Abstract
Most cases of cervical cancer are still caused by persistent infection with high-risk human papillomavirus (HPV) variants, which also drives the development of ~30% of vulvar and ~76% of vaginal malignancies. Implementation of HPV vaccination has significantly decreased the incidence of high-grade pre-cancerous cervical, vulvar and vaginal lesions. However, cervical, vulvar and vaginal cancers can still develop (with or without HPV integration) and treatment options are limited compared to more common cancers. As with many other cancer types, molecular studies should identify targeted agents that could be added to treatment regimens to improve response rates and survival. Combination regimens involving chemoradiotherapy, anti-angiogenics and immune checkpoint inhibitors should be considered in the first instance. Then, depending on the molecular profile of a particular tumour, more targeted therapies should be considered. In particular, HER2-targeted therapies are likely to be a viable treatment option for many individuals, including those with cervical adenocarcinoma of gastric-type and vulvar Paget’s disease. In cervical cancer, TGFβ, PI3K, ATR and PARP inhibitors have shown some benefit and warrant further investigation. In vulvar cancer, combination therapies involving EGFR inhibitors require ongoing evaluation. In vaginal cancer, combination therapies targeting the PI3K and MAPK pathways should be investigated for squamous cell carcinoma and melanoma, respectively. Finally, with the rapid expansion of antibody-drug conjugates in recent years, this is an especially exciting area of investigation. For cervical, vulvar and vaginal cancers specifically, trastuzumab deruxtecan and tisotumab vedotin could be important therapeutic options in the right context. In this review, we describe the molecular features of different cervical, vulvar and vaginal cancer types, current genomically-matched therapies being investigated and discuss treatment strategies with future potential.
Plain language summary
Most cases of cervical cancer are still caused by persistent infection with high-risk human papilloma virus (HPV) variants, which also drives the development of ~30% of vulvar and ~76% of vaginal malignancies. HPV vaccination has been an important preventative mechanism for these types of malignancies, however, they can still arise (with or without HPV infection). Treatment options for cervical, vulvar and vaginal cancers are still limited compared to more common cancers. Investigating changes in the DNA in a particular tumour may indicate therapeutic options involving targeted agents (specific for the DNA alterations). Clinical trials have been developed to test responses to some of these targeted agents in tumours carrying the relevant DNA alterations. Results from these trials indicate that drugs targeting specific molecules, such as HER2, TGFβ, PI3K, ATR and PARP, may be effective against cervical, vulvar or vaginal tumours harbouring relevant DNA alterations. In this review, we describe the most frequent DNA changes observed in the different subtypes of cervical, vulvar and vaginal cancers and discuss different therapies currently being investigated to target these changes, with consideration of what might be possible in the future.
Keywords
Background of cervical, vulvar and vaginal cancer
Incidence
Cervical cancer is the fourth most commonly diagnosed cancer in women, accounting for about 45% of all gynaecological cancers, and is currently responsible for the most gynaecological cancer-related deaths worldwide.1,2 Cervical cancer screening programmes have more than halved the incidence of cervical cancer in developed countries over the past three decades; however, rates remain high in the developing world. 3 Vulvar and vaginal cancer account for just 2%–8% and 1%–2% of all gynaecological malignancies, respectively.1,2
Most cases of cervical cancer are still caused by persistent infection with high-risk human papillomavirus (HPV) variants. 4 Implementation of HPV vaccination programmes has significantly reduced the incidence of pre-cancerous cervical lesions and is expected to prevent up to 90% of cervical cancers in developed countries. 5 Similarly, the incidence of vulvar and vaginal pre-cancerous lesions have significantly reduced since the implementation of HPV vaccination. 6
Pathological subtypes
The two main categories of cervical cancer are squamous cell carcinomas (SCC; ~70%–80%, including keratinising, non-keratinising and small cell squamous carcinomas) and adenocarcinomas (~20%–25%, including mucinous, clear cell and serous), arising from the ectocervix and endocervix, respectively.7 –9 Mucinous adenocarcinomas are the most common type of adenocarcinomas, comprising endocervical, intestinal and gastric subtypes. 9 Gastric-type adenocarcinoma of the uterine cervix (GAS) is a rare HPV-negative variant with a poor prognosis. 10 A third category, called ‘other epithelial cancers,’ contains very rare histological types such as adenosquamous, neuroendocrine, small cell and large cell neuroendocrine carcinomas.7 –9 Cervical sarcomas are extremely rare (<1% of all cervical malignancies). 11
Most vulvar malignancies arise in the labia and the most common subtype is SCC of the vulva, accounting for up to 88% of cases.12 –14 Other, rarer histological types include melanoma (7.5%), adenocarcinoma (4.5%; including adenocarcinoma related to extra-mammary Paget’s disease (EMPD)), basal cell carcinoma, verrucous carcinoma, Bartholin gland carcinoma and sarcoma.12 –14
Vaginal malignancies mostly (in >80% of cases) arise as a result of cervical, endometrial or vulvar cancer recurrence in the vaginal area.13,15 Most invasive vaginal malignancies are SCC (85%–95%), with melanoma and adenocarcinomas accounting for approximately 3% of cases each.13 –15 Rarer histological types include neuroendocrine, papillary squamo-transitional cell carcinoma and sarcoma.14,16
Survival rates
The mean 5-year survival (5-YS) for all types and stages of cervical cancer is 67%; however, the full range is 91% for early stage cervical cancer, 60% when there is spread to local organs, and devastatingly low, at only 19%, for cancer that has spread to distant parts of the body. 17 The histological types adenocarcinoma and small cell carcinoma of the cervix are associated with the poorest survival. 8 The mean 5-YS for vulvar cancer is 60%–80% for early stage disease (International Federation of Gynaecology and Obstetrics (FIGO) stages I and II) and 20%–40% for late-stage disease (FIGO stages III and IV). 13 Similarly, the prognosis of vaginal cancer is also strongly associated with the stage at diagnosis, with a mean 5-YS of 40%–100% for FIGO stages I–II and 0%–50% for stage III–IV.13,14,18
Current standard treatment strategies
The current treatment strategy for early stage disease is surgery and/or platinum-based concurrent chemoradiotherapy (CCRT), and for later-stage disease is CCRT. 19 For advanced cervical cancer, brachytherapy and/or external beam radiotherapy (EBRT) are essential components of treatment. 19 Bevacizumab (an inhibitor of vascular endothelial growth factor (VEGF)-A) and, recently, an immune checkpoint inhibitor (ICI), pembrolizumab (an inhibitor of programmed death (PD)-1 receptor), have been approved as additions to platinum-based chemotherapy in the first-line setting for advanced cervical cancer.20,21 These and other targeted therapeutic strategies will be discussed in more detail below.
For vulvar cancer, the treatment of early stage disease usually involves surgery, with or without CCRT. 22 For locally advanced disease that is initially inoperable, primary treatment may include CCRT to increase surgical feasibility. 22 However, vulvar cancer is considered to be relatively chemoresistant and treatment with chemotherapy alone has not improved survival rates. 23
Due to its location, curative organ-sparing surgery is not often a primary treatment option for vaginal malignancies. 15 Stage I disease may be treated with CCRT alone or following surgery. For stage II–III disease, a combination of brachytherapy and EBRT is most commonly used, and CCRT is sometimes included in treatment strategies. For stage IV disease, CCRT is suggested for optimal tumour control. 15 More effective treatments are desperately needed for cervical, vulvar and vaginal cancer, and, as with many other cancer types, molecular studies should identify targeted agents that could be added to treatment regimens, to improve response rates and survival.
Genomic landscape
Cervical cancer
Persistent HPV infection (due to DNA integration) drives the development of the majority of cervical and vaginal cancers (~90% and ~76%, respectively) as well as ~30% of vulvar cancers.4,24,25 Cancers harbouring HPV integration may have the following biological features: inhibition of apoptosis, telomere shortening, promotion of genomic instability, angiogenesis, invasion and metastasis, and immune response suppression and escape.26,27 Further discussion of these characteristics, and specific HPV-targeting strategies, is beyond the scope of this review (see other Review in this Special Collection). Instead, we will focus on molecular aberrations (some may result from HPV integration) and targeting strategies relevant to these specific changes.
