Abstract
Purpose:
This study aimed to investigate clinical practices and factors related to the outcomes of T-DM1 use in patients with HER2-positive metastatic breast cancer (mBC).
Methods:
We included patients with HER2-positive mBC who received T-DM1 as a palliative therapy between August 2017 and December 2018. The safety and outcomes of T-DM1, including overall response rate (ORR), progression-free survival (PFS), and overall survival (OS), were evaluated. A Cox proportional hazards model was used to estimate the hazard ratio and 95% confidence interval (CI) for mortality or progression to HER2-positive mBC.
Results:
In total, 824 patients were enrolled during the study period. The mean age of patients was 58 years, and 516 (62.6%) patients relapsed after curative treatment. Excluding a history of endocrine therapy, 341 (41.4%) patients previously received none or first-line chemotherapy, 179 (21.7%) received second-line therapy, and 303 (36.9%) received third-or later-line chemotherapy before T-DM1 therapy. During a median follow-up of 16.8 months, the ORR was 35%, the median PFS was 6.6 months, and the median OS was not reached. The clinical factors associated with the hazard of progression were age (<65 years), poor performance status (⩾2), advanced line of palliative chemotherapy (⩾2), prior pertuzumab use, and treatment duration of palliative trastuzumab (<10 months). Common grade 3–4 adverse events were thrombocytopenia (n = 107, 13.2%), neutropenia (n = 23, 2.8%), anemia (n = 21, 2.6%), and elevated liver enzyme (n = 20, 2.5%). Hypokalemia (⩽3.0 mmol/L) and any-grade bleeding events occurred in 25 (3.1%) and 94 (22.6%) patients, respectively.
Conclusion:
This is the first nationwide real-world study of T-DM1 use in patients with HER2-positive mBC in Korea. The effectiveness and toxicity profiles of T-DM1 in real-world practice were comparable to those in randomized trials. Moreover, patient factors and previous anti-HER2 therapy could predict the outcomes of T-DM1 therapy.
Introduction
Overexpression of human epidermal growth factor receptor (HER)2 occurs in approximately 20–30% of breast cancers and is associated with poor prognosis.1–3 However, the advent of HER2-targeted drugs, including anti-HER2 monoclonal antibodies and HER2 tyrosine kinase inhibitors, has revolutionized the treatment of HER2-positive advanced and metastatic breast cancer (mBC).4–10 According to the CLEOPATRA study, the standard first-line systemic treatment for HER2-positive mBC consists of trastuzumab plus pertuzumab combined with a taxane. 9 Although lapatinib was approved by the FDA in 2007 for use in combination with capecitabine for the treatment of patients with HER2-positive mBC who had progressed after initial chemotherapy plus trastuzumab based on a phase III trial, 5 T-DM1 was approved as a second-line therapy for these patients in the EMILIA study in 2014.
T-DM1 was the first antibody–drug conjugate to incorporate the HER2-targeted antitumor properties of trastuzumab with the cytotoxic activity of the microtubule-inhibitory agent, emtansine. 11 T-DM1 significantly prolonged the progression-free survival (PFS) and overall survival (OS) more than lapatinib plus capecitabine (9.6 months versus 6.4 months, and 30.9 months versus 25.1 months, respectively) in patients with HER2-positive advanced breast cancer previously treated with trastuzumab and a taxane. 10 T-DM1 was compared with the choice of the physician in a population with progressive disease who received both trastuzumab- and lapatinib-containing regimens for advanced breast cancer. In that clinical trial, better PFS and OS were observed in the T-DM1 group (6.2 months versus 3.3 months and 22.7 months versus 15.8 months, respectively).12,13 However, these two studies did not provide evidence regarding the efficacy of T-DM1 after a patient had been treated with trastuzumab and pertuzumab because pertuzumab had not received regulatory approval at the time of initiation. In addition, the number of patients who received T-DM1 after the pertuzumab or placebo combination in the CLEOPATRA study was approximately 12% in both groups 9 ; therefore, this was a limitation in determining the efficacy of T-DM1 after pertuzumab.
Trastuzumab deruxtecan (T-DXd) is another antibody–drug conjugate consisting of a humanized anti-HER2 monoclonal antibody linked to topoisomerase I inhibitor payload through a tetrapeptide-based cleavable linker, with a drug–antibody ratio of 7–8.14,15 DESTINY-Breast03 showed that T-DXd treatment led to striking improvements in both progression-free survival [hazard ratio (HR) for progression or death, 0.28; 95% confidence interval (CI), 0.22–0.37; p < 0.001] and OS (HR for death, 0.55; 95% CI, 0.36–0.86; p = 0.007) compared with T-DM1 treatment in patients with HER2-positive mBC who had been previously treated with trastuzumab and a taxane.16,17 Based on this study, the FDA approved T-DXd for the treatment of adults with unresectable or metastatic HER2-positive breast cancer who had received a prior anti-HER2-based regimen either in the metastatic setting or in the neoadjuvant or adjuvant setting and had developed disease recurrence during or within 6 months of completing therapy in May 2022 because T-DXd was only available in approximately 20 countries by May 2023 18 and is rarely reimbursed by national health insurance. Even in countries where T-DXd is available, the standard second-line treatment is still T-DM1 in clinical practice.
