Abstract
Background:
Gastric-type endocervical adenocarcinoma (GAS), the predominant non-human papillomavirus cervical cancer, is highly aggressive with poor prognosis. No drugs are effective against advanced or recurrent GAS.
Objective:
This study aimed to analyze the clinical and pathological characteristics of GAS, evaluate the expression of targets for targeted drug therapy, and assess the predictive value of Immunoscore for prognosis.
Design:
The study retrospectively analyzed the clinical and pathological characteristics and follow-up data of 107 cases of GAS and 523 of human papillomavirus-associated cervical adenocarcinoma (HPVA) treated surgically at Zhejiang Cancer Hospital between January 2015 and December 2022.
Methods:
Immunohistochemistry was used to detect the expressions of Tumor Protein 53 (P53), p16 protein (P16), Human Epidermal Growth Factor Receptor 2, Mucin 6, Programmed Death-Ligand 1 (PD-L1), Claudin 18.2, and immune markers Cluster of Differentiation 3/Cluster of Differentiation 8 in tumors. Ridge regression analysis was employed to obtain an Immunoscore, and receiver-operating characteristic curves were used to calculate the cutoff value to divide the Immunoscores into the low-risk and high-risk groups. The Kaplan–Meier method and Cox proportional hazards regression model were used in the survival and multivariate analyses, respectively.
Results:
Compared with HPVA, GAS was associated with later staging, larger tumor size, deeper invasion, and higher risks of lymph node metastasis, lymphovascular space invasion, parametrial involvement, ovarian metastasis, and peritoneal metastasis (p < 0.05). For patients in stages IA–IIIC2, the risk of recurrence/metastasis in GAS was significantly higher than that in HPVA (43.9% vs 21.2%, p < 0.001). The 5-year rates of progression-free survival (PFS; 44% vs 75.4%, p < 0.001) and overall survival (OS; 47.9% vs 82.5%, p < 0.001) were significantly lower in GAS than in HPVA. In the multivariate analysis, the histological type of GAS was an independent risk factor for OS (hazard ratio (HR) = 1.773, p = 0.023). In GAS, 71.7% and 46.7% were positive for Claudin 18.2 and PD-L1 (combined proportion score ⩾1), respectively. Four (3.7%) patients showed 3+ expression of Her-2. Cox multivariate analysis results indicated the Immunoscore as an independent predictive factor for PFS (HR = 2.532, p = 0.002) and OS (HR = 3.147, p = 0.003) in patients with GAS. The 5-year OS rates for the low- and high-risk groups based on the Immunoscore were 62.1% and 0% (p < 0.001), and the 5-year PFS rates were 57.3% and 9.0% (p < 0.001), respectively.
Conclusion:
GAS is extremely aggressive and prone to recurrence and has a poor prognosis. The Immunoscore is an independent prognostic factor of GAS. Claudin 18.2 is expressed at high rates in GAS and is a potential therapeutic target.
Keywords
Introduction
Cervical cancer is the most common malignant tumor in the female reproductive system. According to the GLOBOCAN 2022, approximately 661,000 new cases of cervical cancer and 348,000 deaths occurred in the world. 1 With the introduction of human papillomavirus (HPV) vaccination and widespread cervical screening, the incidence rate of cervical cancer is generally declining, but the proportion of cervical adenocarcinoma in newly diagnosed cases is increasing. Gastric-type endocervical adenocarcinoma (GAS) is the most common type of non-HPV-related cervical cancers and the second most common type of cervical adenocarcinoma. GAS accounts for 25% of all endocervical adenocarcinomas in Asian populations 2 while constituting approximately 10% in Western countries. 3 Previous studies have reported that GAS is extremely aggressive and has a poor prognosis.3,4 Currently, its treatment follows the NCCN guidelines for cervical cancer management, with surgery as the primary treatment for early-stage patients, concurrent chemoradiotherapy for locally advanced patients, and systemic therapy primarily for recurrent or metastatic patients. However, GAS is not sensitive to chemotherapy or radiotherapy.5,6 Early-stage GAS often recurrence after surgery, and no drugs are effective against advanced or recurrent GAS. In recent years, immunotherapy and targeted therapy have shown promising results for recurrent or metastatic cervical cancer. Notably, the current investigation was constrained by a limited GAS cohort size; the therapeutic efficacy and expression of markers observed in these cases remain unsubstantiated by robust clinical evidence.
The interaction between tumor cells and the tumor microenvironment is crucial in the progression of malignant tumors. The tumor microenvironment consists of tumor cells, immune cells, vascular endothelial cells, fibroblasts, cytokines, chemokines, cellular metabolic products, etc. Tumor-infiltrating lymphocytes (TILs), which are primarily located within the tumor stroma, are key participants in the tumor immune response and are associated with a favorable prognosis.7–9 The Immunoscore is a risk assessment tool that predicts prognosis by quantifying CD3+ lymphocytes and CD8+ cytotoxic T cells in the tumor center (TC) and invasive margins (IM). 10 The prognostic value of this score has been evaluated in patients with colorectal cancer and gastric cancer11,12; however, its status in GAS remains unclear.
