difference, −8.6 months; 95% CI, −16.7 to −0.5 months; p = 0.04) but did not differ significantly between the two treatment groups in patients with LLD >60 mm (RMST difference, 0.9 months; 95% CI, −5.8 to 8.1 months; p = 0.75).
The RMST did not differ significantly between treatment groups among patients with a need for corticosteroid therapy (RMST difference, −2.9 months; 95% CI, −14.3 to 8.6, p = 0.62).
The RMST was significantly shorter in the Pembrolizumab-CT group than in the pembrolizumab monotherapy group among patients with an ECOG performance status of 0 (RMST difference, −12.3 months; 95% CI, −14.9 to −9.8; p < 0.001) but did not differ significantly between the two groups among patients with an ECOG performance status of 1 (RMST difference, −6.1; 95% CI, −13.8 to 1.5; p = 0.12).
No difference in survival times between treatment groups was observed when patients were stratified by the number of metastatic sites (Figure 2).
The RMST was significantly shorter in the Pembrolizumab-CT group among patients with PDL1 tumor proportion scores <90% (RMST difference, −8.9 months; 95% CI, −17.0 to −0.8 months, p = 0.03), but did not differ significantly between the two treatment groups among patients with PDL1 tumor proportion scores ⩾90% (RMST difference, −2.8 months, 95% CI −11.48 to 5.9 months, p = 0.53).
Response rate and progression-free survival
The median PFS was 6 months (95% CI, 4.3–9.7 months) in the pembrolizumab group as a whole and 9.7 months (95% CI, 7.0–14.7 months) in the Pembrolizumab-CT group.
The objective response rate was 48.5% (33 patients) in the Pembrolizumab-CT group and 30.2% (29 patients) in the pembrolizumab monotherapy group (Table 2). Two patients (2.1%) in the pembrolizumab group had hyperprogression compared with none in the Pembrolizumab-CT group.
Tumor response at first follow-up under treatment.

Results are reported as n (%).
Adverse events
Thirty-six patients (21.9%) died before the first follow-up imaging visit. For 12 of these patients (3 in the Pembrolizumab-CT group and 9 in the pembrolizumab monotherapy group), the cause of death was listed as probable disease progression. For six patients, the cause of death could not be clearly determined. Eight deaths (five in the Pembrolizumab-CT group and three in the pembrolizumab monotherapy group) were due to infectious complications (including four documented cases of febrile neutropenia).
Toxicity-related hospitalizations seemed more frequent in the Pembrolizumab-CT group (14/68 patients, 20.5%) than in the pembrolizumab monotherapy group (12 patients, 12.5%; Figure 3).

Adverse events (all grades) in patients treated with pembrolizumab alone and pembrolizumab plus chemotherapy.
Furthermore, 27 patients in the Pembrolizumab-CT group (39.7%) and 5 patients in the pembrolizumab monotherapy group (5.2%) had grade 3 or higher adverse events (Table 3). The most frequent grade ⩾ 3 adverse event in the Pembrolizumab-CT group was neutropenia.
Adverse events in patients who received pembrolizumab alone or Pembrolizumab-CT.
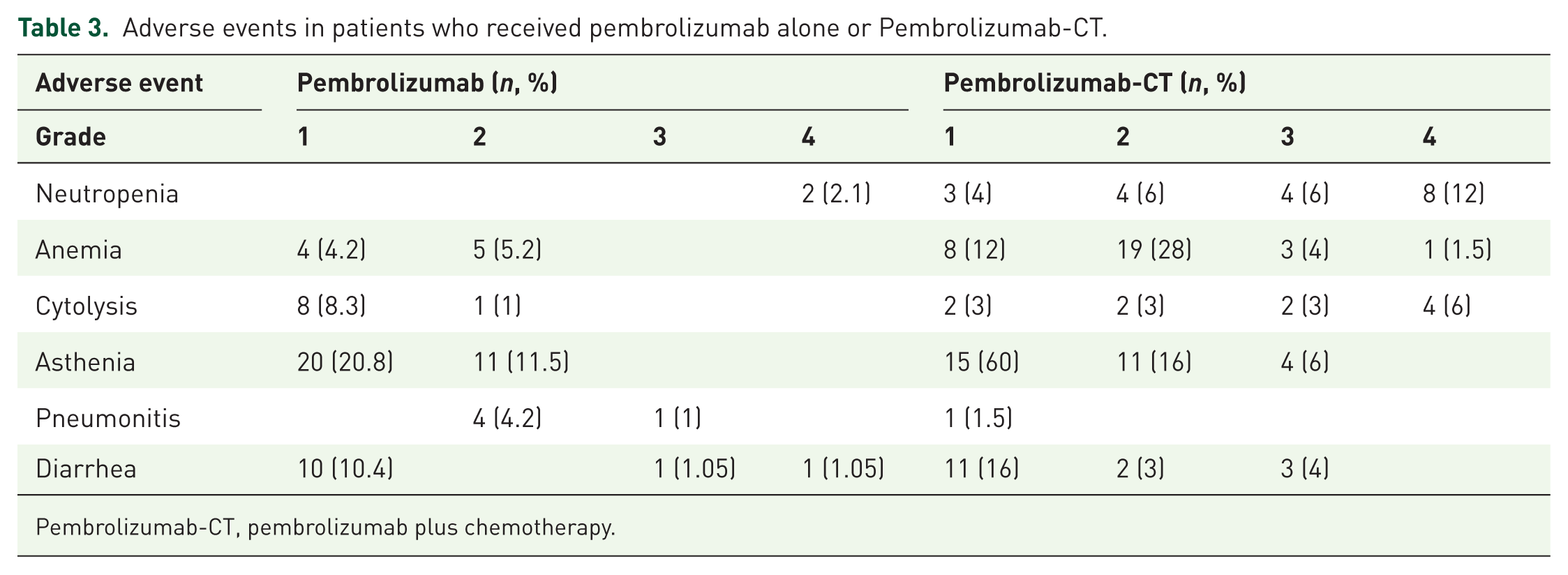
Pembrolizumab-CT, pembrolizumab plus chemotherapy.
