Abstract
Background:
Despite significant advancements in treatment, patients with human epidermal growth factor receptor 2 (HER2)-positive metastatic breast cancer (MBC) continue to face the challenge of drug resistance, highlighting the need to develop new drugs and treatment strategies. Pyrotinib has exhibited notable efficacy in managing HER2-positive MBC, especially in individuals with brain metastasis (BM). However, real-world evidence on the effectiveness of pyrotinib re-treatment remains limited.
Objectives:
This study aims to assess the efficacy of pyrotinib re-treatment—including treatment beyond progression (TBP) and rechallenge—in patients with HER2-positive MBC, particularly those with BMs, in a real-world clinical setting.
Design:
A retrospective, multicenter, real-world study.
Methods:
Patients with HER2-positive MBC who experienced progression during pyrotinib treatment and subsequently continued or resumed pyrotinib-based regimens were enrolled. Patients were divided into two groups according to the re-treatment pattern: the pyrotinib TBP group and the pyrotinib rechallenge group. The endpoints were progression-free survival (PFS) and overall survival (OS).
Results:
A total of 226 participants received pyrotinib TBP, whereas 33 received pyrotinib rechallenge. The median PFS of pyrotinib re-treatment (7.2 vs 6.3 months, p = 0.31) and the median OS, measured from the start of pyrotinib re-treatment (31.6 vs 21.0 months, p = 0.062), were comparable between the two groups. Among patients with BMs before pyrotinib re-treatment, the median PFS for pyrotinib re-treatment was 7.2 months, and the median OS was 25.2 months.
Conclusion:
Pyrotinib TBP and pyrotinib rechallenge both demonstrate potential benefits for patients with HER2-positive MBC, including those with BM.
Plain Language Summary
Patients with HER2-positive metastatic breast cancer (MBC), especially those whose cancer has spread to the brain, often face drug resistance after a period of treatment. Pyrotinib is a targeted drug that has shown benefits in treating HER2-positive MBC, but what happens after the cancer progresses on this drug is unclear. In this study, researchers looked at real-world outcomes from 259 patients who continued using pyrotinib even after their cancer had progressed. Some continued the drug immediately (called “treatment beyond progression”), while others paused and then reused it later (called “rechallenge”). The goal was to understand whether these re-treatment strategies still help. Researchers found that both approaches offered meaningful benefits: patients lived a median of 7.2 and 6.3 months without further disease worsening, and had overall survival times of 31.6 and 21.0 months, respectively. These differences were not statistically significant but suggest that continuing or reusing pyrotinib could still help in some cases. Importantly, patients who had brain metastases also benefited, and those who had longer benefit during their first use of pyrotinib were more likely to do well with re-treatment. This study highlights the potential of pyrotinib as a treatment option even after progression, especially in real-world clinical settings. Further research is needed to identify which patients are most likely to benefit.
Keywords
Background
Human epidermal growth factor receptor 2 (HER2) is overexpressed in approximately 20% of breast cancers (BCs). Up to 50% of patients with HER2-positive metastatic BC (MBC) develop brain metastases (BMs), which significantly contribute to high mortality rates and pose substantial challenges in clinical management.1 –6 Tyrosine kinase inhibitors (TKIs), antibody–drug conjugates (ADCs) such as trastuzumab emtansine (T-DM1) and T-Dxd, along with monoclonal antibodies like trastuzumab and pertuzumab, have shown antitumor activity in the brain.7 –11 The HER2CLIMB trial, which demonstrated the efficacy of tucatinib in patients with BMs, 12 while pooled analyses from DESTINY-Breast01/02/03 13 and findings from the DESTINYBreast-12 trial 14 further confirmed the robust intracranial activity of T-DXd. However, the clinical need for standard care options after progression during anti-HER2 therapy remains unmet.
Over the past few decades, clinical evidence has indicated that consistent anti-HER2 therapy can lead to sustained patient benefits. Moreover, multiple studies have reported favorable responses to re-treatment with the same drug. 15 Re-treatment with trastuzumab has demonstrated clinical benefits in patients who progressed on prior trastuzumab-based therapies.16 –22 In addition, switching to alternative TKI after progression during TKI treatment has shown clinical efficacy.10,23 –29
Pyrotinib is an irreversible, pan-HER receptor TKI that targets HER1, HER2, and HER4. 30 It was approved in China in 2018 and has since been incorporated into treatment guidelines for HER2‑positive MBC. In the first‑line setting, the recommended options are trastuzumab, pertuzumab, and taxane (THP) or pyrotinib combined with taxane plus trastuzumab, the latter being particularly considered for patients with BMs. In the second‑line or later settings, the preferred regimen is T‑DXd, with pyrotinib plus capecitabine as an alternative option. The results from the PHOEBE, 31 PHENIX, 32 and PHILA 33 studies have demonstrated the efficacy of pyrotinib in patients with HER2-positive MBC. The PERMEATE study 34 and our previous real-world studies28,35 have shown the activity of pyrotinib against BMs. Several studies have shown that patients previously treated with lapatinib still benefited from subsequent pyrotinib treatment.23 –25,27 –29
In this context, we conducted a multicenter study to evaluate the efficacy of pyrotinib re-treatment, including treatment beyond progression (TBP) and drug rechallenge, in patients with HER2-positive MBC and BMs. This study aimed to provide further evidence supporting pyrotinib re-treatment and to expand treatment options for these patients.
