Abstract
Background:
Systemic corticosteroids (SCs) are associated with reduced survival in patients with advanced non-small-cell lung cancer (NSCLC) receiving immune checkpoint inhibitor (ICI) monotherapy. However, the current first-line standard of care usually involves combined chemotherapy (CT) and ICIs, and the effect of SCs on survival under combined CT and ICI has never been studied.
Objectives:
To investigate the association between SC therapy and survival under CT-ICI in advanced-stage NSCLC patients.
Design:
We performed a multicenter retrospective cohort study of all advanced-stage NSCLC patients receiving first-line CT-ICI.
Methods:
The primary endpoint was progression-free survival (PFS) according to SC exposure status (⩾10 mg/day), adjusted in a multivariate Cox model for the following confounders: age, performance status, hospital admission prior to treatment, number of metastatic sites, brain metastases, bone metastases, PD-L1 status, and histological subtype. Multivariate analyses also explored the association between dosage and SC exposure duration and PFS.
Results:
Of the 193 included patients, 43 (22.3%) were receiving SCs, mainly because of symptomatic brain metastases (in 25/43 cases, 58%). In multivariate analysis, SC therapy at a 10 mg/day threshold was not associated with PFS (hazard ratio (HR) = 1.25, 95% confidence interval (CI) 0.77–2.03, p = 0.35). However, SC dose was negatively associated with PFS (HR = 1.08 per 10 mg/day increment, 95% CI 1.01–1.16, p = 0.01) especially at doses ⩾60 mg/day (HR = 3.27 per 10 mg/day increment, 95% CI 2.01–5.35, p < 0.001). Duration of SC therapy was not associated with PFS (HR = 0.97, 95% CI 0.81–1.15, p = 0.71), but SC therapy ⩾4 weeks prior to CT-ICI was associated with shorter PFS (HR = 1.07, 95% CI: 1.01–1.14, p = 0.028).
Conclusion:
In this group of patients receiving first-line CT-ICI for advanced NSCLC, SCs at ⩾60 mg/day were associated with shorter PFS, but lower doses were not. Prolonged SC therapy prior to CT-ICI was associated with shorter PFS. Larger studies are required to confirm these results.
Keywords
Introduction
In the absence of oncogene addiction, the management of advanced-stage non-small-cell lung cancer (NSCLC) involves administering either immune checkpoint inhibitors (ICIs; anti-PD1/PD-L1 antibodies) plus platinum-doublet chemotherapy (CT)1,2 or ICIs alone to patients with PD-L1 expression ⩾50%. 3
Because of their mechanism of action, however, ICIs can be affected by systemic corticosteroids (SCs), whose multiple immunosuppressive effects are well known.4–6
Since lung cancers are often diagnosed at an advanced stage, it is not uncommon for patients to be symptomatic from the outset of metastatic disease. Thus, around 25% of NSCLC patients require anti-inflammatory corticosteroid therapy,7–9 mainly in cases of symptomatic brain or bone metastases, 10 or because of dyspnea secondary to carcinomatous lymphangitis. 11
Patients receiving more than 10 mg prednisone equivalents daily have been excluded from trials of ICI monotherapy and CT-ICI combination therapy.1–3,12,13 Several observational studies have found decreased survival, under ICI monotherapy, of patients receiving SCs at >10 mg/day upon ICI initiation, particularly for indications related to lung neoplasia,7–9,14 although the lack of adjustment, in some studies,8,14 for the presence of brain metastases is important to bear in mind given its negative prognostic impact. 15
The effect of SCs in patients receiving current standard of care CT-ICI is unknown, however. This effect may be weaker than in patients receiving ICIs alone because chemotherapy may counter the corticosteroid-induced increase in regulatory T lymphocytes4,5 by decreasing their proportion 16 and creating an immunogenic peritumor environment.17,18 Furthermore, the antitumor action of chemotherapy (immunogenic cell death) may provide a time window in which to decrease SCs to less immunosuppressive dosages.
Finally, SC dose–effect and duration–effect relationships for ICI and CT-ICI efficacy have yet to be studied.
The main objective of the present study was to determine whether prolonged SC administration at ⩾10 mg/day upon initiation of first-line CT-ICI altered the progression-free survival (PFS) of advanced-stage NSCLC patients. Secondary objectives were to investigate potential dose- or duration-deleterious effects in the association between SC therapy and clinical outcomes under CT-ICI.
