Abstract
Background:
Docetaxel, following progression on immunotherapy and platinum-based chemotherapy, remains the standard of care for advanced non-small-cell lung cancer (NSCLC) but offers limited promise. Antibody–drug conjugates (ADCs) may improve outcomes in this population.
Objectives:
Data from the literature, mainly randomized controlled trials (RCTs), have shown discrepancies. This report evaluates the efficacy and safety of ADCs versus docetaxel in previously treated advanced NSCLC.
Design:
The systematic review and meta-analysis was conducted focusing on phase II/III RCTs to synthesize available evidence regarding efficacy outcomes and safety of ADCs compared to docetaxel.
Data resources and methods:
Databases (PubMed (MEDLINE), EMBASE, and Cochrane Library), clinical trial registries, and proceedings of global oncology conferences from January 2015 to November 2024 were screened comparing ADC versus docetaxel. Two researchers independently completed data retrieval and screening work using Covidence. The Cochrane Risk of Bias Tool (RoB 2.0) was used to assess the methodological quality of the included RCTs. The primary outcomes include progression-free survival (PFS) and overall survival (OS), while the secondary outcomes include objective response rate (ORR), disease control rate (DCR), and adverse events (AEs). The pooled hazard ratios (HRs) and odds ratios (ORs) were meta-analyzed using the appropriate generic variance and Mantel-Haenszel methods. Random-effect models were used to compute pooled estimates.
Results:
Of the 212 records screened, three RCTs involving 1597 patients were included. ADCs did not significantly improve PFS (pooled HR: 0.91, 95% CI: 0.73–1.13). For OS, the pooled HR was 0.88 (95% CI: 0.78–1.00, p = 0.06), reflecting a borderline significant trend favoring ADCs, with negligible heterogeneity (I2 = 0%). Furthermore, subgroup analysis demonstrated a significant OS benefit in the nonsquamous cohort (HR: 0.85, 95% CI: 0.74–0.98, p = 0.03). In addition, no difference was observed in ORR (OR: 1.16, 95% CI: 0.54–2.51) and DCR (OR: 1.39, 95% CI: 0.75–2.57). Grade ⩾ 3 treatment-related AEs were significantly lower (pooled OR: 0.49, 95% CI: 0.26–0.90, p = 0.02) with ADCs, despite high heterogeneity (I2 = 87%).
Conclusion:
Overall, there was no difference between the docetaxel and the ADC treatment arms; however, our findings report a significant survival benefit in the subgroup of patients with nonsquamous NSCLC pathology treated with ADCs compared to docetaxel with a manageable safety profile. Further research is needed to address heterogeneity, refine patient selection, and obtain more mature survival data with predictive biomarkers.
Introduction
Lung cancer remains the leading cause of cancer-related mortality worldwide, typically presenting in advanced stages with poor survival and reduced quality of life. 1 Non-small-cell lung cancer (NSCLC) makes up most lung cancer diagnoses, and it is known for its high recurrence rates and limited treatment options following disease progression. In recent years, due to the availability of biomarkers and driver molecular testing via next-generation sequencing (NGS) to select patients for targeted treatment or immunotherapy, successful outcomes in treating these cancers have led to improved survival. Patients with actionable genomic alterations (AGA) often receive personalized treatment with tyrosine kinase inhibitors (TKIs), which target the genomic alterations within their cancer. 2 Available first-line (1L) treatments for metastatic AGA-negative NSCLC, such as platinum-based chemotherapy and immunotherapy, offer short-term benefits; however, the disease progresses in most patients after 6–12 months of therapy, and “resistance” to the original agent often develops.3,4 Based on programmed death ligand 1 (PD-L1) expression score, single-agent immunotherapy or combination chemotherapy and immunotherapy are recommended in the 1L setting for these patients.5 –10
Palliative docetaxel is the current standard of care second-line (2L) chemotherapy in the management of patients with metastatic NSCLC following disease progression on chemotherapy and immunotherapy, given either concurrently or sequentially. 11 However, docetaxel chemotherapy offers limited efficacy, with literature reporting a median progression-free survival (PFS) of approximately 3.0 months and a median overall survival (OS) of 9 months 11 and potential side effects, highlighting an unmet medical need for better and safer treatment options for these patients. Various clinical trials have been conducted to investigate novel therapeutic agents and regimens for advanced NSCLC, yielding promising results. Antibody–drug conjugates (ADCs) comprise an innovative class of biopharmaceutical agents widely used for cancer treatment. Each ADC consists of a monoclonal antibody targeting a tumor-associated antigen linked to a cytotoxic agent (payload) via a chemical linker. This precise mechanism of ADCs allows focused target specificity and strong anti-tumor efficacy, as illustrated in Figure 1. Moreover, because the cytotoxic payload is attached to the hydrophilic antibody, the non-specific uptake of ADC by antigen-negative cells is minimized, resulting in less harm to healthy cells and tissues, thereby enhancing their therapeutic index. 12 Gemtuzumab ozogamicin was the first ADC to receive final approval in 2017 from the U.S. Food and Drug Administration (FDA) for cancer-targeted therapy in acute myeloid leukemia (AML). 13 Since then, many more ADCs have been investigated in clinical trials and received regulatory approvals in North America.

Mechanism of selected ADCs in lung cancer. Sacituzumab govitecan (a) targets TROP2 to deliver SN-38, a topoisomerase I inhibitor. Datopotamab deruxtecan (b) also targets TROP2, releasing a deruxtecan payload that causes DNA damage. Tusamitamab ravtansine (c) targets CEACAM5, DM4 to inhibit microtubule assembly and induces apoptosis, leading to cell death.