Randomized controlled trials (RCTs) continue to provide essential evidence regarding the efficacy of new treatments. However, RCTs are conducted with highly selective populations and specialized environments that differ from those in clinical practice. There may be limited data on the interactions between concomitant illnesses and treatment, and the results may be supported by intensive efforts that are infeasible in practice. RCTs typically require intensive support and expensive resources. 19 Real-world evidence (RWE) refers to information on healthcare derived from multiple sources outside clinical research settings, including electronic health records, claims and billing dates, product and disease registries, and data gathered through personal devices and health applications. 20 It is important to provide valuable insights into treatments and their outcomes in routine daily practice, including for patients excluded or under-represented in RCTs. 21 Moreover, RWE offers an opportunity to complement randomized controlled trial evidence on safety and use in a wider patient population. 22 Patients treated with T-DM1 in second-line trials do not necessarily receive the current standard first-line treatments, including pertuzumab. RWE represents the application of treatment within a specific geographic and economic context and can help produce data regarding the treatment sequence. To date, the RWE from large cohort studies focusing on T-MD1 in HER2-positive mBC is limited.
Therefore, we conducted a nationwide, real-world study to evaluate the effectiveness, safety, usage patterns, and survival outcomes of T-DM1 in patients with HER2-positive mBC who progressed after trastuzumab treatment.
Materials and methods
Study design and patients
In this retrospective, noninterventional study, the effectiveness and safety of T-DM1 were evaluated in a nationwide, real-world setting. The use of T-DM1 in patients with relapsed/metastatic HER2-positive breast cancer has been reimbursed by the National Health Insurance in Korea since 3 August 2017. The recommendations of the Korean Health Insurance Review and Assessment (HIRA) are in accordance with those of the EMILIA and THERESA. The key inclusion criteria were patients who were pretreated with trastuzumab and taxane or had progressed during or 6 months after completing adjuvant chemotherapy for HER2-positive metastatic/relapsed breast cancer between 3 August 2017 and 31 December 2018, and who were registered with the Korean HIRA Service system. Patients receiving T-DM1 before 3 August 2017 were excluded.
The primary endpoint was PFS, and the secondary endpoints were the overall response rate (ORR), OS, time to the next treatment after T-DM1, duration of response to T-DM1, and safety.
This study was conducted by the Breast Cancer Committee of the Korean Cancer Study Group (KCSG) and was funded by the HIRA. This study was registered with ClinicalTrials.gov under identifier NCT04202328 (KCSG BR19-15).
This study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 23
Data collection
Nationwide medical record reviews were conducted in 60 institutions from 3 August 2017 to 30 September 2019, and data recorded on 31 December 2019 were collected for our analysis. Data included baseline patient and disease characteristics, history of prior therapy, response to T-DM1 therapy, adverse events, subsequent therapy, and survival. All data were anonymously collected and managed using an electronic data capture system consisting of filters and a query-generating system to guarantee reliability and control for missing and inconsistent data.
Chemotherapy and endocrine therapy lines were counted separately. A chemotherapy line was defined based on chemotherapy before T-DM1 therapy excluding endocrine therapy. Similarly, the line of endocrine therapy was defined excluding chemotherapy. If an anthracycline had been administered as adjuvant or metastatic setting, it was indicated as a prior anthracycline.
Outcome measures
Data on HER2 status were determined at each institution without a central review and were obtained by reviewing medical records. A positive HER2 status was defined as HER2 immunohistochemistry 3+ or in situ hybridization positivity.
The patients’ baseline clinical characteristics at T-DM1 administration, systemic treatment history before or after T-DM1 administration, and treatment outcomes, such as response rate (RR), PFS, and OS, were collected through a retrospective review of medical records at each institution and recorded in an electronic case report form by trained staff.
