Abstract
Background:
Patients with inoperable or metastatic oropharyngeal squamous cell carcinoma (OSCC) face limited therapeutic options. Nectin-4, an immunoglobulin-like transmembrane adhesion protein, and Trophoblast Surface Antigen 2 (TROP2), a transmembrane glycoprotein, have recently emerged as targets for antibody–drug conjugates (ADCs). Enfortumab Vedotin, an ADC targeting Nectin-4, has been approved for locally advanced or metastatic urothelial carcinoma, while sacituzumab govitecan, targeting TROP2, was approved for metastatic triple-negative breast cancer.
Objectives:
This study aimed to demonstrate expression rates of TROP2 and Nectin-4 in a representative cohort of human papillomavirus (HPV)-positive and HPV-negative OSCC and discuss the relevance of those markers as possible targets for ADCs.
Design:
A retrospective cohort study.
Methods:
We analyzed tissue samples from 226 OSCC patients treated at the University Hospital of Cologne between 2005 and 2020. The expression of Nectin-4 and TROP2 was assessed using immunohistochemistry, and the H-score method was applied to categorize expression levels into four groups: negative (0–10), low (11–100), moderate (101–200), and high (201–300).
Results:
TROP2 expression was positive in 96.5% of the samples, with 84.1% showing moderate to high expression (H-Score 101–300). A total of 38.8% of the cases expressed Nectin-4. Notably, patients with HPV-positive OSCC demonstrated significantly higher Nectin-4 expression compared to those with HPV-negative OSCC (p < 0.001).
Conclusion:
This study is one of the first to investigate the expression of Nectin-4 and TROP2 in a cohort of both HPV-positive and HPV-negative OSCC patients. Our results indicate that TROP2 is almost universally expressed in OSCC, while Nectin-4 expression is less frequent. TROP2 and Nectin-4 are promising therapeutic targets for OSCC, with Nectin-4 being particularly relevant for HPV-positive patients. Clinical trials are necessary to confirm the clinical relevance and efficacy of ADCs targeting TROP2 and Nectin-4 in OSCC treatment.
Introduction
Head and neck squamous cell carcinoma (HNSCC) is the seventh most frequent type of malignancy worldwide. 1 In addition to tobacco and alcohol consumption, persistent infection with high-risk human papillomavirus (HPV) has emerged as a major risk factor for oropharyngeal squamous cell carcinoma (OSCC). OSCC, a subgroup of HNSCC manifesting in the oropharynx, has increased in the last two decades.2 –5 Since the introduction of the eighth edition of the American Joint Committee on Cancer (AJCC), HPV-driven OSCC has been recognized as a distinct entity with distinct biological characteristics and pathogenesis, and a significantly improved prognosis and therapeutic response when compared to HPV-negative OSCC. Therefore, both ongoing and completed trials (ECOG3311 and PATHOS (NCT02215265)) analyze the possibility of therapy de-escalation5 –7 in HPV-positive OSCC. The decreasing incidence of HPV-negative HNSCC attributed to reduced consumption of alcohol and tobacco in the general population contrasts with rising rates of HPV-driven OSCC.2,3
Five-year overall survival (OS) rates of HPV-positive and HPV-negative OSCC differ significantly. According to a study by Mehanna et al., the 5-year OS for patients with p16+/HPV+ OSCC is 81.1%, the 5-year OS for p16−/HPV− patients being merely 40.4%. Capote-Moren et al. demonstrated in an epidemiological study with a follow-up of 39 years that the relapse rate (counting all OSCC patients) only slightly improved from 45.6% in the 1980s to 36.1% between 2010 and 2017. 8 Therapeutic options for patients with unresectable, recurrent, or metastatic OSCC are limited. To date, only three targeted therapies have been successfully established in OSCC: Cetuximab (an anti-epithelial growth factor receptor (EGFR) antibody) or immune checkpoint inhibitors (ICB), pembrolizumab or nivolumab against programmed death 1 (PD1) receptor. However, not all HNSCC patients show a clinical response, and patients’ survival remains limited. To develop and optimize new therapeutic options, the analysis and identification of further biomarkers as possible targets for targeted therapies is indispensable.