In August 2022, Fam-trastuzumab deruxtecan-nxki was the first FDA-approved ADC for metastatic NSCLC. The approval of this drug was based on the objective response rate (ORR: 58%; 95% CI: 43–71) and duration of response (DOR: 8.7 months; 95% CI: 7.1, not estimable (NE)) as per the DESTINY-Lung02 study, which evaluated Fam-trastuzumab deruxtecan activity for previously treated HER2 (ERBB2)-positive advanced nonsquamous NSCLC. 14 Most recently, telisotuzumab vedotin-tllv also received FDA approval in May 2025 based on the LUMINOSITY study, which reported an ORR of 35% (95% CI: 24–46) and median DOR of 7.2 months (95% CI: 4.2–12) in patients with previously treated nonsquamous locally advanced or metastatic lung disease, with high c-Met protein overexpression (⩾50% of tumor cells with strong (3+) staining). 15 Although common adverse events across various ADC combinations include thrombocytopenia, neutropenia, anemia, leukopenia, peripheral neuropathy, nausea, fatigue, diarrhea, skin rash, pneumonitis/interstitial lung disease (ILD), and musculoskeletal and ocular toxicity,16,17 further studies are ongoing to address how to reduce the off-target toxicity of ADCs, thereby allowing for safe dosages and optimal anti-cancer effects. 16
A few randomized clinical trials (RCTs) have compared the survival outcomes and toxicity of different ADCs against docetaxel in patients with pretreated NSCLC and reported their results. A better understanding of ADCs in this population cohort may offer safer subsequent treatment options with novel therapeutic agents after disease progression. This systematic review and meta-analysis (MA) is the first of its kind to evaluate the efficacy and safety of ADCs versus docetaxel in patients with advanced, previously treated NSCLC and explores how this new therapeutic approach can be used to revolutionize treatment options in this patient population.
Materials and methods
We conducted a systematic review using the methods of the Cochrane Database of Systematic Reviews. The meta-analysis follows the guidelines of the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 18 and adheres to the AMSTAR2 checklist 19 (Supplemental Files 1 and 2). The review was registered in the International Prospective Register of Systematic Reviews (PROSPERO) database with the ID CRD420251030747.
Data sources and search strategy
A comprehensive systematic search was conducted in PubMed (MEDLINE), EMBASE, and the Cochrane Library databases to identify relevant RCT publications from January 01, 2015, to November 30, 2024. The search techniques incorporated Medical Subject Headings (MeSH) terms and free-text keywords related to NSCLC, ADCs (e.g., sacituzumab govitecan, datopotamab deruxtecan, tusamitamab ravtansine), and docetaxel. The clinical librarian developed the systematic review search strategy under the guidance of senior authors (Supplemental File 3). The subjects were limited to humans; no filter was applied to the study language. Furthermore, to ensure the inclusion of recent and unpublished data, we also searched for clinical trial registries, including clinicatrails.gov, the World Health Organization (WHO) clinical trial registry platform and proceedings from major oncology conference meetings such as WCLC (World Conference on Lung Cancer), ESMO (European Society for Medical Oncology), and ASCO (American Society of Clinical Oncology). All the abstracts were shortlisted, and the duplicates of relevant studies were removed. Two researchers independently completed data retrieval and screening work using Covidence software, initially blinded for anonymity. Potentially eligible studies underwent a full-text review using the PICOS (population, intervention, comparison, outcome, and study design), as seen in Table 1. Extensive discussions and consensus with a third reviewer resolved the disputes.
PICOS eligibility criteria.

ADC, antibody–drug conjugate; NSCLC, non-small-cell lung cancer; RCTs, randomized controlled trials; TRAEs, treatment-related adverse events.
Study selection
Inclusion criteria
We included the studies if they fulfilled the following criteria: (1) enrolled patients aged 18 and above with histologically proven stage IV NSCLC (adenocarcinoma, squamous cell carcinoma, adenosquamous, or mucinous adenocarcinoma pathology) with or without AGA mutation, who had received at least one prior line of systemic chemotherapy; (2) compared an ADC with docetaxel in a 2L or subsequent line setting; and (3) phase II or III RCTs.
Exclusion criteria
We excluded the studies if they (1) were non-randomized, such as observational studies, case reports/series, or editorials; (2) compared docetaxel to a class of drugs other than ADCs; and (3) were experimental studies with insufficient data.
Study outcomes
The primary outcomes of this systematic review and meta-analysis are progression-free survival (PFS) and overall survival (OS). By contrast, the key secondary outcomes include objective response rate (ORR), disease control rate (DCR), and treatment-related adverse events (TRAEs). We included studies that reported at least one of these outcomes. PFS was defined as the time from the treatment initiation until disease progression or death from any cause. OS was defined as the time from the treatment initiation until death from any cause. ORR was defined as the best overall response with confirmed complete response (CR) or partial response (PR) as per the Response Evaluation Criteria in Solid Tumors (RECIST) v1.1. DCR was defined as the sum of the overall response, with confirmed CR, PR, or stable disease (SD) as the best response, assessed according to the RECIST v1.1. TRAEs were graded as per Medical Dictionary for Regulatory Activities (MedDRA) version 26.0 and National Cancer Institute Common Terminology Criteria for Adverse Events version 5.0.
Data extraction and quality assessment
Two authors performed the data extraction independently following the PRISMA guidelines, and the senior authors reviewed the extracted data for any discrepancies. The key data extraction variables include the study’s first author, publication year, study design, sample size, clinical trial phase, patient demographics (age, gender, smoking history, performance status, disease histology), ADC type and docetaxel, and outcome measures (PFS, OS, ORR, DCR, TRAEs). The extracted data were uploaded onto the Microsoft Excel sheet. The Cochrane Risk of Bias Tool (RoB 2.0) was used to assess the methodological quality of the included RCTs, evaluating randomization, allocation concealment, blinding, incomplete outcome data, selective reporting, and other biases. 20 The conflicts in quality assessment were resolved by joint consensus.