This large-scale retrospective study on GAS in comparison with HPV-associated cervical adenocarcinoma (HPVA) aimed to clarify the clinical and pathological characteristics of GAS. The expression of biomarkers for which targeted therapies are currently available was examined, and the role of Immunoscore in predicting the prognosis of GAS was evaluated to provide a basis for selecting targeted therapies for recurrent or metastatic GAS.
Methods
Patients
This study collected data from 523 HPVA and 107 GAS patients who received surgical treatment at Zhejiang Cancer Hospital between January 2015 and December 2022. Patients with (1) HPVA and GAS confirmed by two gynecologic pathologists (combined with morphological characteristics and RNA scope test results) and (2) complete clinical and pathological data, as well as follow-up information, were enrolled. Patients with other pathological types and those with concurrent primary malignant tumors were excluded. This retrospective study was approved by the Ethics Committee of the Zhejiang Cancer Hospital (approval number IRB-2024-76). Informed consent was waived for this retrospective study. This protocol completely adheres to the Standard Protocol Items: Recommendations for Observational studies statement. 13
Treatment and follow-up
According to the International Federation of Gynecology and Obstetrics (FIGO) 2018 criteria, staging is performed based on postoperative pathological results. All patients underwent radical surgery involving radical hysterectomy and bilateral pelvic lymph node dissection. If lymph node metastasis is detected during surgery, para-aortic abdominal lymph node dissection should be performed. If pelvic or abdominal metastases are identified during surgery, cytoreductive surgery should be conducted. Adjuvant therapy based on pathological results was provided postoperatively. Patients with any high-risk factors, including lymph node metastasis, parametrial involvement, and positive margins, received adjuvant chemoradiotherapy. For patients with intermediate-risk factors, according to the “four-factor model,” tumor diameter ⩾3 cm, lymphovascular space invasion (LVSI), external 1/3 interstitial infiltration, adenocarcinoma, or adenosquamous cell carcinoma, if any two factors are present, adjuvant radiotherapy ± chemotherapy is required.
Adjuvant chemotherapy was offered to patients who declined adjuvant radiotherapy and those whose postoperative staging was upgraded to stage IV. The treatment regimen consists of paclitaxel combined with cisplatin chemotherapy administered every 3 weeks.
Patients were followed up every 3 months for the first 2 years, every 6 months up to 5 years, and annually thereafter. Follow-up examinations included gynecological examinations, transvaginal ultrasonography, vaginal vault cytology, and measurement of serum tumor markers. When recurrent disease was suspected, diagnostic tests such as MRI, CT scans, or PET-CT, and/or biopsies were performed.
Immunohistochemistry and interpretation
Specimens were fixed in 10% neutral formalin and embedded in paraffin. For each case, 1–3 representative paraffin blocks were selected to prepare 10 slides, with a section thickness of 3–5 μm. Immunohistochemical testing was performed using the EnVision two-step method. The antibodies used included Cluster of Differentiation 3 (CD3), Cluster of Differentiation 8 (CD8), Tumor Protein 53 (P53), p16 protein (P16), Human Epidermal Growth Factor Receptor 2 (Her-2), Mucin 6 (MUC6), Programmed Death-Ligand 1 (PD-L1, clone no. 22C3), and Claudin 18.2 (clone no. LBP1-cldn18.2), all purchased from Hangzhou Xibo Pharmaceutical Technology Co., Ltd, Hangzhou, Zhejiang, China. All tests were conducted according to platform standard procedures, and appropriate positive and negative controls were run concurrently for all the tested antibodies.