The transmembrane glycoprotein human trophoblast cell-surface antigen 2 (TROP2) is involved in different oncogenic cell signaling pathways.9 –12 An overexpression of TROP2 has been found in various malignant solid tumors such as ovarian, 13 breast, pancreatic,14,15 gastric, 16 and salivary gland cancer. 17 It also shows overexpression in OSCC, 18 while in normal tissues, TROP2 expression is low or absent.19,20 Fong and Zhang et al. demonstrated that a high expression of TROP2 is correlated with poor survival in OSCC, which makes TROP2 a possible prognostic and predictive biomarker.21,22 Furthermore, TROP2 has proven a successful therapy target for the antibody–drug conjugate (ADC) sacituzumab govitecan (SG) in solid tumors. ADCs are considered promising new drugs for cancer treatment, particularly in the context of recurrent/metastatic HNSCC, due to their ability to combine the specificity of monoclonal antibodies with the potency of cytotoxic agents. This targeted approach allows for the selective delivery of cytotoxic drugs directly to cancer cells, thereby minimizing off-target effects and potentially reducing systemic toxicity.
In 2021, SG was approved by the United States Food and Drug Administration (FDA) for patients with metastatic triple-negative breast cancer.15,23 Recently, SG has also been tested in different epithelial cancers, including HNSCC in a phase I/II study, which demonstrated the safety and tolerability of SG in HNSCC.24,25 However, further research is needed to establish its potential in HNSCC/OSCC therapy.
Nectin-4, an immunoglobulin-like transmembrane cell adhesion protein involved in different tumor-signaling pathways,26,27 is an efficient target for the ADC enfortumab vedotin (EV). EV has been approved by the FDA for locally advanced or metastatic urothelial cancer, either as third-line therapy, in case of progression despite platinum-based and checkpoint inhibitor therapy, or even as first-line therapy in combination with pembrolizumab when patients are not eligible for cisplatin.28,29
An overexpression of Nectin-4 has been found in various tumor entities such as lung, breast, ovarian, bladder, hepatocellular, gastrointestinal, and salivary gland carcinomas.30 –34 Currently, data concerning Nectin-4 expression in OSCC are scarce. Sanders et al. 35 demonstrated an overexpression of Nectin-4 in OSCC and were able to demonstrate a correlation with HPV positivity. The integral membrane protein Claudin 18.2 belongs to the Claudin protein family, which forms key components for tight cell junctions. Due to their involvement in most steps of tumor development and their tissue-specific manner of expression, they represent a useful target for various therapy strategies. 36 Against Claudin 18.2, which is mostly expressed in gastric (cancer) cells, specific antibodies (Claudiximab and Zolbetuximab) were developed recently and are being tested in multiple ongoing trials for advanced gastric cancer.37,38 In OSCC, research on Claudin 18.2 has merely played a minor role thus far. 39
We here aimed to investigate TROP2-, Nectin-4, and Claudin 18.2 expression in a representative cohort of HPV-positive and HPV-negative OSCC using immunohistochemistry (IHC). We furthermore correlated these findings with clinical and pathological data.
Methods
Patient cohort and tumor samples
Patients involved in this retrospective cohort study were diagnosed with OSCC, classified under C09 and C10 according to the International Classification of Diseases for Oncology (ICD-O), and received treatment at the University Hospital Cologne from January 2005 to December 2020. Tissue microarrays (TMA) were constructed from formalin-fixed, paraffin-embedded (FFPE) cancer tissue, resulting in 226 samples. The clinical and pathological characteristics of the patient cohort were obtained from medical records and are summarized in Table 1. The extent of the disease was assessed using the UICC TNM classification, either the 7th or 8th edition, depending on the classification valid at the time of diagnosis. Tumor staging for all patients was determined according to the American Joint Committee on Cancer’s 8th edition (AJCC8 I-IV). The reporting of this study conforms to the STROBE statement for cohort studies (Supplemental Table 1). 40
Clinicopathological features of oropharyngeal squamous cell carcinoma patient cohort according to HPV status.