Data synthesis and statistical analysis
Hazard ratios (HRs) with 95% confidence intervals (CIs) were extracted for time-to-event outcomes (PFS and OS), and odds ratios (ORs) with 95% CIs were computed for dichotomous outcomes (ORR, DCR, and TRAEs). The pooled HR and OR were meta-analyzed using the generic variance and Mantel-Haenszel methods as appropriate. Furthermore, random-effect models were used to derive pooled estimates, which were then visualized through forest plots. The heterogeneity between studies was quantified using the I2 statistics, with range values of 0%–40%, 30%–60%, 50%–90%, and 75%–100%, indicating low/negligible, moderate, substantial, and considerable (high) heterogeneity, respectively. The classification of heterogeneity based on I2 statistics with these ranges is utilized from the Cochrane Handbook of Systematic Review of Intervention (Chapter 10; Version 6.5, 2024). 21 The subgroup analysis was pre-specified to explore potential effect modifiers (squamous vs nonsquamous cell histology).
Ethical considerations
This systematic review and meta-analysis utilized aggregated data from published randomized controlled trial results; thus, institutional ethical review committee (ERC) approval was not required, as no patient identifiers were disclosed. The review adhered to and followed the PRISMA guidelines.
Results
Study search and study characteristics
The initial literature search yielded 224 studies; however, after removing duplicate studies, 212 records remained. Two hundred and six records were excluded after the complete screening of titles and abstracts, and six reports were assessed for eligibility and underwent full-text screening. Furthermore, records were also identified from citation searching and conference reviews; however, these were duplicates of abstracts from the initial literature search and post hoc exploratory reports. Eventually, three studies met our inclusion criteria, as illustrated in Figure 2. The three studies, involving 1597 patients, utilized to extract data are all open-label, multicenter, and multinational, phase III RCTs, namely, EVOKE-01, TROPION-Lung01, and CARMEN-LC03, further characterized in Tables 2 and 3. Overall, the report showed low-risk bias across all domains, with no study identified as high risk of bias.

PRISMA flow diagram of the study selection to be included in the systematic review and meta-analysis.
Baseline patient characteristics of the included studies.
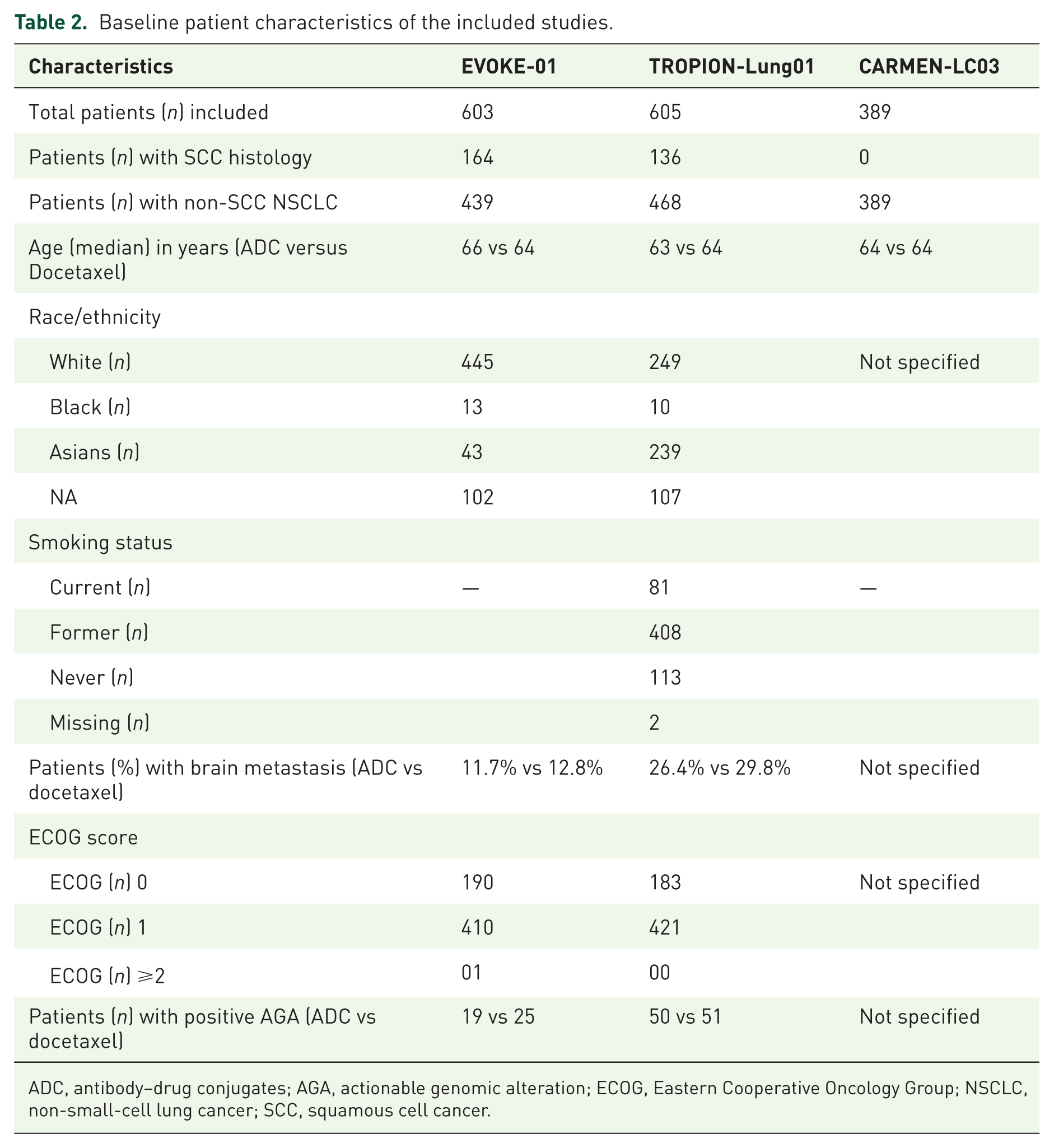
ADC, antibody–drug conjugates; AGA, actionable genomic alteration; ECOG, Eastern Cooperative Oncology Group; NSCLC, non-small-cell lung cancer; SCC, squamous cell cancer.