Seventeen patients (25%) in the Pembrolizumab-CT group and 15 (15.6%) in the pembrolizumab group stopped their treatment because of adverse events (Figure 3). The only adverse event that was more frequent in the pembrolizumab monotherapy group was pneumonitis, which occurred in 5.2% of patients in the pembrolizumab monotherapy group compared with 1 patient (1.4% of patients) in the chemotherapy plus pembrolizumab group (Figure 3).
Discussion
The main result of this study is that Pembrolizumab-CT was not associated with improved survival compared with pembrolizumab alone in the subgroup of patients with PDL1 ⩾ 50% advanced NSCLC whose disease was classified as aggressive. Furthermore, in the study group as a whole, regardless of tumor aggressiveness, pembrolizumab alone was associated with improved survival compared with Pembrolizumab-CT in RMST analysis.
Disease aggressiveness was defined according to the proposed criteria mentioned in ASCO guidelines, based on tumor burden, LLD, and symptom severity deduced from the ECOG performance status scores and need for corticosteroid therapy. In the absence of consensus thresholds, we used the median value for the quantitative factors (SLD and LLD). These criteria may seem restrictive as the aggressiveness of NSCLC presents with heterogeneous characteristics that are difficult to capture entirely. Although increased lactate dehydrogenase (LDH) levels have been shown to correlate with poor prognosis in lung cancer, 36 we could not assess disease severity according to LDH levels in our study because these levels were not routinely sampled in our study population. Similarly, weight loss is associated with decreased survival in advanced NSCLC, 37 and it would have been interesting to analyze this subgroup. However, due to significant missing data, such an analysis could not be reasonably performed.
Evidence supporting the use of Pembrolizumab-CT to treat PDL1 ⩾ 50% advanced NSCLC comes from studies suggesting that high tumor burden may be associated with reduced response to pembrolizumab.17,32,38 The rationale is that reducing the tumor burden with chemotherapy should promote the action of pembrolizumab. In our patients with high tumor burdens, however, RMSTs did not differ between those treated with pembrolizumab alone and those who received Pembrolizumab-CT. This apparent disagreement with previous findings can be explained by the fact that our study group consisted exclusively of patients with PDL1 tumor proportion scores ⩾ 50% (unlike previous studies). It could also be that a small underlying difference was not detected in the present study because of a small sample size. Furthermore, in the absence of a standard definition of tumor burden, we used a threshold SLD of 100 mm, 17 close to the median value in the study group, but published data on this question vary,16,38,39 and no threshold value has been validated. Note also that the RECIST 1.1 criteria for evaluating tumor burden do not encompass certain cancer presentations, such as carcinomatous lymphangitis or bone metastases, 40 which may lead to an underestimation of tumor burden in some patients.
The presence of a large lesion is associated with shorter survival and a less favorable response to immunotherapy. 16 Although the underlying physiopathology is unclear, several hypotheses have been proposed to account for this association, including production of cytokines favoring inflammatory growth and increased aggressiveness due to the presence of a poorly vascularized hypoxic zone in the center of the tumor, with lower antigen exposure. 41 These mechanisms suggest that combining pembrolizumab with chemotherapy may be beneficial in these patients. Our data do not support this hypothesis, however, as they show no superiority of chemotherapy plus pembrolizumab over pembrolizumab alone in patients with LLD ⩾ 60 mm. Furthermore, chemotherapy plus pembrolizumab was associated with shorter survival than pembrolizumab alone in patients with LLD < 60 mm. This may be because smaller tumors are less aggressive,16,17 tipping the benefit/risk balance in favor of pembrolizumab monotherapy.