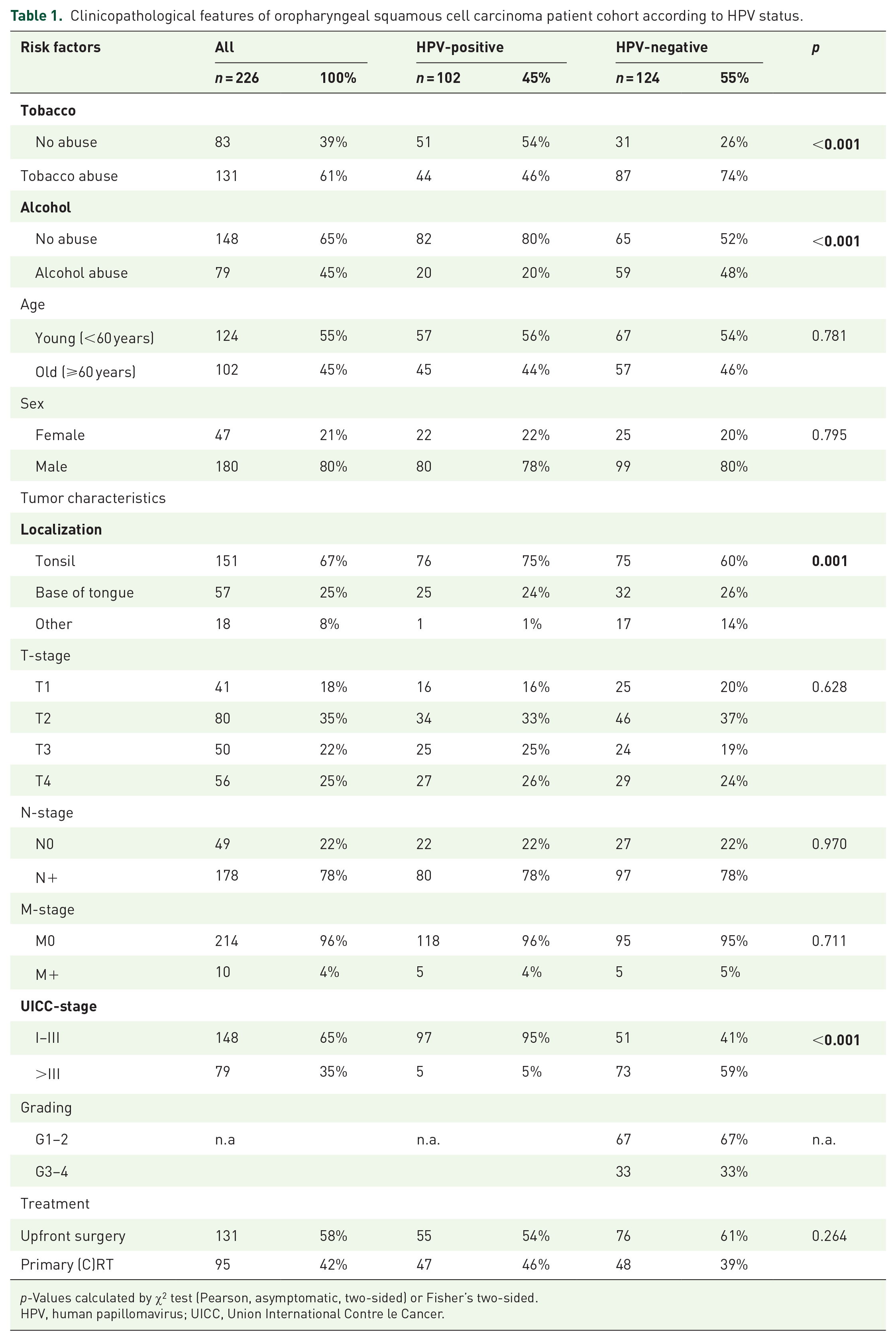
p-Values calculated by χ2 test (Pearson, asymptomatic, two-sided) or Fisher’s two-sided.
