Abstract
Background:
Many studies show that camrelizumab combination therapy can significantly improve progression-free survival (PFS) and overall survival (OS) in non-small cell lung cancer (NSCLC). However, the time of camrelizumab to market is short, and there is no systematic evaluation of camrelizumab-based comprehensive treatment of NSCLC.
Objectives:
To systematically evaluate the efficacy and safety of camrelizumab in comprehensively treating NSCLC.
Design:
A systematic review and meta-analysis.
Data sources and methods:
Databases, including PubMed, Web of Science, Embase, and Cochrane, were searched by computer before August 2023 based on Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) guidelines, and reports on the efficacy and safety of camrelizumab-based treatment for NSCLC were collected, and RevMan 5.4 software was employed for meta-analysis finally.
Results:
Totally, 5 RCTs, 2 cohort studies, and 12 single-arm studies were included. The meta-analysis results revealed that, compared with the treatment without camrelizumab, the camrelizumab-based combination treatment considerably extended the OS (hazard ratio (HR) = 0.60, 95% confidence interval (CI): (0.44–0.82), p < 0.01), PFS (HR = 0.42, 95% CI: (0.28–0.63), p < 0.01), and event-free survival (EFS) (HR = 0.55, 95% CI: (0.44–0.68), p < 0.01). The median objective response rate in single-arm studies was 41% (95% CI: 28%–53%), and the disease control rate was 84% (95% CI: 78%–89%). Furthermore, in terms of the occurrence of grades 3–5 adverse events, the incidence of neutropenia was lower in the camrelizumab combination group than in the control group, while the incidence of leukopenia and rash was higher than in the combination group, and no significant difference was revealed in the incidence of other adverse events. Among single-arm studies, the incidence of grades 3–5 adverse events did not exceed 10%.
Conclusion:
Treatment combined with camrelizumab can effectively prolong OS, PFS, and EFS in NSCLC patients with good safety, camrelizumab combined with chemotherapy is an effective treatment option for NSCLC patients.
Introduction
Lung cancer is the second most prevalent type of malignant tumor in the world and the main reason for cancer death. It has been reckoned that there were 2.5 million new cases and 1.8 million deaths in 2022. 1 The main histological type of lung cancer is NSCLC, which represents approximately 80% of lung cancer cases, with surgical resection being the primary treatment. 2 However, due to the highly invasive nature of the disease and the absence of effective early screening methods, most patients are diagnosed at advanced stages, missing the optimal timing for surgery. Platinum-based chemotherapy is currently used to treat advanced NSCLC patients, but it leads to adverse reactions in the digestive tract, bone marrow suppression, and poor prognosis. Although targeted therapy drugs have demonstrated some therapeutic benefits, their efficacy is limited to patients with advanced lung adenocarcinoma who have sensitive driver gene mutations such as epidermal growth factor receptor, c-ros oncogene 1, and anaplastic lymphoma kinase, with a mutation rate of only 20%–30%.3–5 Therefore, new treatment strategies are urgently needed to extend the survival of patients with advanced NSCLC and minimize adverse reactions.
The emergence of immunotherapy provides a new direction for treating patients with advanced or metastatic malignancies. It has become a crucial treatment method following surgery, radiotherapy, chemotherapy, and targeted therapy. Currently, immune checkpoint inhibitors (ICIs) commonly utilized in clinical practice encompass monoclonal antibodies targeting programmed cell death protein 1 (PD-1), programmed death ligand 1 (PD-L1), and cytotoxic T-lymphocyte-associated antigen-4. Numerous clinical trials have demonstrated the potential benefits of ICIs in treating various malignant tumors. Additionally, combined therapy has been shown to enhance clinical efficacy while simultaneously minimizing side effects, offering patients a more personalized and comprehensive treatment strategy.6–10 KEYNOTE-189 11 is a randomized, double-blind, phase III clinical trial that assessed the effectiveness of pembrolizumab or placebo in combination with pemetrexed and platinum-based therapy for patients with non-squamous stage IV NSCLC. The recently published 5-year follow-up results showed that the 5-year overall survival (OS) rates were 19.4% for the immunotherapy + chemotherapy group and 11.3% for the placebo + chemotherapy group, with controllable toxicity.
Camrelizumab (Erica™, Jiangsu Hengrui Pharmaceuticals Co., Ltd of China, Jiangsu, China) is a humanized anti-PD-1 monoclonal antibody developed by Jiangsu Hengrui Pharmaceuticals Co., Ltd of China. It obtained its first approval from the National Medical Products Administration in May 2019 to treat patients with relapsed or refractory classical Hodgkin’s lymphoma. 12 Later, it was approved for the treatment of non-squamous NSCLC, liver cancer, and esophageal squamous cell carcinoma. At present, it has been confirmed that camrelizumab alone or in combination with other drugs shows significant anticancer activity, and results in a low incidence of adverse reactions. In a study (CameL) 13 evaluating the efficacy of camrelizumab in patients with driver-gene-negative advanced NSCLC, two treatment groups were compared: camrelizumab with carboplatin and pemetrexed versus carboplatin with pemetrexed. The median follow-up time was 11.9 months, and PFS was significantly better in the combination chemotherapy group (11.3 vs 8.3 months), with manageable toxicity. This combination therapy is now the standard treatment for advanced non-squamous NSCLC in China. The CameL study recently published its 5-year follow-up results, showing a 5-year OS rate of 31.2% and 19.3% in the immunotherapy + chemotherapy group and a 5-year PFS rate of 16.1% in the chemotherapy group, with manageable toxicity. Another double-blind, multicenter, phase III trial (CameL-Sq), 14 revealed that camrelizumab combined with carboplatin and paclitaxel significantly extended PFS and OS compared to placebo + chemotherapy in squamous NSCLC patients, establishing immune-based chemotherapy as the first-line treatment for squamous NSCLC.