MUC6 was positive if brown-yellow staining was present in the cytoplasm of tumor cells, P16 was positive when brown-yellow staining was present in both the nuclei and cytoplasm of tumor cells, and P53 results were categorized as mutant or wild type, with the mutant type defined as strong positivity in >80% of the cell nuclei or complete negativity in the tumor, and all other cases were classified as wild type. 14 Her-2 results were interpreted according to the 2016 Gastric Cancer HER2 Testing Guidelines. 15 Assessment of PD-L1 expression used the combined positive score (CPS). CPS was calculated as the number of PD-L1-staining cells (tumor cells, lymphocytes, and macrophages) divided by the total number of viable tumor cells, multiplied by 100. A cutoff score ⩾1 for CPS was used to define PD-L1 positivity. Claudin 18.2 was primarily expressed on the cell membrane, and the staining scoring method was as follows: proportion score of positive cells (0 for ⩽5%, 1+ for 6%–25%, 2+ for 26%–49%, 3+ for 50%–74%, and 4+ for ⩾75%) and intensity score of staining (0 for no color, 1+ for weak staining, 2+ for moderate staining, and 3+ for intensity staining). The product of the two scores was used as the final score; if the final score was ⩾2, Claudin 18.2 was considered expressed. 16
CD3 and CD8 positivities were indicated by brown-yellow staining of the tumor cell nuclei. Scoring was performed by a senior pathologist under the supervision of a senior pathology consultant. If there was no consensus at initial review, the case would be discussed at a multiheaded microscope with another senior pathologist until consensus was reached. Twenty consecutively selected tumor samples were re-examined by a second senior pathologist. The evaluation areas were as follows: IM and TC. IM referred to the area centered on the boundary separating the host tissue from malignant nests, with a range of 1 mm. The TC in all tissues was located within the IM (Supplemental Figure 1). The density of CD3 and CD8 expression was assessed using a semi-quantitative scoring system: (1) 1+, <5%; (2) 2+, 5%–30%; (3) 3+, 30%–80%; and (4) 4+, >80%. The calculation method was as follows: the ratio of the total area occupied by mononuclear lymphocytes to the total stromal/tumor area. 17
Construction of a prognostic score based on immune characteristics
The Immunoscore was derived using ridge regression analysis based on the semi-quantitative scoring of CD3 and CD8 expression levels in patients. Receiver-operating characteristic curves were used to calculate the maximum Youden index to determine the optimal cutoff value, which was −0.41. Patients were divided into low-risk (⩽−0.41) and high-risk (>−0.41) groups, comprising 65 (65%) and 35 (35%) cases, respectively.
RNA scope detection
The RNA scope method was used to detect E6/E7 mRNA of 18 types of high-risk HPV (including HPV16, 18, 26, 31, 33, 35, 39, 45, 51, 52, 53, 56, 58, 59, 66, 68, 73, and 82) in 107 cases. The RNAscope® 2.5 HD Detection Kit was purchased from Beijing Zhongshan Jinqiao Biotechnology Co., Ltd, Beijing, China. The experimental procedures followed the kit instructions, including material preparation, hybridization probes, hybridization Amp1-6, chromogen, and 4′,6-diamidino-2-phenylindole counterstaining of slides. The positive control probe was Hs-PPIB (Catalog No. 313901), and the negative control probe was DapB (Catalog No. 310043). Brown signals within tumor cells were considered positive under optical microscopy.
Statistical analysis
Analyses were performed using IBM SPSS Statistics version 25.0 (IBM Corp., Armonk, NY, USA) and R4.4.0. Normality tests were conducted for continuous data, and normally distributed data are expressed as mean ± standard deviation (x̄ ± s) and analyzed using t-tests. Non-normally distributed data are presented as median (25th–75th percentiles) [M (P25–P75)] and analyzed using nonparametric tests (Mann–Whitney U test). Categorical data are expressed as numbers and percentages and analyzed using Chi-square (χ²) tests or Fisher’s exact probability method. Survival rates were calculated, and survival curves were plotted using the Kaplan–Meier method. Univariate analysis of clinical and pathological factors that affect patient prognosis and multivariate analysis were conducted using the log-rank test and Cox proportional hazards regression model, respectively. A p-value <0.05 was considered significant. The primary outcome measures were progression-free survival (PFS) and overall survival (OS). PFS was defined as the time from initial treatment to recurrence or last follow-up. OS was defined as the time from initial treatment to death or last follow-up. The negative reference range is defined as: cancer antigen (CA) 19-9 ⩽39 U/mL; CA-125 ⩽35 U/mL; carcinoembryonic antigen (CEA) ⩽5 ng/mL. Values above these ranges are considered positive. The last follow-up date for survival analyses was December 2023. In accordance with the journal’s guidelines, we will provide our data for independent analysis by a selected team by the Editorial Team for additional data analysis or the reproducibility of this study in other centers if such is requested.
Results
Comparative study of the clinical and pathological characteristics, recurrence, and prognosis of GAS and HPVA
A total of 107 GAS cases and 523 HPVA cases were included in the study. All GAS cases exhibited at least some regions with typical morphological features (Supplemental Figure 2(A)): (1) tumor cells with abundant clear or eosinophilic cytoplasm, (2) distinct cell membranes, and (3) absence of numerous mitotic figures along the luminal borders and floating apoptotic bodies. All GAS cases tested negative by RNAscope. All HPVA cases showed typical morphological features (Supplemental Figure 2(B)): (1) enlarged, elongated nuclei with coarse chromatin and visible small nucleoli; (2) high nuclear-to-cytoplasmic ratio, cellular crowding, and multilayered arrangement; and (3) active mitosis with numerous floating apoptotic bodies.