PFS after T-DM1 use was defined as the time from the first treatment date with T-DM1 until disease progression or death from any cause. OS was defined as the time from the first treatment date to T-DM1 until death from any cause. Time to the next treatment was defined as the time from the first treatment date with T-DM1 to the first date of the next chemotherapy session. Patients in whom no events were observed were censored on the day of their last follow-up visit. The date of T-DM1 discontinuation was defined as the last administration date. Tumor response was evaluated using the Response Evaluation Criteria in Solid Tumors (RECIST) guideline version 1.1. The overall response rate (ORR) was defined as the proportion of patients with complete response (CR) or partial response (PR) as the best overall response. The disease control rate (DCR) was defined as the proportion of patients with CR, PR, or stable disease (SD) as the best overall response (including non-CR/non-progressive disease). Toxicity was assessed using the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE), version 4.0.
Statistical analysis
The patient demographics and clinical characteristics were summarized using descriptive statistics. Cumulative survival rates were estimated using the Kaplan–Meier method. A Cox proportional hazards model was used to identify independent predictors of PFS and OS. The multivariable model included the patient’s age and variables with a p-value of <0.1 from the simple model. All reported p-values were two-tailed, and a p-value of <0.05 was considered statistically significant. All statistical analyses were performed using SAS v 9.3 (SAS Institute Inc., Cary, NC, USA).
Results
Of the 1037 patients initially identified, 824 from 60 hospitals were enrolled, excluding those from hospitals with fewer than three patients or absence of an IRB (N = 32); those from hospitals with difficulty participating due to other causes (N = 33), such as lack of manpower or difficulty accessing electronic medical records; and those with screening failure or missing data (N = 148) (Figure 1).

Flow diagram of the patient population.
Patients demographics
Table 1 shows the characteristics of the 824 patients. The median follow-up duration was 16.8 months, and the median age was 58 years (range, 28–88 years). The Eastern Cooperative Oncology Group (ECOG) Performance Status (PS) of 0 or 1 was 695 (84%) and that of those with visceral metastasis was 544 (70%). The most frequent metastatic sites are the lungs, bones, lymph nodes, and liver. In total, 401 patients had hormone receptor-positive breast cancer (49%), and 308 had de novo mBC (37%). Regarding the last line of therapy before T-DM1, 766 (93%) patients had previously received palliative chemotherapy. Moreover, 341 (41%) patients had previously received no or first-line chemotherapy, 179 had received second-line chemotherapy, and 133 had received third-line chemotherapy. A total of 171 (21%) patients had previously received endocrine therapy. Of the patients, 60% received first-line endocrine therapy. The percentages of patients with prior pertuzumab, lapatinib, and anthracycline therapy were 22%, 43%, and 48%, respectively.
Baseline characteristics of patients.

Based on chemotherapy before T-DM1 therapy excluding endocrine therapy.
Based on endocrine therapy before T-DM1 therapy excluding chemotherapy.
CNS, central nervous system; ECOG; Eastern Cooperative Oncology Group.
Before the T-DM1 treatment, 161 patients had central nervous system (CNS) metastases. Among them, 83% had brain parenchymal metastasis, 3% had meningeal metastasis, and 14% had metastasis to both regions. Of the patients, 66% were treated with whole-brain radiotherapy, 43% with stereotactic radiosurgery, and 17% underwent surgery.
Effectiveness
At the time of data collection, 707 (86%) patients had discontinued T-DM1 treatment, and 462 (56%) were alive. The most common cause of discontinuation was systemic disease progression (N = 556, 67%), followed by unacceptable toxicity (N = 35, 4%) and CNS progression (N = 25, 3%). Of the 771 patients in whom tumor response was evaluated, 17 (2%) showed CR, 253 (33%) showed PR, and 313 (41%) exhibited SD (Table 2). ORR and DCR were 35% and 76%, respectively. The median PFS was 6.6 months (95% CI: 6.1–7.3; Figure 2). The median time to the next treatment after T-DM1 therapy was 8.1 months (95% CI: 7.6–9.1; Figure 2). The median OS after T-DM1 therapy was not reached at the time of analysis (Figure 2). The DCR of patients previously receiving one or fewer chemotherapy, two, three, four, and beyond were 79%, 78%, 76%, and 66%, respectively. The median PFS of patients who previously received one or fewer chemotherapy, two, three, four, and beyond were 7.9, 7.0, 6.7, and 4.4 months, respectively (Table 3). Figure 3 shows PFS according to prior chemotherapy. The median number of T-DM1 treatment was the eighth cycle (range 1–43).
Response outcomes.

ORR indicates an objective response rate and is defined as the proportion of patients with CR or PR as a best overall response. DCR indicates disease control rate and is defined as the proportion of patients with CR, PR, or SD as best overall response (including non-CR/non-progressive disease).
CR, complete response; PD, progressive disease; PR, partial response; SD, stable disease.
