Abstract
Background:
After disease progression on anti-human epidermal growth factor receptor 2 (HER2) tyrosine kinase inhibitors (TKIs), trastuzumab deruxtecan (T-DXd) is recommended as a subsequent therapy. Because T-DXd has only recently been covered by insurance, patients previously opted for alternative regimens.
Objectives:
This study aimed to report real-world data on HER2-positive metastatic breast cancer (MBC) patients whose disease progressed during TKI therapy and provide a basis for further research.
Design:
This retrospective study included 216 HER2-positive MBC patients who progressed after TKI therapy between July 2014 and February 2025.
Methods:
All patients received anti-HER2 TKI therapy (including pyrotinib and lapatinib) in 28-day cycles. Treatment doses and combinations with chemotherapy, HER2-targeted agents, or endocrine therapy were decided by physicians. The primary objectives were to determine the proportion of patients receiving different regimens after TKI failure and evaluate progression-free survival (PFS). Secondary endpoints included overall survival (OS), objective response rate (ORR), disease control rate (DCR), and adverse events.
Results:
As of April 1, 2025, treatment distribution was 26.9% in the antibody–drug conjugate (ADC) group, 45.8% in the monoclonal antibody (mAb) group, and 27.3% in the TKI group. The expansion of ADC reimbursement under China’s healthcare policy, beginning in 2023, led to increased clinical adoption. Median PFS was 10.2 months (95% confidence interval (CI), 6.7–13.6) in the ADC group, 7.3 months (95% CI, 5.7–9.0) in the mAb group, and 7.2 months (95% CI, 5.8–8.6) in the TKI group, with no significant differences (p = 0.282). T-DXd demonstrated substantial antitumor activity with a 12-month PFS rate of 50.0%. Patients who are acquired-resistant to TKI had better outcomes than primary-resistant ones, particularly among those continuing TKI therapy. No unexpected toxic effects were reported.
Conclusion:
After progression following TKI therapy, T-DXd exhibited notable antitumor activity, although no significant efficacy differences were observed among ADC, mAb, and TKI groups.
Introduction
Breast cancer (BC) is one of the most common malignant tumors and the leading cause of cancer-related deaths among women. 1 Human epidermal growth factor receptor 2 (HER2)-positive BC accounts for approximately 15%–20% of all cases. 2 According to previous studies, HER2 overexpression is associated with poor prognosis in BC. 3 Anti-HER2 therapy is therefore essential for patients with HER2-positive BC.
Currently, based on the results of the PHENIX and PHOEBE studies,4,5 pyrotinib, a pan-HER tyrosine kinase inhibitor (TKI) that irreversibly inhibits HER1, HER2, and HER4, has shown significant efficacy in BC even in trastuzumab-resistant cases. However, pyrotinib was approved for medical insurance only in 2019. Before pyrotinib became available, lapatinib, a reversible inhibitor of HER1 and HER2 tyrosine kinases, also showed good efficacy in treating HER2-positive metastatic breast cancer (MBC). 6 Moreover, although trastuzumab deruxtecan (T-DXd) demonstrated remarkable therapeutic effects in the DESTINY Breast 01-03 studies7–9 and became the preferred option after trastuzumab failure according to the 2024 Chinese Society of Clinical Oncology (CSCO) Guideline, 10 it was only covered by insurance in China in 2025. Therefore, due to the limited accessibility of T-DXd, many patients previously used TKI therapy after trastuzumab failure.
However, an increasing number of patients experience TKI failure in clinical practice. In addition to T-DXd as the preferred treatment option, other anti-HER2 antibody-drug conjugates (ADCs), such as Trastuzumab Emtansine (T-DM1) and Disitamab Vedotin (RC48), trastuzumab plus pertuzumab combined with chemotherapy, and alternative anti-HER2 TKI therapy may be selected according to the 2024 CSCO guidelines. At present, no randomized controlled studies have compared the efficacy and safety of these regimens after TKI failure.
Therefore, our study aimed to report real-world data on patients with disease progression following TKI treatment. We presented the proportions of subsequent treatment regimens and evaluated the efficacy and safety of different anti-HER2 treatment strategies, providing a basis for further research and real-world evidence to support clinical practice.