In recent years, with the in-depth research of immunotherapy, a growing number of studies have focused on the efficacy and safety of camrelizumab combined with chemotherapy or targeted drugs in advanced NSCLC. Many studies show that camrelizumab combined with chemotherapy can significantly improve PFS and OS in advanced NSCLC patients in comparison with chemotherapy.15–17 However, the time of camrelizumab to market is short, and there is no systematic evaluation of camrelizumab-based comprehensive treatment of NSCLC. This study conducted a meta-analysis and systematic review of the relevant published studies on camrelizumab in treatment of advanced NSCLC, with the purpose of providing evidence-based medical evidence for clinical practice.
Materials and methods
The study was conducted in accordance with Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) guidelines, 18 and the respective checklist has been provided as a Supplemental File 1.
Literature search strategy
The literature on the treatment of advanced NSCLC with camrelizumab published up to August 31, 2023 was retrieved. The PubMed, Web of Science, Embase, and Cochrane electronic databases were comprehensively searched using text words and subject headings, and the language was restricted to English language. The keywords included “camrelizumab,” “SHR-1210,” “SHR 1210,” “Carcinoma, Non-Small-Cell Lung,” “Lung Carcinoma, Non-Small-Cell,” and “Non-Small-Cell Lung Cancer.” Pubmed retrieval formula is as follows: (“camrelizumab” [Supplementary Concept]) or (((carrelizumab) or (SHR-1210)) or (SHR 1210)) or (camrelizumab)) and ((“Carcinoma, Non-Small-Cell Lung” [Mesh]) OR ((((((((((((Carcinoma, Non Small Cell Lung) OR (Carcinomas, Non-Small-Cell Lung)) OR (Lung Carcinoma, Non-Small-Cell)) OR (Lung Carcinomas, Non-Small-Cell)) OR (Non-Small-Cell Lung Carcinomas)) OR (Non-Small-Cell Lung Carcinoma)) OR (Non Small Cell Lung Carcinoma)) OR (Carcinoma, Non-Small Cell Lung)) OR (Non-Small Cell Lung Carcinoma)) OR (Non-Small Cell Lung Cancer)) OR (Non small Cell Lung Cancer)) OR (NSCLC))). The complete search strategies for all databases are depicted in Supplemental Table S1.
Literature inclusion and exclusion criteria
Inclusion criteria were as follows: (1) The histological type was NSCLC, and both metastatic and localized NSCLC were included. (2) Types of studies: randomized controlled trial (RCT), cohort study, and single-arm study. (3) Intervention measures of RCT and cohort study: Patients in the experimental group were given camrelizumab on the basis of treatment in the control group. The dosage, usage, and course of treatment of the two groups were not limited. 4 The included articles contained at least one effective outcome indicator: OS, objective response rate (ORR), disease control rate (DCR), progression-free survival (PFS), event-free survival (EFS), or disease-free survival (DFS), or one indicator of safety outcome: leukopenia, neutropenia, nausea, rash, hypothyroidism, and reactive cutaneous capillary endothelial proliferation (RCCEP). Exclusion criteria were as follows: (1) duplicate publications; (2) case reports, reviews, and conference summaries; and (3) literature not in full text and with incomplete information or irrelevant outcome indicators.
Literature screening and data extraction
Literature retrieval, screening, and data extraction were independently carried out by two researchers (Y.L. and N.M.). Then, the results were cross-checked. Doubts or disagreements were resolved after discussion or by a third researcher (Y.C.). The following data were extracted: (1) the first author’s name, publication year, country, and registration number of the included literature; (2) study design, intervention measures, sample size, and patient’s age, gender, and follow-up time; (3) effective outcome indicators: OS, ORR, DCR, PFS, EFS, or DFS; and safety outcome indicators: leukopenia, neutropenia, nausea, rash, hypothyroidism, and RCCEP. PFS, EFS, and DFS are endpoints that share similarities. PFS is defined as the duration from randomization (or initiation of treatment in a single-arm trial) to tumor progression or death, whichever happens first. EFS, on the other hand, is the time from the start of randomization (or treatment initiation in a single-arm trial) to the first occurrence of specific events such as the progression of disease that precludes surgery, local or distant recurrence, and death due to any cause. Lastly, DFS is the duration from the start of randomization (or treatment initiation in a single-arm trial) to the recurrence of the disease or death from any cause, whichever occurs first. Among these, PFS primarily indicates the therapeutic impact of drugs on tumors, encompassing deaths resulting from various causes, which are more closely associated with OS. DFS is primarily employed to assess disease recurrence, particularly in the context of evaluating postoperative or post-radiotherapy adjuvant treatments. EFS is more indicative of surgical interventions than DFS and is predominantly utilized to evaluate neoadjuvant therapies preceding surgical procedures or radiotherapy. DFS and EFS can serve as crucial endpoint indicators in cases where the extended lifetime poses challenges in using the OS as the primary endpoint.