The most commonly mutated genes in cervical cancer are displayed in Table 1.28 –35 For SCC of the cervix these are PIK3CA (13%–38%), KMT2D (14%–15%), FBXW7 (4%–15%) and EP300 (11%–14%); for adenocarcinoma they are PIK3CA (17%–35%), KRAS (7%–24%), ERBB2 (6%–24%), TP53 (4%–18%) and ARID1A (10%–16%); and for ‘other’ cervical cancer types they are PIK3CA (0%–38%), MYC (8%–17%), ARID1A (8%–16%) and TP53 (6%–13%). Mutations in the TERT promoter are more frequent in SCC and ‘other’ cervical cancers (12% and 11%, respectively). 29 HPV integration sites have been commonly found in close proximity to, or within the reading frame of, many genes, including MYC, HMGA2, ERBB2, TP63, FANCC and RAD51B, leading to up or down-regulation of the respective genes, depending on the site of integration.30,32,36 The mean rate of tumour mutational burden (TMB) in cervical cancer is relatively high at 5–6 mutations per megabase. 37 This is thought to be due to APOBEC (Apolipoprotein B mRNA-editing enzyme, catalytic polypeptide) activity as a result of HPV infection. 37 This suggests that cervical cancer may have a high number of neoantigens, which could stimulate the immune system.
Frequencies of mutations in commonly altered genes in the three main groups of cervical cancer (where available).

Only small number of ‘other’ cases in some studies and so a mutation in one case in this group results in an abnormally high frequency.
AC, adenocarcinoma; SCC, squamous cell carcinoma.
The most commonly amplified or deleted genes in cervical cancer are displayed in Table 2.28,35 Copy number (CN) gains in MYC, YAP1 and EGFR, as well as YAP1, EGFR, SRC and MAPK signalling (as indicated by the expression of the relevant phosphorylated proteins), were associated with cervical SCC.28,35 In cervical adenocarcinomas, CN gains in ERBB2, BCAR4 (a gene encoding the BCAR4 protein that has been shown to activate HER2 signalling 38 ), HPV integration near ERBB2 and increased HER2 protein expression were more common.28,32,35 In particular, one study identified GAS as being the subtype most likely to harbour ERBB2 amplification (14.7%). 39 CN losses in TGFBR2 and SMAD4 were also associated with adenocarcinomas.28,35 Taken together, up-regulated signalling involving EGFR, SRC, YAP1 and the MAPK pathway appears to be a key feature of many SCC, whereas up-regulated HER2 signalling is a key feature of many adenocarcinomas. These results agree with a previous study where EGFR, HER3 and HER4 were found to be over-expressed in 63%, 74.4% and 79.5% of cervical SCC cases, respectively, and HER2 was only over-expressed in 21.8% of SCC cases. 40 CN gains in TERC (78%), MECOM (78%), TP63 (77%) and PIK3CA (29%–46%) were common in all types of cervical cancer.28,35 In addition, CN gains were identified in the two genes encoding PD-ligand 1 and 2 (PD-L1 and PD-L2; CD274 and PDCD1LG2, respectively) in 21% of all cervical cancer cases. 28 The tumour proportion score (TPS) and combined positive score (CPS) are the most common methods for assessment of PD-L1 expression (as measured by immunohistochemistry) and refer to the number of PD-L1 positive cells (tumour cells only or tumour cells, lymphocytes and macrophages, respectively) as a percentage of all tumour cells. 41 The cut-off values can be ⩾1% to ⩾10% PD-L1-positive cells for cervical tumours to be counted as PD-L1-positive. 42 Different studies have indicated 32%–70% of cervical cancers are PD-1- or PD-L1-positive. 42 Differences have also been observed between subtypes, with PD-L1 expression being observed in 14% of adenocarcinomas compared to 54% of SCC. 43 Additional standardisation of assessment methods is required to realise the true utility of PD-L1 testing.
Frequencies of amplifications and deletions of commonly altered genes in the three main groups of cervical cancer (or overall, if subtype information not available).
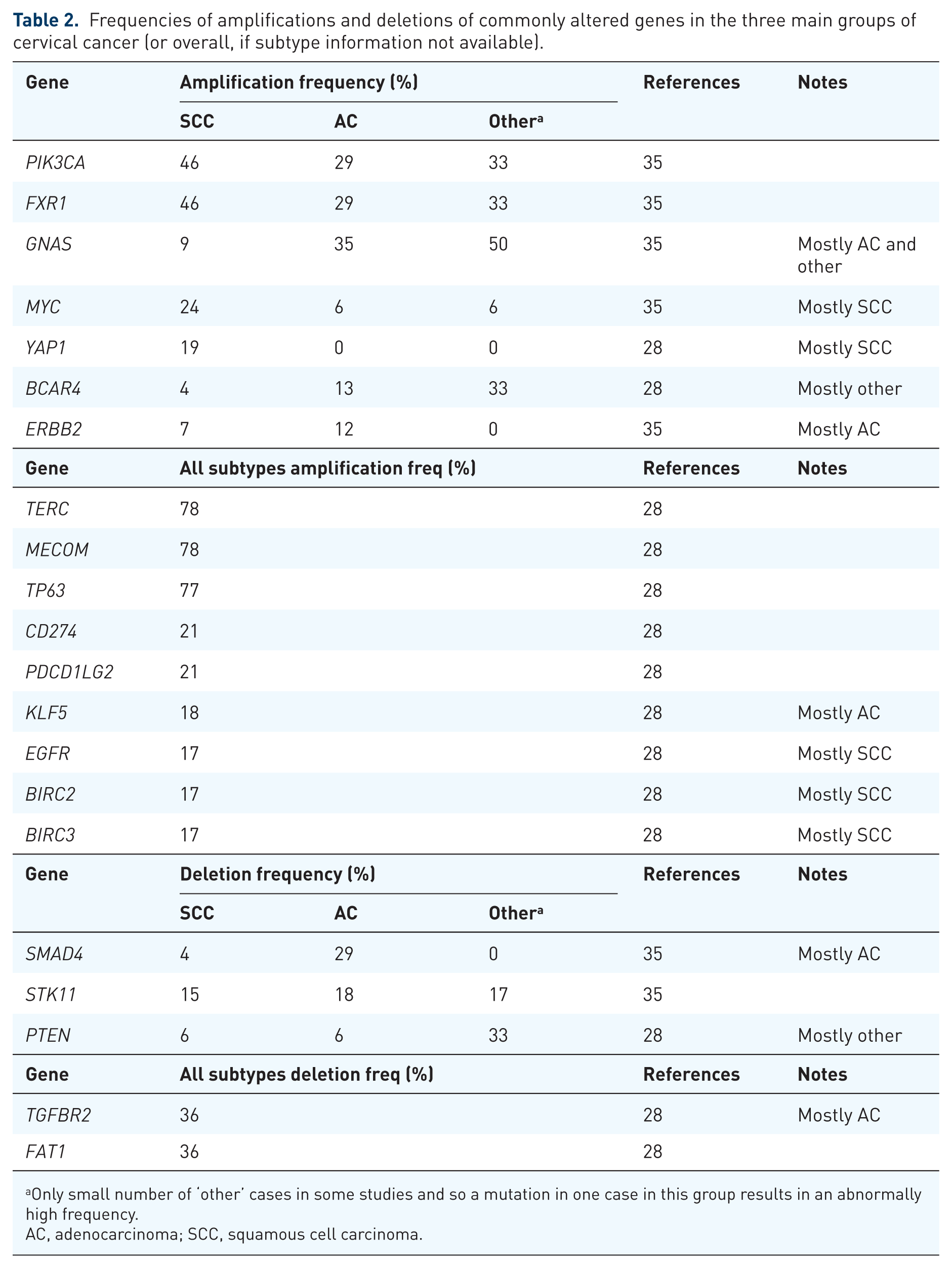
Only small number of ‘other’ cases in some studies and so a mutation in one case in this group results in an abnormally high frequency.
AC, adenocarcinoma; SCC, squamous cell carcinoma.
With the exception of the Friedman study, 29 most molecular data for cervical cancer have been obtained from early stage disease, and so future studies should include samples from later-stage disease and metastatic sites to provide details on genetic heterogeneity and potential targets in advanced cervical cancers.