Materials and methods
Study design and treatment
This was a retrospective real-world study. A total of 216 patients with HER2-positive MBC were enrolled between July 3, 2014 and February 1, 2025 at Jiangsu Cancer Hospital and The First Affiliated Hospital of Nanjing Medical University. The last follow-up was conducted on April 1, 2025. All patients received anti-HER2 TKI therapy (including pyrotinib and lapatinib) in 28-day cycles. Actual doses were standardized. The initial doses of ADC and monoclonal antibody (mAb) were calculated based on patient body weight. The initial TKI dose was the maximum recommended dose (pyrotinib: 400 mg po qd; lapatinib: 1250 mg po qd). Combination therapy with chemotherapeutic drugs, HER2-targeted agents, and/or endocrine therapy was determined by physicians based on previous clinical trial results, patients’ general health status, and patient preference. During the study period, follow-up was conducted through outpatient visits, telephone calls, and other methods to minimize bias from loss to follow-up. This study adhered to the Declaration of Helsinki and was approved by the Ethics Committee of Jiangsu Cancer Hospital (KY-2024-066). Individual consent for this retrospective analysis was waived. The reporting of this study conforms to the STROBE statement. 11
Patient population
Eligibility criteria were as follows: (1) histologically or cytologically confirmed HER2-positive MBC, defined as immunohistochemistry 3+ or fluorescence in situ hybridization positive; (2) disease progression after anti-HER2 TKI treatment (including lapatinib and pyrotinib); and (3) at least one measurable lesion according to the Response Evaluation Criteria in Solid Tumors (RECIST 1.1). Exclusion criteria included: (1) missing treatment information; and (2) history of meningeal metastasis and/or spinal cord metastasis, or other malignancies within 5 years (excluding cured cervical preinvasive carcinoma and skin basal cell or squamous cell carcinoma).
Study endpoints
The primary endpoint was progression-free survival (PFS), defined as the time from disease progression during TKI treatment to disease progression or death during the subsequent line of therapy, whichever occurred first. Secondary endpoints included objective response rate (ORR), defined as the proportion of patients with the best overall response of complete response (CR) or partial response (PR); disease control rate (DCR), defined as the proportion of patients with a best overall response of CR, PR, or stable disease (SD); and clinical benefit rate (CBR), defined as the proportion of patients with a best overall response of CR, PR, or SD lasting ⩾6 months. Tumor response was assessed by investigators in patients with measurable lesions according to the RECIST, version 1.1. Primary resistance to trastuzumab was defined as relapse during or within 12 months after adjuvant trastuzumab or progression within 3 months of trastuzumab treatment for metastatic disease. Primary resistance to TKI was defined as progression within 6 months of TKI treatment for the first time in metastatic disease. Safety assessments were performed using the Common Terminology Criteria for Adverse Events (CTCAE), version 5.0.
Statistical analysis
The Kaplan–Meier method was used to estimate median PFS, and the Cox proportional hazard model was used to estimate hazard ratios and 95% confidence intervals. ORR, DCR, and CBR were estimated using the Clopper–Pearson method. Analyses were performed using SPSS statistical software version 27.0 or R Studio 4.2. A p value < 0.05 was considered statistically significant.
Results
Patients
From July 3, 2014 to February 1, 2025, 216 patients were included. The median line of prior TKI treatment was the second. Among these, 134 (62.0%) patients were HR-positive, and 82 (38.0%) were HR-negative. A total of 31 patients (14.4%) had previously received lapatinib, while 185 (85.6%) had received pyrotinib. The number of patients who developed primary resistance to TKIs was 60 (27.8%). The baseline characteristics are shown in Table 1.
Baseline characteristics.
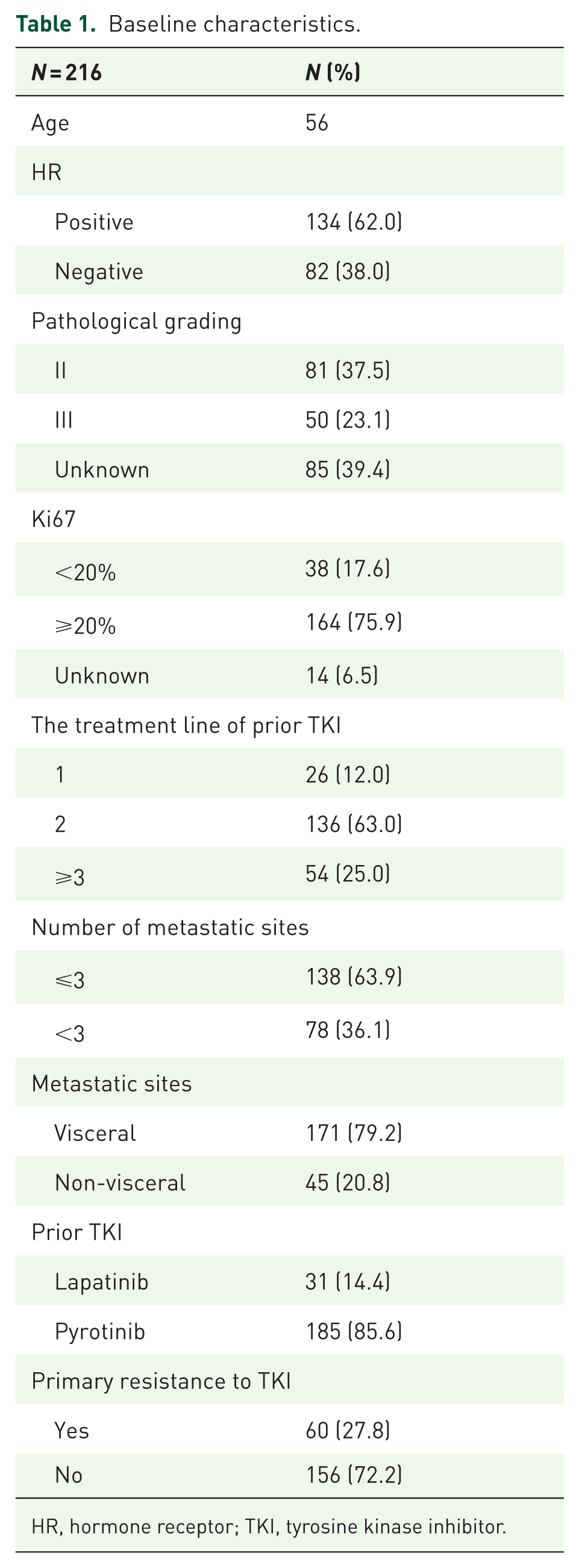
HR, hormone receptor; TKI, tyrosine kinase inhibitor.