HPV, human papillomavirus; UICC, Union International Contre le Cancer.
P16INK4a immunohistochemistry and HPV-DNA genotyping
To identify the association of HPV with OSCCs, p16INK4a (p16) IHC and HPV-DNA genotyping were conducted for all patients. The expression of p16 was assessed using the Zytomed histology kit (Zytomed Systems, Berlin, Germany), following the manufacturer’s instructions and standard protocols. 4 Extracted DNA was examined for the presence of HPV DNA and various HPV genotypes through the amplification of highly conserved regions of the viral genome (L1 Region, using primer pairs MY11/19 and 125′), followed by DNA/DNA hybridization for specificity assessment. Tumors were classified as HPV-positive if p16 IHC staining was observed in 70% or more of tumor cells and high-risk HPV DNA of the specified types was present. Other combinations, such as HPV DNA+/p16−, HPV DNA−/p16−, and HPV DNA−/p16+, were classified as HPV-negative.
Preparation of tissue microarrays and immunohistochemistry
Adequate FFPE cancer tissue, with a minimum thickness of 2 mm, was required for the creation of TMA cores. The process of TMA assembly was carried out as previously described. 41 In brief, 1.2 mm cores were extracted from tumor areas, including the invasive margins, as identified by an experienced pathologist. Using a semi-automated punch press, a 1.2 mm cylinder was removed and placed into a new paraffin block. For staining, 4 µm thick sections were freshly sliced from the TMAs.
Immunohistochemical staining was performed using antibodies specific to Nectin-4 and TROP2, following the protocols recommended by the manufacturers. Details regarding the antibodies (Abcam Cat# ab214488, RRID:AB_2811182, Nectin-4) are provided in Supplemental Table 2. Automated staining was conducted using the Leica BOND-MAX system (Leica Biosystems, Wetzlar, Germany), adhering to the manufacturer’s instructions. The stained slides were then digitized using the Aperio GT 450 DX scanner (Leica Biosystems).
To evaluate TROP2, Nectin-4, and Claudin 18.2 protein expression, the Histo(H)-score method was employed. This involved categorizing the staining intensity into four levels: negative (=0), weak (=1), moderate (=2), and strong (=3). The H-score was calculated by multiplying the percentage of positively stained cells by the respective staining intensity value, using the formula:

The resulting H-score ranged from a minimum of 0 (100% negative cells * 0 = 0) to a maximum of 300 (100% strongly positive cells * 3 = 300) with H-scores of 0–10 being considered negative, 11–100 low, 101–200 intermediate, and 201–300 high.
Statistical analysis
Statistical analyses were performed using SPSS statistical software (IBM SPSS 28.0, Armonk, NY, USA). Comparisons between variable clinical pathological groups were made using the χ2 test (Pearson, asymptomatic) or Fisher’s two-sided exact test. Survival curves were plotted according to the Kaplan–Meier method and analyzed using the log-rank test. Patients for whom the time of death or survival could not be determined were censored at the last known contact. To analyze differences in OS, cox proportional-hazards models were used to estimate hazard ratios (HR) with a confidence interval (CI) of 95%. This was performed as univariate and multivariate analyses. All tests were two-sided. For all tests, p-values < 0.05 were considered statistically significant. Graphs were created using GraphPad Prism V10 (San Diego, CA, USA).
Results
Patient and tumor characteristics
In this study, we included 226 OSCC patients, of whom 102 (45%) had HPV-positive disease, while 124 (55%) of the analyzed cancers were HPV-negative. Comprehensive patient characteristics are displayed in Table 1. Tobacco abuse, defined as ⩾10 pack-years at the time of initial diagnosis, was reported by 131 (61%) patients, while 79 (45%) patients reported alcohol abuse, defined as ⩾2 drinks a day. Both a history of smoking and alcohol consumption were significantly associated with HPV-negative disease (p < 0.001). Forty-seven patients (21%) were female and 124 (55%) were under the age of 60 years. An association of positive HPV status with younger age was not detected in this cohort (p = 0.781).