Vulvar cancer
Vulvar cancers are classified as ‘usual type’, which are HPV-positive, and ‘differentiated type’, which are HPV-negative and mostly attributed to driver mutations in TP53.22,44 While mutations in TP53 are more common in HPV-negative tumours (41%–92%), they do also occur in HPV-positive tumours (0%–50%).45–52 Woelber et al. 53 divided vulvar cancers into three groups based on p16 expression, with p16 over-expression (p16+) as a marker of HPV-mediated transformation. The three groups are p53+ (indicating p53 over-expression as a result of TP53 mutation), p53-/p16- and p53-/p16+, with the latter group having an improved prognosis. 53
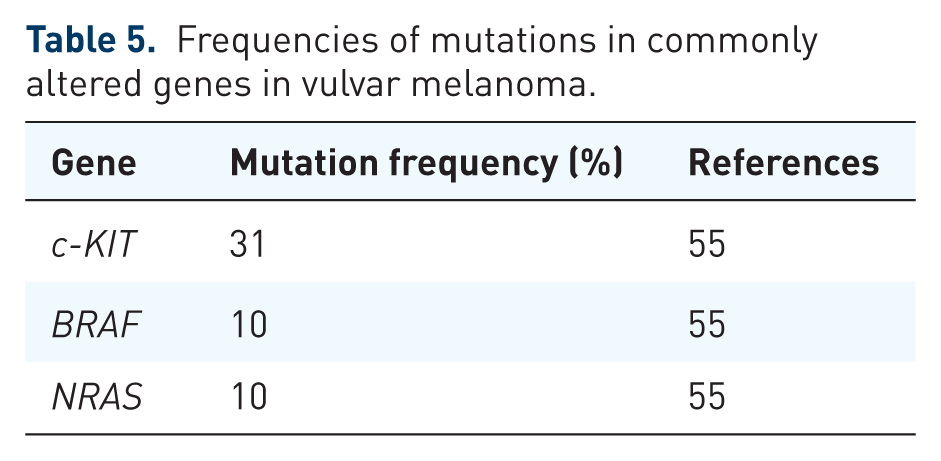
The most commonly mutated genes in SCC of the vulvar (HPV-positive and negative tumours grouped together) are TP53 (33%–82%), CDKN2A (12%–53%), NOTCH1 (11%–41%), FAT1 (11%–41%), HRAS (5%–28%) and PIK3CA (6%–24%; Table 3). Frequent mutations are also observed in the promoter of TERT (19%–82%).46,50 Taking into account HPV status, mutations in TP53, CDKN2A, NOTCH1, FAT1, HRAS and the TERT promoter are more common in HPV-negative tumours, whereas mutations in PIK3CA and c-KIT tend to be more common in HPV-positive tumours (Table 3).45 –51 CN gains in CCND1, EGFR and FGF3/ FGF19/FGF4 (‘FGF cluster’) have been observed in 6%–18% of all vulvar SCC cases, with CCND1 amplifications being more common in HPV-positive tumours (Table 4).46,50 CN gains have also been observed in CD274 and PDCD1LG2 in up to 4% of cases (Table 4).46,50 A recent meta-analysis of 19 studies for PD-L1 expression in vulvar cancer indicated high heterogeneity between studies, with a pooled CPS of 83.4% and a pooled TPS of 53.9%. 54 In addition, high TMB has been observed in >10% of cases, further suggesting these tumours may respond to immunotherapies. 46 However, unlike cervical SCC, HPV-positivity does not appear to be associated with increased immune markers. 46 There have been a few molecular studies carried out on rare vulvar melanomas. 55 Hou et al. 55 carried out their own study, as well as including the data from 17 other studies (~200 cases in total), and found mutations in c-KIT, NRAS and BRAF in 31%, 10% and 10% of cases, respectively (Table 5). The findings from these studies of vulvar cancer genomics are summarised in Tables 3–5.
Frequencies of mutations in commonly altered genes in vulvar SCC (including HPV+ and HPV− frequencies, where available).

HPV, human papilloma virus; SCC, squamous cell carcinoma.
Frequencies of amplifications and deletions in commonly altered genes in vulvar SCC (including HPV+ and HPV− frequencies where available).

FGF cluster includes FGF3, 4 and 19.
FGF, fibroblast growth factor; HPV, human papilloma virus; n/a, not available; SCC, squamous cell carcinoma.
Frequencies of mutations in commonly altered genes in vulvar melanoma.
Vaginal cancer
Molecular data are largely lacking for vaginal cancer, mostly limited to a few molecular studies of rare vaginal melanomas. 55 Hou et al. 55 described findings from their own cohort, as well as including data from 17 other studies (~100 cases in total) and found mutations in NRAS, BRAF and c-KIT in 19%, 6% and 6% of cases, respectively (Table 6). Only one molecular study has analysed the most common vaginal cancer, SCC, reporting on just seven cases. 52 PIK3CA (71%), TP53 (29%), KMT2D (29%) and CREBBP (29%) were the most frequently mutated genes in this small cohort of samples (Table 6). 52 The single HPV-negative case in this study had a mutation in the TERT promoter, mirroring HPV-negative vulvar SCC, where the frequency of TERT promoter mutations is 55%–100%.46,50,52 The most frequently amplified genes in vaginal SCC are CCND1 (14%) and the FGF cluster (14%), and the most frequently deleted gene is FAT1 (14%); however, these numbers are derived from just seven cases, and more molecular studies are undoubtedly required (Table 7). 52 We have molecularly profiled six cases of vaginal cancer, two being SCC and four being adenocarcinoma, which includes two mucinous adenocarcinoma (GAS) cases, adding to the limited data currently available (Table 8).
Frequencies of alterations in commonly altered genes in vaginal cancer.
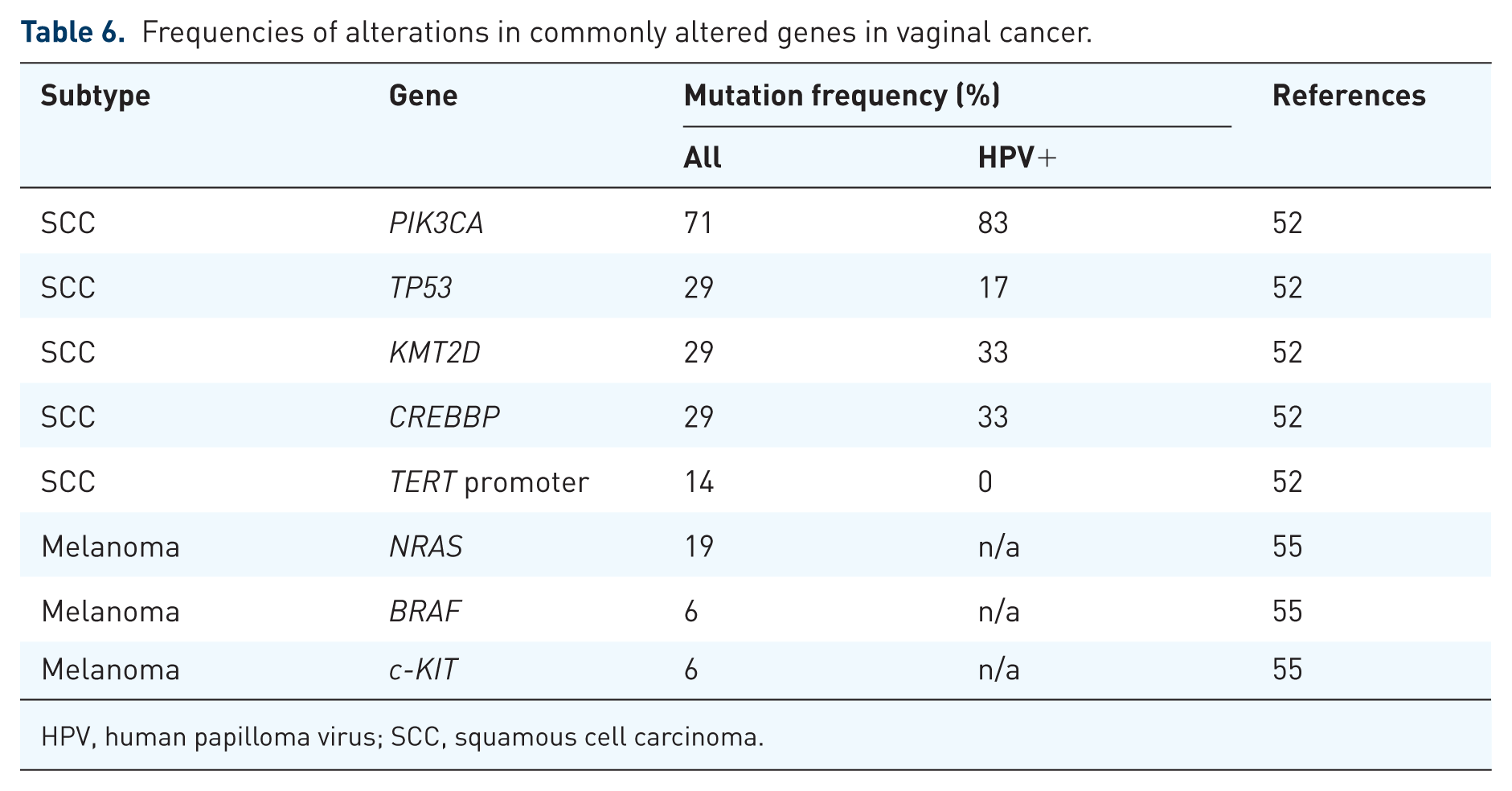
HPV, human papilloma virus; SCC, squamous cell carcinoma.