Literature quality assessment
The risk of bias tool for RCTs recommended by Cochrane Handbook 13.3.5.4 was applied for the risk of bias assessment, which specifically included contents as to whether the random method was accurate, whether the allocation scheme was concealed, whether the blinding method was adopted, whether the result data were integrated, whether the results were selectively reported, and whether there were other sources of bias. Each item was divided into “high bias,” “low bias,” and “unclear.” The Newcastle–Ottawa Scale (NOS) was employed to appraise the quality of the cohort studies, with a total score of 9. According to the NOS scores, the literature was divided into low (0–4), medium (5–6), and high (7–9) quality. The single-arm studies were evaluated by the JBI scale, including 10 items, and each item was input with “Yes” or “No” according to the quality of the articles. Two researchers (Y.L. and N.M.) independently evaluated the quality of the included studies. Disputes were solved via discussion or negotiation with another researcher (Y.C.).
Statistical methods
Revman (Review Manager) 5.4 was utilized for statistical analysis of RCTs and cohort studies. The outcome measures (OS, PFS, EFS, and DFS) were displayed as hazard ratios (HRs) with 95% confidence intervals (CIs), and the binary variables were reported as relative risks (RRs) with 95% CIs. A heterogeneity test was carried out for each analysis. An I2 ⩽ 50% indicated insignificant heterogeneity, and thus a fixed-effects model was used for analysis. An I2 > 50% suggested that heterogeneity existed among the studies, and a random-effects model was used. For the outcome indicators OS, PFS, EFS, and DFS, subgroup analysis was conducted based on various research types. Stata 15.1 was used to combine the outcome indicators and the incidence of adverse events of single-arm studies. For RCT and cohort studies, the publication bias test and sensitivity analysis were carried out by Stata 15.1. Meanwhile, the funnel plot and Egger’s test were selected to evaluate the publication bias of the literature, and a p-value of lower than 0.05 was deemed statistically significant (p < 0.05).
Results
The results of the literature search
Overall, 823 studies were retrieved from the database. A total of 548 studies were obtained after eliminating duplicate studies, and 494 studies were removed through browsing the titles and abstracts of the literature. After reading the full text, 5 RCTs,13,14,19–21 2 cohort studies,22,23 and 12 single-arm studies15–17,24–32 were finally included. The flow chart of literature screening is depicted in Figure 1.

Flowchart of the systematic search and selection process.
Baseline characteristics and quality evaluation of the included studies
Study characteristics
This meta-analysis included 5 RCTs and 2 cohort studies, involving 1104 patients (two studies13,20 were from the same trial with the same sample size, but their follow-up duration and outcome measures were different, so only one of them was involved in the calculation of the overall number of cases, which was different from Table 1). All patients in the included studies were Chinese. The basic characteristics of the included studies are shown in Table 1. In addition, 12 single-arm studies involving 1235 patients were also included in the analysis (Table 2).
Baseline characteristics of included studies.

NSCLC, non-small cell lung cancer; RCT, randomized controlled trial; Q3w, Use once every three weeks.
Baseline characteristics of include studies.

AUC, area under the curve; MWA, microwave ablation; nab-PTX, nab-paclitaxel.
Quality assessment of the included studies
Among the five RCTs, one study did not mention the concealment of the distribution; two studies did not mention the blinding of the investigators and participants; two studies did not mention the adoption of the blinding method for results evaluation, and one did not mention whether the results were selectively reported. All other studies mentioned the specific methods of generating random sequences, and other sources of bias were clear (Figure 2). The scores of the two cohort studies were 8 and 9, respectively, both of which were high-quality studies (Supplemental Table S2). The quality evaluation results of the single-arm studies are displayed in Supplemental Table S3.

Literature quality evaluation.
Results of meta-analysis
Efficacy analysis of RCTs and cohorts
Five studies reported OS, and the results of the included studies were quite heterogeneous (I2 = 60%, p = 0.04). The random-effects model was utilized for meta-analysis. The results revealed that the treatment combined with camrelizumab could significantly prolong the OS of advanced NSCLC patients compared with the treatment without camrelizumab, and statistical significance was noted (HR: 0.60, 95% CI: 0.44–0.82, p = 0.001, Figure 3(a)). Subgroup analysis revealed that the HR for RCT was 0.67 (95% CI: 0.57–0.80) and for cohort studies was 0.13 (95% CI: 0.01–1.91). The higher quality of RCTs compared to cohort studies suggests that camrelizumab combined therapy may be more effective in RCTs.

Forest plot for (a) OS, (b) PFS, (c) EFS, and (d) DFS of treatment combined with camrelizumab versus treatment without camrelizumab in patients with NSCLC.
PFS was reported in three studies, and there was great heterogeneity (I2 = 76%, p = 0.02). The random-effects model was adopted for meta-analysis. The results disclosed that the treatment combined with camrelizumab could significantly extend the PFS of advanced NSCLC patients compared with the treatment without camrelizumab, and statistical significance was noted (HR: 0.42 (95% CI: 0.28–0.63), p < 0.001, Figure 3(b)).