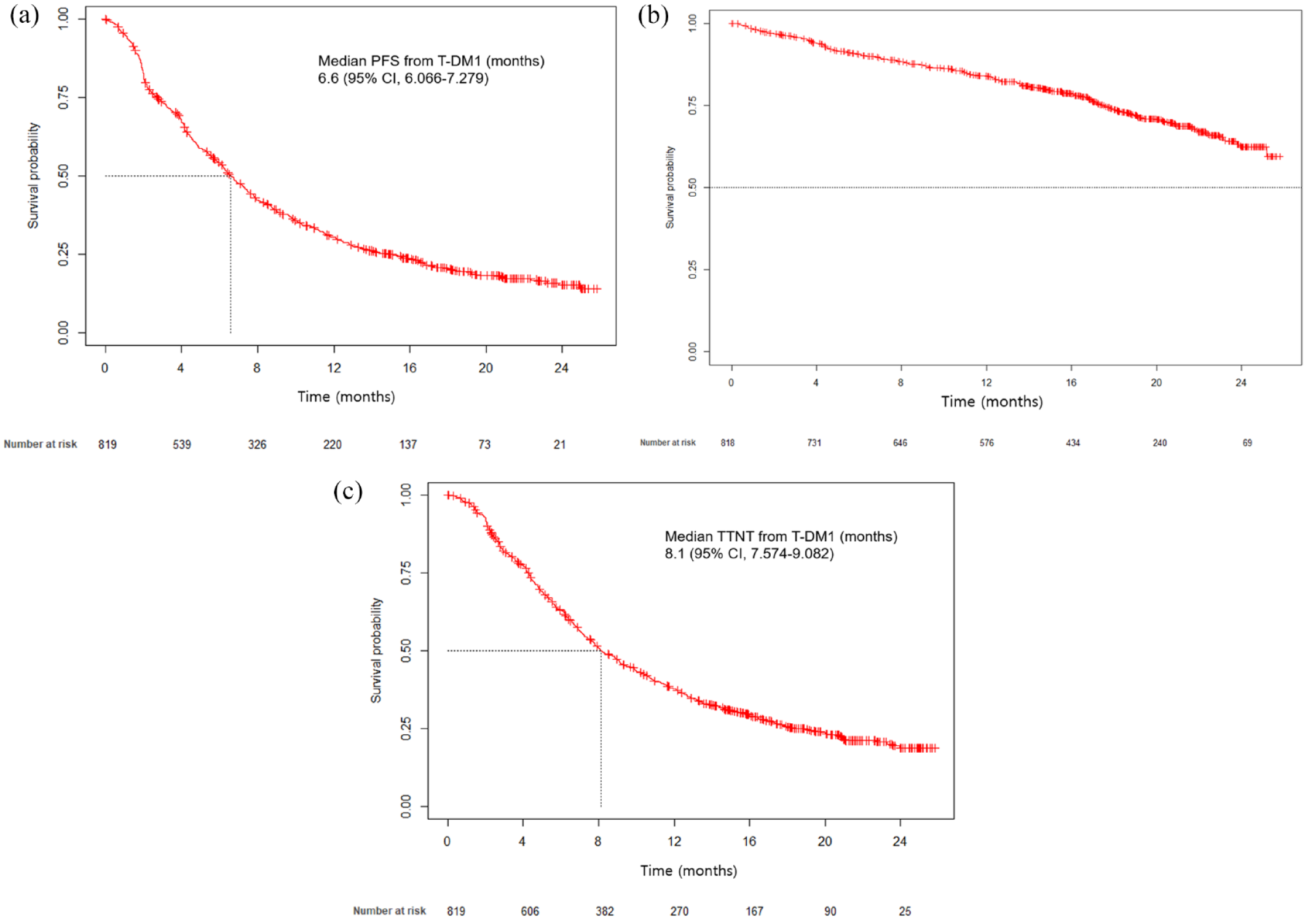
Kaplan–Meier estimates of (a) progression-free survival, (b) overall survival, and (c) time to next treatment.
The DCR and median PFS according to prior chemotherapy before T-DM1.