Methods
Patients
We conducted a multicenter retrospective cohort study at four hospitals in France. Patients who received at least one injection of CT-pembrolizumab between April 2019 and December 2021 were identified with the chemotherapy prescription software of the different hospitals.
Patients were included if they had been diagnosed with stage III NSCLC not amenable to radiotherapy or stage IV NSCLC, and if they had received at least one injection of CT-pembrolizumab (the only CT-ICI treatment available in France at the time) as first-line therapy. Patients were included regardless of the presence of driver alterations. Patients with targetable oncogenic addiction in first-line care (EGFR, ALK, ROS1) were included if they had received CT-ICI after progression under treatment with a tyrosine kinase inhibitor (TKI). Patients with any other oncogenic addiction were included in the first line of treatment.
Patients who received one or two lines of chemotherapy prior to CT-ICI were excluded, except if they had received adjuvant chemotherapy or radio-chemotherapy more than 6 months earlier. Patients with prior exposure to an ICI (e.g., durvalumab after radio-chemotherapy) were included only if they had not displayed tumor progression under that treatment. Patients who received local treatment for oligometastatic disease were excluded. All data were obtained retrospectively from patient medical records.
Exposure to SCs
Patients who had received ⩾10 mg prednisone equivalents/day SC therapy for at least 10 successive days prior to initiation of CT-ICI were considered exposed to SCs, regardless of SC indication. Patients who had received daily corticoid doses <10 mg prednisone equivalents, or who received SCs after CT-ICI initiation were considered not exposed to SCs.
Treatment modalities and follow-up
Patients were treated with carboplatin (AUC 6)—paclitaxel (200 mg/m²)—pembrolizumab (200 mg) in case of epidermoid NSCLC, and with cisplatin (75 mg/m²) or carboplatin (AUC 5)—pemetrexed (500 mg/m²)—pembrolizumab (200 mg), according to local practice, for non-epidermoid NSCLC. Injections were performed every 3 weeks after clinical and biological follow-up.
SC doses were re-evaluated every 3 weeks during CT-ICI administration visits, and the rate of SC dose decrease was determined by attending clinicians. SC doses were generally decreased in increments of 10 mg every 7–15 days provided the symptoms for which they had been prescribed did not recur.
Tumor monitoring involved thoracoabdominal CT and brain imaging (CT or MRI). When no progression was observed after the first four induction cycles, maintenance was carried out with pemetrexed (500 mg/m²)—pembrolizumab (200 mg) every 3 weeks for non-epidermoid NSCLC, and with pembrolizumab (200 mg) every 3 weeks for epidermoid NSCLC, for a maximum of 2 years or until unacceptable toxicity or progression was observed. Adverse events were recorded when they reached or exceeded grade 3.
Endpoints
The study aimed to determine whether SCs affect advanced-stage NSCLC patient outcomes under first-line CT-ICI. The primary endpoint was PFS, defined as the time between the first CT-ICI infusion and either tumor progression or patient death, whichever happened first. PFS was evaluated locally by attending clinicians (real-world PFS). The secondary endpoints were the associations with SC dosage, SC exposure duration under CT-ICI and duration of pre-treatment SC exposure as predictors, and PFS, overall survival (OS), response rate to treatment, and toxicity as outcomes. The radiological response was evaluated with RECIST 1.1 criteria.
Statistical analyses
Continuous variables were summarized as mean ± standard deviation (SD) when normally distributed, and as median with interquartile range (IQR) otherwise. Categorical variables were expressed as numbers and percentages. Continuous variables were not dichotomized and were assumed to follow a linear relationship with PFS in the principal multivariate model. However, the associations between PFS and SC dosage and duration of pre-exposure to SCs were modeled with restricted cubic splines with three break points. Pertinent thresholds were then determined by visual inspection of the corresponding curves. Hazard ratios (HR) were then determined for each dose or time increment unit inside intervals around these thresholds.