Two studies reported EFS. There was no statistical heterogeneity among the studies (I2 = 0%, p = 0.91), so the fixed-effects model was adopted for meta-analysis. The results unveiled that the camrelizumab combination treatment could significantly extend the EFS of advanced NSCLC patients compared with the treatment without camrelizumab, and no statistical significance was revealed (HR: 0.55, 95% CI: 0.44–0.68, p < 0.001, Figure 3(c)).
Two studies reported DFS and the data from these studies were quite heterogeneous (I2 = 73%, p = 0.05). The random-effects model was selected for meta-analysis. The results revealed that the treatment combined with camrelizumab could not prolong the DFS of advanced NSCLC patients, and no significant difference was found between the two groups (HR: 0.21, 95% CI: 0.02–1.88, p > 0.05, Figure 3(d)).
CameL 20 study and CameL-Sq 14 study were included in the analysis, with subgroups analyzed based on the patient’s PD-L1 tumor proportion score, gender, age, ECOG score, liver or brain metastasis status, and smoking status. It can be observed that OS and PFS of patients with advanced squamous NSCLC benefited from camrelizumab combined with chemotherapy, regardless of PD-L1 expression status. In advanced non-squamous NSCLC, patients with PD-L1 expression ⩾1% benefited from combination therapy regarding OS and PFS, whereas those with PD-L1 expression <1% did not benefit from combination therapy in both aspects. The impact of smoking status on patient survival benefits is noteworthy. The combined use of camrelizumab significantly extended the survival of NSCLC smokers who smoke over 20 packs a year compared to chemotherapy. However, it did not enhance the survival of nonsmokers or light smokers. The patients’ benefits from OS and PFS were not impacted by ECOG score, liver metastasis, or brain metastasis. Gao et al.’s 24 research demonstrated that patients with squamous NSCLC benefited from the combination of camrelizumab and apatinib, regardless of PD-L1 expression status. A study by Ren et al. 16 confirmed that the combination of camrelizumab and apatinib improved the ORR and PFS of patients with non-squamous NSCLC, regardless of PD-L1 expression status. Yin et al. 17 discovered that NSCLC patients with high PD-L1 expression had better outcomes when treated with camrelizumab in combination with chemotherapy as a second-line and above-line treatment.
Efficacy analysis of single-arm studies
Among the single-arm studies, nine studies reported the ORR of patients after taking camrelizumab, and the random-effects model was used for analysis (I2 = 93.9%, p = 0). The pooled median ORR was 41% (95% CI: 28%–53%) (Figure 4(a)). Nine studies reported DCR, and the random-effects model was used (I2 = 77.7%, p = 0). The pooled median DCR was 84% (95% CI: 78%–89%) (Figure 4(b)).

Forest plot for ORR (a) and DCR (b).
Among the single-arm studies, two studies reported the OS of patients after taking camrelizumab, and the fixed-effects model was adopted (I2 = 0%, p = 0.86). The pooled median OS was 12.78 months (95% CI: 10.50–15.06 months) (Figure 5(a)). PFS was reported in four studies, and the random-effects model was adopted (I2 = 82.6%, p = 0.001). The pooled median PFS was 7.79 months (95% CI: 4.61–10.97 months) (Figure 5(b)).

Forest plot for OS (a) and PFS (b).
Incidence of adverse events
All RCTs and cohort studies were selected for analysis of grades 3–5 adverse events. The pooled RRs of leukopenia, neutropenia, nausea, rash, hypothyroidism, and RCCEP in the camrelizumab combination group compared with the control group were 1.40 (95% CI: 1.06–1.84; I2 = 0), 0.33 (95% CI: 0.26–0.42; I2 = 31), 0.83 (95% CI: 0.24–2.89; I2 = 0), 6.49 (95% CI: 1.16–36.43; I2 = 0), 3.04 (95% CI: 0.32–29.38; I2 = 0), 7.17 (95% CI: 0.88–58.53; I2 = 0) (Figure 6). The results revealed that the incidence of neutropenia was lower in the camrelizumab combination group than in the control group, while the incidence of leukopenia and rash was higher in the camrelizumab combination group. A significant difference in the incidence of other complications was not found between the camrelizumab combination treatment and the treatment without camrelizumab.

The incidence of AEs in the camrelizumab combination group and the control group. (a) White blood cell count decreased. (b) Neutrophil count decreased. (c) Nausea. (d) Rash. (e) Hypothyroidism. (f) Reactive cutaneous capillary endothelial proliferation.
In the single-arm studies, the most frequent AEs (all grades and ⩾3 grades) related to the treatment of NSCLC with camrelizumab were analyzed (Table 3). Most patients have experienced AEs of unlimited levels and showed favorable tolerance. The results showed that the incidence rates of the three most common adverse events, including anemia, RCCEP, and thrombocytopenia, were 37% (95% CI: 20%–53%), 36% (95% CI: 19%–53%), and 17% (95% CI: 11%–23%), respectively. In addition, the incidence of neutropenia, diarrhea, leukopenia, and lymphopenia reached 11% (95% CI: 5%–17%), 11% (95% CI: 4%–19%), 16% (95% CI: 9%–23%), and 7%, respectively. The incidence of grades 3–5 adverse events was low (all below 10%), even the most common events, such as anemia (3%), RCCEP (1%), and thrombocytopenia (4%).
Adverse events of the studies included in the meta-analysis.