Another finding of this study is the absence of association between systemic corticosteroid therapy (⩾10 mg/day) at the start of treatment and any difference in survival between the pembrolizumab and Pembrolizumab-CT groups. Note, however, that these subgroups were particularly small for this analysis. As reported previously, however,42,43 pembrolizumab was less effective in patients receiving systemic corticosteroid therapy. Although this effect has been reported to be less pronounced in patients receiving Pembrolizumab-CT, 44 our data do not support the addition of chemotherapy in these cases, albeit with the limitations of small sample size.
In patients with ECOG-1 performance status, overall survival did not differ between pembrolizumab monotherapy and Pembrolizumab-CT groups, possibly because the greater toxicity of chemotherapy counterbalances its beneficial effects.
Note that since the aggressiveness of NSCLC can be defined based on other criteria than those used here (e.g., respiratory failure, rapid weight loss), the better response rate associated with Pembrolizumab-CT in studies of NSCLC irrespective of PDL1 expression status1,2 may lead clinicians to offer patients with PDL1 ⩾ 50% advanced NSCLC this treatment option, even though no association with longer survival has been found in our study or elsewhere for this particular type of NSCLC.8,45 The absence of a consensus definition of aggressiveness in NSCLC complicates the identification of aggressive subgroups in clinical studies.
It is also important to note that the subgroup analyses performed in this study only assessed each variable separately, whereas patients with aggressive NSCLC may have several of these characteristics (e.g., high tumor burden and altered performance status). We did not specifically compare patients who did not meet any of the criteria for disease aggressiveness to patients who met one or more of these criteria, whose response to pembrolizumab could possibly be less important due to the accumulation of poor-prognosis factors.
In the study group as a whole, patients who received Pembrolizumab-CT had shorter overall survival than those who received pembrolizumab alone. This result is contrary to those of previous studies of PDL1 ⩾ 50% NSCLC, which have tended to find no difference between the two treatment strategies. A pooled analysis 8 of randomized trial data comparing pembrolizumab and Pembrolizumab-CT in these patients found no difference in survival between the two groups. PFS was slightly longer in the Pembrolizumab-CT group; however, PFS does not evaluate the entire therapeutic sequence and thus does not account for the potential later benefit of second-line chemotherapy. 46 Elsewhere, an observational study focusing exclusively on non-squamous NSCLC 45 found no difference in overall survival between patients treated with Pembrolizumab-CT and pembrolizumab alone, and a third retrospective, single-center study 47 likewise found no difference in overall survival between pembrolizumab monotherapy and Pembrolizumab-CT, albeit with a very small sample size. Another retrospective study 38 focusing on patients with SLD ⩾ 50 mm receiving either chemotherapy plus immune checkpoint inhibitors (ICIs) or ICIs alone found better survival in the chemotherapy plus ICIs group overall, but found no difference between the two treatment groups among patients with PDL1 tumor proportion scores ⩾ 50%.
Our findings, in contrast with previous studies, show that the combination of chemotherapy plus pembrolizumab has a harmful effect on overall survival compared with pembrolizumab alone, while both response rates and PFS seem superior in the combination group. While this may seem counterintuitive, it is important to note that both the response rate and PFS were descriptive analyses with no statistical testing or adjustment for confounding factors. Therefore, direct comparison should be avoided. In addition, response rates and PFS rarely correlate with overall survival when comparing two treatment sequences (concomitant Pembrolizumab-CT or sequential pembrolizumab followed by chemotherapy). 46 Furthermore, the decreased survival in the Pembrolizumab-CT group is likely not due to poor antitumor activity but rather, it can be explained in light of the more frequent adverse events in the pembrolizumab chemotherapy group, notably those of grade ⩾3 events requiring hospitalization or discontinuation of treatment. Accordingly, overall survival may be impacted by toxicity beyond progressive disease and, therefore, without impacting PFS. Note also that some prognostic factors, such as brain metastases, PDL1 expression, and ECOG performance status, were slightly unbalanced between the groups, although we did not test the statistical significance of these differences to limit alpha inflation and because of little interest in such systematic tests. Furthermore, these differences were adjusted for in all the multivariate analyses. Smoking status was not adjusted for because of the very small number of non-smoking patients. It was therefore unlikely to have affected the results. However, our multivariate analysis adjusted for only a few prognostic variables and did not account for other possible prognostic factors that could have influenced treatment choices. Therefore, unmeasured selection bias remains possible and could explain the decreased survival observed under chemotherapy plus pembrolizumab.