Most primary tumors were located at the tonsil (67%), followed by the base of tongue (25%) and only 8% of cancers arose from other areas of the oropharynx. Tonsillar location was associated with HPV-positive disease (p = 0.001). Only 5 (5%) of HPV-positive cancers were classified as stage IV (UICC TNM 7th or 8th edition according to the valid classification at the time of diagnosis), while 73 (59%) of HPV-negative cancers were stage IV (p < 0.001). In this cohort, 131 (58%) of patients received upfront surgery, while 95 (42%) were treated with primary (chemo)radiotherapy.
Expression of TROP2, Nectin-4, and Claudin 18.2 in OSCC
To evaluate the expression of target molecules for the ADCs SG, EV, and SHR-A1904, we performed IHC staining for TROP2, Nectin-4, and Claudin 18.2 (Figure 1).

TROP2, Nectin-4, and Claudin 18.2 immunhistochemistry in OSCC. Representative images of TROP2, Nectin-4, and Claudin 18.2 expression: (a) low TROP2 expression, (b) high TROP2 expression, (c) low Nectin-4 expression, (d) high Nectin-4 expression, and (e) Claudin 18.2 expression (no expression).
In the following tissue analysis, we classified patients according to H-Score as described in the methods section. For TROP2, 8 (3.5%) patients were classified as negative, while 28 (12.4%), 90 (39.9%), and 100 (44.2%) were classified as low, intermediate, or high, respectively (Table 2).
Relation of TROP2 and Nectin-4 expression to clinicopathological characteristics (n = 226).

p-Values calculated by X2 test (Pearson, asymptomatic, two-sided), significant p-values (p ⩽ 0.05) in bold.
HPV, human papillomavirus; OSCC, oropharyngeal squamous cell carcinoma; TROP2, Trophoblast Surface Antigen 2; UICC, Union International Contre le Cancer.
No association with HPV status or clinicopathological features was found. Collectively, 218 out of 226 (96.5%) of evaluable tumor samples showed positive expression of TROP2. No expression of Nectin-4 was detected in 131 (61.2%) patients. Seventy-three (34.1%) tumors exhibited low expression of Nectin-4, while 9 (4.2%) and 1 (0.5%) expressed intermediate and high levels. Expression of Nectin-4 was significantly associated with positive HPV status (p < 0.001), and 51 (51.5%) of HPV-positive tumors expressed Nectin-4. Association with clinicopathological features was not detected. Claudin 18.2 was not expressed in oropharyngeal cancer tissue irrespective of HPV status.
Survival analysis of TROP2 and Nectin-4 expression
For survival analysis, we combined patients with a TROP2 H-score of 0–200 (n = 120) and compared them to patients with a high expression (H-score > 200, n = 96). High expression of TROP2 was not associated with survival, even though a statistically nonsignificant trend of impaired survival (p = 0.233, Figure 2(a)) was detected. Similarly, TROP2 expression was not associated with OS in HPV-positive or HPV-negative subgroups (Figure 2(b) and (c)). Patients with detectable expression of Nectin-4 (defined as H-Score > 10) were associated with improved overall survival (p = 0.018, Figure 2(d)) compared to patients with no measurable expression. In the HPV-positive cohort, Nectin-4 expression was not associated with OS (Figure 2(e)). In the HPV-negative cohort, improved survival remained a statistically nonsignificant trend (p = 0.159, Figure 2(f)). As Nectin-4 expression was associated with positive HPV status, which in itself is a well-established predictor of improved survival in OSCC, 5 we performed survival analysis by multivariate Cox regression (Table 3). In this analysis, positive HPV status was an independent predictor of improved survival (HR 0.378, p < 0.001), but Nectin-4 was not (HR 0.737, p = 0.175).