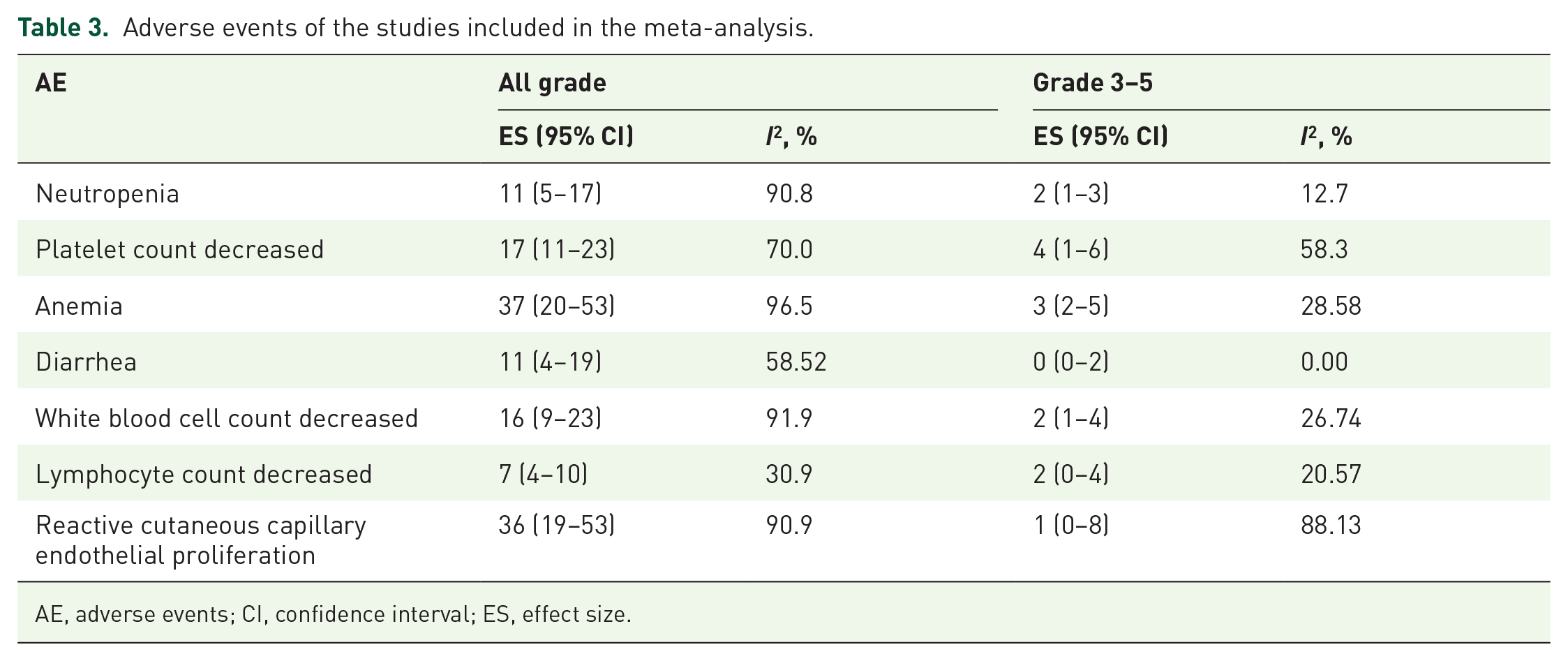
AE, adverse events; CI, confidence interval; ES, effect size.
Publication bias
Cochrane Handbook (13.3.5.4) 33 highlights the need to assess publication bias when there are over 10 comprehensive analysis studies. Since this paper included fewer than 10 RCT and cohort studies, publication bias analysis was not conducted.
Sensitivity analysis
Stata 15 software was adopted to investigate the sensitivity of OS and PFS and to detect the change of the results after eliminating a certain study. After analyzing sensitivity, the results disclosed that the statistical difference between OS and PFS was not changed by excluding any study, suggesting stable results of OS and PFS (Figure 7). The sensitivity analysis of OS for RCTs shows that OS results are stable. There are only two RCTs reporting PFS, so sensitivity analysis for PFS was not conducted (Supplemental Figure S1).

Sensitivity analysis. (a) OS. (b) PFS.
Discussion
At present, NSCLC is usually treated with platinum-based dual-drug chemotherapy. Although such treatment has a certain effect, its serious adverse reactions are unbearable for most patients. Besides, it is difficult for advanced NSCLC patients who have failed chemotherapy numerous times to benefit from chemotherapy. Therefore, it is necessary to discover new drugs and treatment schemes to prolong patients’ survival.
