Abstract
Cervical cancer (CC) is still characterized by a poor prognosis despite the progress made in its treatment in recent years. Although immunotherapy has improved outcomes for advanced/recurrent disease, there is a significant gap in addressing patients’ needs when they progress after platinum and immunotherapy treatments. In this setting, traditional chemotherapy showed limited effectiveness. In this context, antibody–drug conjugates (ADCs) emerged as a promising tool within targeted cancer therapies. Tisotumab vedotin (TV), an ADC targeting tissue factor, represents the first ADC approved by the US Food and Drug Administration for the treatment of recurrent or metastatic CC with disease progression on or after chemotherapy. In phase I–III published trials, TV has already demonstrated an advantage in terms of objective response rate (17.8%–54.4%) and progression-free survival (3.1–6.9 months) in patients who progressed to the first-line standard therapy. Data concerning the addition of TV to platinum/pembrolizumab first-line chemotherapy are still under analysis and strongly expected. However, several questions are still unresolved: (1) the identification of the most suitable timing for ADCs administration in the treatment sequence of advanced/recurrent CC; (2) the evaluation of combination therapies as a tool to minimize the emergence of resistant clones and to enhance overall effectiveness; and (3) the assessment of tolerability and correct management of special toxicities (e.g. ocular and neurological adverse events). In the near future, an improvement in patient selection via biomarker-driven strategies should be crucial for optimizing both treatment benefits and maintaining an acceptable toxicity profile.
Keywords
Introduction
Despite the widespread diffusion of human papillomavirus (HPV) vaccination in the past few years, cervical cancer (CC) still impacts public health care, especially in developing countries, with a worldwide new estimated case of 660,000 and 350,000 deaths in 2022. Indeed, CC represents the fourth most common cancer in women globally. 1 Until 2014, therapeutic options for metastatic/recurrent CC were significantly limited, primarily relying on platinum-based combinations with paclitaxel, topotecan, gemcitabine, or vinorelbine. The results of the GOG-240 showed a significant improvement in the median overall survival (OS) by the addition of bevacizumab (16.8 vs 13.3 months; hazard ratio (HR): 0.765; 95% confidence interval (CI): 0.62–0.95; p = 0.0068). These results made paclitaxel and cisplatin combined with bevacizumab the preferred first-line regimen in metastatic/recurrent CC.2,3 In 2021, KEYNOTE-826 showed unprecedented results in terms of OS by the addition of pembrolizumab to standard platinum-based chemotherapy (CT), rapidly becoming the new standard of care in first-line treatment for persistent, recurrent, or metastatic CC in women with tumors expressing programmed death-ligand 1 (PD-L1) with a Combined Positive Score (CPS) of 1 or higher. 4 In subsequent lines of therapy, in KEYNOTE-158 study, Pembrolizumab showed quite interesting objective response rate (ORR) in several solid tumors expressing PD-L1, including patients with recurrent CC (ORR 12%), leading in 2018 to approval by the US Food and Drug Administration (FDA). 5 The efficacy of checkpoint inhibition in CC was reinforced by the findings of the phase III EMPOWER-Cervical 1 study, in which the anti-PD-1 Cemiplimab demonstrated an advantage in median OS (12 vs 8.5 months) compared to standard CT, regardless of PD-L1 status. 6 Based on trial results, the FDA and the European Medicine Agency (EMA) approved Cemiplimab in immune-naïve patients with disease progressing after first-line platinum-containing CT. Finally, in January 2024, the results of the BEATcc study were published. This randomized trial enrolled patients with recurrent or metastatic CC candidate to treatment with cisplatin plus paclitaxel and mandatory bevacizumab with or without atezolizumab, an anti-PD-L1 antibody. The addition of atezolizumab resulted in significantly higher progression-free survival (PFS) and OS with a 38% reduction in the risk of progression and a 32% reduction in the risk of death. 7
Additionally, KeynoteA18 demonstrated an advantage in terms of PFS in locally advanced CC treated with chemo-radiotherapy (CT/RT) in combination with pembrolizumab (HR for disease progression or death was 0.70 (95% CI: 0.55–0.89, p = 0.0020)). 8 While the introduction of immunotherapy has enhanced the outcomes of advanced/recurrent disease, a significant unmet need persists for patients experiencing progression after platinum and immunotherapy. In this setting, standard CTs exhibit a suboptimal response rate (<15%), thereby resulting in a median OS of approximately 9 months. 9 In this scenario, antibody–drug conjugates (ADCs) represent a promising approach in the field of targeted cancer therapy. This review provides a general overview of ADCs and, more specifically, discusses the current evidence and potential future developments of tisotumab vedotin (TV) in treating recurrent or advanced CC.