Overall survival according to TROP2 and Nectin-4 expression and HPV status. Overall survival in patients with TROP2 expression (a–c) and Nectin-4 expression (d–f): (a, d) in the total cohort, (b, e) in patients with HPV-positive OSCC, and (c, f) in patients with HPV-negative OSCC.
Univariate and multivariate survival analyses according to HPV status and Nectin-4 expression.

p-Values calculated by log-rank (Mantel-Cox) test; univariate; p-values (p ⩽ 0.05) in bold; pa-values estimated by Cox proportional-hazards model, uni- and multivariate analyses; p-values (p ⩽ 0.05) in bold.
CI, confidence interval; HPV, human papillomavirus; HR, hazard ratio estimated by cox proportional-hazards models; n.a., not applicable; OSCC, oropharyngeal squamous cell carcinoma; TROP2, Trophoblast Surface Antigen 2.
Discussion
We investigated the expression of Nectin-4, TROP2, and Claudin 18.2C in a representative cohort of primary OSCC cases. We observed that TROP2 was expressed in the vast majority of tumors (96.5% of included cancer specimens were positive for TROP2). The nearly ubiquitous expression of TROP2 in OSCC, compared to its low or absent expression in tumor-free tissue, makes TROP2 a highly favorable candidate to deliver a payload specifically into cancer tissue. We found a higher rate of TROP2 expression in comparison to previous studies of Tang et al. and Fong et al., who reported an overexpression in 62.5% and 58% of cases.21,42 This difference may be explained by varying sensitivities of the IHC analysis as there is no standardized definition of cut-offs: Tang et al., for example, defined the H-Score 0–120 as low or no expression (and <130 as high expression), whereas Fong et al. did not use the H-score System at all. Therefore, the comparability of the expression rates of TROP2 with those of different working groups is limited. Alternative quantification methods may better reflect the functional relevance of TROP-2 expression. These include digital image analysis, which allows for objective and continuous quantification of staining intensity and area, and automated machine-learning-based tissue classification, which may capture complex spatial expression patterns. 43 Moreover, methods such as quantitative immunofluorescence (QIF) or mass spectrometry-based proteomics can provide more precise or multiplexed protein quantification.44,45 On the transcriptomic level, RNA in situ hybridization (e.g., RNAscope) may complement protein data and clarify expression in specific cellular compartments. 46 These approaches could be employed in future studies to validate our findings and explore the prognostic and predictive value of TROP2 expression more thoroughly.
With the emergence of individualized tumor therapy for many cancers, treatment options for patients with advanced-stage, recurrent, metastatic, or inoperable HNSCCs remain limited. Available therapies (surgery, chemotherapy, and radiation therapy) often yield suboptimal results in advanced OSCC and other HNSCC. While the use of immune checkpoint inhibitors like pembrolizumab and nivolumab has revolutionized the treatment for certain cancers (malignant melanoma, Hodgkin’s lymphoma), only a subset of patients responds to these therapies.47 –49 This highlights the need for further exploration of targeted therapies, including ADCs. SG was developed as an ADC for TROP2 and was approved by the FDA for triple-negative breast cancer in 2021. In the ASCENT trial, whose results led to SG’s FDA approval, greater efficacy of SG was observed among patients with medium or high TROP2 expression (14.2 months OS for high TROP2 expression, 14.9 months for medium TROP2 expression, and 9.3 months OS for low TROP2 expression).15,23 Datopotamab deruxtecan (Dato-DXd), another TROP2-directed ADC currently investigated in phase II clinical trials, has several advantages over SG in the treatment of TROP2-positive cancers. Dato-DXd features a highly stable construct 50 in circulation with a longer half-life compared to SG. Dato-DXd utilizes a more potent topoisomerase I inhibitor (DXd) 51 as its cytotoxic payload, which has been shown to induce significant DNA damage and apoptosis in TROP2-expressing tumor cells, leading to promising results in the phase I clinical trial TROPION-PanTumor01. 52 Dato-DXd has shown a manageable safety profile with common adverse events being low-grade nausea and stomatitis. This contrasts with SG, which is associated with higher rates of neutropenia and diarrhea. 52 In the majority of cases in our study, there was an intermediate or high expression (84.1%) of TROP2. Our findings suggest that TROP2 may serve as an effective target for ADCs like SG or Dato-DXd in OSCC, warranting further investigation in clinical trials.