ICIs have greatly changed the prospect of cancer treatment since their emergence. As a kind of immunosuppressive molecule, it can regulate immune activation. However, the growth and proliferation of tumors are linked to the deactivation of certain immune checkpoints by tumor cells, causing an imbalance in antitumor immunity, which is a significant mechanism in lung cancer. PD-1 inhibitors (nivolumab and pembrolizumab) and PD-L1 inhibitors (durvalumab and atezolizumab) showed significant activity against tumor and tolerable toxicity in treating advanced NSCLC, which provided a new direction for immunotherapy of advanced NSCLC. As a new monoclonal antibody of humanized immunoglobulin G4, camrelizumab binds to the PD-1 receptor to inhibit ligands PD-L1 and PD-L2 interaction, block the immunosuppressive reaction mediated by the PD-1 pathway, mobilize the autoimmune system, identify and destroy tumor cells, and exert antitumor effect. Because PD-L1 and PD-L2 are primarily expressed on tumor cells’ surface, the binding of PD-1 to its ligand happens in the tumor immune microenvironment. Using PD-1 inhibitors leads to fewer immune-related side effects, thus enhancing patients’ survival and prognosis. 34
Visser et al. 35 find that initially, the survivorship curve of patients receiving combination therapy is significantly different from that of patients receiving single-agent therapy, indicating that combination therapy is a better treatment model for cancer, and is more beneficial to patients than single-drug therapy in the treatment of various tumors. An RCT by Wang et al. 36 reveals that the combination of tislelizumab and standard chemotherapy can significantly slow disease progression and reduce mortality of patients with advanced NSCLC. A phase III trial 37 shows that the outcome is significantly improved in the atezolizumab and chemotherapy combination group than in the chemotherapy alone group, and the median PFSs were 6.3 and 5.6 months in the two groups, respectively. It was further found that for metastatic, squamous NSCLC, atezolizumab could benefit patients with high expression of PD-L1. Again, in the trial by IMPower150 38 on the same population, atezolizumab combining bevacizumab and chemotherapy drastically extended the PFS and OS of patients. In the KEYNOTE-407 39 experiment, pembrolizumab combined with chemotherapy prolonged PFS and OS in NSCLC patients. The efficacy of camrelizumab combined with chemotherapy in treating NSCLC has been confirmed in previous studies. In an RCT involving patients with resectable NSCLC at stage IIIA or IIIB (T3N2), camrelizumab combined with chemotherapy significantly improved the pathologic response rate than chemotherapy alone. 19 In the study by CameL, 13 412 patients were divided into two groups randomly and received camrelizumab combining chemotherapy and chemotherapy, respectively. The results showed that PFS was 11.3 and 8.3 months, the PFS rate at 12 months was 49.6% and 35.1%, and the PFS rate at 48 months was 15.6% versus 0%, respectively. The experiment confirmed that the application of camrelizumab significantly prolonged PFS, providing a theoretical basis for camrelizumab combining chemotherapy for advanced or metastatic squamous NSCLC as the first-line treatment. A recent meta-analysis 40 on the efficacy of ICIs combined with chemotherapy for advanced NSCLC found that camrelizumab combined with chemotherapy was more effective in improving PFS compared to other combinations. Therefore, we conducted a systematic review and meta-analysis of camrelizumab combined with advanced NSCLC to enhance treatment strategies for NSCLC patients and establish a solid theoretical foundation for larger clinical research studies.
Our study included 19 studies, including 5 RCTs, 2 cohort studies, and 12 single-arm studies, involving 2339 people. After combining RCTs and cohort studies, we conducted a systematic evaluation. The meta-analysis indicated that the HR for OS, PFS, EFS, and DFS were 0.60 (95% CI: 0.44–0.82), 0.42 (95% CI: 0.28–0.63), 0.55 (95% CI: 0.44–0.68), and 0.21 (95% CI: 0.02–1.88), respectively. It was suggested that compared with the treatment without camrelizumab, camrelizumab combination treatment could significantly prolong the OS, PFS, and EFS of patients, and no statistical significance was noted in DFS after pairwise comparisons. This paper also included 12 single-arm studies, and the results showed that the median ORR, DCR, OS, and PFS were 41%, 84%, 12.78 months, and 7.79 months, respectively. Most of the studies included in this paper focused on the combination of camrelizumab and chemotherapy in treating advanced NSCLC. The mechanism underlying the improvement in patients’ survival with this combination therapy may be related to the fact that camrelizumab can inhibit tumor immune escape and further reshape the tumor microenvironment, and PD-1 and chemotherapy may exert a certain synergistic effect in enhancing the function of the immune system, further eliminating tumor cells. In this study, ORR (41%) and DCR (84%) were lower than those of the first-line test (CameL: non-squamous NSCLC; ORR = 60.5%, DCR = 87.8%; CameL-Sq: squamous NSCLC; ORR = 64.8%, DCR = 88.1%). This may be related to the individual differences of patients, drug types, and dosages.
The combination therapy involving immunotherapy shows improved clinical outcomes, with ongoing research to investigate its underlying mechanism. When immunosuppressants are combined with chemotherapy, chemotherapy may increase the immunogenicity of tumors by enhancing the recognition of new antigens by T-cells. Immunotherapy, on the other hand, may boost the antitumor immune response by inhibiting T-cell checkpoints, resulting in a synergistic effect. 41 Moreover, chemotherapy-induced increase in antigen-presenting cells in tumor tissue may enhance the antitumor activity of immunosuppressants. 42 When immunotherapy is combined with targeted therapy, their synergistic effect may be due to targeted drugs altering the tumor microenvironment, enhancing the immunogenicity of tumor cells, and improving the efficacy of immunotherapy. Immunotherapy combined with targeted therapy can improve the curative effect and is promising to achieve accurate treatment of tumors. Preclinical studies indicated that low-dose apatinib can enhance the remodeling of the immunosuppressive microenvironment. When combined with camrelizumab, which boosts T cell function, it shows a synergistic antitumor effect. Qin et al.’s 43 research revealed that in first-line treatment of advanced NSCLC, the combination of camrelizumab and apatinib achieved a median progression-free survival (mPFS) of 8.3 months and an ORR of 78.1%. This suggests that nearly 8 out of 10 patients experienced relief with this treatment, highlighting its tumor reduction effect. This also provides a promising treatment option for NSCLC patients, and more large-scale clinical experiments are needed to verify the efficacy of camrelizumab combined with apatinib.