The clinical and pathological characteristics of patients with GAS and HPVA are shown in Table 1. Patients with HPVA were younger at diagnosis than those with GAS, with median ages of 47 and 51 years, respectively (p < 0.001). Compared with the HPVA group, the GAS group had a later stage (stages II–IV: 33.8% vs 73.8%, p < 0.001), larger tumors (>4 cm: 32% vs 58.9%, p < 0.001), deeper invasion (>2/3 layers: 48.4% vs 92.5%, p < 0.001), incidence of lymph node metastasis (27% vs 55.1%, p < 0.001), higher incidence of LVSI (41.3% vs 70.1%, p < 0.001), and incidence of parametrial involvement (1.7% vs 5.6%, p = 0.016). Ovarian and peritoneal metastases are also more common in the GAS group. Among 107 patients with GAS, 21 (19.6%) had ovarian metastasis, compared with only 14 (2.7%) out of 523 patients with HPVA (p < 0.001). Regarding tumor markers, the abnormal increase rate of CA 199 was higher in the GAS group (51.4%) than in the HPVA group (21.6%; p < 0.001).
Comparison of clinical and pathological features between gastric-type endocervical adenocarcinoma and HPV-associated adenocarcinoma.
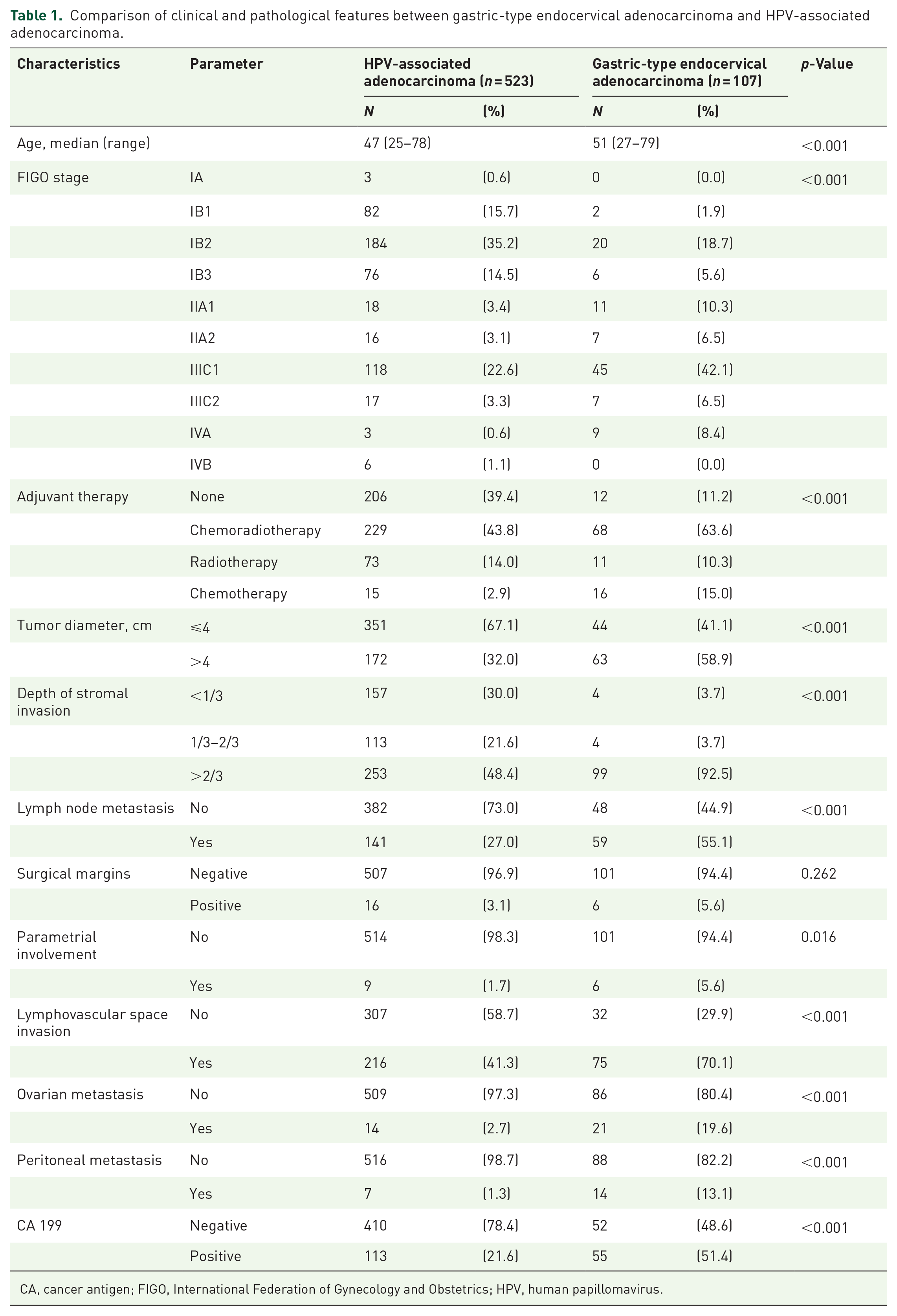
CA, cancer antigen; FIGO, International Federation of Gynecology and Obstetrics; HPV, human papillomavirus.