Methods
Patient eligibility
This retrospective, multicenter, real-world study was conducted by the Second Xiangya Hospital of Central South University in Hunan Province, China. We analyzed data from 259 patients with MBC who received pyrotinib for TBP or were rechallenged with the drug between August 3, 2018, and November 15, 2023, across 17 institutions in China, including the Second Xiangya Hospital of Central South University; Hunan Cancer Hospital; the First People’s Hospital of Xiangtan City; Central South University Third Xiangya Hospital; Yiyang Central Hospital; the First People’s Hospital of Changde; Chenzhou No. 1 People’s Hospital; the First Affiliated Hospital of University of South China; the Central Hospital of Yongzhou; the Affiliated ZhuZhou Hospital of Xiangya School of Medicine Central South University; Hunan Provincial People’s Hospital; Loudi Central Hospital; the First Hospital Hunan University of Chinese Medicine; the First Affiliated Hospital of Gannan Medical University; Ganzhou People’s Hospital; Jiangxi Cancer Hospital; and Xiangya Hospital of Central South University.
The inclusion criteria were as follows: (i) patients aged over 18 years; (ii) patients with a confirmed histological diagnosis of HER2-positive MBC, defined as immunohistochemistry results of category 3+ or positive results of fluorescence in situ hybridization 36 ; and (iii) patients who either continued or resumed pyrotinib-based therapy after experiencing progression during initial pyrotinib treatment. The exclusion criteria were as follows: (i) the use of pyrotinib as neoadjuvant or adjuvant therapy; (ii) the presence of other tumors; (iii) a diagnosis of inflammatory BC; and (iv) incomplete medical records. No restrictions were placed on the number of prior systemic or local treatment regimens for metastatic disease. No restrictions were placed on participant sex during enrollment. However, due to the low incidence of HER2-positive BC in males,37,38 all enrolled participants were female. Given the retrospective and single-arm design of the study, randomization was not applicable.
Pyrotinib TBP was defined as the ongoing use of pyrotinib after disease progression, while pyrotinib rechallenge was defined as patients switching to non-pyrotinib regimens after disease progression before resuming pyrotinib therapy (Figure 1). Treatment-free interval (TFI) was defined as the period from the termination of initial pyrotinib treatment to the beginning of re-treatment.39,40

Pyrotinib treatment regimens: detailed pathways for pyrotinib TBP and rechallenge therapies.
To be classified as trastuzumab sensitive, patients were either diagnosed with de novo stage IV BC without prior exposure to anti‑HER2 therapy, or experienced recurrence more than 12 months after completing trastuzumab‑based neoadjuvant or adjuvant therapy. First-line treatment refers to the initial therapy received by patients after the diagnosis of MBC, with subsequent therapies classified as second-line and third-line treatments.
Procedures
As recommended by the guidelines, pyrotinib was administered at a clinically recommended dose of 400 mg orally once daily. Progression was assessed by clinicians on the basis of imaging test results, including ultrasound and enhanced magnetic resonance imaging (MRI)/computed tomography (CT). Enhanced MRI/CT was performed for intracranial lesions, while ultrasound and enhanced MRI/CT were performed for extracranial lesions. As this was a retrospective real-world study, the timing of progression assessments was not predefined and was determined by the treating investigators based on clinical practice. Clinical data were manually extracted from electronic medical records by trained researchers. To ensure data accuracy, a second researcher independently verified the records, and discrepancies were resolved through discussion.
Endpoints
The primary endpoint of this study was progression-free survival (PFS). PFS was defined as the duration from the first oral dose of pyrotinib to either disease progression or death according to the Response Evaluation Criteria in Solid Tumors version 1.1 (RECIST 1.1) criteria. PFS1 referred to the initial pyrotinib treatment, while PFS2 referred to pyrotinib re-treatment. Disease progression was defined as progression in either intracranial or extracranial lesions.
The secondary endpoint of this study was overall survival (OS). OS was defined as the duration from the first oral dose of pyrotinib re-treatment to death from any cause. OS1 represented the duration from the first oral dose of initial pyrotinib treatment to death from any cause.