For patients eligible for surgery, the benefits of traditional adjuvant and neoadjuvant chemotherapy are limited. Camrelizumab combined with chemotherapy plays an important role in the neoadjuvant treatment of surgically resectable or possibly resectable NSCLC. Hou et al.’s 23 research found that combining camrelizumab with neoadjuvant chemotherapy resulted in a better clinical response and higher ORR, complete pathological response, and major pathological response (MPR) compared to neoadjuvant chemotherapy alone in patients with resectable stage IIIA–IIIB NSCLC. Subsequently, the TD-FOREKNOW trial, 19 a randomized controlled prospective phase II clinical trial, divided patients into two groups: one receiving neoadjuvant camrelizumab combined with chemotherapy and the other receiving neoadjuvant chemotherapy alone, followed by surgery. It was found that, compared with chemotherapy, neoadjuvant camrelizumab combined with chemotherapy for NSCLC significantly improved the pathological complete response (pCR) rate (32.6% vs 8.9%), and also improved the MPR rate (65.12% vs 15.56%) and ORR (72.09% vs 53.33%). Additionally, the EFS rates in patients receiving immune-based chemotherapy for 1 and 2 years were 93.0% and 76.9%, respectively, while those in the chemotherapy group were 76.9% and 67.6%. Based on this study, neoadjuvant camrelizumab combined with chemotherapy for surgically resectable stage IIIA–IIIB (T3N2M0) NSCLC has been included in the B-level recommendation of early NSCLC guidelines in China. The synergistic effect of neoadjuvant camrelizumab combined with chemotherapy for NSCLC may be achieved by blocking immune escape and enhancing chemotherapy sensitivity, thus improving the efficacy of neoadjuvant chemotherapy. 44 The second phase clinical trial by Zhao et al. 31 confirmed that combining neoadjuvant camrelizumab with apatinib, a small molecule anticancer drug, showed promising activity in patients with stage IIA–IIIB NSCLC. Patients with squamous NSCLC had a better pathological response compared to those with adenocarcinoma, with 19% achieving pathological complete response (pCR) and 47% achieving MPR. Therefore, neoadjuvant camrelizumab combined with apatinib is a potential treatment option for patients with resectable NSCLC.
In terms of grades 3–5 adverse events, the incidence of leukopenia and rash was found to be lower in the camrelizumab group than in the control group, and the incidence of neutropenia was higher in the control group. No statistical difference was noted in the incidence of hypothyroidism and RCCEP between the two groups. The combined results of the single-arm studies suggested that the most common unclassified AEs were anemia, RCCEP, and thrombocytopenia, with incidences of 37%, 36%, and 17%, respectively. The incidence of grades 3–5 adverse events was low (all <10%). The incidence of some treatment-related adverse events in the camrelizumab combination group was higher than that of the control group. This may be due to the fact that PD-1 inhibitors target T-cells, suppressing the patient’s autoimmunity. And camrelizumab itself has hematologic toxicity, which can cause leukopenia. According to the results of this study, most adverse events were grades 1–2, and the number of grades 3–5 adverse events was small, which can be improved after symptomatic treatment or discontinuance of camrelizumab, so the adverse events are generally controllable. A recent meta-analysis examining the use of ICIs in combination with radiotherapy or radiotherapy and chemotherapy for advanced non-small cell lung cancer (NSCLC) revealed that the prevalence of common adverse events such as nausea (47.6%), anemia (46.6%), and rash (36.4%). The incidence of anemia was higher than 37% in this study (nausea and rash were excluded due to incomplete data), which could be attributed to the predominance of studies focusing on the combination of radiotherapy and chemotherapy, as well as the incorporation of diverse ICIs. 45 The results of the phase III controlled trial KEYNOTE-189 46 revealed that the incidence of anemia, diarrhea, and thrombocytopenia in NSCLC patients treated with a combination of pembrolizumab and chemotherapy was 47.7%, 32.3%, and 18.8%, respectively. These data were found to be higher compared to the results of our analysis. Additionally, the occurrence of nausea (57.3%) and fatigue (43.2%) was relatively elevated. The findings from the China subgroup analysis of the phase III trial CheckMate-816 46 revealed that the predominant adverse reactions of any grade in the nivolumab + chemotherapy group were anemia (37%), leukopenia (28%), and neutropenia (21%). The incidence of anemia was comparable to that observed in our study, while the rates of leukopenia and neutropenia were higher in comparison. The incidence of adverse reactions to treatment combining camrelizumab is generally low, and all related adverse reactions are controllable.
From a medical and health perspective in China, numerous studies48–51 indicate that the combination of camrelizumab and chemotherapy offers substantial cost-effectiveness for patients with NSCLC when compared to standard chemotherapy. Zhou et al. 52 found that camrelizumab, developed in China, is more available and cost-effective compared to imported drugs while offering a favorable curative effect. Liang et al.’s 49 research showed that the incremental cost-effectiveness ratio (ICER) of camrelizumab combined with chemotherapy is $25,674/QLY (quality-adjusted life year), and the probability of cost-effectiveness is >90% regardless of the expression level of PD-L1. Wu et al. 53 found that in the treatment of advanced NSCLC, the ICER of camrelizumab was lower than that of atezolizumab, pembrolizumab, and nivolumab. However, the latest study 54 found that pembrolizumab combined with chemotherapy is the most cost-effective option for patients with advanced squamous NSCLC. In China, camrelizumab combined with chemotherapy in the treatment of non-squamous NSCLC and squamous NSCLC has been included in the medical insurance, reducing the economic burden for NSCLC patients. Generally speaking, camrelizumab combined with chemotherapy is an economical and effective first-line treatment for advanced squamous NSCLC in China.