After progression on TKI therapy, 58 patients received ADC, 99 received mAb, and 59 received TKI therapy (Figure 1(a)). In the ADC group, T-DXd accounted for 25.9%, while T-DM1 and RC48 accounted for 32.8% and 34.5%, respectively (Figure 1(b)). During 2020–2022 and 2023–2025, the proportions of TKI and ADC use increased significantly compared with earlier periods (Figure 1(c)).
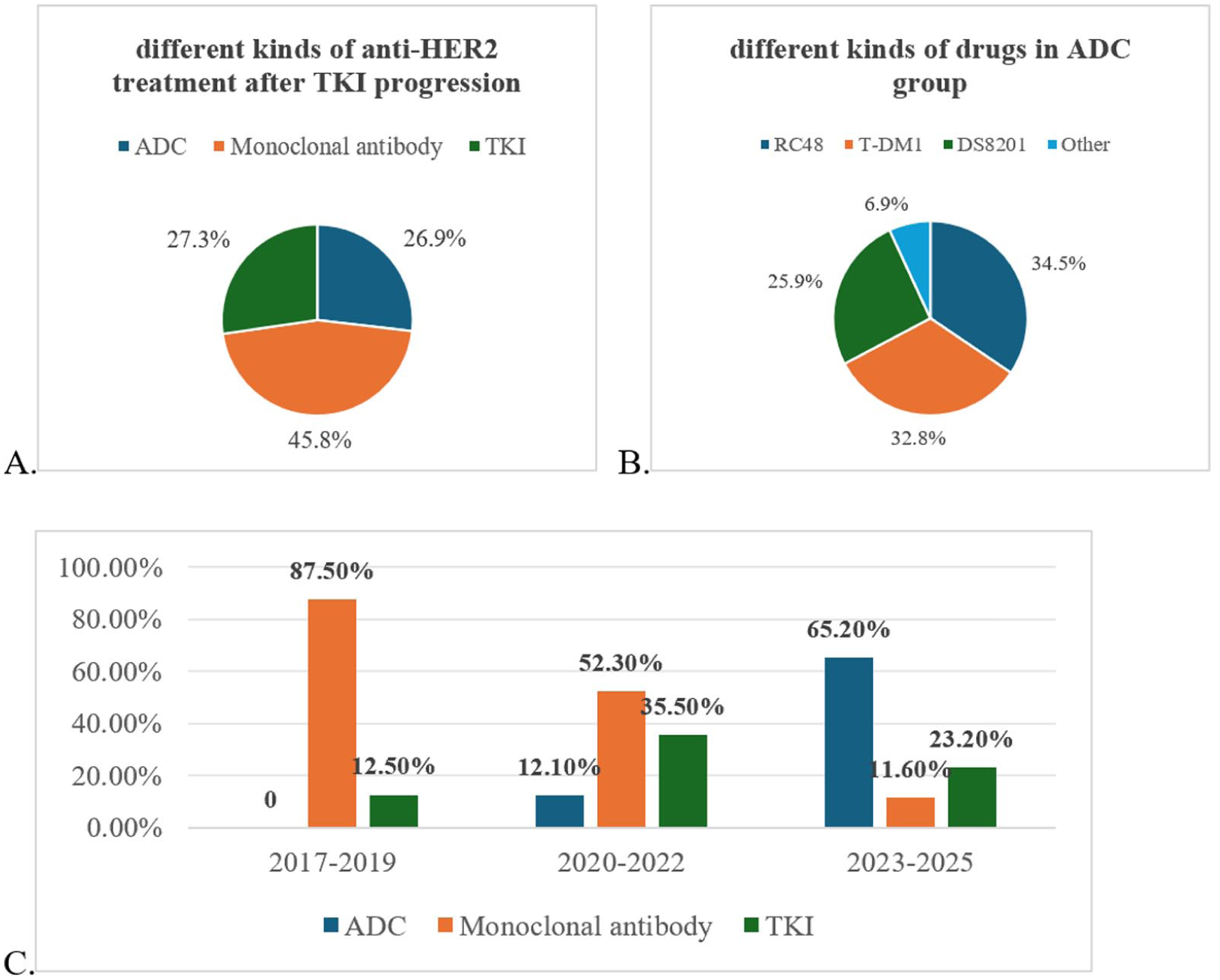
Different types of anti-HER2 treatment after TKI progression (a). Different types of anti-HER2 ADC drugs after TKI progression (b). Distribution of different types of anti-HER2 therapy over time (c).