Statistical analysis
Categorical variables across different patient groups were compared using Pearson’s χ2 test and Fisher’s exact test. Continuous variables with non-normal distributions were analyzed using the non-parametric Mann–Whitney U test. Kaplan–Meier survival curves were employed for estimating PFS and OS, with group differences assessed by the log-rank tests. Statistical analyses and visualizations were performed using R software (version 4.3.3, https://www.r-project.org/, RRID:SCR_001905). A two-sided p value < 0.05 was considered statistically significant.
To minimize potential bias, strict adherence to the clearly defined inclusion and exclusion criteria for patient selection ensured the representativeness within the study population. Patients with incomplete baseline data or unavailable follow-up information were excluded from the analysis. In addition, all data underwent cross-verification against clinical records for accuracy and consistency.
Reporting guideline
The reporting of this study conforms to the Strengthening the reporting of observational studies in epidemiology (STROBE) statement 41 (Supplemental File).
Results
Baseline characteristics
Among 259 female participants, 226 received pyrotinib TBP therapy while 33 underwent pyrotinib rechallenge (Figure 2). Dose reductions occurred in 9% (24/259) of patients, adjusted at the treating physician’s discretion from 400 to 320 mg and subsequently to 240 mg. During re-treatment with pyrotinib, 186 patients received pyrotinib combined with different chemotherapy regimens compared with their initial treatment, 57 received pyrotinib combined with another anti-HER2 agent, and 16 received pyrotinib combined with endocrine therapy, 57 with another anti-HER2 agent, and 16 with endocrine therapy. A total of 128 were diagnosed with hormone receptor (HR)-positive MBC, 24 of whom received a CDK4/6 inhibitor in addition to endocrine therapy, with the CDK4/6 inhibitor administered concurrently with pyrotinib. After the diagnosis of MBC, 113 patients received pyrotinib as first-line treatment, including 54 who were trastuzumab sensitive. In addition, 89 patients initiated pyrotinib as second-line treatment, 37 as third-line, and 20 as later-line therapy. The median TFI in the rechallenge group was 231 days, with an interquartile range (IQR) of 104–440 days. The baseline characteristics and p values for comparisons between the pyrotinib TBP group and the rechallenge group are presented in Table 1. No significant differences were observed in the baseline data between the two groups.
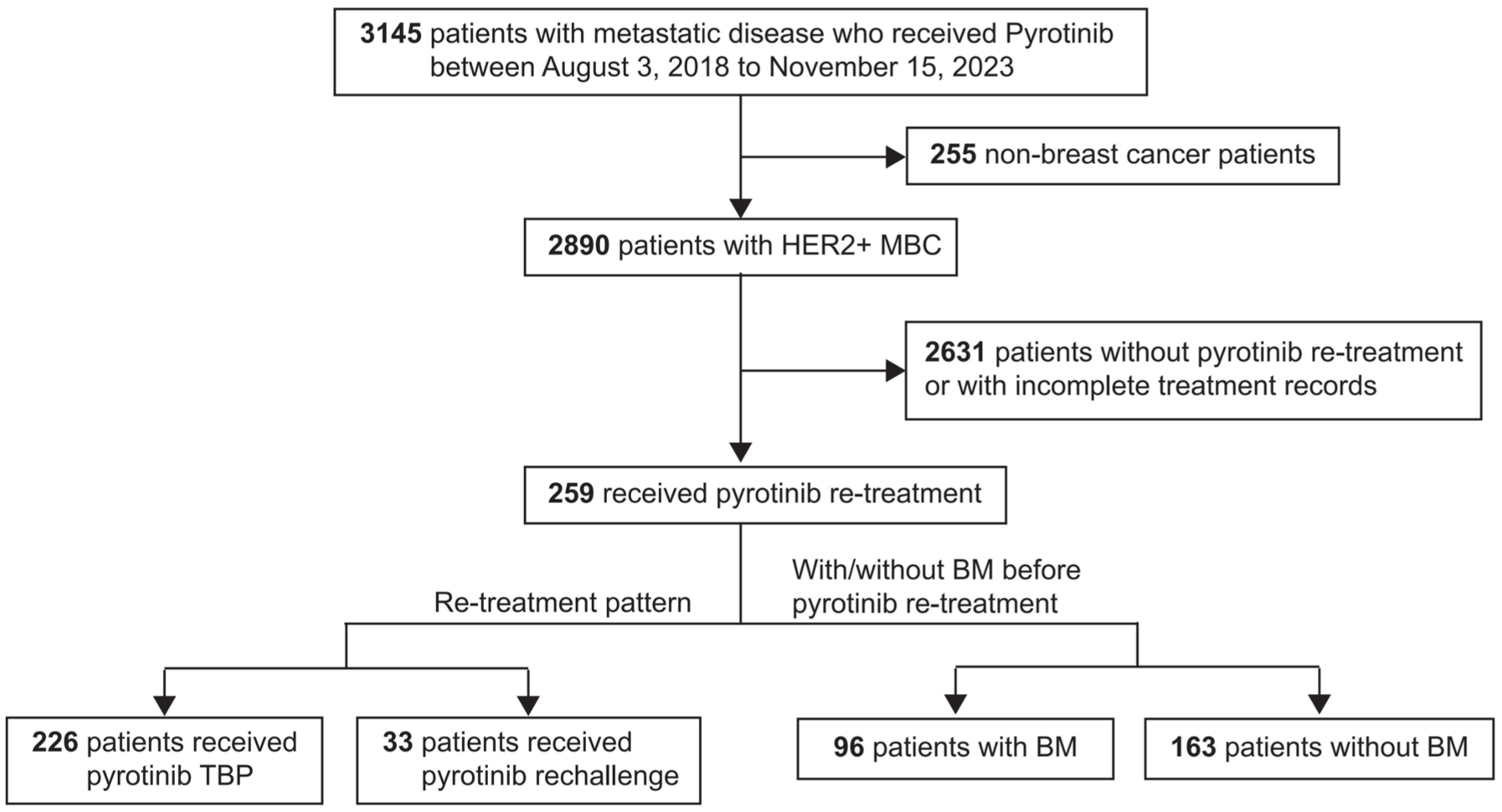
Study design: overview of patient selection and treatment pathways.
Baseline characteristics of patients undergoing pyrotinib TBP and rechallenge.

From Fisher’s exact test.