The associations with PFS of SCs, SC dosage, and SC therapy duration prior to treatment were studied with multivariate Cox proportional-hazards models. The association with PFS of the duration of exposure under CT-ICI was studied with a time-dependent multivariate Cox model to control for immortal time bias. 19 All analyses were multivariate, with adjustment for the following confounders: age, Eastern Cooperative Oncology Group (ECOG) performance status, hospital admission prior to treatment, number of metastatic sites, brain metastases, bone metastases, PD-L1 status, and histological subtype. We estimated that at least 90 PFS events would be required to achieve sufficient statistical power for these analyses. We estimated that about 180 patients would be required based on an estimated PFS rate of 50% per year of follow-up. Missing data were handled by available-case analysis.
Given the likely different effects of ICIs in patients with oncogene addictions (EGFR, ALK), a sensitivity analysis was performed with these patients excluded.
HRs are reported with 95% confidence intervals (95% CIs). Results were considered statistically significant at a two-sided p-value <0.05.
Ethics
Because this study only involved a retrospective analysis of medical records, no ethics committee approval was required under French law. A data protection report was submitted to the Data Protection Delegate of Lille University Hospital under the no. DEC23-095, in agreement with MR004 (CNIL Reference Methodology 004). In keeping with French law, patients were informed by letter that their anonymized data would be used for research purposes unless they objected.
This manuscript was written in accordance with the STROBE guidelines for observational studies 20 (STROBE statement in Supplemental Material).
Results
Patients
Two hundred ninety-one patients who had received pembrolizumab–CT were identified and 193 patients were included. The 98 other patients were excluded either because they had received at least one previous line of treatment (n = 79), had oligometastatic disease with subsequent local treatment by radiation therapy (n = 13), or carried an EGFR mutation but had received a CT-ICI injection before their EGFR status was known, and TKI treatment thereafter (n = 6; Figure S1).
Patient baseline characteristics are presented in Table 1. The mean age was 62.3 ± 8.5 years; 70.5% of patients were male, 92.2% were active or past smokers, and 85.5% had an ECOG performance status of 0 or 1.
Baseline characteristics of included patients.
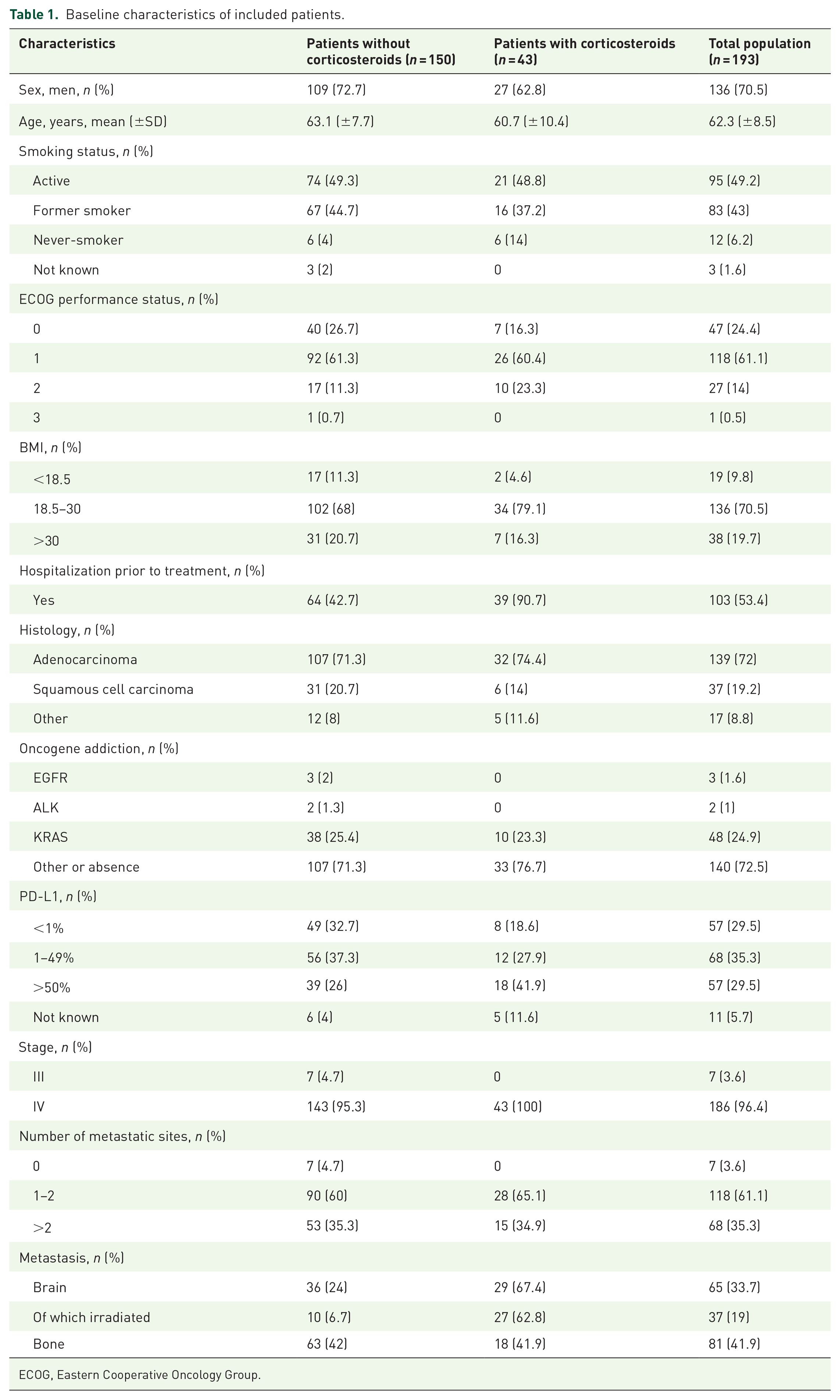