Efficacy
Among the 216 patients, the median PFS was 7.3 months (95% confidence interval (CI), 6.3–8.2). All three groups had a median treatment line of three. The ADC group showed a median PFS of 10.2 months (95% CI, 6.7–13.6), and the mAb group 7.3 months (95% CI, 5.7–9.0), both longer than the TKI group, which had a median PFS of 7.2 months (95% CI, 5.8–8.6; p = 0.151, HR = 0.735; p = 0.873, HR = 0.972), with no significant difference among groups (p = 0.282; Figure 2(a)). The TKI-acquired resistant subgroup showed a numerically longer median PFS than the TKI-primary resistant subgroup (7.7 vs 6.0 months; p = 0.147; HR = 0.794; 95% CI, 0.572–1.101; Figure 2(b)). In addition, in the post-progression TKI group, patients with acquired resistance to TKI experienced significantly better outcomes than those with primary resistance (median PFS: 7.9 vs 5.7 months, p = 0.018).
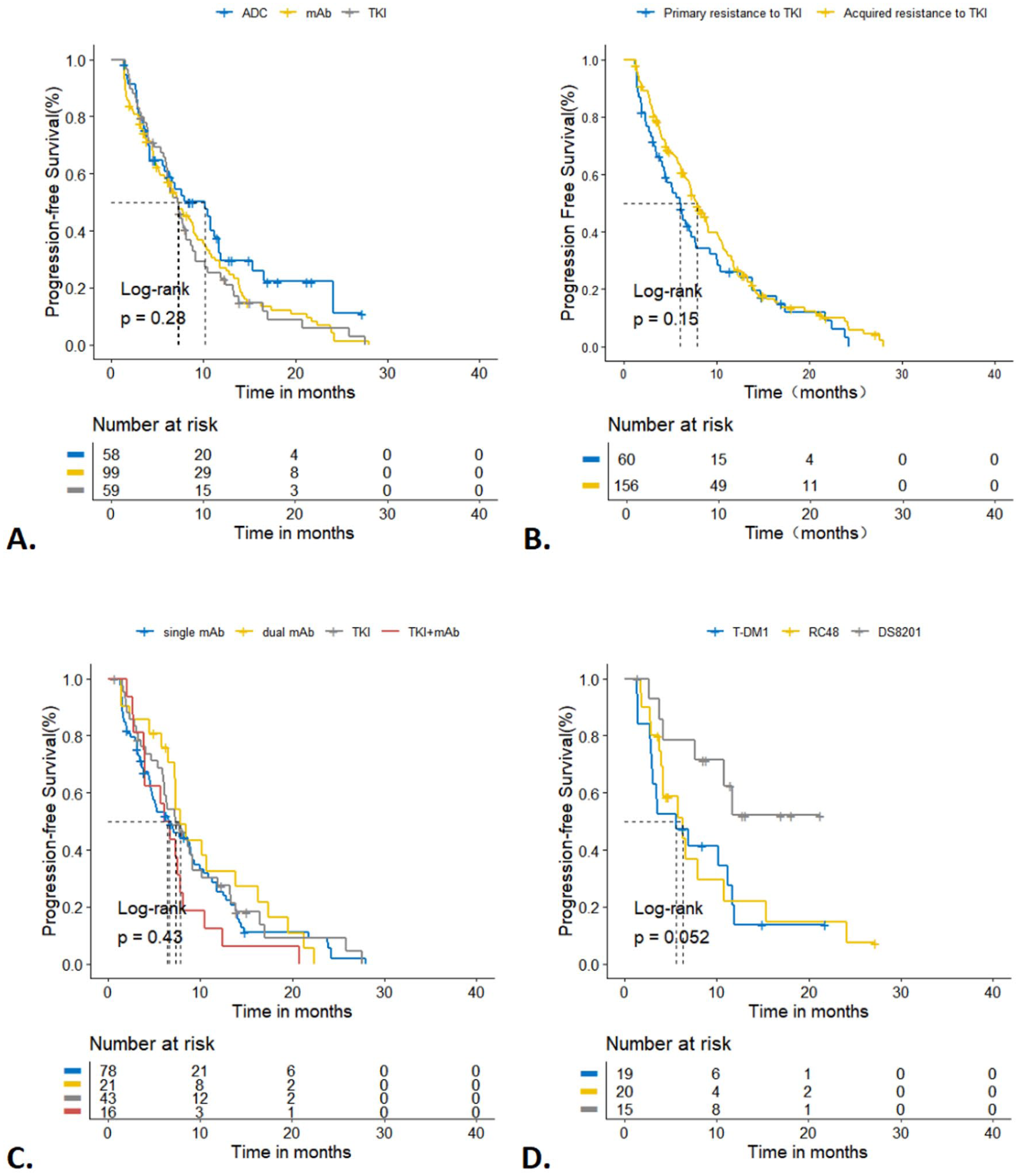
Progression-free survival for patients according to treatment regimens (a), resistance to TKI (b), different types of ADC (c), and single or dual-target anti-HER2 regimens (d).