ADCs: bridging the gap between precision and potency
One of the primary goals of cancer research is the development of anticancer drugs capable of eradicating tumor cells while preserving normal tissues, thereby minimizing potential side effects. ADCs are a novel and revolutionary class of drugs combining the antigen selectivity of monoclonal antibodies (mAbs) with potent cytotoxic agents, that are bringing us closer to achieve the goal of enhancing both the survival and quality of life of patients. 10
A typical ADC consists of three key components: the targeting antibody, the cytotoxic payload, and the linker. Following intravenous (IV) administration, the ADC circulates into the bloodstream and reaches cancer tissues, where the antibody component recognizes its target antigen. The antibody–antigen complex is then internalized and the linker undergoes selective cleavage by lysosomal enzymes, allowing the release of the payload. 11 Moreover, the toxic payloads released can induce a “bystander effect,” consisting in the diffusion of the payload from antigen (Ag)-positive tumor cells to neighboring Ag-negative tumor cells, thereby increasing cell death. To achieve the bystander effect, the intracellularly released payload must possess high membrane permeability. This requires it to be hydrophobic, lipophilic, and uncharged. 12
The majority of ADC targets are expressed in a wide range of tumor. ADCs can target any molecules found on the surface of cancer cells, such as polypeptides, proteins, carbohydrates, and lipids. Optimal targets are antigens expressed exclusively on tumor cell surfaces, with minimal expression in normal tissues. In the context of solid tumors, ADCs currently approved by the FDA target Human Epidermal Growth Factor Receptor 2 (HER2), trophoblast antigen 2 (TROP-2), Nectin-4, folate receptor alpha (FRα), and tissue factor (TF). 13 The linkers play a key role in the success of ADCs, and they must have sufficient plasma stability to prevent the premature release of the payload and avoid undesired damage to noncancerous tissues. 13
Depending on their stability, they can be classified into two main types: cleavable and non-cleavable linkers. Non-cleavable linkers, being the most stable type, generally result in better tolerability, but ADCs with cleavable linkers typically demonstrate superior efficacy, partly attributable to the bystander effect. 14
The payload typically consists of a highly potent small molecule with nanomolar to picomolar half-maximal inhibitory concentration (IC50) values. The payloads of FDA-approved ADCs for solid tumors currently fall into three classes: microtubular inhibitors (auristatins, maytansinoids), topoisomerase inhibitors (deruxtecan, SN-38—active form of irinotecan), and alkylating agents (duocarmazine). 15 The drug–antibody ratio (DAR) refers to the average number of drug molecules conjugated to an antibody in an ADC. It is a critical parameter in ADC design, influencing the therapeutic efficacy and safety of the conjugate. A higher DAR may increase the drug payload, potentially enhancing therapeutic efficacy, but it could also impact stability and increase the risk of off-target effects. On the other hand, a lower DAR may provide better stability but may limit the overall drug payload. Finding the optimal DAR is crucial in ADC development to balance therapeutic benefits and potential challenges. 16 ADCs were first approved for hematologic malignances (lymphoma, multiple myeloma), breast cancer, and gastric cancer. 17 Among gynecological cancers, mirvetuximab soravtansine has been recently approved by the FDA for relapsed platinum resistant ovarian cancer. 17
Concerning CC several targets are under evaluation, but TF is the most investigated.
TV: the first ADC approved in CC
TV is composed of a human mAb against tissue factor (TF) linked to the monomethyl auristatin E (MMAE), a microtubule-disrupting agent. 18 TF, also called platelet tissue factor, factor III or CD142, is a transmembrane glycoprotein that has an important role in starting the extrinsic coagulation pathway (by binding activated factor VII (FVIIa)), resulting in thrombin generation and then, in the hemostatic clot formation. In healthy conditions, TF is absent from blood and endothelial cells, being expressed in many extravascular cells, such as fibroblasts, pericytes, and vascular smooth muscle cells. In these cells, TF is intracellular and inactivated until vascular integrity is interrupted or its expression is induced. 19 Furthermore, the TF-FVIIa complex starts an internal signaling pathway by the activation of protease-activated receptor 2, resulting in gene transcription, pro-angiogenic factors, cytokines (such as interleukin-1 and interleukin-8) production, and cell survival. 20 The activation of these signaling pathways ultimately culminates in the inhibition of apoptosis, activation of migration processes, and induction of angiogenesis.21,22 A high expression of TV was described in several solid tumors, including CC (94%–100%). 23
TV can bind TF with high affinity, interfering with its signaling pathway and inhibiting cell proliferation. Additionally, TV is conjugated with the microtubule-disrupting agent MMAE and a protease-cleavable valine–citrulline linker. Auristatins are potent cytotoxic drugs that disrupt microtubules, leading to cell death. 24
After binding TF, the complex TV-TF is internalized and transported to the lysosome. Here, the linker is enzymatically cleaved and MMAE is released. MMAE can bind to tubulin and interfere with microtubule polymerization, inducing G2/M cell cycle arrest and cell apoptosis. 25 Moreover, MMAE has a high membrane permeability, thus conferring the ability to induce bystander killing effect. This attribute enables the agent to affect not only the targeted cells but also neighboring cells, contributing to a broader cytotoxic off-target activity.26,27 These antitumor effects are further enhanced by the capacity of TV to bind FcγRIIIa on adjacent natural killer cells, which leads to antibody-dependent cellular cytotoxicity of TF-expressing tumor cells. MMAE-based ADCs have also been shown to induce immunogenic cell death, which can activate innate and adaptive immune responses to tumor antigens.28,29
The efficacy and safety of TV have been investigated in ovarian cancer (OC), endometrial, and CC leading to FDA approval in 2021 for the treatment of recurrent or metastatic CC with disease progressing on or after CT. Recently, EMA has validated for review the marketing authorization application of this drug in the same setting. 30
TV’s mechanism of action is shown in Figure 1.