The PFS and OS curves for the GAS and HPVA groups are shown in Figure 1(a) and (b). The 5-year PFS and OS rates of the GAS group were 44% and 47.9%, both significantly lower than those of the HPVA group, with 75.4% and 82.5%, respectively (both p values <0.001). Among patients in stages IA–IIIC2 (patients whose postoperative staging was upgraded to stage IV were excluded from the recurrence pattern analysis), 152 experienced recurrence/metastasis, including 43 out of 98 (43.9%) patients with GAS and 109 out of 514 (21.2%) with HPVA, indicating a significantly higher recurrence risk in GAS (p < 0.001). Failure patterns between the two groups are shown in Table 2. The local recurrence and distant metastasis of GAS are significantly higher than those of HPVA. Univariate analysis of all patients revealed that histological type, FIGO stage, ovarian metastasis, peritoneal metastasis, CA199, CA125, CEA, tumor size, invasion depth, LVSI, lymph node metastasis, and parametrial involvement were all associated with OS (all p < 0.05, Table 3). Multivariate analysis indicated that GAS (p = 0.023), lymph node metastasis (p = 0.035), and parametrial involvement (p = 0.015) were independent risk factors for OS (Supplemental Table 1).

(a) PFS curves for GAS and HPVA. (b) OS curves for GAS and HPVA. (c) PFS curves for the Immunoscore. (d) OS curves for the Immunoscore. (e) PFS curves for FIGO stage. (f) OS curves for lymph node metastasis.
Comparison of failure patterns between patients with stage IA–IIIC2 gastric-type endocervical adenocarcinoma and HPV-associated adenocarcinoma.

HPV, human papillomavirus.
Univariate analysis of gastric-type endocervical adenocarcinoma.

CA, cancer antigen; FIGO, International Federation of Gynecology and Obstetrics; Her-2, human epidermal growth factor receptor 2; HPV, human papillomavirus; MUC6, Mucin 6; OS, overall survival; p53, tumor protein 53; PFS, progression-free survival.
Immunohistochemical characteristics of GAS
The expression levels of mucin epithelium-related markers, including Claudin 18.2 and MUC6, were evaluated in 107 GAS cases. Among the 106 patients examined, a remarkable 76 (71.7%) tested positive for Claudin 18.2. A significant proportion of patients exhibited robust labeling of Claudin 18.2. Specifically, 57 patients (53.8%) displayed a staining intensity of ⩾2+ in at least 50% of cells, while 47 patients (44.3%) achieved a staining intensity of ⩾2+ in ⩾75% of cells. (Supplemental Figure 2(C)). Moreover, 90 (84.1%) patients expressed MUC6 to varying degrees, with 32 (29.9%) showing expression levels of <10%, 35 (32.7%) expressing between 10% and 50%, and 23 (21.6%) exhibiting expression levels >50%. This further confirms that GAS possesses characteristics similar to those of gastric-type mucinous epithelium.
Among the 107 patients with GAS, only 1 (0.9%) showed a speckled expression pattern in P16 immunohistochemistry, and the remaining 106 cases were negative. HPV RNA scope testing was performed on all cases, and the results were negative (100%), indicating the absence of replication-competent HPV in the tumor cells. In addition, 61 cases (57%) exhibited a mutant phenotype P53 immunohistochemistry in 107 GAS (Supplemental Figure 2(D)).
The expression levels of two treatment-related markers, Her-2 and PD-L1, in GAS were detected simultaneously. In addition, 41.1% of the patients expressed Her-2, with 4 (3.7%) showing 3+ expression, 21 (19.6%) exhibiting 2+ expression, and 19 (17.8%) showing 1+ expression. Using a cutoff value of CPS ⩾1, 50 (46.7%) patients exhibited positive expression of PD-L1.
Immunoscore for GAS
The expression ratios of CD8 and CD3 in TILs at both the TC and IM were evaluated in 100 GAS cases. An Immunoscore was calculated based on CD3 and CD8 results, with 65 (65%) and 35 (35%) in the low-risk and high-risk groups, respectively. The recurrence rates for the low- and high-risk groups were 38.5% (25/65) and 78.4% (24/35), respectively (p = 0.004), and the 5-year PFS rates were 57.3% and 9.0%, respectively (p < 0.001; Figure 1(c)). The mortality rates for the low- and high-risk groups were 32.3% (21/65) and 54.3% (19/35), and the 5-year OS rates were 62.1% and 0%, respectively (p < 0.001; Figure 1(d)).