In the ADC cohort, the median follow-up time was 13.1 months (95% CI, 11.0–15.1). The median PFS for T-DXd was not reached, but the 12-month PFS rate for T-DXd was 50.0%, which was significantly longer than that of RC48 (6.3 months; p = 0.047; HR = 2.66; 95% CI, 1.01–7.00) and T-DM1 (5.5 months; p = 0.024; HR = 2.98; 95% CI, 1.15–7.71; Figure 2(c)).
In the mAb group, HP (trastuzumab + pertuzumab) regimens did not confer a significant benefit compared to H (trastuzumab)-based regimens (median PFS: 7.8 vs 6.6 months; p = 0.537). Similarly, in the TKI group, TKI combined with mAb therapy (median PFS: 6.1 months) showed no advantage over TKI-based regimens (median PFS: 7.3 months; p = 0.173). No difference was observed between dual-target and single-target regimens in either group (Figure 2(d)).
Within the TKI subgroup, no significant difference in median PFS was observed between patients who received pyrotinib after lapatinib failure (6.0 months) and those who continued pyrotinib after progression (6.7 months; p = 0.799; HR = 0.922; 95% CI, 0.494–1.722).
We further analyzed survival outcomes of the next-line treatment. Among these patients, 28.6% received ADC, 47.1% received mAbs, and 24.3% received TKI therapy. The median PFS was 6.1, 5.7, and 6.7 months in the ADC, mAb, and TKI groups, respectively (p = 0.817), with no significant difference among groups. Furthermore, patients who received ADC2 after ADC1 (median PFS1 + PFS2: 14.3 months) showed better clinical outcomes than those who received mAb after ADC (median PFS1 + PFS2: 8.0 months; p = 0.424). We also compared patients who received mAb after T-DM1 (N = 9) with those who received T-DM1 after mAb (N = 8). The results showed no significant difference between the two treatment sequences (Figure 3).

PFS1 + PFS2 of different treatment regimens after TKI progression: different treatment regimens after ADC (a), different treatment sequences of T-DM1 and mAb (b).
The median follow-up time was 17.0 (95% CI, 13.0–20.9), 46.0 (95% CI, 35.1–56.8), and 40.0 (95% CI, 27.9–52.1) months in the ADC, mAb, and TKI groups, respectively. The median overall survival (OS) in the mAb group was 31.6 months (95% CI, 25.3–37.9). In the ADC group, the 1-year OS rate was 80.0%. In the TKI group, the 1- and 2-year OS rates were 81.6% and 48.7%, respectively (Table 2).
OS rate, ORR, CBR, and DCR of different groups.