ECOG, Eastern Cooperative Oncology Group.
Almost all the NSCLCs were metastatic (96.4%) and more than two-thirds were adenocarcinomas (72%). Oncogene addiction was found in 28.7% of patients, with KRAS mutation the most frequent (25.4%). The level of tumor PD-L1 expression was <1% in 29.5% of patients, between 1% and 49% in 35.3% of patients, and ⩾50% in 29.5% of patients.
Forty-three patients (22.3% of the study group) were receiving SCs at ⩾10 mg prednisone equivalents/day at the start of CT-ICI, in all but three cases (40, 93%) for the treatment of cancer-related symptoms. The main indication for SCs was the presence of symptomatic brain metastases (25 patients, 58% of indications). Only 7% of SC indications were unrelated to lung cancer. The median duration of SC exposure was 13.3 weeks (IQR, 7.2–21 weeks). The mean duration of exposure to SCs prior to CT-ICI was 5.7 ± 4.5 weeks (Table 2).
Details regarding corticosteroid therapy.

SD, standard deviation.
Six of the 43 patients receiving SCs were non-smokers (14%), 10 (23%) had an ECOG performance status of 2, and 18 (42%) had high PD-L1 expression levels (⩾50%). All but four of the patients receiving SCs (91%) had been hospitalized prior to initiation of CT-ICI. Twenty-nine of the patients receiving SCs (67%) had brain metastases and 27 (63%) required radiotherapy. Other variables were similar in the two patient groups (Table 1).
PFS and association with SC therapy
One hundred thirty-nine PFS events including 89 deaths occurred during follow-up. The median follow-up of event-free patients was 10.3 months (IQR, 7.8–13 months).
The median PFS was 5.9 months (95% CI, 5.1–7.4 months) overall. The median PFS was 6.8 months (95% CI, 5.5–8.5 months) among patients who did not receive SCs and 4.1 months (95% CI, 2.7–5.4 months) among patients who were receiving SCs.
After adjusting for confounders, there was no statistically significant difference in PFS between the SC and no-SC groups (HR = 1.25, 95% CI 0.77–2.03, p = 0.35; Figure 1). The same result was obtained in a sensitivity analysis excluding ALK and EGFR patients (HR = 1.31 in the SC vs no-SC group, 95% CI 0.91–2.13, p = 0.27).

Progression-free survival curves and corresponding confidence intervals for corticosteroid-treated and non-corticosteroid-treated groups (using the 10 mg/day threshold). Results were adjusted in multivariate analyses for WHO performance status, age, pre-treatment hospitalization history, number of metastatic sites, brain metastases, bone metastases, PD-L1 status, and histology.