Analysis of prognostic factors for GAS
The median follow-up time for the GAS group was 46 (range, 9–107) months. Univariate analysis of 100 patients with GAS showed that FIGO stage (p = 0.001), LVSI (p = 0.015), lymph node metastasis (p = 0.001), and Immunoscore (p < 0.001) were associated with PFS, whereas FIGO stage (p = 0.004), ovarian metastasis (p = 0.009), peritoneal metastasis (p = 0.033), CA125 (p = 0.002), lymph node metastasis (p < 0.001), and Immunoscore (p < 0.001) were associated with OS (Table 3). Expression levels of Claudin 18.2, PD-L1, Her-2, P53, and MUC 6 were not related to PFS or OS (p > 0.05). Multivariate Cox proportional hazards model results indicated that the Immunoscore is an independent predictive factor for both PFS (hazard ratio (HR) = 2.532, p = 0.002) and OS (HR = 3.147, p = 0.003). In addition, the FIGO stage (HR = 1.482, p = 0.046) is an independent prognostic factor for PFS, and lymph node metastasis (HR = 1.89, p = 0.01) is an independent prognostic factor for OS (Table 4). The 5-year PFS rates for patients in FIGO stages I, II, III, and IV were 69.9%, 47.9%, 32.8%, and 11.1%, respectively (p = 0.001; Figure 1(e)). The 5-year OS rates for patients with and without lymph node metastases were 31.8% and 69%, respectively (p < 0.001; Figure 1(f)).
Multivariate analysis of gastric-type endocervical adenocarcinoma.
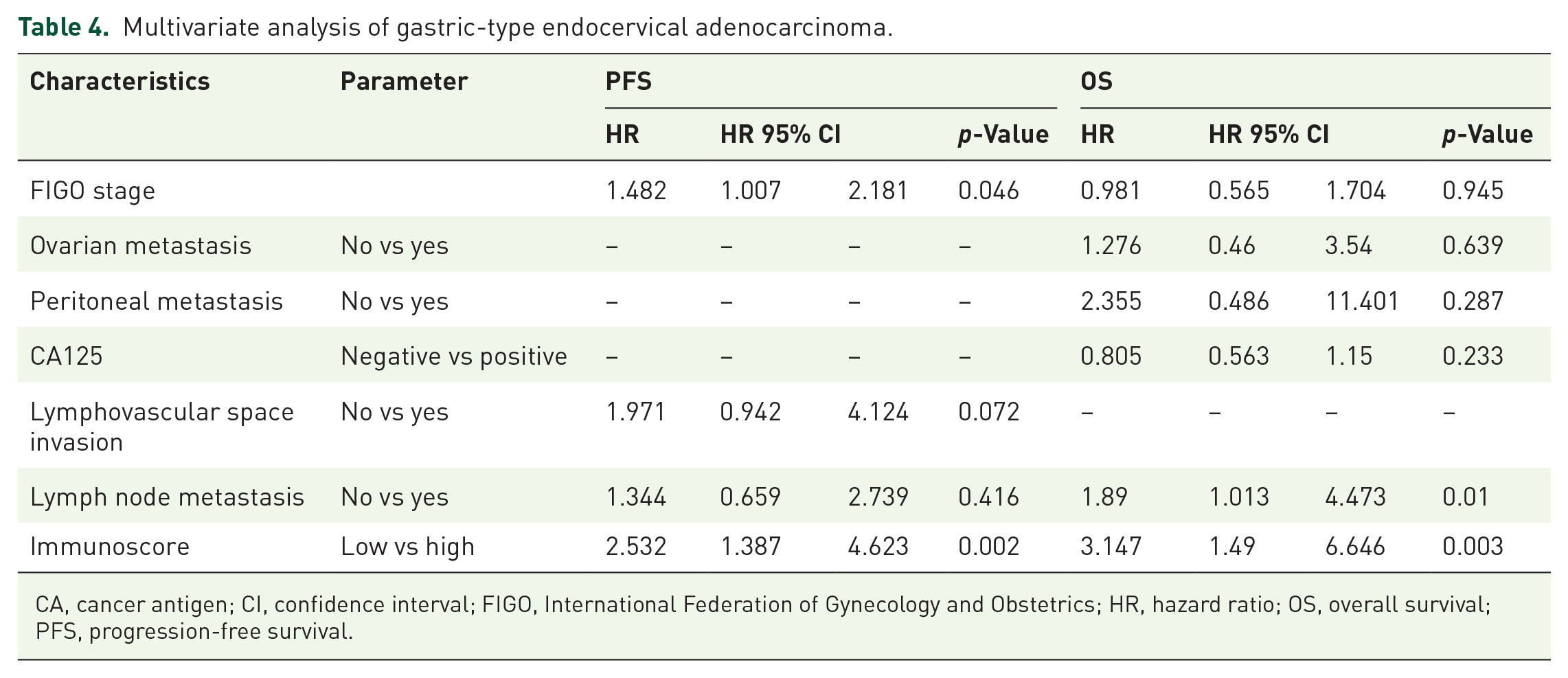
CA, cancer antigen; CI, confidence interval; FIGO, International Federation of Gynecology and Obstetrics; HR, hazard ratio; OS, overall survival; PFS, progression-free survival.