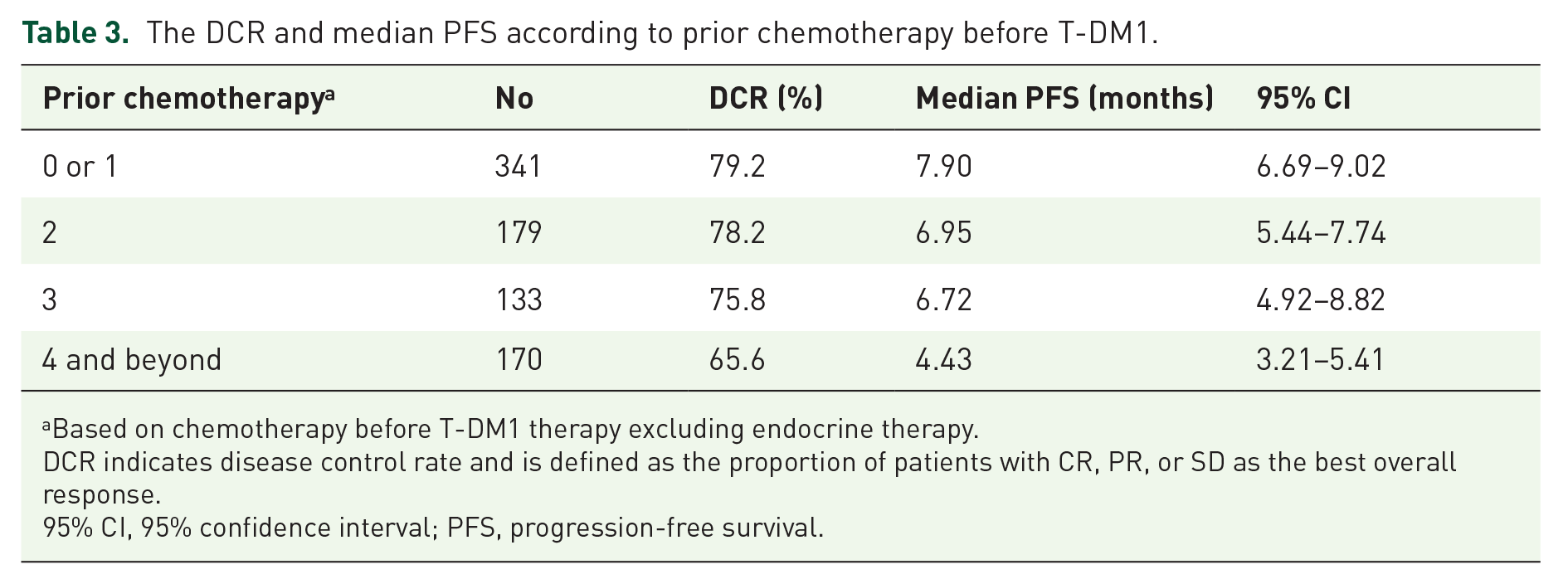
Based on chemotherapy before T-DM1 therapy excluding endocrine therapy.
DCR indicates disease control rate and is defined as the proportion of patients with CR, PR, or SD as the best overall response.
95% CI, 95% confidence interval; PFS, progression-free survival.

Kaplan–Meier curve of progression-free survival according to the number of prior chemotherapy.
Tables 4 and 5 show the clinical predictors of PFS and OS, respectively. The age [<65 years (HR) 1.581, p = 0.001], poor ECOG performance status (PS ⩾ 2, HR 2.199, p < 0.001), an advanced line of palliative chemotherapy before T-DM1 therapy (⩾2, HR 1.362, p = 0.030), prior pertuzumab use (HR 1.375, p = 0.015), and shorter treatment duration of palliative trastuzumab (<10 months, HR 1.623, p < 0.001) were associated with poor PFS. Poor ECOG performance status (PS ⩾ 2, HR 4.517, p < 0.001), prior pertuzumab use (HR 1.623, p = 0.040), prior lapatinib use (HR 2.118, p = 0.001), and shorter treatment duration of palliative trastuzumab (<10 months, HR 2.092, p < 0.001) were associated with poor OS.
Univariate and multivariate analyses for PFS from T-DM1 treatment.

CNS, central nervous system; ECOG PS, Eastern Cooperative Oncology Group Performance Status; HR, hazard ratio; PFS, progression-free survival.
Univariate and multivariate analyses for OS from T-DM1 treatment.