However, higher corticosteroid doses were associated with shorter PFS (HR = 1.08 per 10 mg/day increment, 95% CI 1.01–1.16, p = 0.01; Figure 2). The dose–effect relationship was nonlinear with an apparent threshold for increased risk around 60 mg/day (Figure 3(a)). Analyses accordingly showed no association with PFS for doses of 10–60 mg/day (HR = 1.01 per 10 mg/day increment, 95% CI 0.65–1.57, p = 0.96) but a significant association with PFS for doses of 60–120 mg/day (adjusted HR = 3.27 per 10 mg/day increment, 95% CI 2.01–5.35, p < 0.001) and for doses ⩾120 mg/day (HR = 3.85 per 10 mg/day increment, 95% CI 2.16–6.85, p < 0.001; Figure 2). This nonlinear relationship was observed in patients both with and without brain metastases (Figure 3(a)).
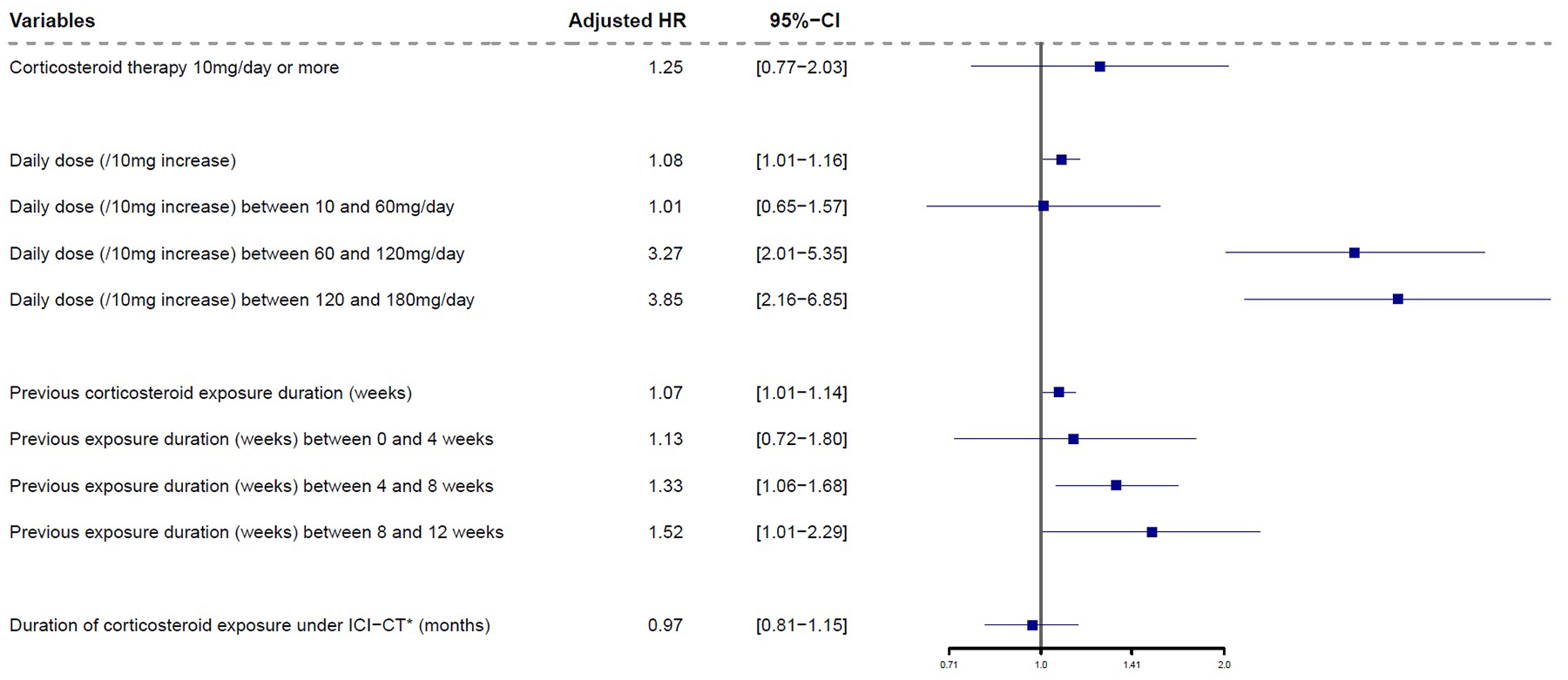
Forest plot showing the effect of systemic corticosteroid therapy >10 mg/day, dose, cumulative duration, and pre-exposure duration on progression-free survival. Hazard ratios were adjusted in multivariate analyses for WHO performance status, age, pre-treatment hospitalization history, number of metastatic sites, brain metastases, bone metastases, PD-L1 status, and histology.