Discussion
This study compared the clinical and pathological characteristics and prognosis of 107 cases of GAS and 523 cases of HPVA. In this study, GAS had a later FIGO stage, with approximately 73.8% being stages II–IV, whereas 66.2% of patients with HPVA were diagnosed at stage I. Similarly, a study reported that approximately 59% of GAS cases were discovered at stages II–IV, whereas 89% of patients with HPVA were at stage I, with only 11% in advanced stages. 3 This may be related to the fact that GAS tumors are usually located in the upper part of the cervix, making it difficult to obtain tumor tissue during cervical screening and biopsy. Improving the early detection rate of GAS may be one method to improve prognosis. Compared with HPVA, GAS is more aggressive and is associated with larger tumors, deeper invasion, higher risks of lymph node metastasis, LVSI, and parametrial involvement. A previous retrospective study including 95 cases of GAS reported similar results. 18
In addition, patients with GAS have a high risk of pelvic and abdominal metastasis, with ovarian metastasis rates as high as 19.6% and peritoneal metastasis rates at 13.1%. Furthermore, patients with ovarian or peritoneal metastasis have poorer OS than those without metastasis. The research results from the MSK Center in the United States show that the incidence of ovarian metastasis in patients with GAS is 19% (13/70). 18 Nishio et al. 18 reported that the rate of ovarian metastasis in GAS is significantly higher than in usual-type adenocarcinomas (5.3% vs 1.3%). Karamurzin et al. 3 reported that ovarian metastasis in GAS can reach up to 35%, significantly higher than in HPVA. In the study by Kojima et al., 4 the risk rates of ovarian metastasis for mucinous adenocarcinoma in GAS versus non-GAS were 6% (1/16) and 3% (1/37), respectively. Therefore, combining our data, for the surgical treatment of GAS, ovarian preservation is not recommended, and omentectomy should be considered.
The OS of the GAS group was poorer than that of the HPVA group which underwent surgical treatment during the same period, with 5-year OS rates of 47.9% and 82.5%, respectively. A previous study reported 5-year OS rates of 25%–42% and 90% for GAS and HPVA, respectively.18,19 The poor prognosis of GAS may be related to the aggressive characteristics of the tumor and delayed diagnosis. GAS is a different type, and current treatment standards should be unique rather than referencing HPVA.
The biological behavior and prognosis of GAS differ significantly from those of HPVA, and it tends to recur and metastasize easily. The clinical challenge in treating recurrent or metastatic GAS is the lack of effective drugs. Several clinical trials have indicated that PD-L1-positive cervical cancer benefits from immunotherapy. PD-L1 expression is an important immune-related biomarker for the benefit of patients with cervical cancer. Meta-analysis shows that the positive rate of PD-L1 expression (calculated with CPS ⩾1) in patients with cervical cancer ranges from 8.5% to 93.3%, with a pooled positive rate of 58.1%. Among these, squamous cell carcinoma is expressed at 67.8% and adenocarcinoma at 36.6%.20,21 In the present study, PD-L1 detection in GAS using CPS scoring yielded a positivity rate of 46.7%, whereas in the study by Chen et al., 22 this rate was as high as 87.5%. Thus, whether patients with PD-L1-positive GAS can benefit from immunotherapy awaits further drug clinical studies for confirmation.
Traditional targeted drugs for Her-2, such as trastuzumab and pertuzumab, have been used in recent years. More recently, novel antibody–drug conjugates (ADCs) targeting Her-2, such as Trastuzumab deruxtecan, have demonstrated significant and durable disease relief in various refractory solid tumors expressing Her-2, with particularly notable efficacy in cervical. The DESTINY-PnTomor02 trial included 40 patients with advanced recurrent cervical cancer (IHC 3+/2+) with HER2 expression, with an Objective Response Rate (ORR) of 50%, median Duration of Response (DOR) of 14/2 months, mPFS of 7.0 months, and mOS of 13 months. 23 Currently, targeted therapies primarily focus on Her-3+ or Fluorescence In Situ Hybridization (FISH)-positive cases, whereas ADC drugs targeting Her-2 can be used in tumors with only 1+ expression. In this study, only 4 (3.7%) patients exhibited strong positive (3+) expression, whereas 44 (41.1%) patients had 1+ or higher expression, a ratio similar to that reported in a previous study (3+, 3.8% 24 ; 3+, 3.7%; 1+ and above, 55.6% 16 ). However, one small-sample study reported significantly different results, possibly due to bias related to the small sample size. 25 The efficacy of targeted drugs against the Her-2 target and current ADCs in GAS requires further clinical validation.