Frequencies of amplifications and deletions in commonly altered genes in vaginal cancer.

FGF cluster includes FGF3, 4 and 19.
CN, copy number; HPV, human papilloma virus; SCC, squamous cell carcinoma.
Alterations identified in vaginal cancer cases (2 × SCC and 4 × AC) in the Stafford Fox Rare Cancer Program.
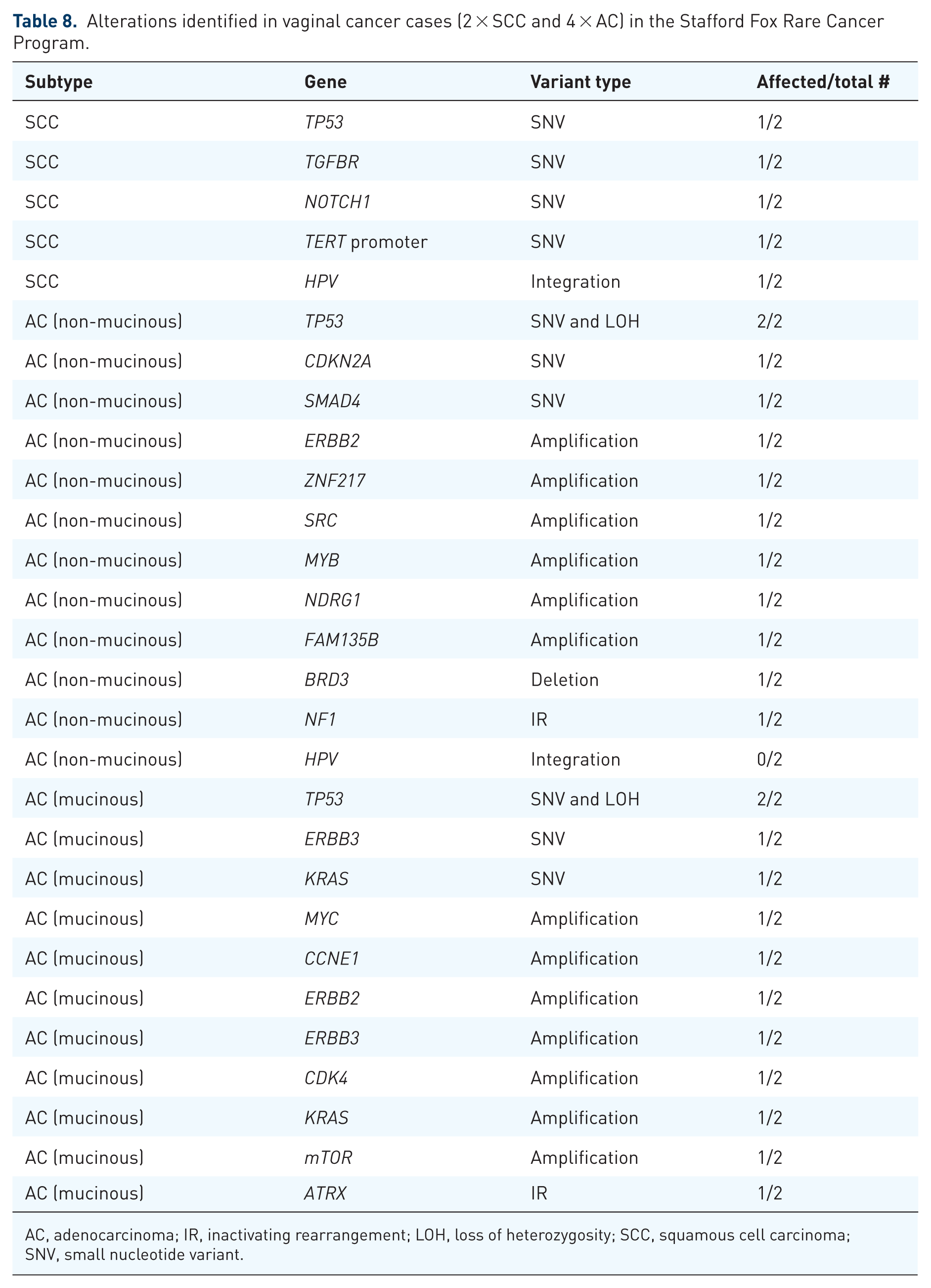
AC, adenocarcinoma; IR, inactivating rearrangement; LOH, loss of heterozygosity; SCC, squamous cell carcinoma; SNV, small nucleotide variant.
Targeted therapies for the treatment of cervical cancer
Although matching targeted therapies based on molecular profiling has changed the treatment landscape for many cancer types, cervical cancer has lagged behind. Therefore, the prognostic value of cervical cancer genomics is yet to be determined. The National Cancer Institute-Molecular Analysis for Therapy Choice (NCI-MATCH) phase II trial (NCT02465060), which investigated multiple treatment options based on tumour genomic testing, enrolled 25 individuals with cervical cancer in specific trial arms. 56 Analysis indicated that 28.4% of the 25 individuals with cervical cancer had an actionable molecular alteration, suggesting tumour genomic testing was worthwhile. However, whether there have been responses to matched treatments is yet to be reported. 56
Based on the available molecular data for cervical cancer, combination therapeutic strategies involving EGFR, FGFR3, SRC, TGFβ, MAPK and PI3K inhibitors may benefit individuals with SCC of the cervix. For adenocarcinomas of the cervix, relevant combinations of drugs targeting HER2, HER3 and PI3K may be effective. Inhibitors of the PI3K and MAPK pathways, as well as of ATR and PARP (for tumours harbouring ARID1A mutations), may be included in combination strategies for ‘other’ cervical cancers. As HPV infection is prevalent in all histological types of cervical cancer, except for GAS, cell cycle checkpoint and DNA repair inhibitors, as well as immunotherapies, may be effective irrespective of molecular profile. In addition, high expression of PD-L1 and PD-L2 in about a fifth of all cervical cancers further suggests these tumours may respond to immunotherapies. 28 Below, we will discuss targeted therapies with observed patient benefit in cervical cancer, with full details provided in Table 9.
Clinical trials involving cervical cancer and targeted therapies where clinical benefit has been observed.