Relationships between corticosteroid dose and excess risk of progression or death (a) and between duration of corticosteroid pre-exposure and excess risk of progression or death (b) according to the presence of brain metastases. Curves were adjusted in multivariate analyses on WHO performance status, age, number of metastatic sites, bone metastases, PD-L1 status, and histology.
Duration of SC therapy during CT-ICI was not associated with PFS (HR = 0.97 per additional month under SC, 95% CI 0.81–1.15, p = 0.71; time-dependent Cox model, Figure 2).
By contrast, the duration of SC exposure prior to CT-ICI was significantly negatively associated with PFS (HR = 1.07 per additional week of SC exposure, 95% CI 1.01–1.14, p = 0.028; Figure 3(b)), after adjustment for confounders. The relationship between prior SC exposure time and PFS was nonlinear, with a potential threshold of about 4 weeks since pre-exposure to SCs <4 weeks was not associated with shorter PFS (HR = 1.13 per week of SC exposure, 95% CI 0.72–1.80, p = 0.59), but SC exposures of 4–8 weeks (HR = 1.33 per week of SC exposure, 95% CI 1.06–1.68, p = 0.014) and 8–12 weeks (HR = 1.52 per week of SC exposure, 95% CI 1.01–2.29, p = 0.047) were (Figure 2). This nonlinear relationship was observed in patients both with and without brain metastases (Figure 3(b)).
OS and association with SC therapy
The median OS of the entire study group was 15.1 months (95% CI, 13.2–16.7 months).
The median OS of patients who did not receive SCs was 15.5 months (95% CI, 13.7–22.2 months), and the median OS of patients who received SCs was 14.5 months (95% CI, 5.8 months–not reached). Multivariate analysis adjusted for the same confounders as for PFS revealed no significant difference in OS between these two groups (HR = 1.43 in the SC vs no-SC group, 95% CI 0.81–2.52, p = 0.22).
Response rate at first tumor response evaluation
Seventy-three patients in the no-SC group (50%) and 16 patients in the SC group (37%) had an objective response at the first tumor response evaluation (Table S1).
Toxicities
Twenty-one patients (10.9%) developed febrile neutropenia (15 patients in the no-SC group (10%) and 6 patients in the SC group; Table 3). Twenty-three patients in the no-SC group (15.3%) and nine patients in the SC group (20.8%) had grade ⩾3 immune-related adverse events (irAEs). These adverse events were cutaneous in 11 cases (5.7% of the entire study group), pulmonary (immune-induced diffuse interstitial lung disease) in 7 cases (one patient in the SC group and six in the no-SC group, 3.6% of the entire study group), and thyroid-related in 5 cases (2.6% of the study group; Table 4).
Results of first disease assessment according to RECIST 1.1.

Toxicities of chemoimmunotherapy in included patients.

irAE, immune-related adverse event.
Discussion
In this group of advanced NSCLC patients receiving CT-ICI in a real-world setting, the confounder-adjusted analysis did not reveal any significant difference in PFS between patients receiving SCs at ⩾10 mg/day and those receiving no SCs or less than 10 mg/day SCs.
It is notable, however, that the PFS observed in our study group was shorter than the values observed in the KEYNOTE-407 2 et KEYNOTE-189 1 studies. This could be because patients in these studies were selected based on performance status and did not have brain metastases (or at least no treated brain metastases), whereas ours were recruited in a real-world clinical setting.