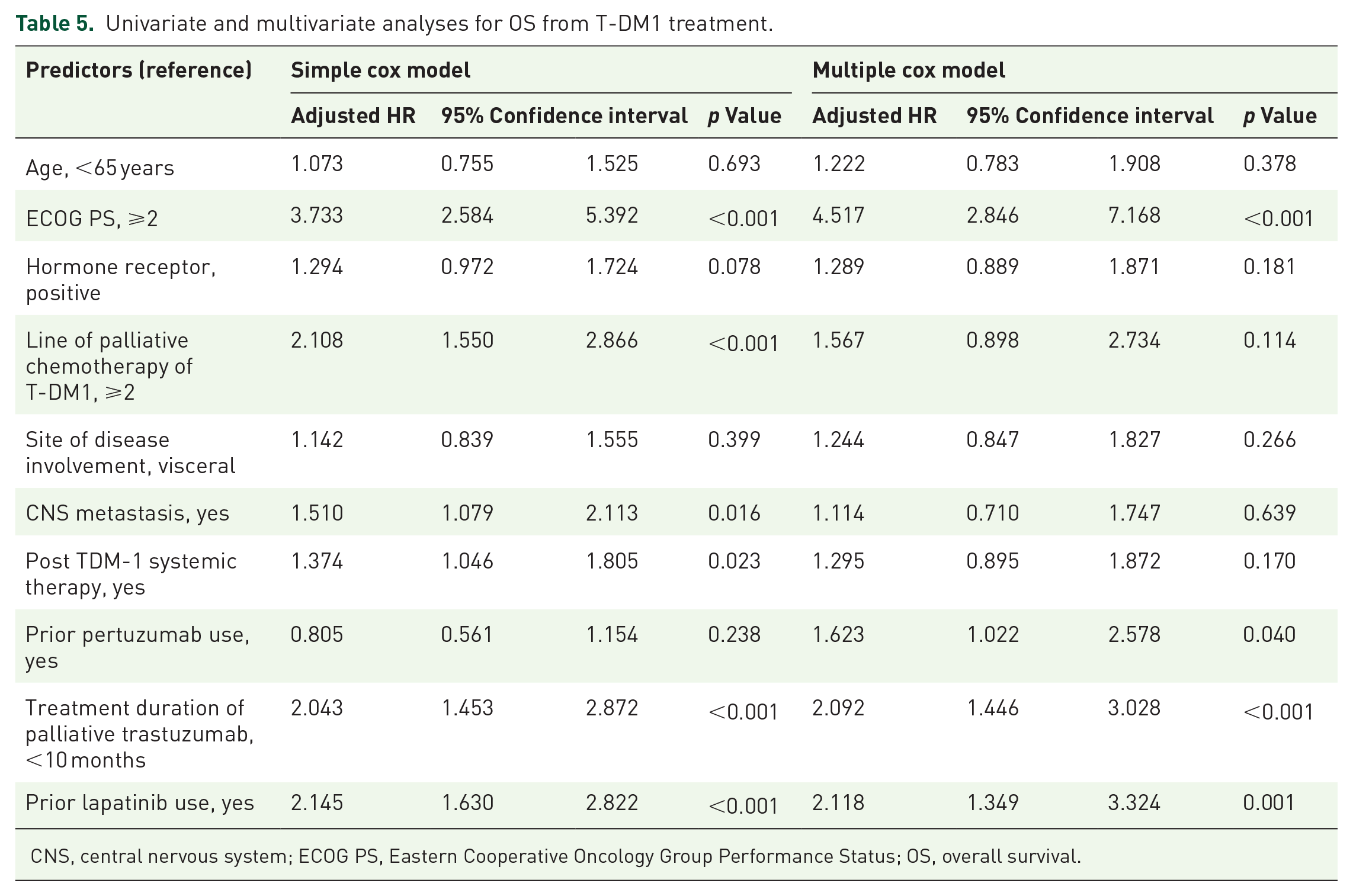
CNS, central nervous system; ECOG PS, Eastern Cooperative Oncology Group Performance Status; OS, overall survival.
Safety
A total of 154 (19%) patients underwent a dose reduction during T-DM1 therapy owing to toxicity. Patients who needed a dose reduction of T-DM1 received a median of five doses (range 1–31). Drug discontinuation owing to toxicity occurred in 76 patients who discontinued T-DM1, either temporarily (n = 28) or permanently (n = 48).
The most common adverse events were elevated AST/ALT levels (n = 318), thrombocytopenia (n = 324, 39.9%), anemia (n = 213, 26.4%), and fatigue (n = 203, 25.8%). Common grade 3–4 adverse events included thrombocytopenia (n = 107, 13.2%), neutropenia (n = 23, 2.8%), and elevated liver enzyme levels (n = 20, 2.5%). Hypokalemia (⩽3.0 mmol/L) was observed in 25 patients (3.1%). Bleeding events included epistaxis [61 (7.5%)], gum bleeding [13 (1.6%)], and others [20 (2.5%)]. Among 405 patients undergoing T-DM1 therapy whose ejection fractions were evaluated, a decreased ejection fraction was reported in 12 (3.0%). Table 6 shows the adverse events associated with T-DM1 use.
Incidence of adverse events with T-DM1 treatment.