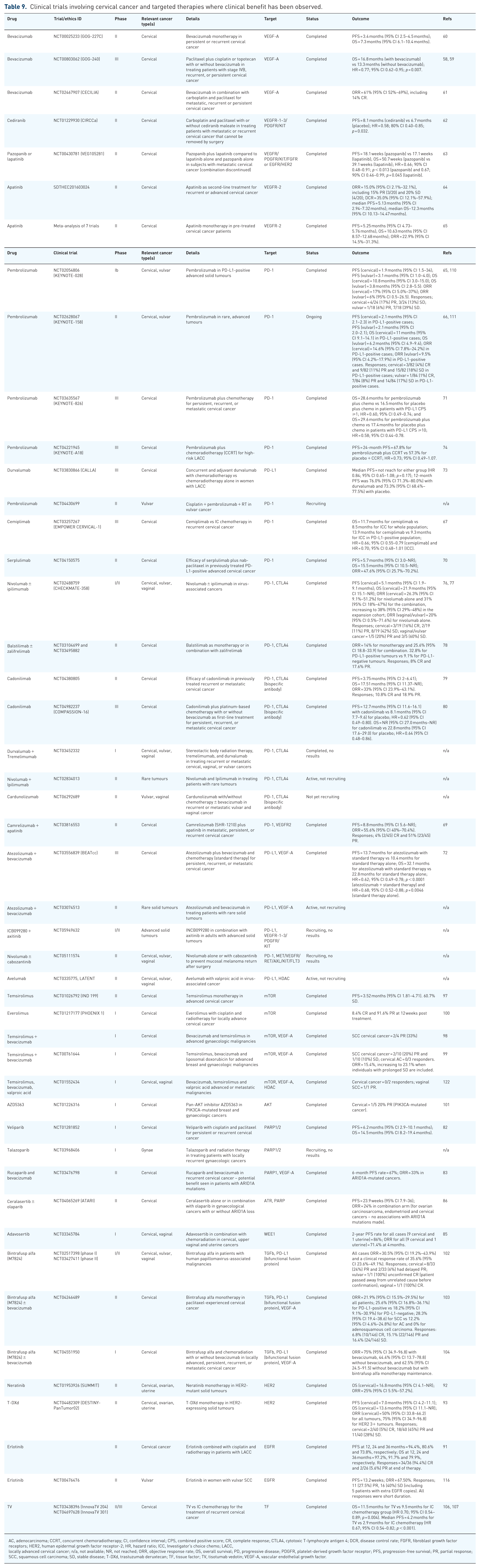
AC, adenocarcinoma; CCRT, concurrent chemoradiotherapy; CI, confidence interval; CPS, combined positive score; CR, complete response; CTLA4, cytotoxic T-lymphocyte antigen 4; DCR, disease control rate; FGFR, fibroblast growth factor receptors; HER2, human epidermal growth factor receptor-2; HR, hazard ratio; ICC, Investigator’s choice chemo; LACC, locally advanced cervical cancer; n/a, not available; NR, not reached; ORR, objective response rate; OS, overall survival; PD, progressive disease; PDGFR, platelet-derived growth factor receptor; PFS, progression-free survival; PR, partial response; SCC, squamous cell carcinoma; SD, stable disease; T-DXd, trastuzumab deruxtecan; TF, tissue factor; TV, tisotumab vedotin; VEGF-A, vascular endothelial growth factor.
Vascular endothelial growth factor/R
Inhibitors of VEGF and its receptors target a feature that is present in all solid cancers, namely angiogenesis. A promising phase II trial of bevacizumab monotherapy for persistent or recurrent cervical cancer (NCT00025233), demonstrated progression-free survival (PFS) of 3.4 months. 57 The GOG 240 trial (NCT00803062) then investigated combination strategies, with a statistically significant improvement in median overall survival (OS) being observed (16.8 months for cisplatin/paclitaxel plus bevacizumab vs 13.3 months for chemotherapy alone). 58 This resulted in the approval of the addition of bevacizumab to standard platinum-based chemotherapy for advanced cervical cancer in 2014.58,59 Similar benefits were observed in the global, single-arm phase II CECILIA trial (NCT02467907), which showed that the addition of bevacizumab to carboplatin/paclitaxel chemotherapy was safe, providing an alternative chemotherapy backbone for patients who may not tolerate cisplatin. 60
The small-molecule tyrosine kinase inhibitor (TKI), cediranib, has anti-angiogenic activity through inhibition of VEGFR-1/2/3, platelet-derived growth factor receptor (PDGFR) and KIT. Its efficacy has been investigated in combination with chemotherapy for the treatment of cervical cancer in the CIRCCa phase II trial (NCT01229930). A significantly longer PFS was observed for the cediranib arm compared to placebo (8.1 vs 6.7 months). 61 Increased toxicity was observed with cediranib, but this was thought to be manageable as no reduction in overall quality of life occurred. 61 The TKI, pazopanib, which targets VEGFR-1/2/3, PDGFR-α/β, KIT and fibroblast growth factor receptors (FGFR)-1/2, has also been investigated for the treatment of patients with advanced and recurrent cervical cancer in a phase II trial (NCT00430781). Patients who received pazopanib had a longer median OS compared with patients who received single-agent lapatinib, a dual EGFR/HER2 inhibitor (50.7 vs 39.1 weeks). 62 Due to toxicity issues and minimal efficacy, pazopanib has not been investigated further with chemotherapy or included in other treatment strategies for cervical cancer since.
Another VEGFR-2 inhibitor, apatinib, has also been investigated for the treatment of advanced cervical cancer in a phase II trial, where chemotherapy with or without bevacizumab had previously been unsuccessful. Modest benefit was observed in this study, with median PFS of 5.13 months and an objective response rate (ORR) of 16.7%. 63 A meta-analysis has since been carried out to assess the efficacy of apatinib monotherapy in seven studies of 243 pre-treated Chinese patients with cervical cancer. 64 The combined ORR was 22.9%, which increased to 24.5% when selecting patients without prior exposure to bevacizumab. 64 Taken together, there is clear evidence supporting the inclusion of anti-angiogenic therapy, most clearly for bevacizumab at this stage, in combination strategies for the treatment of all cervical cancer subtypes.
Immunotherapy
ICIs are used to treat cervical cancer due to specific features discussed earlier, such as amplification of the two genes encoding PD-L1 and PD-L2.28,32 Similarly, a relatively high TMB and enrichment of APOBEC mutational signatures further support the use of ICIs. 28 Consequently, many clinical trials including ICIs have had promising results, with a number of PD-1 inhibitors (PD-1i) receiving approval in various cervical cancer settings. Early trials tested the efficacy of single-agent ICI therapy in the advanced cervical cancer setting in patients who had received prior systemic chemotherapy.
The KEYNOTE-028 phase Ib trial (NCT02054806), which investigated the efficacy of single-agent pembrolizumab in individuals with advanced PD-L1-positive cervical cancer (96% of whom had received prior platinum chemotherapy), achieved an ORR of 17%. 65 Consistent results were obtained in the KEYNOTE-158 phase II basket trial (NCT02628067), where an ORR of 14.6% was achieved in patients with previously treated, PD-L1-positive, advanced cervical cancer treated with pembrolizumab monotherapy. 66 Impressively, for a monotherapy following other lines of treatment, three patients achieved complete response (CR) and nine patients achieved partial response (PR; out of 98 patients treated), resulting in rapid approval of pembrolizumab in this setting. 66 The phase III EMPOWER CERVICAL-1 trial included patients with recurrent cervical cancer who had progressed on platinum-based therapy and compared treatment with single-agent PD-1i, cemiplimab, versus Investigator’s Choice (IC) chemotherapy (NCT03257267). An OS of 11.7 months was achieved for the overall population treated with cemiplimab compared with 8.5 months for the IC chemotherapy arm. 67 Median OS was even longer when selecting for patients with PD-L1-positive tumours. 67 This led to approval of cemiplimab in the recurrent cervical cancer setting in various countries around the world. 68 These encouraging single-agent ICI results led to the investigation of ICI combination therapies in cervical cancer, initially in the subsequent line setting.
The combination of an ICI with an anti-angiogenic drug was studied in a phase II trial of the PD-1i, camrelizumab (SHR-1210), combined with apatinib in metastatic, recurrent or persistent cervical cancer, following at least one line of systemic chemotherapy (NCT03816553). An impressive ORR of 55.6% was achieved, including two CR and 23 PR (out of 45 patients treated). 69 In previously treated patients with PD-L1-positive advanced cervical cancer, in a phase II trial investigating response to the PD-1i, serplulimab, in combination with nab-paclitaxel (NCT04150575), an ORR of 47.6% was achieved. 70
These encouraging results in the previously treated advanced cervical cancer setting led to two first-line phase III studies including ICI combinations with chemotherapy. The first-line phase III KEYNOTE-826 trial (NCT03635567) compared the addition of ICI to chemotherapy, with or without bevacizumab, in persistent, metastatic or recurrent cervical cancer. This trial observed an improvement in OS from 16.5 months for placebo plus chemotherapy, with or without bevacizumab, to 28.6 months for pembrolizumab plus chemotherapy, with or without bevacizumab, in the PD-L1 CPS ⩾1% cohort. 71 Even greater benefit was observed in the PD-L1 CPS ⩾10% cohort, with OS increasing from 17.4 to 29.6 months when ICI was included. 71 This study led to approval of pembrolizumab with chemotherapy, with or without bevacizumab, in the first-line cervical cancer setting, and as ~90% of cases had a PD-L1 CPS of ⩾1%, some approvals based on this study were independent of PD-L1 status. 21
The first-line phase III BEATcc trial (NCT03556839) studied the triple combination of the PD-L1 inhibitor (PD-L1i), atezolizumab, with bevacizumab and platinum/paclitaxel chemotherapy, in patients with metastatic, resistant or recurrent cervical cancer. A significant improvement in median PFS was observed (13.7 vs 10.4 months). 72 The benefits of adding anti-angiogenic drugs and ICIs to therapeutic strategies for cervical cancer are clear, with both types of targeted therapies approved in multiple settings.