To our knowledge, our study is the first to evaluate the association between SC therapy and NSCLC patient outcomes under CT-ICI. These results go against the majority of those obtained for the effect of SCs in patients receiving ICIs alone.7–9,14 Our study group is nevertheless similar to those of previous studies, notably regarding the proportion of patients receiving SCs,7,8 with the exception of Skribek et al.’s, 9 which included a higher proportion of patients receiving SCs, albeit in some cases to treat an irAE. Exposure to SCs was likewise defined in the same way as previously.7–9,14
Some studies seem to suggest that SCs only have a negative effect on patient outcomes when used for palliative indications.8,14 This was the case for the vast majority of patients in our study group, but the association with SC indication could not be analyzed because too few patients were receiving CSs for non-cancer-related symptoms. It is notable however that after adjustment for confounders, and despite a predominance of palliative indications for SCs, exposure to SCs at ⩾10 mg/day was not associated with shorter PFS in our study group. The lack of association is thus not due to non-palliative indications for SC.
The seemingly different effects of SCs on CT-ICI and ICI monotherapy may result from the direct tumoricidal and anti-tumor immunological effects of CT: decreased immunoregulatory cells (notably regulatory T lymphocytes), immunogenic cell death, and increased expression of PD-L1. This could explain the synergistic effects of CT and ICI16,17,21–23 and may explain why this combination remains effective even alongside SCs, except at doses above 60 mg/day. It is also possible that the anti-tumor action of CT provides tumor control in the time required to decrease SC doses.
It is also important to note that some previous studies did not adjust for the presence of brain or bone metastases8,14 and/or PD-L1 expression in their analyses.7,14 In our study, most SC-exposed patients were receiving SCs for lung-cancer-related indications, associated with poorer prognosis.8,14 Accordingly, in comparison with patients in the no-SC group, these patients seemed to have been hospitalized more often prior to CT-ICI initiation, with a lower performance status and more brain metastases. It is thus possible that the results obtained for SCs in the context of ICI monotherapy are at least partly due to non-adjustment for these confounders and to possible differences in the proportion of patients exposed to high SC doses.
Nevertheless, although exposure to SCs ⩾10 mg/day was not associated with shorter PFS in these patients under CT-ICI, those who received higher SC doses or were exposed to SCs for longer before CT-ICI did have shorter PFS. This brings into question the relevance of the 10 mg/day threshold typically used as a trial exclusion criterion1,2 and the absence of conditions regarding the duration of pre-exposure, notably for patients receiving CT-ICI. The observed dose–deleterious effect relationship was not linear (no effect between 10 and 60 mg/day but a significant effect above that range) and the fact that it was observed after adjustment for confounders, notably regardless of the presence of brain metastases, suggests it does not merely reflect disease aggressiveness. A threshold of around 60 mg/day may therefore be more appropriate but to our knowledge, this has never been investigated: existing studies all consider the effects of SCs ⩾10 mg/day and not the dose–deleterious effect relationship.7–9,14 Most available data on the effects of SCs concern rheumatic diseases and suggest that corticosteroid-induced immunosuppression, notably in terms of opportunistic infections, is mainly linked to the dose administered,24,25 with higher doses typically used for aggressive inflammatory or auto-immune disease. 26 This is also the case for ICI toxicities, for which higher doses are recommended for grade-3 or grade-4 irAEs.27,28 One therefore supposes that the degree to which SCs antagonize ICIs depends in large part on corticosteroid dosage, with relatively higher thresholds under CT-ICI.
In patients receiving CT-ICI, longer pretreatment exposure to SC was non-linearly associated with shorter PFS, the association only becoming significant for pretreatment exposures ⩾4 weeks. By contrast, the cumulative duration of SC therapy under CT-ICI was not associated with PFS in time-dependent Cox analysis controlling for immortal time bias. A possible explanation for these results is that corticosteroid-induced lymphocyte apoptosis depends on the dosage and the duration of exposure. 29 Furthermore, data on anti-SARS CoV2 vaccination have highlighted the effects of prior immunosuppression on the immunogenicity of antigens in patients receiving immunosuppressants. Accordingly, both EULAR and ACR recommend interrupting immunosuppressant administration during vaccination periods (if possible without compromising later resumption) and vaccinating before introducing any immunosuppressant treatment.30,31 In the context of lung cancers moreover, SCs introduced to treat toxicity after the start of ICI therapy have been shown to have no effect on survival. 32 This supports the idea that pre-treatment exposure to SCs is a stronger prognostic factor than later exposure. However, the biological rationale in the context of anti-tumor immunotherapy remains unclear. This finding could also be explained by multiple testing resulting in alpha inflation and false-positive statistical significance. However, the fact that the duration of exposure under CT-ICI was not associated with PFS may also be due to the concurrent tapering in SC doses in our patients. Data suggest that the risk of infection under SCs remains low at doses not exceeding 10 mg/day, even when treatment is prolonged. 24 How SC duration alone affects the risk of infection is unclear. 25
The two groups of patients did not differ in terms of the frequencies of CT-linked toxicity or irAEs. The overall frequency of irAE in our study is lower than in the princeps studies,1,2 but this was probably due to shorter PFS (shorter exposure to ICI) and possible underestimation of irAEs in retrospective real-life data.