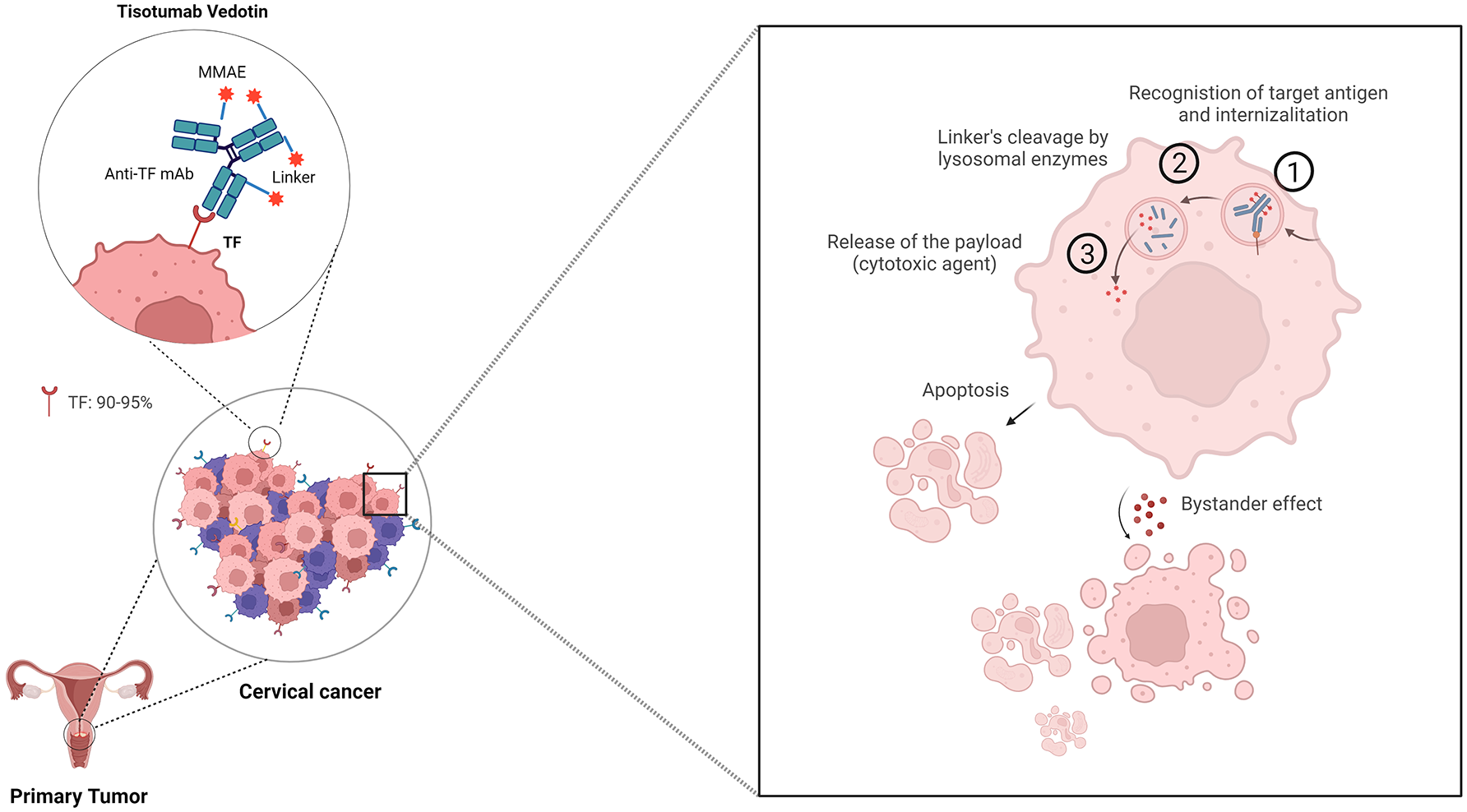
ADCs’ mechanism of action: a magnifying glass on TV is composed of a human monoclonal antibody against TF linked to the MMAE, a microtubule-disrupting agent. TF, also called platelet tissue factor, is expressed in 90%–95% of cancer cells in cervical cancer. TV mechanisms of action consist of three phases: (1) recognition of the target and internalization; (2) linker’s lysosomal degradation; and (3) release of payload. Besides the direct cytotoxic effect, TV is characterized by a bystander effect on neighboring cancer cells, which enhances its efficacy.