Anti-HER2 treatment before initial pyrotinib treatment for metastatic disease. Some patients received more than one anti-HER2 treatment.
Sites of metastasis that occurred during the entire course of treatment. Some patients developed more than one metastasis.
BM, brain metastasis; ER, estrogen receptor; HER2, human epidermal growth factor receptor 2; HR, hormone receptor; PR, progesterone receptor; TBP, treatment beyond progression; T-DM1, trastuzumab emtansine.
Efficacy of pyrotinib TBP and rechallenge in all patients
The median follow-up time was 36.9 months. Disease progression occurred in 229 patients (88%) during pyrotinib re-treatment, and 113 patients (44%) died during the overall follow-up period. Among patients treated with pyrotinib TBP and rechallenge, the median PFS1 was 11.7 months (CI: 10.2–13.2) and 9.8 months (8.3–15.0), with no significant difference observed (p = 0.08; Supplemental Figure 1(A)). For PFS2, the median durations were reported as 7.2 months (6.2–8.3) and 6.3 months (5.1–10.1), respectively (p = 0.31; Figure 3(a)). No significant differences were observed between the two groups for either PFS1 or PFS2. A trend toward improved OS was noted in the TBP group, with a median OS of 31.6 months (26.3–not available [NA]), compared to 21.0 months (14.2–NA) in the rechallenge group. Although this difference was not statistically significant (p = 0.062; Figure 3(b)). The median OS for all patients was 30.1 months (25.4–NA; Supplemental Figure 1(B)). HR-positive and HR-negative patients exhibited no statistically significant variations in the median PFS2 (p = 0.19; Supplemental Figure 2(A)) and OS (p = 0.24; Supplemental Figure 2(B)).

Kaplan–Meier curves for survival outcomes. (a) PFS2 and (b) OS for all patients who received TBP/rechallenge with pyrotinib.
Efficacy of pyrotinib TBP and rechallenge for patients with BMs
Among patients with BMs, 72 of them developed BMs before pyrotinib treatment, while 24 developed BMs during or after their initial pyrotinib treatment, resulting in 96 patients developing BMs before pyrotinib re-treatment. The baseline characteristics of patients with and without BMs before the initial pyrotinib treatment are presented in Table S1.
In patients with BMs before initial pyrotinib treatment, the median PFS2 and OS were 7.6 months (5.8–9.1; Figure 4(a)) and 25.2 months (19.6–NA; Figure 4(b)), respectively. At the time of pyrotinib re-treatment, 96 patients had BMs, with a median PFS2 of 7.2 months (5.7–8.5; Figure 4(c)), and the median OS was 25.2 months (21.0–NA; Figure 4(d)). Among all enrolled patients, 105 were eventually diagnosed with BMs, with a median OS of 26.1 months (23.5–NA; Supplemental Figure 3(A)).

Kaplan–Meier curves for survival outcomes. (a) PFS2 and (b) OS for patients with BMs before initial pyrotinib treatment. (c) PFS2 and (d) OS for patients with BMs before pyrotinib re-treatment.