Study characteristics of the included studies.

Clinically relevant and most commonly seen with datopotamab deruxtecan.
Clinically relevant and commonly reported with sacituzumab govitecan.
ADC, antibody–drug conjugate; DCR, disease control rate; DOR, duration of response; FN, febrile neutropenia; ILD, interstitial lung disease; ORR, objective response rate; OS, overall survival; PFS, progression-free survival; QOL, quality of life; RCT, randomized controlled trial; TRAEs, treatment-related adverse events.
Studies comparing ADCs and docetaxel in recurrent advanced NSCLC
Luis G. Paz-Ares et al. conducted an open-label phase III randomized controlled study (EVOKE-01) that evaluated the safety and efficacy of the antibody–drug conjugate sacituzumab govitecan (SG) compared with docetaxel, a standard subsequent-line treatment after progression on platinum-based chemotherapy/chemoimmunotherapy or targeted drugs in patients with stage IV NSCLC. 22 This study enrolled 603 patients, 299 of whom were assigned to SG, and 304 were assigned to docetaxel. Patients in the SG arm received one 10 mg/kg intravenous infusion on day 1 and day 8, and patients in the docetaxel arm received one 75 mg/m2 intravenous infusion on day 1 of every 21-day cycle. The study had a median follow-up of 12.7 months (range, 6.0–24.0). The median OS, the study’s primary endpoint, was 11.1 months in the SG arm, compared to 9.8 months with docetaxel, with an HR of 0.84 (95% CI: 0.68–1.04) and a p-value of 0.0534. Although the trial did not meet the statistical significance threshold, it showed a numerical improvement of 1.3 months in the median OS, favoring SG. The study also reported consistent OS results across both squamous and nonsquamous histologies. A modest and nonsignificant improvement in median PFS of 4.1 months with SG compared to 3.9 months with docetaxel (HR = 0.92, 95% CI: 0.77–1.11) suggested that the clinical benefit may not be fully captured in the study’s heavily pretreated population. The ORR was 13.7% (95% CI: 10.0–18.1) with SG and 18.1% (95% CI: 13.9–22.9) with docetaxel, with a difference of 4.3% (95% CI: 10.1–1.5). Interestingly, in the subgroup of patients who were non-responsive to their last PD-1/PD-L1-based treatment, a clinically significant benefit was observed, with a median OS of 11.8 months with SG versus 8.3 months with docetaxel and an HR of 0.75 (95% CI: 0.58–0.97). The safety and tolerability favor SG, with fewer grade 3 or more TRAE observed than with docetaxel (52% vs 60%). Despite narrowly missing statistical significance for the primary endpoint, the EVOKE-01 study presents consequential insights.
In another phase III study (TROPION-Lung01), Myung-Ju Ahn et al. assessed the efficacy and safety of a TROP2 (trophoblast cell-surface antigen 2)-directed ADC, datopotamab deruxtecan (Dato-DXd), compared to docetaxel in patients with previously treated stage IV NSCLC. 23 The study targeted a biomarker-unselected cohort, adding to its generalizability across various NSCLC populations. The study enrolled 604 patients, 299 in the Dato-DXd group and 305 in the docetaxel group. Most (>76%) patients had nonsquamous cell histology, and over 90% had received prior immune checkpoint inhibitors. Actionable genomic alterations were present in 16.7% of patients with NSCLC in both groups. Almost an equal number of patients in both groups have brain metastasis. Patients randomized to the Dato-DXd arm received a 6 mg/kg intravenous infusion, and patients in the docetaxel arm received a 75 mg/m2 intravenous infusion once every 3 weeks. Dato-DXd significantly improved PFS, with a median PFS of 4.4 months compared to 3.7 months with docetaxel and an HR of 0.75 (95% CI: 0.62–0.91; p = 0.004). The early part of the Kaplan–Meier (KM) curve showed a clear separation between the two arms that is consistent throughout. Although the absolute median PFS difference is slight (0.7 months) with a 6-month PFS rate of approximately 40% versus 25%, respectively, the tail of the curve suggests prolonged disease control in a subset of patients who received Dato-DXd. Additionally, PFS was significantly longer in the nonsquamous cohort with an HR of 0.63 (95% CI: 0.50–0.79). The ORR with Dato-DXd was 26.4% (95% CI: 21.4–31.8) compared to 12.8% (95% CI: 9.3–17.1) with docetaxel, reflecting a rate of tumor shrinkage more than double that of docetaxel. Although the study was not powered to calculate OS superiority at the data cutoff, a numerically insignificant trend favored Dato-DXd with a median OS of 12.9 months compared to 11.8 months with docetaxel (HR = 0.94; 95% CI: 0.78–1.14). The benefit of Dato-DXd was observed regardless of PD-L1 expression and in patients with AGA alterations within the nonsquamous subgroup. Interstitial lung disease (ILD) risk was relatively high with Dato-DXd, reaching approximately 8.8% of the treated patients, necessitating close monitoring to minimize the risk; however, grade 3 TRAEs were fewer with Dato-DXd compared to docetaxel (25.6% vs 42.1%). The significant improvement in response rate, PFS, and acceptable safety and tolerability highlighted the potential use of Dato-DXd in nonsquamous NSCLC. The lack of benefit in squamous cell histology necessitates further exploration regarding biological differences and predictive biomarkers for response.
By contrast, Benjamin Besse and colleagues did a biomarker-selective study (CARMEN-LC03) assessing the safety and efficacy of tusamitamab ravtansine (tusa rav), a CEACAM5 (carcinoembryonic antigen-related cell adhesion molecule 5)-targeting ADC, compared to docetaxel in previously treated advanced nonsquamous NSCLC patients. 24 One hundred and ninety-four patients were randomized to receive an intravenous tusa-rav infusion of 100 mg/m2 once every 2 weeks, and 195 received an intravenous docetaxel infusion of 75 mg/m2. Based on immunohistochemical (IHC) expression, patients with high CEACAM5 expression levels were included in the study. The median PFS was not significantly improved with tusa-rav compared to docetaxel, with comparable ORR between the two groups. However, a numerically insignificant trend toward improved median OS (12.8 vs 11.5 months; HR = 0.85; 95% CI: 0.64–1.11) was observed with tusa-rav in the entire cohort with a favorable safety profile. Tables 2 and 3 summarize the patient’s baseline, disease characteristics, and outcomes.