Despite the high expression of TROP2 in this cohort, our survival analysis did not reveal a statistically significant association between high TROP2 expression and OS, regardless of HPV status. This contrasts with the findings of Zhang et al. and Fong et al., who demonstrated an association of decreased OS with TROP2 overexpression.21,22 This discrepancy may be attributed to differences in the cut-off values used in the respective studies. Larger longitudinal studies are needed to validate the clinical relevance of TROP2 as a prognostic biomarker in OSCC.
Claudin 18.2 showed no protein expression in OSCC. Thus, clinical testing of individual OSCC tumor specimens for Claudin 18.2 is not warranted. Further analyses are needed to confirm this result in other anatomical regions of HNSCC, particularly in nasopharyngeal carcinoma due to the distinct tumor biology of these cancers.
Nectin-4 expression was detected in 38.8% of OSCC cases, with a significantly higher prevalence in HPV-positive tumors (51.5%). The correlation with HPV positivity is consistent with a study in a smaller cohort of HNSCC patients by Sanders et al. 35 that also reported Nectin-4 overexpression in HPV-positive OSCC. Sanders et al. are, next to us, the only other working group that analyzed Nectin-4 expression in OSCC in correlation with clinicopathological data. Grass et al., who demonstrated this association between HPV and penile squamous cell carcinoma, suggested that Nectin-4 expression is triggered by the oncogenic activity of the oncoproteins E6 and E7, which are known to be mainly responsible for HPV-induced carcinogenesis.53,54 The E6 and E7 oncoproteins of HPV interact with cellular proteins involved in epigenetic regulation, such as DNA methyltransferases and histone-modifying enzymes, leading to alterations in DNA methylation and histone modifications. These epigenetic alterations may contribute to Nectin-4 expression in HPV-positive OSCC. 55 Another explanation for the higher prevalence of Nectin-4 in HPV-positive tumors could be a possible biological connection between HPV and Nectin-4: Nectin-4 is a cancer-specific TIGIT (inhibitory receptor T-cell immunoreceptor with Ig and ITIM domains) ligand, and the only Nectin family member that interacts solely with TIGIT. 56 TIGIT expression is prevalent in the tumor immune microenvironment of HPV-positive HNSCC, which includes OSCC. 57 Further research on this topic is needed. Despite using the same antibody as Sanders et al., our analysis revealed notable differences in protein distribution. Sanders et al. reported expression levels as follows: 13.8% negative, 53.5% low, 27.0% medium, and 5.7% high. 35 By contrast, we employed an H-score system and categorized the results as follows: 61.2% negative, 34.1% low, 4.2% intermediate, and 0.5% high expression. These discrepancies can be attributed primarily to differences in cohort composition, sample size, and scoring methodology. While Sanders et al. analyzed a heterogeneous cohort of HNSCC (n = 159), which included tumors in the oral cavity, oropharynx, and hypopharynx, as well as cases of unknown localization (49 cases) and laryngeal SCC (28 cases), our study exclusively focused on OSCC. In addition, cases categorized as “weak” by Sanders et al. may correspond to our “negative” classification (H-score 0–10). These methodological and cohort-specific differences provide a plausible explanation for the observed variation in Nectin-4 expression patterns between the two studies.