Claudin 18.2 is a member of the claudin protein family and is specifically expressed in cancer cells of the stomach, pancreas, and esophagus. 26 Claudin 18.2 positivity occurred primarily in gastric adenocarcinoma and other tumors with gastric epithelium-like differentiation. Evaluating claudin 18.2 expression in these tumors can guide targeted therapy, also in Gastric-type cervical adenocarcinoma. The monoclonal antibody claudiximab, targeted against Claudin 18.2, improves the prognosis of patients with gastric adenocarcinoma in multiple clinical trials.27–29 The SPOTLIGHT and GLOW studies included a total of 1072 locally advanced unresectable or metastatic gastric cancer patients who were CLDN18.2 positive and HER2 negative. They were randomly assigned to receive cetuximab combined with chemotherapy or placebo combined with chemotherapy. The results showed that compared with placebo + chemotherapy, cetuximab + chemotherapy significantly prolonged PFS and OS. The median PFS of the treatment group and placebo group were 9.2 and 8.2 months, respectively, and the median OS was 16.4 and 13.7 months, respectively. 29 The present study indicates that Claudin 18.2 has a positivity rate of up to 72.6% in GAS. Other studies have reported positivity rates of 95.5% 30 and 95.2%. 16 The strong expression of Claudin 18.2 in patients with GAS proposes the potential for anti-Claudin 18.2 therapy. Clinical trials for Claudin 18.2–ADC drugs are ongoing, and this drug has the potential to become the most promising and effective treatment for GAS.
TILs play a crucial role in tumor immunity. A study showed that TIL infiltration was associated with improved clinical outcomes in cancer patients. 8 The Immunoscore, an important prognostic indicator, has been previously validated in colorectal cancer.10,12 In this study, the Immunoscore combines the location and quantity of CD3+ and CD8+ cells. The results indicate that the Immunoscore is an independent predictive factor for PFS and OS, with the low-risk group showing significantly better PFS and OS. This risk assessment tool reliably identifies subgroups of patients with an increased risk of recurrence or death, for whom more aggressive treatment and intensified follow-up may be recommended.
Lymph node metastasis is a recognized risk factor for poor prognosis in patients with cervical cancer and is involved in the determination of FIGO stages. In this study, lymph node metastasis is also an independent risk factor for GAS, with a significantly higher risk of lymph node metastasis than HPVA, which is consistent with the results of Nishio et al. 18 This indicates that patients with GAS having lymph node metastasis have a very poor prognosis, with a 5-year OS rate of only 31.8%, further emphasizing the importance of early diagnosis.
This study is limited by the single-center setting, which may introduce some bias, and the use of the Immunoscore to predict the prognosis of GAS, which requires further external data validation.
Conclusion
Compared with HPVA, GAS exhibits more aggressive biological behavior, with a higher likelihood of recurrence and a poorer prognosis. Claudin 18.2 is highly expressed in GAS and is considered the most promising therapeutic target. The Immunoscore is a newly identified independent prognostic factor for GAS.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251361880 – Supplemental material for Clinical and pathological analyses of gastric-type cervical adenocarcinoma and its prognostic relevance
Supplemental material, sj-docx-1-tam-10.1177_17588359251361880 for Clinical and pathological analyses of gastric-type cervical adenocarcinoma and its prognostic relevance by Yun Xi, Fang Zhou, Yulan Liu, Haifei Zhou, Xiaoyan Lu, Xianhua Fang, Lifeng Yan, Jianqing Zhou, Tao Zhu and Huarong Tang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-png-2-tam-10.1177_17588359251361880 – Supplemental material for Clinical and pathological analyses of gastric-type cervical adenocarcinoma and its prognostic relevance
Supplemental material, sj-png-2-tam-10.1177_17588359251361880 for Clinical and pathological analyses of gastric-type cervical adenocarcinoma and its prognostic relevance by Yun Xi, Fang Zhou, Yulan Liu, Haifei Zhou, Xiaoyan Lu, Xianhua Fang, Lifeng Yan, Jianqing Zhou, Tao Zhu and Huarong Tang in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-png-3-tam-10.1177_17588359251361880 – Supplemental material for Clinical and pathological analyses of gastric-type cervical adenocarcinoma and its prognostic relevance
Supplemental material, sj-png-3-tam-10.1177_17588359251361880 for Clinical and pathological analyses of gastric-type cervical adenocarcinoma and its prognostic relevance by Yun Xi, Fang Zhou, Yulan Liu, Haifei Zhou, Xiaoyan Lu, Xianhua Fang, Lifeng Yan, Jianqing Zhou, Tao Zhu and Huarong Tang in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