Among 96 patients diagnosed with BMs prior to pyrotinib re-treatment, 58 received local treatment targeting the BM lesions (either surgery or radiation) in addition to pyrotinib, while 38 did not. Comparing the survival outcomes between these two groups revealed no significant differences in PFS2 (p = 0.54; Figure 5(a)) or OS (p = 0.19; Figure 5(b)).

Kaplan–Meier curves for survival outcomes. (a) PFS2 and (b) OS in patients with BMs prior to pyrotinib re-treatment, comparing those who received local treatment with those who did not.
Optimal candidates for pyrotinib TBP and rechallenge
In the subgroup analysis, 259 participants were divided into two groups based on the median PFS1 of 11.5 months to evaluate comparative treatment outcomes. Among patients with a shorter PFS1, the median PFS2 was 7.1 months (5.6–8.4), whereas for those with a longer PFS1, it was 7.6 months (6.2–9.3). No significant difference in PFS2 was observed between the two groups (p = 0.22; Figure 6(a)). However, a significant difference in OS1 was identified, with patients having longer PFS1 demonstrating an OS1 of 60.9 months (50.4–NA) compared to an OS1 of 37.0 months (33.5–NA) for those with shorter PFS1 (p = 0.00014; Figure 6(b)). To further assess whether PFS1 correlated with OS1, univariate Cox regression analysis was performed. Among the variables examined, only PFS1 showed a statistically significant association with OS1 (HR = 0.49, 95% CI: 0.33–0.71, p < 0.001; Table S2). As no other variables were significantly associated with OS1, a multivariate model was not constructed. In addition, a modest extension of 2.8 months was noted in the median OS (31.6 months (23.5–NA) vs 28.8 months (25.2–NA); p = 0.85; Supplemental Figure 3(B)).
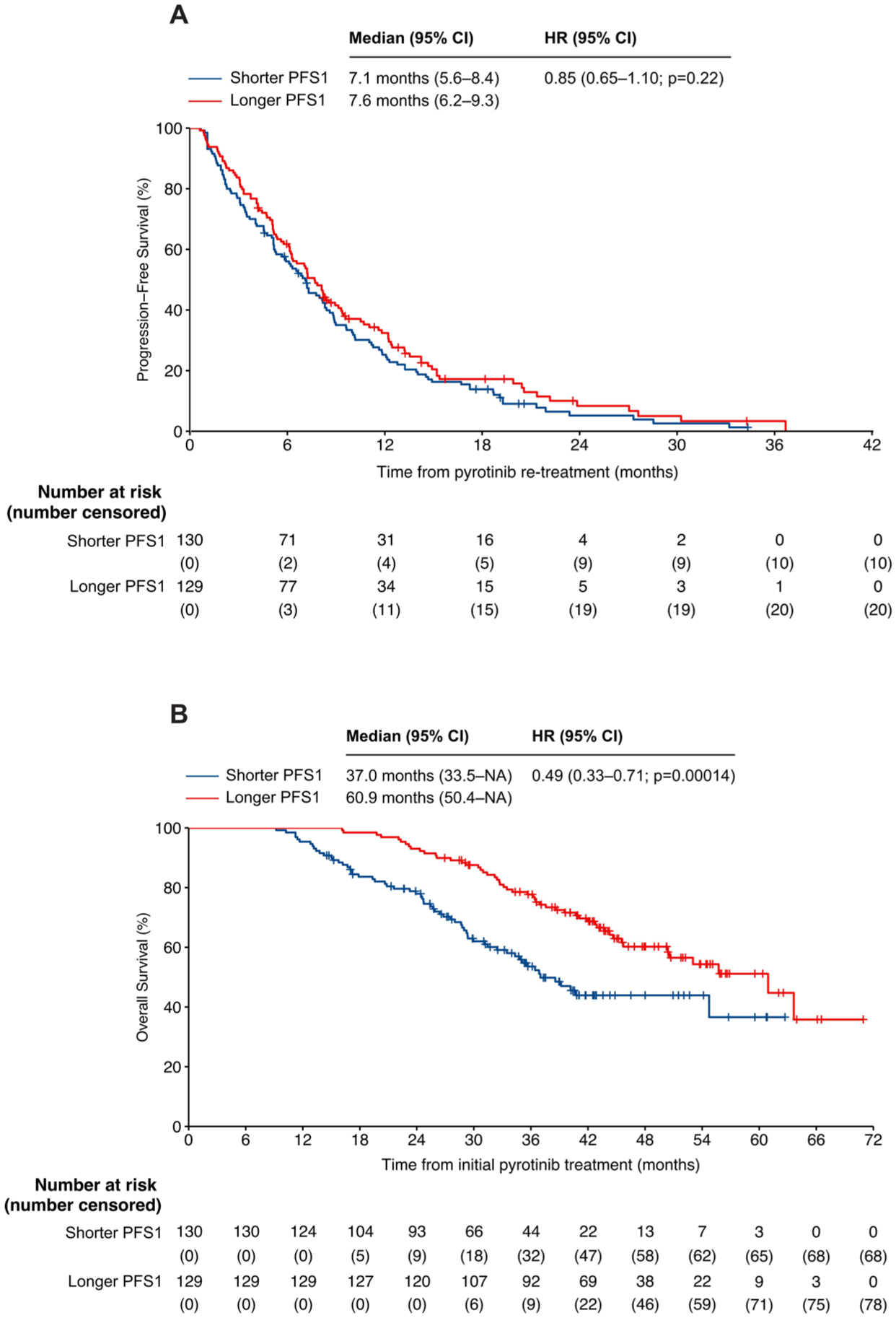
Kaplan–Meier curves for survival outcomes. (a) PFS2 and (b) OS1 for patients stratified by longer and shorter PFS1.