Primary outcomes of meta-analysis
Progression-free survival
The forest plot (Figure 3(a) and (b)) summarizes the HRs from three studies (CARMEN-LC03, EVOKE-01, and TROPION-Lung01) evaluating the effectiveness of ADCs compared to chemotherapy. The pooled analysis of all studies yielded an HR of 0.91 (95% CI: 0.73–1.13), suggesting a slight reduction in risk with ADCs compared to chemotherapy; however, this result was not statistically significant. Heterogeneity analysis revealed substantial heterogeneity (I2 = 67%, Chi2 = 6.12, p = 0.05), showing divergence among the included studies.

Forest plots of pooled analysis of PFS. (a) Forest plot of pooled analysis of PFS from studies included in the meta-analysis. (b) Forest plot of a pooled analysis of PFS based on histological subtypes from all studies included in the meta-analysis.
In the squamous subgroup, data from two studies (EVOKE-01 and TROPION-Lung01) yielded a pooled HR of 1.14 (95% CI: 0.77–1.68), indicating no statistically significant difference between the two treatments. Similarly, in the nonsquamous subgroup, which includes data from all three studies, the pooled HR is 0.87 (95% CI: 0.62–1.21), again showing no significant advantage for either treatment. Moderate heterogeneity is observed in the squamous subgroup (I2 = 57%, p = 0.13). In comparison, high heterogeneity is noted in the nonsquamous subgroup (I2 = 83%, p = 0.003) and the overall analysis (I2 = 77%, p = 0.002), reflecting variability among the included studies. These findings imply no clear superiority of ADCs over chemotherapy; however, the significant heterogeneity, particularly in the nonsquamous subgroup, warrants further investigation.
Overall survival
The forest plot (Figure 4(a) and (b)) summarizes the HRs from three studies for OS. None of these individual studies demonstrated statistically significant differences. When pooled, the overall HR was 0.88 (95% CI: 0.78–1.00), indicating a trend favoring ADCs over chemotherapy, although the result narrowly missed statistical significance (p = 0.06). Heterogeneity was negligible (I2 = 0%, Chi2 = 0.77, p = 0.68), suggesting that the study results were consistent.

Forest plots of pooled analysis of OS. (a) Forest plot of pooled analysis of OS from studies included in the meta-analysis. (b) Forest plot of pooled analysis of OS based on histological subtypes from all studies included in the meta-analysis.
Squamous subgroup analysis of OS from two studies (EVOKE-01 and TROPION-Lung01) yielded a pooled HR of 1.05 (95% CI: 0.66–1.66), indicating a statistically nonsignificant difference between the two treatments, with substantial heterogeneity observed (I2 = 66%, p = 0.09). Interestingly, nonsquamous subgroup data from three studies (CARMEN-LC03, EVOKE-01, and TROPION-Lung01) revealed a pooled HR of 0.85 (95% CI: 0.74–0.98, p = 0.03), representing a statistically significant survival benefit for ADCs in this subgroup, underscoring their therapeutic promise beyond traditional chemotherapy. No heterogeneity was noted (I2 = 0%, p = 0.98), indicating consistency across studies. Furthermore, no significant heterogeneity was found between the squamous and nonsquamous subgroups (I2 = 0%), indicating that the observed differences in treatment effect between subgroups are consistent.
Secondary outcomes of meta-analysis
Objective response rate
The pooled analysis from all three studies showed an overall odds ratio (OR) of 1.16 (95% CI: 0.54–2.51), indicating no statistically significant difference in the ORR between the two treatment modalities (Figure 5). High heterogeneity (I2 = 89%) among the studies suggests considerable variability in the results, likely due to differences in study populations. The overall effect (Z = 0.37, p = 0.71) further confirms the lack of statistical significance in the pooled analysis. While TROPION-Lung01 (OR: 2.46, 95% CI: 1.61–3.75) shows a significant benefit for ADCs over chemotherapy, the pooled analysis findings do not consistently support an advantage, and the high heterogeneity warrants cautious interpretation.

Forest plot of pooled analysis of ORR from all studies included in the meta-analysis.
Disease control rate
The pooled analysis of the studies (EVOKE-01 and TROPION-Lung01) compares the effectiveness of ADCs versus chemotherapy in terms of DCR based on outcomes measured by OR, with 95% CI revealing no statistically significant difference across the studies (OR: 1.39; 95% CI: 0.75–2.57) (Figure 6). Although the TROPION-Lung01 study demonstrated a clear DCR benefit of ADC over chemotherapy (OR: 1.91; 95% CI: 1.34–2.71), the overall odds ratio and high heterogeneity (I2 = 84%, Chi2 = 6.23, p = 0.01) do not provide conclusive evidence due to inconsistencies between the studies.

Forest plot of pooled analysis of DCR from studies included in the meta-analysis.
Treatment-related adverse events
The pooled analysis of all studies revealed no significant differences in the occurrence of TRAE of any grade between ADCs and chemotherapy, with an overall OR of 1.06 (95% CI: 0.39–2.84) and a trend favoring ADCs. However, a statistically significant reduction in grade ⩾ 3 TRAE was observed with ADCs (OR: 0.49; 95% CI: 0.26–0.90; p = 0.0004). High heterogeneity was observed (I2 = 87%). Nevertheless, the forest plot (Figure 7(a) and (b)) favored ADCs with the pooled effect, illustrating a clinically meaningful reduction in severe adverse events with ADCs.