These results indicate that ICIs are indeed useful for treating cervical cancer in the first-line setting, delivered in combination with chemotherapy, with or without bevacizumab, and with or without prior chemotherapy exposure, for persistent, metastatic or recurrent cervical cancer. The strength of these results led to an investigation of ICI in treatment regimens during and following initial CCRT for high-risk locally advanced cervical cancer (LACC). While the phase III CALLA study (NCT03830866) found no benefit of adding the PD-L1i, durvalumab, with and following CCRT in LACC, 73 the phase III KEYNOTE-A18 study (NCT04221945) observed benefit for the addition of pembrolizumab in a similar setting, but included patients with higher-risk LACC. 74 In KEYNOTE-A18, a 24-month PFS of 68% for pembrolizumab plus CCRT was achieved, compared with 57% for placebo plus chemoradiotherapy. 74 The conflicting results between the CALLA and KEYNOTE-A18 trials could be due to the different drug actions of durvalumab and pembrolizumab (i.e. PD-L1i vs PD-1i), however, the KEYNOTE-A18 trial enrolled patients with significantly higher-risk disease, requiring ⩾2 positive lymph nodes with the shortest diameter ⩾1.5 cm. 74 Indeed, relevant sub-groups of the CALLA trial, including lymph node-positive Stage III patients, supported the KEYNOTE-A18 finding. 73 The interim results from KEYNOTE-A18 led to approval of pembrolizumab with and following chemoradiotherapy for high-risk LACC in January 2024. 75
Targeting two immune checkpoint proteins simultaneously, both PD-1 and cytotoxic T-lymphocyte antigen 4 (CTLA4), has also been studied in advanced cervical cancer in patients who had received prior systemic therapy. The CHECKMATE-358 phase I/II trial (NCT02488759) studied the PD-1i, nivolumab, with or without the CTLA4 inhibitor (CTLA4i), ipilimumab, in patients with virus-associated recurrent or metastatic cervical cancer (36%–79% of patients had received prior systemic therapy in the metastatic setting).76,77 An ORR of 26.3% was achieved for cervical cancer patients treated with nivolumab compared with an initial ORR of 31% for the combination, followed by an ORR of 38% in the expansion cohort.76,77 The combination of two newer ICIs, the PD-1i, balstilimab and the CTLA4i, zalifrelimab, was investigated in pre-treated recurrent or metastatic cervical cancers in a phase II trial (NCT03495882). An ORR of 25.6% was achieved for the combination arm, which increased to 32.8% when selecting for patients with PD-L1-positive tumours. 78 Excitingly, there were 10 CR and 22 PR (out of 125 patients treated). 78 Similar impressive results were obtained in a phase II trial of the anti-PD-1/CTLA4 bispecific antibody, cadonilimab, for previously treated recurrent and metastatic cervical cancer (NCT04380805). 79 This was followed by the phase III COMPASSION-16 trial (NCT04982237), where individuals with previously untreated persistent, recurrent or metastatic cervical cancer received either cadonilimab or placebo plus platinum-based chemotherapy, with or without bevacizumab, followed by maintenance therapy for up to 2 years. 80 The dual primary end-points were positive, with median PFS of 12.7 months for the cadonilimab group and 8.1 months for the placebo group. 80 Toxicity of this compound will be important to evaluate across ethnicities. In summary, it appears that in cervical cancer, early delivery of ICI, including in the adjuvant setting, improves efficacy, delivering the hope of increasing cure in this otherwise devastating cancer.
DNA repair
A number of features of cervical cancer indicate that PARP inhibitors (PARPi) may be an effective treatment. Not only does expression of the HPV oncoproteins lead to inhibition of the guardian of the genome, p53, and increased DNA damage, but also the RAD51B locus is a common site for viral integration.30,32,36 Disruption of RAD51B function has been found to lead to deficient homologous recombination (HR). 81 Clinical trials investigating PARPi for the treatment of cervical cancer have so far been limited and only one completed trial showed benefit. The PARPi, veliparib, was investigated in combination with paclitaxel and cisplatin for the treatment of persistent or recurrent cervical cancer in a phase I trial (NCT01281852), achieving a PFS of 6.2 months. 82 Interestingly, response to this combination was not associated with HR status, as determined by the presence or absence of FANCD2 foci. 82 A phase II trial investigated the combination of rucaparib and bevacizumab in recurrent cervical cancer (NCT03476798), and while overall there didn’t appear to be improved anti-tumour activity, there did seem to be some benefit in ARID1A-mutated cancers. 83 There are two current trials underway for cervical cancer patients investigating PARPi combinations: pembrolizumab with olaparib (NCT04483544) and the new selective PARP1-specific PARPi, saruparib (AZD5305), as monotherapy or in combination with anti-cancer agents (NCT04644068).
Cell cycle checkpoints
Along with impaired repair of DNA damage, HPV infection also causes reduced activation of cell cycle checkpoints.26,27 This is primarily due to loss of p53 function, which maintains the G1/S checkpoint.26,27 This vulnerability can be exploited by inhibitors of G2 checkpoint components, such as WEE1 and ATR, leading to increased sensitivity to chemotherapy and radiation, and subsequent cell death. 84 A phase I study of the WEE1 inhibitor, adavosertib, in combination with chemoradiation, included nine patients with LACC (NCT03345784). Dose-limiting toxicities were an issue; however, four patients achieved CR and PFS at 2 years was 86%. 85 Response to the ATR inhibitor, ceralasertib, is currently being investigated alone and in combination with olaparib in gynaecological cancers in the ATARI trial (NCT04065269). Cohort three includes 10 cervical cancer patients, with preliminary analysis indicating an ORR of 24%. 86 Future analysis will involve comparisons between patients with and without ARID1A loss. 86 Clearly, this is an avenue that warrants further investigation.
EGFR family
With up-regulated EGFR and HER2 signalling being key features of cervical cancer, there have been a number of studies involving drugs that target these pathways. Studies involving EGFR inhibitors (EGFRi), such as gefitinib, erlotinib and cetuximab, as monotherapies or in combination with standard chemotherapy, have been mostly disappointing.87 –90 The one exception was a phase II study of erlotinib combined with CCRT, reporting PFS at 24 months of 80.6%, 91 compared to other studies involving CCRT alone, where PFS at 24 months ranged from 66% to 76%. 91
There have been fewer cervical cancer clinical trials involving HER2-targeted therapies. These therapies would be most relevant for cervical adenocarcinomas (i.e. GAS), which are most likely to have high expression of HER2.28 –30,32,35 The Friedman study included six patients enrolled in clinical trials based on ERBB2 alterations. 29 Four patients were enrolled in a trial involving the pan-HER TKI, neratinib, with two patients achieving SD, one for longer than 5 years. 29 Similarly, the phase II SUMMIT trial (NCT01953926) included individuals with HER2-mutant cervical cancers, achieving an ORR of 25% to neratinib monotherapy. 92 The new HER2 antibody-drug conjugate (ADC), trastuzumab deruxtecan (T-DXd), involving a potent topoisomerase I inhibitor payload, is currently being investigated in HER2-expressing solid tumours (NCT04482309; DESTINY-PanTumor02). For the cervical cancer cohort, an ORR of 50% was achieved for all tumours and 75% for HER2-high (3+) tumours. 93 These impressive results led to approval of T-DXd in 2024 for any HER2-positive solid tumour (pan-tumour approval), the first ADC to be approved for tumour-agnostic therapy. T-DXd is now recommended as a second-line treatment option for patients with HER2-high (3+ as well as 2+) cervical cancers. 94 A number of related studies are currently recruiting, including the HER2-targeting ADCs A166 (NCT03602079) and RC48 (NCT06155396), a bispecific molecule that targets both HER2 on tumour cells and CD137 on immune cells (PRS-343; NCT03330561) and a highly HER2-selective TKI, tucatinib, in combination with trastuzumab (NCT04579380). These are likely to demonstrate efficacy in HER2-high cervical cancers and are promising therapeutic innovations.