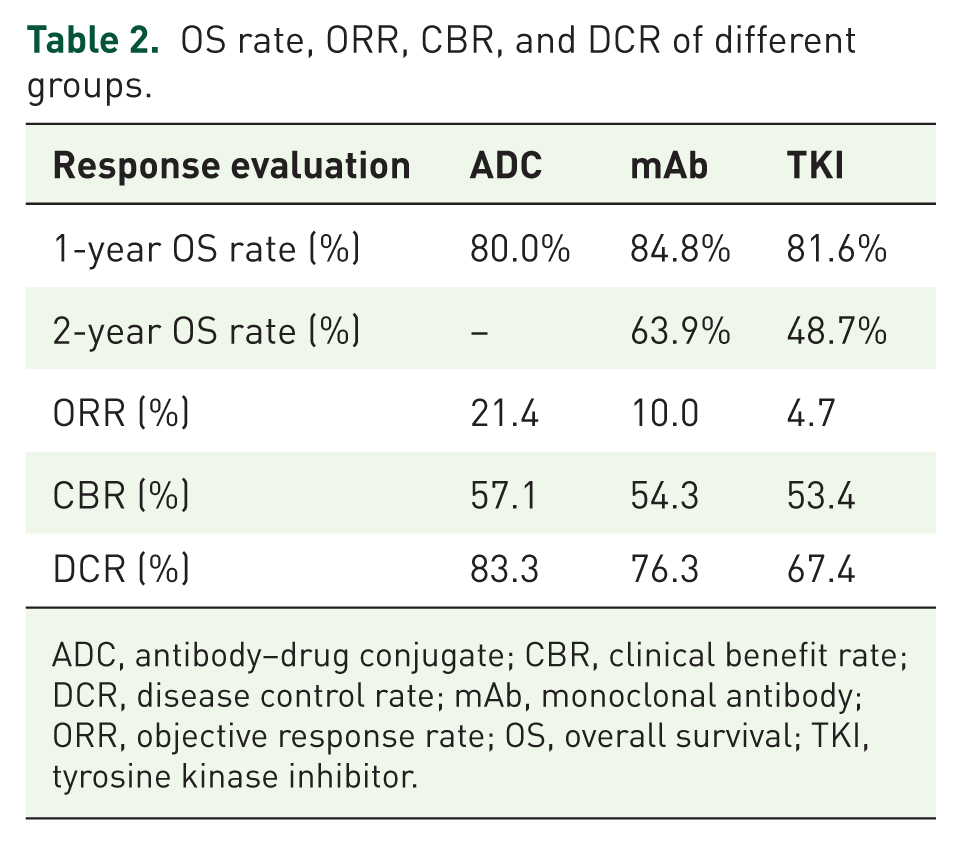
ADC, antibody–drug conjugate; CBR, clinical benefit rate; DCR, disease control rate; mAb, monoclonal antibody; ORR, objective response rate; OS, overall survival; TKI, tyrosine kinase inhibitor.
In the ADC, mAb, and TKI treatment groups, 9, 8, and 2 patients achieved PR, respectively; SD was observed in 26, 53, and 27 patients, and PD occurred in 7, 19, and 14 patients. The ORRs were 21.4%, 10.0%, and 4.7% for the three groups, respectively. The CBRs were 57.1%, 54.3%, and 53.4%, while the DCRs were 83.3%, 76.3%, and 67.4%. Notably, ADC therapy showed a clinical advantage over other therapies in patients who had failed prior TKI therapy (Table 2).
Multivariate analysis showed that only the number of metastatic sites was an independent prognostic factor for PFS. For OS, primary resistance to TKI became the independent prognostic factor in TKI-pretreated patients (Figure 4).
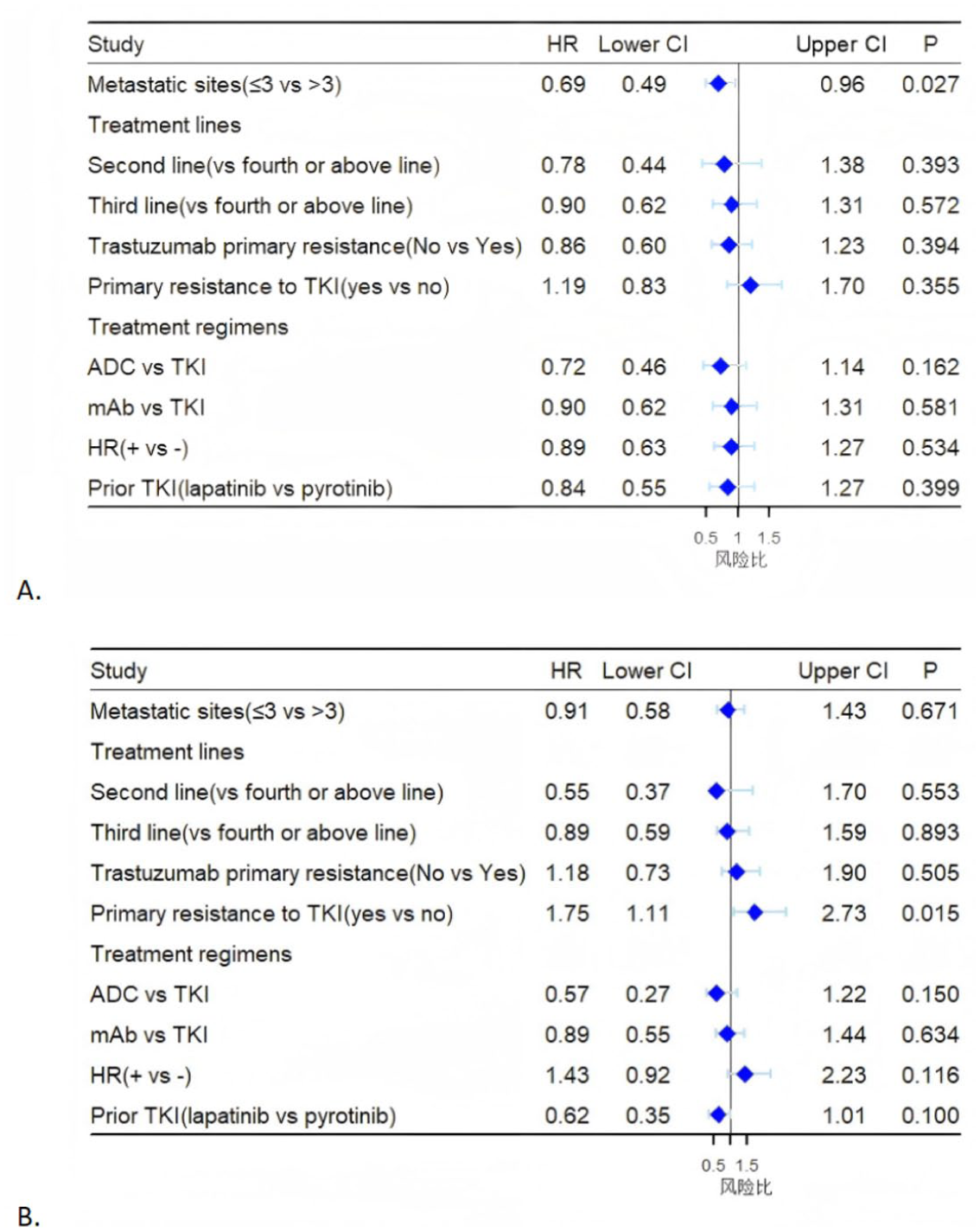
Multivariate analysis for PFS (a) and OS (b). HRs are derived from unstratified analyses.