EV, a Nectin-4-directed ADC, was evaluated in a phase II trial involving patients with previously treated advanced head and neck cancer. 58 The study demonstrated that EV exhibited antitumor activity in heavily pretreated HNSCC patients, with a confirmed objective response rate (ORR) of 23.9% and a disease control rate (DCR) of 56.5%. The safety profile was consistent with known adverse events associated with EV, including alopecia, fatigue, and peripheral sensory neuropathy. The median H-Score of Nectin-4 expression in the trial population was 180 (range between 20 and 300). 58 This contrasts with findings from our analyses, where, as already mentioned above, Nectin-4 expression was negative in 61.2% of tumor specimens. As noted by the authors themselves, one limitation of the phase II trial was its small sample size of only 46 patients. The cohort composition differs entirely from ours: In the phase II trial, patients with heterogeneous tumor localization (oral cavity, oropharynx, larynx) were included. All patients were from North America or Asia. This might explain the discrepancies in Nectin-4 expression compared to our results. Nevertheless, our results imply the importance of assessing Nectin-4 expression before including patients in the trial. In the phase II trial, Nectin-4 expression was not an inclusion criterion, but a retrospective analysis of the Nectin-4 protein expression was conducted. Larger prospective studies are necessary to assess Nectin-4 expression in OSCC/HNSCC and to evaluate its relevance for the success of ADC therapy, such as EV.
In the survival analysis, we demonstrated improved OS of patients with Nectin-4 positive OSCC compared to those negative for Nectin-4 in the univariate analysis. This finding aligns with the results of Sander et al., 35 who also reported longer survival times in patients with Nectin-4 positive HNSCC. In our case, this might be explained by the association of Nectin-4 with HPV positivity, which is itself a well-established predictor of improved survival in OSCC. 5 In the multivariate Cox regression, we demonstrated that positive HPV status was an independent predictor of improved survival, but Nectin-4 was not. In other tumor entities (e.g., lung cancer, 34 bladder cancer, 59 and esophageal cancer 60 ), decreased OS is associated with Nectin-4 overexpression. Due to the lack of further studies on Nectin-4 expression in OSCC, its prognostic role in OSCC remains inconclusive.
Conclusion
In summary, we demonstrated an almost ubiquitous expression of TROP2 in OSCC while Nectin-4 expression was detectable in some patients, and Claudin 18.2 was not expressed. Nectin-4 expression was associated with HPV-positive OSCC. These results provide further rationale for conducting clinical trials of TROP2- and Nectin-4-directed ADCs in OSCC.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251361877 – Supplemental material for Expression patterns of TROP2 and Nectin-4 in oropharyngeal squamous cell carcinoma in relation to HPV status: potential biomarkers for targeted therapy
Supplemental material, sj-docx-1-tam-10.1177_17588359251361877 for Expression patterns of TROP2 and Nectin-4 in oropharyngeal squamous cell carcinoma in relation to HPV status: potential biomarkers for targeted therapy by Charlotte Klasen, Hans N. C. Eckel, Nora Wuerdemann, Joseph Böckelmann, Karl Knipper, Malte Suchan, Kariem Sharaf, Shachi Jenny Sharma, Helen Abing, Arthur Charpentier, Julia Esser, Kevin Hansen, Marcel Mayer, Louis Jansen, Jens Peter Klußmann, Alexander Quaas and Su Ir Lyu in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251361877 – Supplemental material for Expression patterns of TROP2 and Nectin-4 in oropharyngeal squamous cell carcinoma in relation to HPV status: potential biomarkers for targeted therapy
Supplemental material, sj-docx-2-tam-10.1177_17588359251361877 for Expression patterns of TROP2 and Nectin-4 in oropharyngeal squamous cell carcinoma in relation to HPV status: potential biomarkers for targeted therapy by Charlotte Klasen, Hans N. C. Eckel, Nora Wuerdemann, Joseph Böckelmann, Karl Knipper, Malte Suchan, Kariem Sharaf, Shachi Jenny Sharma, Helen Abing, Arthur Charpentier, Julia Esser, Kevin Hansen, Marcel Mayer, Louis Jansen, Jens Peter Klußmann, Alexander Quaas and Su Ir Lyu in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