Clinical trials of tisotumab vedotin in CC
Methods
We searched on PubMed and CENTRAL databases from their inception up to the 20 of February 2024, without language restrictions or any filters. The following keywords were used: “cervical cancer,” “antibody drug conjugate,” and “tisotumab vedotin.” The reference section of previous relevant reviews on the topic was searched for other potentially pertinent papers. Published, completed, and ongoing trials were considered. For the latter, we queried ClinicalTrials.gov. The following inclusion criteria were applied: diagnosis of CC, treatment with TV, recruiting, and not yet recruiting studies without study results. Similarly, we excluded other ADCs, other gynecological malignances.
Phase I/II
The InnovaTV 201 trial (NCT02001623), comprising a phase Ib/II design, encompassed a dose-escalation phase followed by a dose-expansion phase. It enrolled patients with advanced or metastatic tumors who either relapsed after or were ineligible for standard treatments. The expansion phase included various cancer types, including cervical, ovarian, endometrial, prostate, bladder, esophageal, and non-small cell lung cancer (NSCLC). In the CC cohort, eligible patients had undergone at least four prior lines of CT. Of interest, TF expression did not represent an inclusion criterion, although its expression was confirmed in most evaluable patients (100% membrane expression and 95% cytoplasmic expression). The trial enrolled 55 patients who received TV at a dosage of 2 mg/kg every 3 weeks for 4 cycles, with the option of continuing treatment for up to 12 cycles in case of stable disease, partial, or complete response, and beyond progression if a clinical benefit occurred (NCT03245736). Safety represented the primary endpoint and patients received prophylactic ocular medication to mitigate the risk of ocular adverse events (AEs). Notably, 56% of patients experienced Grade 3 or worse AEs, with anemia (11%), fatigue (9%), and vomiting (7%) being the most common. All patients experienced at least one AE of any grade, with fatigue (51%), epistaxis (51%), nausea (49%), and conjunctivitis (42%) being the most prevalent. Bleeding events, neuropathy, and ocular events (conjunctivitis, ulceration, keratitis, and symblepharon) were reported as adverse events of special interest (AESIs). Epistaxis was the most common bleeding-related AE (98% grade 1), 7% reported a grade >3 neuropathy and 60% had an ocular event (any grade). The most common ocular AE was conjunctivitis (43%), the incidence of which was reduced by the implementation of ocular mitigation strategies. The assessment of antitumor activity, a secondary endpoint, revealed a median progression-free survival (mPFS) of 4.2 months and an investigator-evaluated response rate (ORR) of 24%, confirmed (ORR 22%) in the evaluation by the independent review committee.31,32
InnovaTV206 (NCT03913741), a single arm phase I/II trial, evaluated TV’s data of safety and efficacy in a Japanese population with recurrent and metastatic CC. The dose escalation phase (part 1) included patients with several solid tumors, while the dose expansion phase (part 2) included 17 patients with pretreated metastatic CC (at most two prior lines of CT including platinum doublet or paclitaxel and nogitecan plus bevacizumab were allowed) receiving the recommended phase II dose (RP2D) of TV settled at 2 mg/kg in the escalation phase. Implementation strategies for ocular toxicity prevention were mandatory. Primary endpoint was safety; secondary endpoints included ORR, duration of response (DOR), and time-to-response (TRR). OS, PFS, and TF expression measurements on tissue and blood samples were exploratory endpoints. In part 2, the most common treatment-emergent adverse events (TEAEs) were anemia (58.8%), nausea (58.8%), alopecia (47.1%), and epistaxis (47.1%). The most common grade 3 TEAEs were anemia (35.3%), tumor hemorrhage (11.8%), and leukopenia (11.8%). No TEAEs were associated with death. AESIs included grade 1–3 bleeding events (76.5%), grade 1–2 ocular events (35.3%), and grade 1 peripheral neuropathy (17.6%). Ocular toxicities accounted for 35.3% of events; the incidence of peripheral neuropathy was 17.6%. Efficacy data showed an ORR of 29.4% and a median DOR of 7.1 months. The efficacy data were consistent with data from the European and American populations, with a median PFS of 3.1 months (95% CI, 1.2–7.1) and a median OS of 11.4 months (95% CI, 6.2–not reached). Patients’ responses were reported regardless of tumor cells’ TF expression. 33