Discussion
The prognosis for patients with HER2-positive MBC has markedly improved owing to the advent of HER2-targeted agents. 42 Despite these advancements, patients with BMs still experience poorer survival outcomes. Pyrotinib, a novel irreversible pan-HER TKI, has been shown to significantly improve the prognosis of patients with BMs.34,35
The classic Goldie–Coldman hypothesis of drug resistance 43 recommends that patients who develop resistance to a specific treatment should switch to a different therapy. However, clinical evidence accumulated over the past decades indicates that patients may still benefit from agents, particularly HER2-targeted agents for BC, despite initial disease progression.18,19,21,22,44 –51 TKIs, such as lapatinib, have been reported to have sustained efficacy upon re-treatment beyond previous disease progression.29,44,52,53 The Trastevere study demonstrated that lapatinib remained efficacious among 73 patients who experienced progression during prior lapatinib-based therapy, with a median PFS and OS of 3.9 and 21.6 months, respectively. 52 Another real-world study reported a median PFS of 4.0 months in 21 patients who were retreated with lapatinib. 44 In addition, the latest data from the DESTINY-Breast02 study (NCT03523585) revealed a median PFS of 4.5 months in patients with HER2-positive unresectable or MBC previously treated with T-DM1 who were treated with trastuzumab plus capecitabine or lapatinib plus capecitabine, compared to 11.3 months in those who received T-Dxd. 54 Moreover, a previous study reported that in patients with HER2-positive MBC who had received prior TKI therapy, the median PFS was 7.0 months with novel ADCs—including 13.0 months with T-Dxd—versus 4.0 months with T-DM1. 55 In our study, we reported a longer median PFS of 7.2 months for patients who received pyrotinib TBP, with a median OS of 31.6 months. Moreover, the pyrotinib rechallenge groups presented a similar PFS2 of (6.3 months, p = 0.31). A direct comparison of these results is summarized in Table S3. Based on the current therapeutic landscape, T-Dxd has demonstrated superior efficacy and is considered the optimal treatment option for HER2-positive MBC previously treated with TKIs. Nevertheless, pyrotinib re-treatment also showed promising efficacy in this setting and may serve as a viable therapeutic alternative.
TKIs have a unique ability to penetrate the blood–brain barrier and have been widely demonstrated to exert favorable therapeutic effects in HER2-positive MBC patients with BMs.56,57 The PERMEATE study reported a median PFS of 10.9 months and a median OS of 35.9 months in radiotherapy-naïve patients,34,58 while a recent study demonstrated that combining radiotherapy with pyrotinib plus capecitabin yielded a superior median PFS of 17.6 months. 59 For TBP in the HER2CLIMB trial, a median PFS2 of 7.6 months was reported among patients with isolated brain progression who received tucatinib‑based TBP after local therapy. 12 Our study extended these findings by demonstrating that among patients with BMs before pyrotinib re-treatment, those who received local treatment in addition to pyrotinib achieved a median PFS2 of 7.2 months (4.7–9.1), which is comparable to the HER2CLIMB result, with differences in study design and treatment context, and a median OS of 23.5 months (17.2–NA). No statistically significant difference in either PFS2 or OS was observed between patients who received local therapy and those who did not. These results suggest that pyrotinib maintains its efficacy in patients with BMs, regardless of whether local treatment for BM lesions is administered. Our findings provide support for further randomized clinical trials and suggest that pyrotinib may continue to be beneficial in the re-treatment of HER2-positive MBC patients, including those with BMs.