Forest plots of pooled analysis of TRAE. (a) Forest plot of pooled analysis of TRAE of any grade from studies included in the meta-analysis. (b) Forest plot of a pooled analysis of TRAE of grade ⩾3 from studies included in the meta-analysis.
Discussion
The recent advent of ADCs as an innovative therapeutic class has revamped the management landscape of advanced NSCLC, specifically for patients with limited options following the progression of chemotherapy and immunotherapy. This systematic review and meta-analysis consolidated data from three crucial trials, namely, EVOKE-01, TROPION-Lung01, and CARMEN-LC03, delivering a timely synthesis of evidence comparing ADCs with standard-of-care chemotherapy (docetaxel) in a subsequent-line setting in NSCLC.
Most patients with AGA-negative advanced NSCLC received a combination of platinum-based chemoimmunotherapy or single-agent immunotherapy, depending on the PD-L1 TPS (tumor proportion score) status. In selected patients with oligoprogressive disease, local therapy with continued maintenance treatment can be considered 25 ; for other patients who have progressed on 1L chemoimmunotherapy, palliative docetaxel chemotherapy is preferred. 11 In prospective RCTs, second-line docetaxel improved OS by 2.9 months compared with corresponding best supportive care patients (7.5 vs 4.6 months; log-rank test, p = 0.010; 1-year survival, 37% vs 11%; chi (2) test, p = 0.003), with less deterioration in quality of life (QOL).26,27 Although limited data are available, single-agent gemcitabine or vinorelbine may be considered as a subsequent-line treatment in patients for whom other options have been exhausted.28,29 In a randomized phase III study (REVEL) by Garon EB et al., patients post-progression on platinum-based systemic treatment received a survival benefit with a median OS of 9.1 months with docetaxel and 10.5 months with docetaxel and ramucirumab (HR: 0.86, 95% CI 0.75–0.98; p = 0.023). The subgroup analysis of this study by Paz-Ares LG et al. showed consistent benefits across histological subtypes. 30 Moreover, in further efforts to improve survival, Capelletto et al. investigated docetaxel plus nintedanib in nonsquamous NSCLC in a second-line setting and beyond; however, the study (SENECA) uncovered results with modest efficacy, significantly affecting QOL. 31 Hence, there is an unmet clinical need for a safe and effective therapy that extends survival for such a cohort with the preservation of QOL.
This systematic review and meta-analysis investigated safety and efficacy outcomes stratified by histo-pathological subtypes and treatment-related toxicities. It offered thoughtful insights into the therapeutic value and limitations of ADCs in the rising context of NSCLC treatment. The overall survival, the primary outcome of this meta-analysis, demonstrated a pooled HR of 0.88 (95% CI: 0.78–1.00), suggesting a favorable trend for ADCs over systemic chemotherapy. Although the finding narrowly escapes statistical significance (p = 0.06), the consistency across the included studies, displayed by negligible heterogeneity (I2 = 0%), strengthens the reliability of the notable trend. Individual studies such as EVOKE-01 and CARMEN-LC03 reported HRs of 0.84 for OS, accentuating a reproducible direction of benefit. The findings from TROPION-Lung01 (HR: 0.94; 95% CI: 0.78–1.14) further reinforce this trend, although it did not reach significance individually. These results insinuate a substantial survival advantage trend with ADCs, which may become significant in a more refined patient cohort or with longer follow-ups.
Moreover, subgroup analysis of histologic subtypes exhibits more captivating evidence of differential treatment effects. In the nonsquamous NSCLC cohort, a statistically significant OS benefit appeared with a pooled HR of 0.85 with a 95% CI of 0.74–0.98 (p = 0.03). A negligible heterogeneity (I2 = 0%) further endorsed complete consistency across the studies, contributing to this subgroup analysis. Such uniformity bolsters confidence in the solidarity of survival benefits in the nonsquamous NSCLC. These findings suggest that ADCs benefit selected cohorts due to the differential expression of target antigens such as CEACAM5 or TROP2, as well as pharmacokinetic and pharmacodynamic properties and the tumor microenvironment.
Different studies have evaluated the role of biomarker expression in tumor tissue and plasma, which may benefit in strategizing the treatment paradigm in NSCLC.32,33 ICARUS-LUNG01, which is a single-arm phase II study currently evaluating biomarkers of response and resistance to Dato-DXd in heavily pretreated NSLC patients by utilizing TROP2 tumor membrane expression, TROP2-dynamics and spatial distribution (by AI-digital pathology), genomics, transcriptomic, spatial proteomics (by imaging mass cytometry), and circulating tumor cells. 34 The survival advantage observed in our meta-analysis is aligned with the findings from the phase II non-randomized trials, ICARUS-LUNG01 (median OS of 12.6 months in nonsquamous NSCLC with a 95% CI of 9.5–15.4) and TROPION-PanTumor02 (ORR of 56.5% in the nonsquamous NSCLC cohort). Although these studies provided supportive signals, they are primarily focused on response outcomes and cannot offer confirmatory evidence of a survival benefit.34 –36
The included studies in this meta-analysis also showed heterogeneity concerning individual subgroup study analysis, such as prior responses to anti-PD-L1 treatment and the presence or absence of AGA or biomarker expression. EVOKE-01 study demonstrated a meaningful OS benefit of 3.5 months with SG over docetaxel, with a 25% reduction in the risk of death in the subgroup of patients who were non-responsive to their last anti-PD-L1 treatment. 22 Likewise, TROPION-lung01 reported median OS of 15.6 months (95% CI, 12.0–16.9) with Dato-DXd compared to 9.8 months (95% CI, 6.2–14.8) with docetaxel in the nonsquamous NSCLC cohort in the AGA-positive subgroup (HR, 0.65; 95% CI, 0.40–1.08). 23 TROPION-Lung05 also reported similar results with a confirmed ORR of 35.8% (95% CI, 27.8–44.4) and a DCR of 78.8% (95% CI, 71.0–85.3) in recurrent NSCLC patients with AGA, treated with Dato-DXd. 37 Pons-Tostivint et al. 38 reported intracranial efficacy of Dato-DXd in a post hoc analysis of TROPION-Lung01, with median PFS of 4.9 months versus 3.6 months with docetaxel in patients with brain metastasis (HR: 0.59; 95% CI, 0.35–1.00). 38 Such data cannot be consolidated because of the limited information and differences in the stratification of individual studies. 38
Progression-free survival data, when analyzed, offer additional granularity to the efficacy evaluation. Although the pooled HR of all the studies was 0.91 (95% CI: 0.73–1.13), TROPION-Lung01 reported a significant PFS advantage of Dato-DXd (HR: 0.75; 95% CI: 0.62–0.91). The heterogeneity (I2 = 67%) of the pooled PFS may be reflected because of differences in the trial design, ADC constructs, and patient and disease characteristics, showcasing short-term disease control. Stratifying PFS by histology adds no further context in terms of survival advantage. This led to an open discussion and further exploration of predictive biomarkers or histologic correlates that might explain the differential impact on PFS, especially in the context of the appealing pooled OS outcome. Similarly, no difference was found in DCR (pooled OR: 1.39; 95% CI: 0.75–2.57) and ORR (pooled OR: 1.16; 95% CI: 0.54–2.51). In TROPION-Lung01, although a small PFS gain is statistically significant, in CARMEN-LC03, docetaxel yielded a numerically non-significant higher PFS and comparable ORR. Hence, the pooled results should be interpreted more cautiously.