The strengths of our study include its relatively large size, which allowed multivariate analysis and precise estimates, and its multicenter design, which limited treatment-center bias and ensured the study group was representative of advanced NSCLC patients in general. The small number of exclusion criteria ensured good extrinsic validity and adjusting for major confounders made the results more interpretable. Furthermore, this study is the first, to our knowledge, to have analyzed the association between SCs and PFS under CT-ICI and the dose–deleterious effect and duration–deleterious effect relationships of SCs under CT-ICI. Nonlinear modeling revealed the complexity of these relationships and raised the question of new inclusion thresholds, notably regarding corticosteroid doses. Lastly, the use of a time-dependent Cox model limited immortal time bias.
The main limitations of this study are its retrospective nature and the lack of multiple imputation for missing data. The date at which SC therapy was stopped was not always precisely recorded in medical records. Furthermore, while it appears that patients receiving antibiotic or proton pump inhibitor treatments have a diminished response to immunotherapy,33–35 information on these treatments was not collected. Since patients receiving SCs were presumably more likely to have been exposed to proton pump inhibitors and antibiotics, this may have led to omitted confounders. Other potentially important variables that were not included in the models include comorbidities, which can alter survival, co-interventions such as early palliative care, which is known to be associated with longer survival, 36 and radiation therapy, which may also increase survival. 37
Another limitation of our study is the inclusion of patients with an EGFR mutation, treated by CT-ICI upon progression after, or intolerance to, TKI treatment. Our study was conducted before the publication of the KEYNOTE-789 study, which showed that CT-ICI was not beneficial in these patients. 38 However, excluding patients with an EGFR mutation from the analysis did not change our results, indicating that our findings were not biased by the inclusion of these patients.
Two final limitations are (i) that we considered SC doses at the start of CT-ICI in our analyses without accounting for the reductions in doses that generally occurred during follow-up and (ii) that PFS was estimated from medical records (real-world PFS, necessarily subjective), rather than according to an objective metric.
The practical implications of our findings are that they suggest that SCs at less than 60 mg/day SCs should not be considered a contraindication for CT-ICI in advanced NSCLC patients, nor should these patients be excluded from clinical trials, especially since SC doses are usually reduced rapidly during treatment. On the other hand, our results suggest other antitumor treatments should be proposed to patients receiving higher SC doses (⩾60 mg/day) when no decrease in SC doses is expected, and to reserve immunotherapy for possible later-line treatment once corticosteroid weaning has been achieved. Larger prospective studies are required to confirm our results and identify the highest corticosteroid dose that does not alter the effects of CT-ICI.
Conclusion
In this group of patients receiving first-line CT-ICI for advanced-stage NSCLC, SCs at ⩾10 mg/day were not associated with shorter PFS. Higher doses, in particular ⩾60 mg/day, were associated with shorter PFS, as was prolonged (⩾4 weeks) pre-treatment exposure to SCs.
Larger prospective studies are required to confirm these findings and optimize therapeutic strategies for advanced NSCLC patients receiving SCs.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251318160 – Supplemental material for Impact of corticosteroids on the efficacy of first-line pembrolizumab plus chemotherapy in patients with advanced non-small-cell lung cancer
Supplemental material, sj-docx-1-tam-10.1177_17588359251318160 for Impact of corticosteroids on the efficacy of first-line pembrolizumab plus chemotherapy in patients with advanced non-small-cell lung cancer by Amytis Roboubi, Eric Wasielewski, Soraya Bordier, Amélie Turlotte, Geoffrey Pavaut, Arnaud Scherpereel, Alexis Cortot and Clément Gauvain in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