PI3K/AKT/mTOR pathway
PIK3CA and PTEN are frequently mutated in all types of cervical cancer,28 –30,32 and phosphorylation of AKT and mTOR (indicating PI3K/AKT/mTOR pathway activation) has been observed in up to 85% and 53% of cases, respectively.95,96 Activity of the mTOR inhibitor (mTORi) temsirolimus was investigated as monotherapy in patients with advanced cervical cancer in a phase II trial (NCT01026792). Almost two-thirds of patients experienced SD, with a modest median PFS of 3.52 months. 97 Temsirolimus has also been combined with bevacizumab in a phase I study, with two patients with SCC of the cervix experiencing PR (out of four patients treated). 98 Neither of the individuals with cervical adenocarcinoma or neuroendocrine tumour responded to this therapy. 98 The addition of liposomal doxorubicin to temsirolimus and bevacizumab in a phase I study (NCT00761644) also resulted in two patients with SCC achieving PR and one achieving SD (out of 10 patients treated). 99 None of the three individuals with adenocarcinoma responded in this trial. 99 It is not clear whether the responses observed were associated with PIK3CA mutations. 99 The mTORi everolimus has been investigated in combination with cisplatin and radiotherapy in the phase I PHEONIX 1 trial (NTC01217177). There was no mention of histological type, or whether patients had molecular aberrations in the PI3K pathway; however, an impressive 91.6% and 8.4% of patients achieved CR and PR, respectively, when assessed 12 weeks after treatment completion. 100 A phase I study of the pan-AKT inhibitor AZD5363 (NCT01226316) included five patients with cervical cancer, two of whom had PIK3CA-mutated tumours. 101 Both of these tumours reduced in size, with one being classified as a PR. 101 These results indicate that targeting the PI3K/AKT/mTOR pathway, particularly with mTORi, may be an effective treatment option for some individuals with cervical cancer harbouring relevant molecular aberrations. These trials have been too small to date to be certain about implications for specific histologic subtypes.
TGFβ pathway or EMT
As SRC, TGFβ and YAP-mediated signalling are often important in SCC of the cervix, targeting these aberrations is of interest. Two studies have investigated the activity of the bifunctional fusion protein that targets both TGFβ and PD-L1, bintrafusp alfa (M7824), in HPV-associated malignancies (NCT02517398 (phase I) and NCT03427411 (phase II)). The ORR for all patients in the study (including non-cervical HPV-associated cases) was 30.5%, with 24% achieving PR in the cervical cancer cohort. 102 Two additional studies have investigated the activity of bintrafusp alfa in advanced cervical cancer. A phase II study (NCT04246489) investigating bintrafusp alfa monotherapy resulted in an ORR of 21.9%. 103 Slightly better responses were observed in the PD-L1-positive cohort, and markedly better responses were observed for SCC compared with adenocarcinoma, with no responses observed in the five individuals with adenosquamous carcinoma. 103 At the same time, a phase I study (NCT04551950) resulted in a notable ORR of 75% when bintrafusp alfa was combined with chemoradiation and bevacizumab. 104 These impressive responses indicate this avenue warrants further investigation, especially for individuals with PD-L1-positive SCC.
Antibody-drug conjugates
Apart from the HER2-targeted ADCs mentioned previously, there has been an expansion of ADCs targeting other tumour biomarkers in recent years. So far, there have been 11 ADCs approved for the treatment of solid malignancies and almost 190 in the pipeline. 105 The main ADC shown to have activity in cervical cancer to date is tisotumab vedotin (TV). 106 TV involves a payload of monomethyl auristatin E linked to an antibody specific for tissue factor (TF), which is highly expressed in cervical cancer. 107 TV received accelerated approval in September 2021, following positive results from the phase II innovaTV 204 trial (NCT03438396). 107 A recently conducted phase III study, innovaTV 301 (NCT04697628), compared responses in patients who received TV with those who received IC chemotherapy. The TV group achieved a significantly longer OS of 11.5 months compared with 9.5 months for the IC chemotherapy group. 106 These results likely position TV as a new standard second- or third-line therapy for recurrent or metastatic cervical cancer, once reimbursement is more widely available. Multiple other ADCs are in phase I and II trials, including PHN010 (NCT06457997), LY4101174 and LY4052031 (NCT06238479 and NCT06465069, respectively), LCB84 (NCT05941507) and XB002 (NCT04925284). The results of these studies are eagerly awaited.
Targeted therapies for the treatment of vulvar cancer
The molecular data for vulvar cancers indicate that combination therapeutic strategies involving inhibitors of KIT and the PI3K pathway may be relevant for HPV-positive tumours, whereas inhibitors of the MAPK pathway may be relevant for HPV-negative tumours, which are more likely to have TP53 mutations and a worse prognosis/be harder to treat. 46 Inhibitors of EGFR, FGFR, immune checkpoints and the cell cycle may warrant investigation for the treatment of all vulvar SCC. Inhibitors of KIT and the MAPK pathway, such as imatinib and vemurafenib, may be relevant for vulvar melanomas. The rarer histological types of vulvar cancer will require a more personalised approach, given that molecular data is so far mostly limited to studies of single cases.
Matching targeted therapies based on molecular profiling of vulvar cancer has been largely lacking compared to common cancers and even cervical cancer. Therefore, the prognostic value of vulvar cancer genomics is yet to be determined. The NCI-MATCH phase II trial (NCT02465060) only screened three patients with vulvar cancer and enrolled just one in a specific trial arm. 56 Below, we will discuss a few targeted therapies with evidence of patient benefit in vulvar cancer (often just from case studies, as clinical trial data is mostly not available), with a summary provided in Table 9.
Vascular endothelial growth factor/R
With high VEGF expression and high microvessel density being significant prognostic factors in vulvar cancer, 108 and bevacizumab being approved for the treatment of persistent, recurrent or metastatic cervical cancer, 58 anti-angiogenic drugs should be investigated for the treatment of vulvar cancer. Indeed, based on extrapolation of the phase III trial data in cervical cancer (NCT00803062), 58 cisplatin/paclitaxel/bevacizumab combination therapy is now included as a first-line treatment option for recurrent and/or metastatic vulvar cancer. 108 In a small retrospective study, nine individuals with vulvar SCC had received bevacizumab maintenance therapy following first-line chemotherapy, with two patients (22.2%) achieving CR and one achieving PR. 109 Some long-term responses (at least 12 months in four patients) were observed, which is remarkable for a single agent following other lines of chemotherapy and/or radiation. 109 Despite this data coming from a small patient cohort, it further supports the inclusion of bevacizumab in treatment strategies for vulvar cancer going forward.
Immunotherapy
Given that PD-L1-positivity and high TMB are features of some vulvar cancers,46,108 ICI may demonstrate efficacy. Similar to anti-angiogenic therapy, the ICI responses observed in cervical cancer support the investigation of ICI in vulvar cancer. Three patients with vulvar cancer (at least two having PD-L1-positive tumours) were included in the CheckMate-358 trial (NCT02488759). In the cohort of five vulvar/vaginal cases, one patient achieved PR (unknown whether vulvar or vaginal cancer). 77 There were 18 patients with PD-L1-positive vulvar cancer included in the KEYNOTE-028 phase Ib trial (NCT02054806), with one patient achieving PR and seven patients achieving SD. 110 A larger cohort of 84 patients with PD-L1-positive vulvar cancer was included in the KEYNOTE-158 phase II trial, with one patient experiencing CR, seven patients experiencing PR and 14 patients experiencing SD. 111 ORR was actually greater in the PD-L1-negative population (28.6% compared to 9.5%), although this included just seven individuals and TP53 status was unknown. 111 There are other case reports of PD-L1-positive vulvar cancers responding to pembrolizumab,112,113 however, in most cases responses to monotherapy have been far from inspiring and not significantly associated with PD-L1 expression. Although these results are derived from generally small patient cohorts, it is likely that combination therapies will be required for improved efficacy in vulvar cancer. Currently, clinical trials are investigating the ICIs pembrolizumab, nivolumab, durvalumab, atezolizumab, ipilimumab or tremelimumab (CTLA4i), alone or in combination with radiotherapy (NCT04430699, NCT03452332 and NCT02834013). 114 Trials are also underway to study the ICIs nivolumab, cardunolizumab (bispecific antibody targeting PD-1 and CTLA4), atezolizumab, INCB099280 or avelumab (both PD-L1i), with or without bevacizumab, the multiple TKIs axitinib and cabozantinib, or the HDAC inhibitor valproic acid (NCT06292689, NCT03074513, NCT05949632, NCT05111574 and NCT03357757). However, most trials also include more common gynaecological and other cancer types, and consequently, individuals with vulvar cancer are likely once again to be under-represented.