The post hoc biomarker-based exploratory analysis of CARMEN-LC03 and TROPION-Lung01 revealed interesting results. However, the primary analysis of CARMEN-LC03 showed no PFS advantage with a trend toward improved OS; both PFS (HR = 0.87 (0.60–1.26)) and OS (HR = 0.71 (0.49–1.03)) favored tusa rav in participants with CEACAM5 ⩾ 2+ in ⩾80% tumor cells compared to the population with CEACAM5 ⩾ 2+ in 50%–79% tumor cells (PFS: HR = 1.38 (0.92–2.07), and OS: HR = 1.02 (0.69–1.50)). 39 In addition, when dividing patients into cohorts by CEACAM5 expression, it was found that patients with >90% achieved the most significant benefit with tusa rav, with a median OS (in months) of tusa rav versus docetaxel of 18.63 versus 15.24, respectively (HR = 0.65 (0.39–1.08)). 39 Likewise, Dato-DXd demonstrated strong efficacy (ORR was higher and median PFS was longer) in patients with TROP2 Quantitative Continuous Scoring (QCS)-Normalized Membrane Ratio (NMR)-positive nonsquamous, non-AGA advanced/metastatic NSCLC. 40 Although these biomarkers predict clinical outcomes, further investigation is required to inform clinical benefit in such patients, and subsequently, the data need specialized interpretation. Many clinical trials are still ongoing (Table 4).
Ongoing clinical trials comparing ADC versus docetaxel for previously treated advanced NSCLC.

ADC, antibody–drug conjugate; c-MET, mesenchymal epithelial transition factor; DOR, duration of response; EGFR, epidermal growth factor receptor.; ORR, objective response rate; OS, overall survival; PFS, progression-free survival; QOL, quality of life.
ADCs have unique adverse events and could be highly specific to their molecular targets. Known hematological and non-hematological adverse events include, but are not limited to, infusion reaction, alopecia, nausea, vomiting, stomatitis, diarrhea, fatigue, arthralgia, hepatitis, dyspnea, pneumonia, interstitial lung disease (ILD), urinary infections, neutropenia, anemia, thrombocytopenia, and endocrine abnormalities. 17 In our meta-analysis, TRAEs are the key discriminator between ADCs and chemotherapy. Even though the pooled OR for all-grade adverse events was 1.06 (95% CI: 0.39–2.84) with high heterogeneity (I2 = 92%), suggesting mixed safety signals. For grade ⩾ 3 TRAE, ADCs consistently triumph over chemotherapy. A 51% reduction in the odds of severe toxicity was observed when pooled with an OR of 0.49 (95% CI: 0.26–0.90; p = 0.0004), significantly favoring ADC. Dato-DXd was associated with an increased risk of grade ⩾ 3 stomatitis and ILD, an important AE that needs close monitoring and appropriate management; however, the consistency of direction and magnitude of benefit with pooled results for all ADCs demonstrated a clinically meaningful benefit. In addition, decreased rates of anemia, neutropenia, hospitalization, dose reductions, and treatment interruptions with ADCs contribute to an acceptable safety profile that may translate into better compliance and QOL. It is also important to note that our analysis pooled ADCs directed against different targets (TROP2, CEACAM5). Given the distinct adverse event profiles associated with each target, separating these agents in future analysis may provide clinically meaningful insights.