Among the three groups, no unexpected toxic effects were observed. The most common grade ⩾3 adverse events were neutropenia, leukopenia, and thrombocytopenia. Furthermore, the most frequent adverse event in the ADC group was an increase in alanine transaminase (ALT) levels (Table 3).
AEs in the three groups.

ADC, antibody–drug conjugate; AE, adverse events; ALT, alanine transaminase; mAb, monoclonal antibody; TKI, tyrosine kinase inhibitors.
Discussion
As is well known, anti-HER2 therapy is the cornerstone of treatment for HER2-positive MBC. With the widespread use of TKI, resistance is inevitable. Previous studies have demonstrated several mechanisms underlying TKI resistance, including aberrant activation of the PI3K/AKT/mTOR pathway, changes in the tumor immune microenvironment, and HER2-positive BC heterogeneity. 12 Therefore, selecting the appropriate next-line therapy after TKI failure to overcome resistance remains a significant challenge in clinical practice.
Our data showed that after TKI progression, nearly 50% of patients chose mAb therapy. This phenomenon may be due not only to the availability of new drugs but also to China’s medical insurance policy. Before ADCs were covered by medical insurance, patients had to pay the full cost themselves. However, once these drugs were included in China’s medical insurance, depending on regional policies, the reimbursement rate for most patients could range from 50% to 70%. For some employee medical insurance programs, it could even exceed 70%. This substantially alleviates the financial burden on patients and enables more patients to afford new drugs. Given that T-DM1 was included in medical insurance in 2023 and T-DXd in 2025, the proportion of ADC use increased significantly from 2023 to 2025. This trend mirrors the pattern observed for TKIs from 2020 to 2022 (pyrotinib was included in insurance coverage in 2019).
In our study, the median PFS for ADC, mAb, and TKI cross-line cohorts was 10.2, 7.3, and 7.2 months, respectively (p = 0.282). ADC (especially T-DXd) demonstrated clinically meaningful efficacy in TKI-pretreated patients, consistent with previous studies.7,13–16 With a median follow-up of 15.0 months, T-DXd showed a more favorable trend than T-DM1 and RC48 (12-month PFS rate: 50.0%), consistent with the DESTINY-Breast02 findings (12-month PFS rate: 62.3%). 7 Among patients who received T-DM1 after TKI progression, the median PFS was 5.5 months, similar to that reported in the TH3RESA study (median PFS, 6.2 months). 15 This may be attributed to the high drug-to-antibody ratio and the precise targeting ability of ADCs to kill cancer cells, which differ from the mechanism of TKIs and may overcome resistance.17,18 In particular, the compound released from T-DXd through the “Bystander effect” can diffuse to adjacent tumor cells. 19
Although ADCs are increasingly covered by China’s medical insurance, their higher cost compared to TKIs and mAbs remains a barrier. The concepts of trastuzumab rechallenge and TKI cross-line therapy in HER2-positive MBC have emerged as therapeutic strategies because of the limited treatment options available. According to previous studies, TKIs form irreversible bonds with the intracellular domains of HER proteins, thereby inhibiting downstream signaling pathways and blocking these receptors through a mechanism distinct from that of mAbs.20,21 Thus, TKI can overcome resistance to trastuzumab,21,22 which also suggests the potential value of mAb rechallenge after TKI failure. Our study confirms that mAbs retain therapeutic value for ADC-ineligible patients after TKI failure (median PFS: 7.3 months). In addition, the median OS of the mAb group was comparable to that reported in another study (31.6 vs 33.7 months). 23 For cross-line TKI therapy, our result (median PFS: 7.2 months) was consistent with previous studies.24,25 However, the ORR was lower than that in some reports, likely because most patients (76.4%) in our study did not change the TKI after progression.