The mechanism of drug resistance is always a topic of interest in oncology research. Several hypotheses have been proposed regarding the use of drug TBP and rechallenge. Some researchers suggest that resistance resulting from non-heritable cellular adaptation might be reversed by a drug holiday, whereas others argue that the altered ratio of fast-growing/sensitive to slow-growing/resistant cells leads to tumor regrowth during a drug holiday or intervention therapy, but the cells retain their drug sensitivity upon re-treatment. Moreover, at the time of initial progression, tumors may have developed resistance to the chemotherapy agents rather than to pyrotinib itself; therefore, switching chemotherapy while continuing pyrotinib re-treatment could still be beneficial. 15 Furthermore, patients with longer PFS1 also experienced longer OS1 (60.9 vs 37.0 months, p = 0.00014), which is consistent with results from other studies.60,61 Univariate Cox regression analysis further supported this association, identifying PFS1 as the only variable significantly correlated with OS1. However, as PFS1 is intrinsically a component of OS1 by definition, the observed association may partly reflect this structural relationship rather than an independent predictive effect. Although multivariate modeling was not performed due to the lack of other significant covariates, these findings suggest that longer PFS1 may indicate a more favorable baseline prognosis. Further studies with adjusted analyses are warranted to clarify whether PFS1 can serve as an independent predictor of re-treatment benefit. These findings offer guidance for identifying optimal candidates for pyrotinib re-treatment, although further exploration through prospective trials is necessary.
To our knowledge, this study included the largest sample size and the highest percentage of BM patients among studies focusing on TKI re-treatment for HER2-positive MBC. Furthermore, it represents the most extensive analysis of pyrotinib treatment in patients with BMs. However, several limitations must be considered when interpreting our findings. First, as a multicenter retrospective study, it is susceptible to inherent selection and data collection biases. Second, the lack of a control group is a major limitation. Without a comparator arm, it is difficult to determine whether the observed outcomes were attributable to pyrotinib re-treatment or to baseline patient characteristics, such as disease biology or prior treatment responsiveness. This limitation restricts the ability to draw definitive causal inferences and highlights the exploratory nature of our findings. Third, pyrotinib was approved for marketing in China only in August 2018, resulting in a relatively short follow-up period. Therefore, further high-quality randomized clinical trials are necessary to validate our findings.
Conclusion
In conclusion, pyrotinib re-treatment presents promising treatment strategies for patients with HER2-positive MBC, irrespective of the presence of BMs and the stages of local treatment for BMs.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251387965 – Supplemental material for Treatment beyond progression and rechallenge with pyrotinib in HER2-positive metastatic breast cancer and brain metastases: a multicenter real-world study
Supplemental material, sj-docx-1-tam-10.1177_17588359251387965 for Treatment beyond progression and rechallenge with pyrotinib in HER2-positive metastatic breast cancer and brain metastases: a multicenter real-world study by Jing Peng, Qitong Chen, Liping Liu, Ning Xie, Mingwen Liu, Xuan Liu, Liyuan Qian, Kaili Lu, Tao Wu, Haiqing Xie, Haifan Xu, Zecheng Hu, Sijuan Ding, Wei Zhou, Chaojie Zhang, Yan Li, Jinhui Hu, Ruilian Xie, Jing He, Zhengkui Sun, Feng Xu, Danhua Zhang, Qian Long, Jinfeng Zhu, Xi Chen, Quchang Ouyang, Shouman Wang and Wenjun Yi in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251387965 – Supplemental material for Treatment beyond progression and rechallenge with pyrotinib in HER2-positive metastatic breast cancer and brain metastases: a multicenter real-world study
Supplemental material, sj-docx-2-tam-10.1177_17588359251387965 for Treatment beyond progression and rechallenge with pyrotinib in HER2-positive metastatic breast cancer and brain metastases: a multicenter real-world study by Jing Peng, Qitong Chen, Liping Liu, Ning Xie, Mingwen Liu, Xuan Liu, Liyuan Qian, Kaili Lu, Tao Wu, Haiqing Xie, Haifan Xu, Zecheng Hu, Sijuan Ding, Wei Zhou, Chaojie Zhang, Yan Li, Jinhui Hu, Ruilian Xie, Jing He, Zhengkui Sun, Feng Xu, Danhua Zhang, Qian Long, Jinfeng Zhu, Xi Chen, Quchang Ouyang, Shouman Wang and Wenjun Yi in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-3-tam-10.1177_17588359251387965 – Supplemental material for Treatment beyond progression and rechallenge with pyrotinib in HER2-positive metastatic breast cancer and brain metastases: a multicenter real-world study