Patient-reported outcomes (PROs) in EVOKE-01 and TROPION-Lung01 demonstrated a higher compliance rate for SG (>84%) and Dato-DXd (>94%), with a delayed time to deterioration (TTD). As compared to docetaxel, SG significantly delayed key NSCLC symptoms, including shortness of breath, fatigue, and total symptom burden (NSCLC-SAQ (Symptom Assessment Questionnaire), QLQ-C30) (p < 0.05) with least squares mean change favoring SG (p < 0.01) at week 25, regardless of high treatment-related nausea, vomiting, and diarrhea. 41 Likewise, Dato-DXd demonstrated reduced risk of deterioration compared to docetaxel in dyspnea at rest (HR: 0.56), overall dyspnea (HR: 0.61), physical functioning (HR: 0.57), social functioning (HR: 0.66), peripheral neuropathy (HR: 0.35), pain (HR: 0.55), and diarrhea (HR: 0.43) despite increased risk of nausea/vomiting (HR: 1.67) and constipation (HR: 1.64). Although we cannot pool the data, this exploratory analysis of individual studies suggests that both SG and Dato-DXd improved cancer-related symptoms, delayed deterioration, and enhanced QOL, complementing their efficacy in previously treated NSCLC patients. 42
An important area to explore further is strategies to “re-stimulate” the immune system in a particular subset of patients who initially derived long-term benefit from immunotherapy but eventually experienced disease progression. These long responders represent a clinical course that suggests a period of sustained immune surveillance, which later becomes exhausted through mechanisms of immune escape, including T-cell fatigue. Although limited, emerging evidence indicates that rechallenge with an immune checkpoint inhibitor (ICI) after a treatment-free interval may provide antitumor activity in selected patients. 43 Another promising approach includes the use of oncolytic viruses to reshape the tumor microenvironment, therapeutic vaccines to re-priming the immune system against cancer-associated neoantigen, and cytokine-based therapies to sustain the effector T-cell function.44,45 Furthermore, tumor-infiltrating lymphocyte (TIL) transfer or engineered T-cell receptor techniques are also being investigated to reintroduce immune effector cells with enhanced specificity. 46 These methods collectively reflect growing evidence for the future long-term management of advanced NSCLC, which involves re-stimulating the immune system to overcome adaptive resistance.
Limitations
This systematic review and meta-analysis provide key observations into the relative efficacy and safety of ADCs versus docetaxel in pretreated NSCLC patients; however, it has several limitations that need careful consideration. The number of eligible RCTs included in the meta-analysis is limited, as only three studies, EVOKE-01, TROPION-Lung01, and CARMEN-LC03, met our inclusion criteria. Although these are extensive, randomized, well-planned studies, the smattering of data sources limits the overall power of this meta-analysis, increases the vulnerability to random error, or overanalyzes the subgroup effect. Furthermore, an open-label design was implemented across all the studies, which may introduce a risk of performance or detection bias. Moreover, although most efficacy outcomes were assessed independently, some PROs and unintended therapy-related AEs may have been influenced by the knowledge of drug assignment. This is pertinent when comparing ADCs, which may cause unique adverse events such as ocular toxicity or ILD, to docetaxel, a historical agent with a more familiar side-effect safety profile.
Another notable limitation is the substantial heterogeneity in various endpoints, including PFS, DCR, and ORR. This dictates considerable variability between studies, likely due to differences in the study population, the mechanism of action of ADC (antigen target, linker stability, payloads, and internalization efficiency), biomarker thresholds, and so forth that may influence the overall treatment efficacy and toxicity, perplexing cross-comparison. Moving on, histologic subtype and biomarker variability further limit the interpretation. The subgroup analysis of our MA revealed that the nonsquamous NSCLC cohort benefited more with ADCs; however, not all studies are stratified by outcomes based on histology or biomarker expression. CARMEN-LC03 utilized the CEACAM5 expression level of ⩾2+ in ⩾50% of the tumor cells as the inclusion criteria, while other studies did not specifically target the TROP2 expression cutoff; thus, there was an inconsistency in assessment methods. In addition, a lack of uniform individual patient data restricts the subgroup analysis or adjustment for confounding baseline characteristics such as smoking status, presence or absence of AGA, targeted treatment, and PD-L1 expression levels. The data cutoff timing and follow-up duration also varied across the studies, which may affect the outcome measures. For instance, although the survival data are not mature, the median follow-up for OS in EVOKE-01 is approximately 12.7 months; however, it is longer in CARMEN-LC03 and TROPION-Lung01. These temporal differences may impact the credibility of pooled OS outcomes. Adding to this, post-progression therapies are not uniformly documented or adjusted for; thus, it could bias the OS report in favor of either treatment arm.
While our report reconciles the best applicable evidence from the key RCTs, its conclusion is restricted by the small number of trials, heterogeneity in the results, and discrepancies in stratification, study design, biomarker expressions, and follow-up. New clinical trials, real-world data, and future meta-analyses are required to validate the clinical significance of our observations.
Conclusion
Overall, there was no difference between the docetaxel and the ADC treatment arms; however, our findings report a significant survival benefit in the subgroup of patients with nonsquamous NSCLC pathology treated with ADCs compared to docetaxel with a manageable safety profile. Further research is needed to address heterogeneity, refine patient selection, and obtain more mature survival data with predictive biomarkers.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251387393 – Supplemental material for Antibody–drug conjugates versus docetaxel for previously treated advanced non-small-cell lung cancer: a systematic review and meta-analysis of randomized controlled trials
Supplemental material, sj-docx-1-tam-10.1177_17588359251387393 for Antibody–drug conjugates versus docetaxel for previously treated advanced non-small-cell lung cancer: a systematic review and meta-analysis of randomized controlled trials by Saqib Raza Khan, Laís Marques Eiras, Gabriel Boldt, Jacques Raphael and Daniel Breadner in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251387393 – Supplemental material for Antibody–drug conjugates versus docetaxel for previously treated advanced non-small-cell lung cancer: a systematic review and meta-analysis of randomized controlled trials
Supplemental material, sj-docx-2-tam-10.1177_17588359251387393 for Antibody–drug conjugates versus docetaxel for previously treated advanced non-small-cell lung cancer: a systematic review and meta-analysis of randomized controlled trials by Saqib Raza Khan, Laís Marques Eiras, Gabriel Boldt, Jacques Raphael and Daniel Breadner in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-3-tam-10.1177_17588359251387393 – Supplemental material for Antibody–drug conjugates versus docetaxel for previously treated advanced non-small-cell lung cancer: a systematic review and meta-analysis of randomized controlled trials
Supplemental material, sj-pdf-3-tam-10.1177_17588359251387393 for Antibody–drug conjugates versus docetaxel for previously treated advanced non-small-cell lung cancer: a systematic review and meta-analysis of randomized controlled trials by Saqib Raza Khan, Laís Marques Eiras, Gabriel Boldt, Jacques Raphael and Daniel Breadner in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