InnovaTV 205/GOG-3024/ENGOT-cx8 (NCT03786081) is a phase Ib/II open-label trial evaluating TV as monotherapy (experimental ARM G) and in combination with bevacizumab (experimental arm A), pembrolizumab (experimental arm B, arm E, arm F), carboplatin (experimental arm C and D) or with carboplatin, pembrolizumab and optionally bevacizumab (experimental arm H). The study design was articulated in a dose escalation phase (arm A, B, and C) and a dose expansion phase (arm D, E, F, G, and H) enrolling both patients with recurrent and metastatic (IVB) CC previously treated according to the standard of care (arm A, B, C, F, and G) and treatment naïve patients (arm D, E, and H). TF and PD-L1 expression did not represent an inclusion criterion. The primary endpoint of the dose-escalation phase was the incidence of dose-limiting toxicities (DLTs) and AEs with TV used in combination with bevacizumab (arm A), pembrolizumab (arm B), and carboplatin (arm C). The primary endpoint of the dose expansion phase was the ORR according to RECIST 1.1 in treatment-naïve patients (TV in combination with carboplatin arm D, or pembrolizumab arm E). Patients who have progressed on or after standard-of-care treatment received TV in combination with pembrolizumab (arm F) or TV alone (arm G). The preliminary results of InnovaTV 205 concerning dose escalation arms and a part of dose expansion arms (arm D, E, and F) have been recently published. A total of 142 patients were enrolled, 41 of which in the dose escalation phase and 101 in the dose expansion phase. The defined RP2D of TV was 2 mg/kg for every combination. Concerning the primary endpoint of the dose expansion phase, the ORR in untreated patients was 54.5% (95% CI, 36.4–71.9) with carboplatin (arm D) and 40.6% (95% CI, 23.7–59.4) with pembrolizumab (arm E). In the pretreated population, ORR with pembrolizumab (arm F) was 35.3% (95% CI, 19.7–53.5). The median DOR was 8.6 months, not reached, and 14.1 months, for carboplatin and pembrolizumab (arm D, E, and F), respectively. Concerning the safety profile, the more frequently reported grade <3 events were: anemia (39.4%), diarrhea (15.2%), thrombocytopenia (15.2%), nausea (15.2%) in arm D; anemia (12.1%), asthenia (9.1%), hypokalemia (9.1%) in arm E; and anemia (28.6%), intestinal obstruction (11.4%), weight decreased (11.4%) in arm F. The most common AEs leading to TV discontinuation were ocular and neurological events. The approval of KEYNOTE286 led to a protocol amendment adding another cohort (arm H) to homologate the trial to the new standard of care (platinum doublet plus pembrolizumab ± bevacizumab); data from arm H are not available yet. 34
Phase II
Within the InnovaTV204/GOG3023/ENGOTcx6 phase II trial, Coleman et al. assessed the antitumoral activity and safety of TV in advanced CC patients who progressed after no more than two prior treatment lines. The primary endpoint was ORR, with secondary endpoints including safety, tolerability, PFS, OS, time to response (TTR), and DOR. The correlation between TF expression and ORR was an exploratory endpoint. At a median follow-up of 10 months, 4 patients out of the 101 enrolled were still on treatment. The ORR was 24%, with 7 complete responses and 17 partial responses. The disease control rate (DCR) was 72%, with a median TTR of 1.4 months. Kaplan–Meier analysis yielded a median PFS of 4.2 months and a median OS of 12.1 months. No correlation between TF membrane expression and TV response was found in the exploratory analysis. The safety profile was consistent with previous reports: treatment-related AEs occurred in 93% of patients, with grade 3 or worse AEs observed in 28%. Notably, neutropenia (3%), fatigue (2%), ulcerative keratitis (2%), and peripheral neuropathies (2%) were among the most common grade 3 or worse AEs. Among ocular AEs, conjunctivitis (23%), dry eye (23%), and keratitis (11%) were the most common. Only two patients experienced a grade 3 ocular AE, with 86% of events fully resolved 30 days post-treatment. Bleeding AEs affected 39% of patients, with two cases of grade 3. Epistaxis (30%), vaginal hemorrhage (7%), and hematuria (3%) were the most frequent bleeding events, with over 90% resolving by the 30-day follow-up. Peripheral neuropathy treatment reactions were noted in 33% of patients, with 7 experiencing grade 3 events. Unlike previously reported AESIs, neuronal damage persisted in 79% of patients 30 days post-treatment. Thirteen percent of patients experienced serious AEs, with sensorimotor neuropathy (2%) being most common. 35
Phase III
InnovaTV301/ENGOT cx-12/GOG-3057 (NCT04697628) is a phase III open label trial enrolling patients with pretreated (second or third line) metastatic/recurrent CC randomized to TV 2.0 mg/kg 1q3w or investigators’ choice CT among topotecan (1 or 1.25 mg/mq IV on Days 1–5, every 21 days), vinorelbine (30 mg/mq IV on Days 1 and 8, every 21 days), gemcitabine (1000 mg/mq IV on Days 1 and 8, every 21 days), irinotecan (100–125 mg/mq IV weekly for 28 days, every 42 days), or pemetrexed (500 mg/mq IV on Day 1, every 21 days). The study had a hierarchical design with OS as primary endpoint; secondary endpoints included PFS, ORR, DOR, TTR, and health-related quality of life. Of interest, patients had to experiment disease progression on or after CT doublet with bevacizumab and an anti-PD-L1 agent, if eligible and available, according to the new first-line standard of care. ECOG