Discussion
In this real-world study, we analyzed the data of 824 patients with HER2-positive advanced breast cancer who were treated with T-DM1 at 60 Korean medical centers. The efficacy and toxicity of T-DM1 were also evaluated. In the present study, the RR and median PFS of patients with T-DM1 were 35% and 6.6 months, respectively. Young age, poor ECOG performance status, a later line of palliative chemotherapy before T-DM1 administration, prior pertuzumab use, and shorter treatment duration with palliative trastuzumab were associated with poor PFS. Moreover, poor ECOG performance status, prior pertuzumab and lapatinib use, and shorter treatment duration with palliative trastuzumab were associated with poor OS.
We found that prior pertuzumab exposure was associated with a lower PFS. There have been no published RCTs on the efficacy of T-DM1 after pertuzumab–trastuzumab therapy. The available data are mainly from real-world studies with controversial results.24–28 A metanalysis to determine the efficacy of T-DM1 after first-line trastuzumab and pertuzumab use in HER2-positive breast cancer reported that the efficacy of T-DM1 after pertuzumab use was similar to that of the EMILIA study in the third line or more lines, but not in the second line. 29 Sanglier et al. reported that pertuzumab-treated patients entering the T-DM1 cohort earliest had more aggressive disease and poorer outcomes than patients entering the study in the most recent years and described this result as a selection bias. 30 The control group in DESTINY-Breast03, with 75% of patients receiving a prior second or later line, was similar to that of our study, with approximately 65% of patients receiving a prior second or later line. DESTINY-Breast03 reported that prior pertuzumab treatment had no impact on either T-DM1 or T-DXd outcomes in terms of absolute point estimates and HR. 16 Observation studies need caution in reporting the association between prior use of ‘new’ treatment and worse subsequent outcomes and interpreting them as generally rigorous causal effects.
In previous trials of T-DM1, patients’ age was not described as an independent factor for PFS. Cil et al. reported that the efficacy of T-DM1 was acceptable and that was well tolerated among older patients with HER2-positive mBC. 31 From the study of differential treatment effects between younger and older patients for new cancer therapies in solid tumors, a trial evaluating treatments for breast cancer showed a favorable trend among older patients compared with young patients. However, clinical trial participants differ from real-world patients older than 65 years, especially if they received second-line or later-line chemotherapy. Because older patients are relatively vulnerable to cytotoxic chemotherapy, those with good performance and less aggressive biology survived until they received T-DM1 after taxane and trastuzumab. Therefore, older age may be associated with a good prognosis in the real world data. The real-world data (RWD) of eribulin used as second- or later-line chemotherapy in mBC leads to a better prognosis in older patients. 32 Given the risk of false discoveries owing to complex factors, the results should be interpreted with caution, and further research is necessary.
With the development of powerful first-line therapeutic agents, the RCT results of second-line therapy in patients not receiving newly standardized first-line therapy could differ from those of patients who received second-line therapy after receiving the new standard therapy. Moreover, in DESTINY-Breast03, it was observed that approximately 25% of the patients in the T-DM1 group experienced progression in the second month, and their PFS decreased sharply at the beginning of the study. A similar initial drop has also been observed in patients with hormone receptor-positive and HER2-negative mBC using second-line hormone therapy after endocrine and cyclin-dependent kinase 4/6 (CDK4/6) inhibitor therapy, 33 and in patients with mBC resistant to aromatase inhibitors, such as those with ESR1 mutation. 34 The mechanisms underlying T-DM1 resistance are related to trastuzumab resistance and ADC (Antibody Drug Conjugate) and HER2 heterogeneity. Structural modifications of the trastuzumab-binding site and low tumor HER2 expression lead to poor internalization and defective intracellular trafficking of the T-DM1. 35 Moreover, increased activity and expression of drug efflux pumps and altered lysosomal transport are unique mechanisms of resistance to antibody–drug conjugates. 36 When T-DM1 administration immediately follows pertuzumab–trastuzumab combinations due to resistance, the cytotoxic effects of T-DM1 may be impaired by inefficient internalization in cancer cells. Lapatinib targets the tyrosine kinase (TK) domain, and its resistance is mainly related to alternative TK activation, the cross-talk between HER2 and estrogen receptors, or HER2 mutations (such as L755S), rather than extracellular domain abnormalities. 37
HER2 intratumoral heterogeneity is defined as the presence of at least two distinct clones of cells with varying HER2 statuses within the same tumor. The prevalence of HER2 heterogeneity is up to 40% of breast cancers. 38 This could be another potential factor contributing to resistance to T-DM1 treatment. The KRISTINE study compared neoadjuvant T-DM1 plus pertuzumab with docetaxel, carboplatin, trastuzumab, and pertuzumab for the treatment of HER2-positive stage II/III breast cancer. T-DM1 plus pertuzumab resulted in a higher risk of event-free survival owing to locoregional progression before surgery. Tumors from patients who experienced locoregional progression had lower HER2 expression and higher HER2 heterogeneity than those from other patients in the T-DM1 arm, suggesting that these patients may require conventional systemic chemotherapy combined with HER2 targeting agents rather than T-DM1 combined with pertuzumab. 39 In the metastatic setting, the phase III MARIANNE study examined the effects of HER2-related biomarkers on PFS in patients administered T-DM1 as a first-line chemotherapy. The performance of the T-DM1 arms was poor in patients with HER2 intratumoral heterogeneity compared to patients with homogeneous HER2 expression. 40 By contrast, T-DXd effectively targets tumor cells that express low levels of HER2 and delivers a potent cytotoxic payload (drug-to-antibody ratio of 8:1) through a bystander effect on neighboring tumor cells heterogeneously expressing HER2. 41 In the DESTINY-Breast04 trial involving patients with HER2-low metastatic breast cancer, T-DXd resulted in a significantly longer PFS and OS than the physician’s choice of chemotherapy. 42 Further understanding of HER2 heterogeneity within a tumor is important to guide treatment decisions including anti-HER2-targeted therapy, improve clinical outcomes, and develop novel anti-HER2 agents to overcome resistance.