In the mAb and TKI cohorts, we also evaluated the efficacy difference between dual anti-HER2 therapy and single anti-HER2 therapy. The results showed no significant survival benefit between the two regimens in late-line settings (median PFS: 6.2 vs 5.8 months, p = 0.147), consistent with previous studies.26,27 However, trastuzumab-pertuzumab dual blockade demonstrated superior efficacy compared with TKI-mAb combinations (median PFS: 8.4 vs 5.8 months; HR = 0.69, 95% CI: 0.52–0.91; p = 0.027). This may reflect the fact that only 23.1% of patients had previously received pertuzumab.
According to previous studies, we defined PFS ⩾6 months during initial TKI treatment as TKI-primary resistance.26,27 Multivariate analysis revealed that whether TKI was primary resistant or not did not significantly influence first-line post-TKI outcomes (p = 0.355). However, it emerged as an independent prognostic factor for long-term survival (p = 0.015), suggesting a poorer prognosis for TKI-refractory patients. Furthermore, in the cross-line TKI group, patients with acquired resistance to TKI showed a significant benefit compared with those with primary resistance (median PFS: 7.9 vs 5.7 months, p = 0.018), findings similar to those of a previous study. 27 Hence, our results further support the potential of cross-line TKI treatment, even with the same TKI.
We further explored the subsequent treatment line. As ADC drugs are gradually included in medical insurance in China, more patients have opted for them in recent years. However, resistance to ADCs inevitably develops. One key question is whether continuing another ADC after failure of a prior ADC provides clinical benefit. Efficacy was maintained in patients treated with a second ADC in the DESTINY-Breast02 study. 7 Similarly, in our study, 11 patients received a different ADC after progression on the first ADC and achieved a median PFS of 5.8 months, which was longer than that reported in another study (median PFS: 4.8 months), indicating potential therapeutic benefit. 28 Based on our analysis, there was a significant difference in clinical outcomes between patients who received an ADC immediately after TKI progression and those who tried other treatments first (median PFS: 10.2 vs 6.1 months, p = 0.039). These results suggest that earlier use of ADC therapy may lead to better efficacy.
Furthermore, because the median PFS of T-DXd was immature, we selected T-DM1 as a representative ADC. T-DM1 was the first anti-HER2 ADC approved in China. 29 We compared patients who received mAb after T-DM1 with those who received T-DM1 after mAb. The results showed no significant difference in survival outcomes between the two treatment sequences in the late-line setting. However, the sample size was small, and these findings should be confirmed in larger studies.
Our data demonstrate that all three treatment types were well tolerated in patients pretreated with TKIs, and no unexpected toxic effects were observed.5,30–32 Hematologic abnormalities were the most common adverse event overall. Notably, the potential for overlapping toxicities should be considered when switching TKIs. However, our data showed a relatively mild safety profile for cross-line TKI therapy. In the ADC group, elevated transaminase levels were common and consistent with previous studies,9,33 regardless of whether patients received T-DXd, T-DM1, or RC48. For example, T-DM1 can cause hepatotoxicity through a HER2-dependent pathway, and hepatocellular injury mediated by T-DM1 may be enhanced by the proinflammatory cytokine TNF-α. 34 Therefore, liver function should be closely monitored during ADC treatment. This study had several limitations. First, although it was a retrospective, multicenter study, the data were derived from only two centers, which may limit generalizability. In addition, real-world data are inherently less rigorous than those from randomized controlled studies. Second, because T-DXd was only recently included in medical insurance, the available data remain immature, and continued follow-up is required. Finally, the sample size was relatively small and imbalanced across the three subgroups, which may have introduced bias.
Conclusion
Our study demonstrated the real-world status of BC patients in China after TKI failure. The data showed that T-DXd demonstrated notable antitumor efficacy after TKI progression, although differences in efficacy among the ADC, mAb, and TKI groups were not significant. Furthermore, in TKI-treated HER2-positive MBC patients, resistance to TKI may serve as an independent long-term prognostic factor.
Supplemental Material
sj-pdf-1-tam-10.1177_17588359261442687 – Supplemental material for A real-world study on treatment after TKI progression in HER2-positive MBC
Supplemental material, sj-pdf-1-tam-10.1177_17588359261442687 for A real-world study on treatment after TKI progression in HER2-positive MBC by Shiyi Li, Lili Zhang, Ting Xu, Jiayuan Huang, Chengjun Zhu and Yuan Yuan in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors are grateful to the Jiangsu Cancer Hospital and the other participating hospitals for their support and to Professor Deng Rong for his guidance on our manuscript. The content of this manuscript is solely the responsibility of the authors and does not necessarily represent the official views of the Jiangsu Cancer Hospital.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