Another important result of our study is that Cox analysis did not identify any difference in survival outcomes between the treatment groups, in keeping with previous studies, but contrary to our main RMST analysis. This is probably because RMST analysis was able to account for early events, such as hyperprogression and early toxicity-related death, whose impact is underestimated by proportional hazards analysis because their effects vary over time. These results highlight the importance of the choice of statistical model used and of accounting for non-proportional hazards, as described previously in immunotherapy studies. 14
The poor tolerance of chemotherapy observed in this real-world study is notable in comparison with those reported in randomized controlled trials for these treatments, where events leading to discontinuation of treatment were twice as less frequent (13%).1–3 Unfortunately, these trials did not report unscheduled hospitalization, which is likely to occur less frequently in clinical trials because they have restrictive inclusion/exclusion criteria. These criteria result in the inclusion of fewer frail patients than those treated in a real-world setting.48,49 This raises questions about the results’ extrinsic validity. Previous observational studies of pembrolizumab and chemotherapy in NSCLC have not reported tolerance data45,47 and our study is the first to address this issue. The excess of severe adverse events in the Pembrolizumab-CT group may account for some of the relative benefit of pembrolizumab monotherapy observed here. Differences in tolerance profiles between clinical trials and real-world settings should be considered when choosing the best treatment options for unselected PDL1 high NSCLC patients.
In summary, given the absence of a randomized trial directly comparing these two treatments in patients with PDL1 ⩾ 50% advanced NSCLC, the findings of the present study, the greater risk of adverse events associated with Pembrolizumab-CT,1,3 and the higher cost of this combination therapy, choosing Pembrolizumab-CT over pembrolizumab alone in patients with PDL1 ⩾ 50% advanced NSCLC does not seem justified.
The ongoing phase III PERSEE trial 50 aims to determine the best treatment strategy for these patients, but questions will very likely remain in specific situations.
One justification for the use of Pembrolizumab-CT may have been the perceived risk of hyperprogression under pembrolizumab alone.9–11,41 There were only two documented cases of hyperprogression in this study, both in the pembrolizumab group. However, there is no consensus definition of hyperprogression, 33 and our approach to recording it may have underestimated its incidence. In addition, approximately 20% of patients in both groups died before the first follow-up imaging visit and therefore could not be assessed for hyperprogressive disease. Thus, these data are insufficient to conclude the impact of chemotherapy plus pembrolizumab on preventing hyperprogressive disease. Nevertheless, given the similar early death rates in both groups, these results suggest that chemotherapy does not reduce the risk of early death in these patients, possibly due to higher toxicity as previously mentioned.
The strengths of our study include its multicenter design, including university hospitals, as well as general hospitals and private clinics. Recall bias was limited by using lists of patients exposed to the different treatments. Confounding was limited by adjustment in multivariate analysis. We used RMST analysis to account for non-proportional hazards, and between-observer variability in tumor burden assessments was limited by performing independent blinded analyses of imaging data.21–25
The main limitations of the study are its retrospective nature and the associated missing data, which reduced the sample size for multivariate analysis. Furthermore, due to the limited sample size, our multivariate analysis was only adjusted for a limited number of prognostic variables and did not account for other prognostic factors that might have influenced treatment choice. It is also important to note that, although proper matching through propensity score or inverse probability of treatment weighting is recommended in order to account for the lack of randomization, we were unable to use these techniques because they are difficult to implement on RMST analyses. Therefore, they are not routinely combined despite recent work suggesting this as a possible option. 51 Consequently, confounding bias or potential unmeasured selection bias remains possible in our study. In addition, subgroup analyses were limited by the small sample size, and therefore may be underpowered to accurately assess differences between groups, which could lead to false negative results. Besides, the number of aforementioned subgroup analyses results in alpha risk inflation and should be interpreted with this limitation in mind. The results should be considered as exploratory only.
Conclusion
In this group of NSCLC patients with PDL1 tumor proportion scores ⩾50%, chemotherapy plus pembrolizumab was associated with shorter survival than pembrolizumab alone under RMST analysis. Adding chemotherapy to pembrolizumab did not seem to improve survival compared with pembrolizumab alone in cases of aggressive disease or high tumor burden, but analyses were subject to limited sample size. Identifying new predictive markers of pembrolizumab response, with or without chemotherapy, or predictive of hyperprogression under pembrolizumab, should facilitate the selection of patients liable to benefit from one or other of these treatment strategies.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251396266 – Supplemental material for Pembrolizumab versus pembrolizumab plus chemotherapy in patients with PDL1 ≽50% advanced non-small-cell lung cancer, depending on tumor aggressiveness and clinical impact
Supplemental material, sj-docx-1-tam-10.1177_17588359251396266 for Pembrolizumab versus pembrolizumab plus chemotherapy in patients with PDL1 ≽50% advanced non-small-cell lung cancer, depending on tumor aggressiveness and clinical impact by Marine Lejeune, Eric Wasielewski, Vincent Leroy, Soraya Bordier, Arnaud Scherpereel, Alexis Cortot and Clément Gauvain in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