PS (0 vs 1), prior bevacizumab exposure, prior anti-PD-L1 therapy, and geographic region were stratification factors. Although the trial is still formally recruiting on ClinicalTrial.gov with an estimated enrollment’s number of 556 patients, preliminary results were presented as oral late breaking abstract at ESMO Congress in October 2023. With a median follow-up of 10.8 months, TV showed a 30% of reduction in risk of death (HR: 0.70; 95% CI: 0.54–0.89; p = 0.0038) with a significantly longer median OS (11.5 months (95% CI: 9.8–14.9) vs 9.5 months (95% CI: 7.9–10.7)). Even the secondary endpoints favored TV in terms of PFS (HR: 0.67 (95% CI: 0.54–0.82); p < 0.0001), ORR (17.8% vs 5.2% odds ratio: 4.0; 95% CI, 2.1–7.6; p < 0.0001), and DCR (75.9% vs 58.2%). The median duration of response (mDOR) was 5 months in each arm. Data on safety were consistent with previous reported AEs; no grade 4 or 5 AESIs were reported. The most common grade ⩾ 3 AESIs with TV were peripheral neuropathy (5.2%), ocular toxicities (3.2%), and bleeding (0.8%). Two patients receiving TV died for treatment related events (acute kidney injury and Stevens–Johnson syndrome); one patient receiving CT died for pancytopenia. Dose discontinuation due to ocular and peripheral neuropathy events occurred in 5.6% of patients. 36
Focus on the safety profile and AEs of TV
According to the available data on CC from clinical trials, TV presents a manageable and tolerable safety profile. The most common AEs reported were nausea (54%), alopecia (39%), conjunctivitis (30%), fatigue (26%), and dry eye (23%).31–36
Since TV is a TF-directed ADC containing MMAE, the most common AESIs were related to ocular events and neuropathy, typical toxicity of MMAE-containing drugs, and bleeding or hemorrhage, given the TF coagulation properties. AESIs’ incidence within clinical trial is reported in Table 1. As mentioned, the evaluation of the safety profile represented the primary endpoint of innovaTV201 and innovaTV206 trials. Among AEs, bleeding events, neuropathy, and ocular events were reported as AESIs, and as shown in Table 1, in both trials, most of the events were mild (grade 1–2). In the phase II study innovaTV 204, 138 ocular treatment related adverse events (TRAEs) were observed in 54 (53%) patients, mostly grade 1 or 2. Because ocular TRAEs presented signs and symptoms easily recognizable both by patients and by healthcare professionals, the majority (86%) resolved within 30 days after the last dose of TV. Generally, ocular TRAEs were manageable with ophthalmic care (prophylactic and symptom management) and TV dose modifications. To help reduce the risk of ocular AEs, an eye care plan based on clinical trial experience was developed. This involves an oncology care team collaborating with an ophthalmologist, integrating eye examinations at baseline (according to the trial mitigation measures) and before administering each dose, providing eye drops and cold compresses, discouraging the use of contact lenses, and recommending immediate referral for any new or worsening ocular signs and symptoms. Additionally, guidelines for adjusting dosage have been formulated to address possible ocular AEs. Overall, the grade 3 AESI detection rate was very low (ocular events 3%, neuropathy 7%, bleeding 6%).
Phase I–III clinical trial investigating Tisotumab Vedotin in cervical cancer.

Trial value reported are referred to dose expansion cohorts.
Trial value reported are referred to TV arm.
Estimated number.
AESI, adverse event of special interest; CBDCA, carboplatin; CC, cervical cancer; CI, confidence interval; CR, complete response; DOR, duration of response; IC of CHT, investigator choice of chemotherapy; NCT, national clinical trial; ORR, objective response rate; OS, overall survival; PFS, progression free survival; PD, progression disease; PR, partial response; Q3W, every 3 weeks; SD, stable disease; TV, tisotumab vedotin.
However, despite the measures implemented to prevent toxicities, ocular and neurological events were the most common AEs leading to TV discontinuation within innovaTV205 trial. Of interest, grade 3 ocular and neurological events were frequently observed in arm D (TV + carboplatin in chemo naïve patients), with an incidence rate of 9.1% and 12.1%, respectively.
In the InnovaTV301 interim analysis presented at ESMO 2023, the most common TRAEs in the TV arm were conjunctivitis (30%), nausea (29%), peripheral sensory neuropathy (27%), alopecia (24%), and epistaxis (23%). Among AESIs, 50% were ocular AEs (30% conjunctivitis, 16% keratitis, 13% dry eyes), 36% were AEs related to peripheral neuropathy (27% peripheral sensory neuropathy, 3% paresthesia, 2% muscular weakness, 2% peripheral sensorimotor neuropathy), and 28% were AEs related to bleeding (23% epistaxis, 3% hematuria, and 3% vaginal hemorrhage). AESIs were mostly grade 1–2. There were no grade 4 or 5 AESIs. Dose discontinuation due to ocular and peripheral neuropathy events occurred in 5.6% of patients.