Grade 3–4 adverse events, including thrombocytopenia (13.2%), neutropenia (2.9%), elevation of liver enzymes (2.5%), and hypokalemia (3.1%), were comparable to those reported in the EMILIA study (14.2%, 2.2%, 4%, and 2%, respectively). 43 The bleeding was not severe. In the DESTINY-Breast03 trial, drug-related treatment-emergent adverse events led to discontinuation in 20% of patients in the T-DXd group and 7% of patients in the T-DM1 group. The most common drug-related treatment-emergent adverse events that led to discontinuations of T-DXd were pneumonitis, interstitial lung disease, and pneumonia. Drug-related treatment-emergent adverse events that led to drug interruption occurred in 42% and 17% of patients in the T-DXd and T-DM1 groups, respectively. 17 T-DXd requires a safety profile with longer follow-ups. Therefore, T-DM1 will still play an important role in patients living in countries where T-DXd is not available or in patients who have discontinued the drug owing to T-DXd-related adverse events.
The present study has some limitations, mostly related to its retrospective design and the heterogeneity of the study population. The large number of participating centers and real-world practice settings added heterogeneity to the study population. Furthermore, the RECIST criteria and timing of tumor reassessment were less stringent and precise than those in patients from randomized clinical studies. Initially, we proposed a data-collection framework for EMR-based RWD to evaluate the effectiveness and safety of cancer drugs. This framework included reliability tests for investigators and collected data, site training programs, an EDC (Electronic Data Capture) system consisting of filtering and a query-generating system, and verification of the reproducibility of the data analysis. 44 This systematic collection process was based on the principles of the FDA’s RWE program and was prospectively planned and conducted to minimize bias and increase reliability. As described in the Data Collection section, this study was based on the response evaluation of RECEIST. Response evaluation in HER-derived RWD has many limitations, and response evaluation standards for RWD have not yet been proposed in Korea, as in the United States. 45 Second, the toxicity profile obtained using CTCAE with retrospective data was not ideal. In this study, subjective symptoms were collected only for nausea, vomiting, diarrhea, and fatigue, and most toxicities depended on laboratory findings. Subjective toxicities could be at high risk of under-reporting by physicians, even when prospectively collected within randomized trials. 46 Directly collecting this information from patients through patient-reported outcomes or digital healthcare data can improve the reliability and precision of symptomatic adverse events detection. Nevertheless, this study enrolled more than 800 patients with HER2+ mBC treated with T-DM1 and analyzed the effectiveness and predictors of the outcomes of T-DM1 therapy in a real-world setting. These results will contribute to drug selection after second-line treatment or in countries or cases where the use of T-DXd is restricted, as various new HER2-targeted drugs are being developed.
In conclusion, this large-scale analysis suggests that the efficacy and safety of T-DM1 in HER2+ mBC in real-world practice are comparable to those reported in RCTs. Our results also offer interesting clues regarding the treatment sequences and prognosis in the population treated with T-DM1 after multiple HER2-targeted therapies using clinical predictors. Various HER2-targeted therapies and new treatments have been developed. Further studies exploring the multiple mechanisms of resistance to these agents and developing potential strategies to overcome them are crucial to improve the outcomes of HER2+ mBC treatment.
Footnotes
Acknowledgements
This work was supported by the Korean Cancer Study Group and the Health Insurance Review & Assessment Service. The views expressed are those of the authors and not necessarily those of the Health Insurance Review & Assessment Service. We thank all the investigators and their support staff who generously participated in this study.