Supplemental material, sj-docx-3-tam-10.1177_17588359251387965 for Treatment beyond progression and rechallenge with pyrotinib in HER2-positive metastatic breast cancer and brain metastases: a multicenter real-world study by Jing Peng, Qitong Chen, Liping Liu, Ning Xie, Mingwen Liu, Xuan Liu, Liyuan Qian, Kaili Lu, Tao Wu, Haiqing Xie, Haifan Xu, Zecheng Hu, Sijuan Ding, Wei Zhou, Chaojie Zhang, Yan Li, Jinhui Hu, Ruilian Xie, Jing He, Zhengkui Sun, Feng Xu, Danhua Zhang, Qian Long, Jinfeng Zhu, Xi Chen, Quchang Ouyang, Shouman Wang and Wenjun Yi in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-4-tam-10.1177_17588359251387965 – Supplemental material for Treatment beyond progression and rechallenge with pyrotinib in HER2-positive metastatic breast cancer and brain metastases: a multicenter real-world study
Supplemental material, sj-jpg-4-tam-10.1177_17588359251387965 for Treatment beyond progression and rechallenge with pyrotinib in HER2-positive metastatic breast cancer and brain metastases: a multicenter real-world study by Jing Peng, Qitong Chen, Liping Liu, Ning Xie, Mingwen Liu, Xuan Liu, Liyuan Qian, Kaili Lu, Tao Wu, Haiqing Xie, Haifan Xu, Zecheng Hu, Sijuan Ding, Wei Zhou, Chaojie Zhang, Yan Li, Jinhui Hu, Ruilian Xie, Jing He, Zhengkui Sun, Feng Xu, Danhua Zhang, Qian Long, Jinfeng Zhu, Xi Chen, Quchang Ouyang, Shouman Wang and Wenjun Yi in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-5-tam-10.1177_17588359251387965 – Supplemental material for Treatment beyond progression and rechallenge with pyrotinib in HER2-positive metastatic breast cancer and brain metastases: a multicenter real-world study
Supplemental material, sj-jpg-5-tam-10.1177_17588359251387965 for Treatment beyond progression and rechallenge with pyrotinib in HER2-positive metastatic breast cancer and brain metastases: a multicenter real-world study by Jing Peng, Qitong Chen, Liping Liu, Ning Xie, Mingwen Liu, Xuan Liu, Liyuan Qian, Kaili Lu, Tao Wu, Haiqing Xie, Haifan Xu, Zecheng Hu, Sijuan Ding, Wei Zhou, Chaojie Zhang, Yan Li, Jinhui Hu, Ruilian Xie, Jing He, Zhengkui Sun, Feng Xu, Danhua Zhang, Qian Long, Jinfeng Zhu, Xi Chen, Quchang Ouyang, Shouman Wang and Wenjun Yi in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-6-tam-10.1177_17588359251387965 – Supplemental material for Treatment beyond progression and rechallenge with pyrotinib in HER2-positive metastatic breast cancer and brain metastases: a multicenter real-world study
Supplemental material, sj-jpg-6-tam-10.1177_17588359251387965 for Treatment beyond progression and rechallenge with pyrotinib in HER2-positive metastatic breast cancer and brain metastases: a multicenter real-world study by Jing Peng, Qitong Chen, Liping Liu, Ning Xie, Mingwen Liu, Xuan Liu, Liyuan Qian, Kaili Lu, Tao Wu, Haiqing Xie, Haifan Xu, Zecheng Hu, Sijuan Ding, Wei Zhou, Chaojie Zhang, Yan Li, Jinhui Hu, Ruilian Xie, Jing He, Zhengkui Sun, Feng Xu, Danhua Zhang, Qian Long, Jinfeng Zhu, Xi Chen, Quchang Ouyang, Shouman Wang and Wenjun Yi in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We thank the Second People’s Hospital of Hunan Province, the General Hospital of Hunan Medical College, the First Hospital of Hunan University of Chinese Medicine, the Second People’s Hospital of Huaihua, the Fifth People’s Hospital of Huaihua, the First Affiliated Hospital of Jishou University, the First Affiliated Hospital of Nanchang University, Nanchang People’s Hospital, the Central Hospital of Shaoyang, Shaoyang Hospital of Traditional Chinese Medicine, the Second People’s Hospital of Xiangtan City, the Maternal and Child Health Care Hospital of Xiangtan City, Xiangtan Central Hospital, Xiangya Changde Hospital, Yichun People’s Hospital, Yiyang Kangya Hospital, the Third People’s Hospital of Yongzhou, Yongzhou Hospital of Traditional Chinese Medicine, Yugan People’s Hospital, the Second People’s Hospital of Yueyang, the Fourth People’s Hospital of Yueyang, Yueyang Central Hospital, Changsha Kexin Cancer Hospital, the Fourth Hospital of Changsha, and Changsha Central Hospital for providing patient medical records.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