Discussion
In recent years, the integration of immunotherapy in metastatic/recurrent CC therapeutic algorithm has led to a paradigmatic change in first-line standard of care. As expected, the close relationship between persistent HPV infection, present in up to 99% of cases in squamous CC, 37 and the high rate of immune infiltration and PD-L1 expression (close to 90% in registration trial), 38 has represented the rational for the introduction of immunotherapy in the treatment algorithm of CC and has led to a significant survival improvement. The KEYNOTE-826 had shown practice changing results in metastatic/recurrent CC. In the intention-to-treat population in fact the median PFS was 10.4 months (HR 0.65; 95% CI: 0.53–0.79; p < 0.001), and the median OS was 24.4 months with 50.4% of patients estimated to be alive at 24 months (HR, 0.67; 95% CI, 0.54–0.84). However, based on the subgroup analyses according to PD-L1 expression, the HR for disease progression or death in the CPS < 1 population was 0.94 (95% CI, 0.52–1.70), leading to a final restricted approval to the CPS > 1 population. 4 However, despite an observed decrease in death rate risk of 36% in the PD-L1 positive population, 38% of patients experienced disease progression, suggesting the necessity to identify better treatments for these patients. 4
In addition, the definition of the correct therapeutic algorithm has been further complicated by the recent approval, to date exclusively by the FDA, of pembrolizumab in combination with chemoradiation therapy (CCRT) in locally advanced disease, based on ENGOT-cx11/GOG-3047/KEYNOTE-A18 trial. Preliminary data, presented at ESMO 2023, have indeed shown an advantage in terms of PFS (24-month PFS was 67.8% with pembrolizumab + CCRT vs 57.3% with placebo + CCRT; HR = 0.70 (95% CI, 0.55–0.89; p = 0.0020)) and a benefit trend in OS (HR = 0.73 (95% CI, 0.49–1.07); 42.9% maturity) in favor of immunotherapy integration, regardless of PD-L1 expression. 8
So, in this intricate labyrinth, several questions remain unanswered, including the impact of ADCs combined with other agents, especially in the first-line setting where early progression must be avoided. On the other hand, the investigation of novel targets for ADCs could further expand the therapeutic options with the aim of reducing AEs through greater specificity.
Concerning the first issue, within innovaTV 205/GOG-3024/ENGOT-cx8, the combination of TV with CT, bevacizumab, and/or immunotherapies had demonstrated promising efficacy in the absence of increased AE rate. For patients treated with TV plus carboplatin in the first line, the ORR was 55%, and the median DOR was 8.6 months. With a median follow-up of 17.8 months, the median PFS was 6.9 months. The PFS rate was 28% and 5% at 1 and 2 years, respectively. The median OS was NR at the data cutoff. For chemo-naïve patients who received TV plus pembrolizumab in the first line, the ORR was 41% and median DOR was NR. With a median follow-up of 21.7 months, the median PFS was 5.3 months; the PFS rate was 37% at 1 year and 29% at 2 years. The median OS was NR. Among patients who received second or third-line treatment with TV plus pembrolizumab, the ORR was 35% with a median DOR of 14.1 months. Responses were maintained in 4 of the 12 responders at the last assessment, 3 of whom remained on study treatment. With a median follow-up of 15.0 months, the median PFS was 5.6 months. The 1- and 2-year PFS rates were 35% and 15%, respectively. The median OS was 15.3 months. 34
Considering that both pembrolizumab and TV are already approved in the United States (US) as monotherapies for patients with recurrent and metastatic CC in the second-line setting, the observed additive clinical benefit and favorable safety profile of TV in combinatorial regimens support further evaluation. Data from InnovaTV205 arm H could reveal an intriguing enhanced efficacy rate in chemo naïve patients, although the evaluation of side effects and the financial toxicity resulting from the combination of the four/five drugs would be determinant to assess the translation in clinical practice. In this regard, data concerning combination therapy in other neoplasms are controversial. To date, the combination of ADC with CT has shown negative results in HER2-positive breast cancer. The KAITLIN 39 and KRISTINE 40 trials did not meet their primary endpoint, showing no advantage in the addition of trastuzumab emtansine (TDM-1) in adjuvant and neoadjuvant setting, respectively, compared to standard therapy. Efficacy data from DESTINY Breast 07 41 and DESTINY Breast 09 (ClinicalTrials.gov identifier: NCT04784715) would further clarify the role of ADC, specifically trastuzumab deruxtecan (T-Dxd), in addition to standard CT in HER2-positive metastatic breast cancer. On the other hand, the BEGONIA trial 42 showed encouraging results in triple-negative breast cancer (TNBC) with the association of datopotamab-deruxtecan and durvalumab in the metastatic