EGFR family
With EGFR being amplified in up to 10% of vulvar cancers (CN increases observed in up to 40%), and high EGFR protein expression (3+ staining) being present in about 50% of cases,50,115 this appears to be a key target for therapeutic strategies. The only phase II trial testing EGFRi in individuals with vulvar SCC to date investigated responses to monotherapy erlotinib (NCT00476476). In cohort 1 (early stage disease), 5/17 cases had extra copies of EGFR (either by amplification or high polysomy); however, all 17 tumours displayed 2–3+ EGFR staining. 116 An ORR of 67.5% was achieved for both cohorts (an additional 24 patients with measurable metastatic disease were enrolled in cohort 2). 116 However, all responses were short-lived. 116 Since then, studies have been limited to case studies or very small cohorts as part of larger studies, with dramatic responses observed in some instances.108,117 In a small retrospective study, five patients with vulvar SCC had received erlotinib, with two patients achieving PR and two achieving SD. 109 However, responses were again short-lived (2–6 months), 109 indicating EGFRi combination therapies should be designed for future clinical trials.
Although ERBB2 amplifications are not common in vulvar cancer, they appear to be relatively common in the super rare vulvar Paget’s disease (VPD), which represents ~60%–80% of EMPD cases.118,119 In addition, at least 25% of VPD cases over-express HER2 (3+ staining), 119 indicating HER2-targeted therapies should be considered in treatment strategies of this rare subtype of vulvar cancer. There have been a few case reports of patients with HER2-positive VPD having marked responses to HER2-targeted therapies.119,120 Most patients had been treated with HER2-targeted agents in combination with taxanes in the first-line setting, with a median PFS of 12 months achieved. 119 Inclusion of HER2-positive VPD (including 1+) in basket trials of HER-2-targeted ADCs will be important.
TGFβ pathway or EMT
Although TGFβ-related targets have not been identified in vulvar cancer, one case was included in the phase II trial investigating bintrafusp alfa in HPV-associated malignancies (NCT03427411). This individual had an unconfirmed CR and unfortunately passed away from an unrelated medical event. 102 However, this notable response indicates further investigation is required in this area.
Antibody-drug conjugates
Apart from the HER2-targeted ADCs mentioned above, which are likely to be important treatment options for individuals with VPD, TROP2-targeted ADCs, such as sacituzumab govitecan (SG) are likely to be relevant for the treatment of vulvar SSC. This is due to the high prevalence of TROP2 expression that has been observed in these tumours (almost all tumours have some expression, with about 75% having high (2+ or 3+) expression). 121
Targeted therapies for the treatment of vaginal cancer
The limited molecular data for vaginal cancers indicate that inhibitors of the MAPK pathway may be relevant for the melanoma histological type. In addition, inhibitors of the PI3K/AKT/mTOR pathway may be relevant for vaginal SCC. Apart from specific targets, anti-angiogenics and ICIs are worth considering given their demonstrated efficacy in cervical cancer. Disappointingly, despite a search on https://wwww.clinicaltrials.gov retrieving 357 studies involving the search term ‘vaginal cancer’, only about five cases of vaginal cancer were actually found to be enrolled in these studies (with numbers unknown for trials not yet completed). Furthermore, the NCI-MATCH phase II trial (NCT02465060) only screened seven patients with vaginal cancer and enrolled just one patient on a treatment arm. 56 Below, we will discuss the limited targeted therapies, which have demonstrated some patient benefit in vaginal cancer, or which have trial results pending, with a summary provided in Table 9.
Immunotherapy
Two patients with vaginal cancer (at least one having a PD-L1-positive tumour) were included in the CheckMate-358 trial (NCT02488759), but as mentioned previously, it is unknown whether the single case of PR was vulvar or vaginal cancer. 76 There is one case report of PD-L1-positive vaginal cancers responding to pembrolizumab; however, another PD-L1-positive case did not respond. 112 Currently, individuals with vaginal cancer are included in clinical trials investigating ICIs, such as durvalumab, tremelimumab and cardunolizumab (NCT03452332 and NCT06292689). Patients with vaginal cancer may also be eligible for the clinical trials previously mentioned, investigating nivolumab, ipilimumab, atezolizumab, INCB099280 and avelumab alone or in combination with bevacizumab, axitinib, cabozantinib or valproic acid (NCT02834013, NCT030745513, NCT05949632, NCT05111574, NCT03357757). However, as with vulvar cancer, most trials also include more common gynaecological and other cancer types, meaning individuals with vaginal cancer are likely once again to be under-represented. Nevertheless, given the promising results for combinations of ICI, anti-angiogenics and chemoradiotherapy in cervical cancer, similar combination strategies warrant assessment for vaginal cancer.
Other targets and ADCs
Extrapolating from cervical cancer, the PI3K/AKT/mTOR pathway may be an important target in vaginal cancer. In phase I study (NCT01552434) investigating temsirolimus, bevacizumab and valproic acid, the single individual with vaginal SCC included in the study experienced a PR. 122 With respect to targeting TGFβ, one patient with vaginal cancer was included in the phase II trial investigating bintrafusp alfa in HPV-associated malignancies (NCT03427411), with this patient experiencing a CR. 102 This remarkable response mirrors that of the patient with vulvar cancer, indicating further investigation into targeting TGFβ in combination with PD-L1 is definitely warranted for vulvar and vaginal cancers.
As with cervical and vulvar cancers, ADCs are also likely to become important therapeutic options for patients with vaginal cancer in the future. Many targets, such as HER2, AXL, TROP2 and TF, are relevant to vaginal cancer, indicating patients should be enrolled in current trials involving T-DXd, TV, SG and other ADCs.
Summary
While clinical trial reporting of cervical cancer and targeted therapies has increased in recent years, results lag behind those for other types of gynaecological cancer and are vanishingly rare for vulvar and vaginal cancers. Combination regimens with CCRT, anti-angiogenic therapy and ICI are indicated for cervical cancer and should be considered, if accessible, for vulvar and vaginal cancers in the first-line. More complex combination strategies involving ICIs and genomically-directed targeted therapies may be required for vulvar cancer, which appears less responsive to ICI than cervical cancer.
In cervical cancer, initial trials involving TGFβ and PI3K pathway inhibitors have shown some benefit and warrant further investigation. HER2-targeted ADCs have shown efficacy in small trials of HER2-positive cases and the results of current trials involving newer HER2 ADCs are eagerly awaited. In particular, HER2-targeted therapies are likely to be a viable treatment option for many individuals with the GAS type of cervical adenocarcinoma. Further investigation of targeting ATR and PARP is also warranted, particularly in those with ARID1A aberrations. These treatments may constitute important therapeutic options for cervical cancer in the future, as may the ADC, TV and potentially other novel ADCs.
In vulvar cancer, combination therapies involving EGFRi require ongoing evaluation. HER2-targeting therapies are clearly important in VPD, and it will be particularly interesting to study responses to the HER2 ADC, T-DXd, in this context. In vaginal cancer, combination therapies involving PI3K inhibitors should be explored in SCC and targeting of the MAPK pathway should be investigated in melanomas. Further investigation of bintrafusp alfa in both vulvar and vaginal cancer will be of great interest. Due to their rarity, molecular studies of vaginal cancers and clinical trials including patients with vaginal cancer have been severely limited to date and, unsurprisingly, prognosis remains poor. More comprehensive molecular profiling is required to determine whether vulvar and vaginal cancers are indeed most similar to cervical cancers. If so, then individuals with vulvar and vaginal cancers should be prioritised in cervical cancer trials to generate urgently needed evidence to advance therapeutic options for these women. Genomically-directed decision-making has the potential to increase the available treatment options for women with cervical, vulvar and vaginal cancers, and should support the design of future clinical trials.