In addition, there are some limitations in this study. First, some of the included studies are single-arm studies, which are of lower quality and evidence level compared with RCTs and cohort studies. Second, the sample size of RCTs and cohort studies is small, the literature data is limited, and some indicators are heterogeneous, which may lead to publication bias. Third, although this study confirmed that therapy combined with camrelizumab prolonged OS, PFS, and EFS. Due to limited literature availability and insufficient data for subgroup analysis, conducting a comprehensive analysis regarding differences in gender, age, smoking, specific drug regimen, NSCLC pathological types, PD-L1 expression, and organ metastasis is not feasible. This limitation may impact the conclusion’s reliability and hinder the determination of the optimal treatment plan, necessitating further experiments to address data gaps. 4 Camrelizumab has not been approved for the treatment of lung cancer in foreign countries, and there is no relevant research abroad at present. Therefore, the clinical studies included are all from China, which will affect the representativeness of the research and may have some bias, thus affecting the generalization of the conclusion. This paper has the following strengths. First, it is the first meta-analysis to explore the efficacy of camrelizumab comprehensive treatment for NSCLC by including RCTs, cohort studies, and single-arm studies. Second, most of the included RCTs and cohort studies are prospective studies with high quality, and low-quality studies such as retrospective and cross-sectional studies were not included. Third, the conclusion of this article further supports the efficacy and safety of camrelizumab in treating NSCLC and also provides theoretical support for the popularization of camrelizumab in NSCLC patients and the further development of large-scale clinical trials.
To sum up, this paper systematically analyzed the efficacy and safety of camrelizumab in treating advanced NSCLC. The clinical impact of combining camrelizumab treatment is satisfactory, offering significant benefits for patients in OS, PFS, and EFS, but shows no clear improvement in DFS. Regardless of PD-L1 expression in squamous NSCLC patients, combining immunotherapy with chemotherapy can enhance OS and PFS. However, the survival advantage is not significant for non-squamous NSCLC patients with less than 1% PD-L1 expression when treated with combined chemotherapy. However, patients with advanced NSCLC can benefit from camrelizumab combined with apatinib regardless of PD-L1 expression status. In addition, therapy combining camrelizumab may increase the risk of adverse reactions such as leukopenia and capillary endothelial hyperplasia, which, although not severe, should not be overlooked. Among them, RCCEP is a distinct adverse reaction associated with camrelizumab, characterized by skin manifestations such as capillary hyperplasia and endothelial cell proliferation in the dermis. Camrelizumab-induced RCCEP may be related to the curative effect and can be used as a predictor of the curative effect. 55 This necessitates the timely monitoring of hematological indicators, observation of patient reactions, provision of guidance to patients on symptom management, and adjustment of dosages as necessary. Although our study has some limitations, it is the first to integrate RCTs, cohort studies, and single-arm studies to investigate the efficacy of camrelizumab. More well-conceived, large-scale prospective RCTs are necessary to thoroughly analyze the efficacy of camrelizumab in treating NSCLC, accurately locate the population that is most suitable to be treated with camrelizumab combination therapy, make an objective and effective medication plan for patients, and monitor the side effects, with the aim to provide a more reliable reference for clinical treatment of NSCLC.
Conclusion
This comprehensive analysis shows that camrelizumab combination treatment can well improve the OS, PFS, and EFS of NSCLC patients at advanced stages, and has good safety and controllable drug-related adverse reactions. The expression status of PD-L1, smoking history, and the occurrence of RCCEP can predict the clinical benefit of camrelizumab combination therapy. Clinicians should exercise caution in prescribing medication to patients with PD-L1 expression below 1% who are nonsmokers or light smokers. Considering the inadequate sample size of the included literature, well-designed RCTs with a large sample size are warranted to further evaluate more effective combination therapy for advanced NSCLC without increasing adverse reactions, and further confirm its feasibility of worldwide popularization.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241284904 – Supplemental material for Efficacy and safety of camrelizumab-based comprehensive treatment for non-small cell lung cancer: a systematic review and meta-analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359241284904 for Efficacy and safety of camrelizumab-based comprehensive treatment for non-small cell lung cancer: a systematic review and meta-analysis by Nuerbiyamu Maimaitiyiming, Yue Li, Yunfeng Cao and Yanwei Li in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-pdf-2-tam-10.1177_17588359241284904 – Supplemental material for Efficacy and safety of camrelizumab-based comprehensive treatment for non-small cell lung cancer: a systematic review and meta-analysis
Supplemental material, sj-pdf-2-tam-10.1177_17588359241284904 for Efficacy and safety of camrelizumab-based comprehensive treatment for non-small cell lung cancer: a systematic review and meta-analysis by Nuerbiyamu Maimaitiyiming, Yue Li, Yunfeng Cao and Yanwei Li in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