setting. Focusing on other neoplasms, the association in first line of pembrolizumab plus enfortumab vedotin in urothelial cancer showed a statistically significant improvement in OS vs CT in patients with previously untreated locally advanced or metastatic urothelial carcinoma (mUC), according to data from the phase III EV-302 trial. Based on these results, the FDA in April 2023 granted accelerated approval in patients ineligible for cisplatin-containing CT. 43 Moving to the second topic, in the past few years, several targets for ADCs have been investigated in CC. A great interest was raised by T-Dxd, a combination of a mAb (Trastuzumab) targeting HER2 linked to a topoisomerase I inhibitor (Deruxtecan). T-Dxd is currently approved for the treatment of advanced breast cancer expressing HER2, as well as advanced gastric or gastroesophageal junction adenocarcinoma and NSCLC that are HER2-positive, in both the US and the European Union.44–46 Preliminary results from DESTINY-PanTumor02 Phase II Trial showed an impressive improvement in terms of outcome (ORR, 37.1% 95% CI, 31.3–43.2), median DOR 11.3 months (95% CI, 9.6–17.8), median PFS 6.9 months (95% CI, 5.6–8.0), and median OS was 13.4 months (95% CI, 11.9–15.5). Subgroups analyses according to HER2 expression showed a significant advantages in immunohistochemistry (IHC) 3+populations (ORR (61.3% (95% CI, 49.4–72.4), median DOR 22.1 months (95% CI, 9.6–not reached), median PFS 11.9 months (95% CI, 8.2–13.0), and median OS 21.1 months (95% CI, 15.3–29.6)), leading to an accelerated agnostic approval from the FDA in April 2024 restricted to this setting. Looking at results in the CC cohort, ORR was directly proportional to HER2 expression assessed by immunohistochemistry (75% for IHC3+ and 40% for IHC2+) with an overall ORR of 50%. 47
Based on data shown in breast cancer, 48 an increasing attention has been lately addressed to sacituzumab govitecan, an ADC comprising a humanized antibody targeting TROP-2 linked to the active metabolite of irinotecan, SN-38. Initially described in trophoblastic tissue, TROP-2 was later found to be highly expressed in several solid tumors, with prevalence ranging from 44% to 88.7%. Its limited expression in normal tissues makes it an appropriate target for ADCs in cancer treatment.49,50 Considering that the expression rate of TROP-2 in CC (88.7%) surpasses that in TNBC (78.1%), TROP-2-positive CC could emerge as a promising area for future research.51,52 To date, sacituzumab govitecan is under investigation in a non-randomized phase II trial enrolling patients with recurrent or persistent CC who have progressed following at least one prior CT treatment regimen (ClinicalTrials.gov identifier: NCT05838521). Additionally, the ENGOT-cx20/MK-2870-020 (NCT trial is scheduled to start in June 2024: it is a phase III randomized, active-controlled, open-label, multicenter study designed to compare the efficacy and safety of MK-2870 monotherapy (an anti-TROP-2 ADC with a belotecan-derived payload) versus treatment of physician’s choice as a second-line or third-line treatment for participants with recurrent or metastatic CC (ClinicalTrials.gov identifier: NCT06459180). The main characteristics of anti-TF, anti-HER2, and anti-TROP-2 ADCs are summarized in Figure 2.

Antibody drug conjugates in cervical cancer: mechanism of action and current drugs under investigation. (a) The binding between antibodies and cervical cancer cells’ receptors is highly specific. In detail in a clockwise direction: trastuzumab recognizes ERBB2, tisotumab and sacituzumab recognize the corresponding antigens (TF and TROP-2) on cell’s surface. (b) On the left ADC mechanism of action and anti-TF, anti-HER2, anti-TROP-2 specific components.
Conclusion
TV is the first ADC approved in advanced/recurrent CC. The drug demonstrated a significant increase in OS and PFS with respect to standard CT with a manageable toxicity profile. The era of ADC in CC and gynecologic malignancies is just started, but several issues need to be solved.
Enhancing the selection of patients eligible for ADCs through biomarker-driven approaches is essential to maximize treatment benefits while ensuring an acceptable toxicity profile. Determining the optimal timing for ADCs treatment in CC and the optimal schedule, whether alone or in combination with CT or immunotherapy, necessitates a careful consideration of (1) tolerability; (2) biological changes that could influence subsequent treatments; and (3) treatment costs. Conducting translational studies is crucial to enhance the effectiveness and positioning of these drugs and to better evaluate their mechanism of resistance. In general, promoting combinations is essential to reduce the selection of resistant clones and enhance overall efficacy. However, toxicity profile and financial toxicity may represent a major issue and patients’ reported outcomes, and pharmacoeconomical evaluations should be incorporated in final evaluations.
