Abstract
Background:
Immunotherapy combined with chemotherapy is the first-line treatment for extensive-stage small cell lung cancer (ES-SCLC). However, the effect of delayed initiation of immunotherapy on its efficacy remains unclear.
Objectives:
This study aimed to investigate the impact of the delayed addition of programmed death receptor 1/programmed death ligand 1 (PD-1/PD-L1) inhibitors on treatment outcomes in patients with ES-SCLC.
Design:
This retrospective cohort study used propensity score matching (PSM) to balance baseline characteristics.
Methods:
This study included 416 patients with ES-SCLC who received first-line immunotherapy between January 2020 and December 2022. Patients were categorized into two groups: delayed-immunotherapy (IO) (PD-1/PD-L1 inhibitors initiated during the 2nd or 3rd chemotherapy cycle) and early-IO (immunotherapy initiated during the first cycle). A 1:1 PSM was performed to balance baseline characteristics. The primary endpoints were overall survival (OS) and progression-free survival (PFS), which were analyzed using the Kaplan-Meier method and compared using log-rank tests.
Results:
Owing to the exclusion of PD-L1/PD-1 inhibitors from medical insurance, financial constraints, and poor physical condition of some patients, 72 patients were included in the delayed-IO group (second cycle: 41; third cycle: 31), while 344 were in the early-IO group. Before PSM, median OS and PFS in the delayed-IO group were 24.00 and 8.75 months, compared to 18.59 and 7.57 months in the early-IO group, with no significant differences (OS: HR 0.72, p = 0.054; PFS: HR 0.86, p = 0.281). After PSM (72 patients per group), the delayed-IO group showed significantly longer median OS (24.00 vs 18.79 months, HR 0.60, 95% CI 0.38–0.97, p = 0.037) and PFS (8.75 vs 6.49 months, HR 0.69, 95% CI 0.48–0.99, p = 0.044) compared to the early-IO group. These results suggest that, in specific patient populations, delayed immunotherapy may improve survival outcomes by optimizing patient condition or treatment response.
Conclusion:
In patients with ES-SCLC, delayed administration of PD-1/PD-L1 inhibitors during the second to fourth chemotherapy cycles improves survival outcomes compared to concurrent administration during the first cycle, with a similar safety profile. These results suggest that, in specific patient populations, delayed immunotherapy may improve survival outcomes by optimizing patient condition or treatment response.
Plain language summary
This study looks at the best time to start immunotherapy for small-cell lung cancer, which is a very aggressive type of lung cancer often found in its advanced stage. The standard treatment now is to combine immunotherapy with chemotherapy. However, it’s unclear if when you start immunotherapy affects how well it works. The study included 416 patients divided into two groups: one group started immunotherapy during the 2nd or 3rd cycle of chemotherapy (delayed immunotherapy group), and the other group started it with the first cycle of chemotherapy (early immunotherapy group). After matching patients’ basic characteristics, the delayed group had longer survival (median overall survival of 24 months) compared to the early group (median overall survival of 18.8 months). The delayed group also had longer progression-free survival (median 8.75 months) compared to the early group (median 6.49 months). This suggests that starting immunotherapy later, during the 2nd to 4th chemotherapy cycles, may be more effective than starting it with the first cycle. Importantly, delayed immunotherapy did not increase safety concerns. This finding offers new insights for treating small-cell lung cancer, especially for patients who might face financial or health-related reasons for not starting immunotherapy immediately.
Keywords
Introduction
Small cell lung cancer (SCLC), a distinct subtype of lung cancers, represents approximately 10%–15% of pulmonary malignancies and exhibits extraordinary biological aggressiveness, marked by rapid disease evolution and early metastatic dissemination. 1 The rapid disease progression pattern frequently culminates in late-stage diagnoses, with over two-thirds of patients presenting with extensive-stage disease (ES-SCLC) at the initial evaluation, indicating dismal clinical outcomes. 2 Therapeutic paradigms have long been anchored on cytotoxic regimens, particularly platinum-containing protocols that integrate cisplatin/carboplatin with etoposide.1,2 Notwithstanding the notable initial tumor regression observed in 60%–80% of cases, therapeutic durability remains suboptimal, as evidenced by frequent relapse patterns and a stagnant 5-year survival plateau at 7%. 3 For ES-SCLC cohorts, the historical survival benchmark under conventional chemotherapy hovers between 8 and 10 months.4,5
The immunotherapeutic era has fundamentally reshaped ES-SCLC management, with programmed death receptor 1/programmed death ligand 1 (PD-1/PD-L1) axis inhibitors demonstrating paradigm-shifting efficacy. Pivotal phase III investigations, including IMpower133 and CASPIAN, have established the survival superiority of chemoimmunotherapy combinations over traditional chemotherapy alone, achieving statistically significant enhancements in both the overall survival (OS) and progression-free survival (PFS) endpoints.6,7 Subsequent confirmatory trials have expanded this evidence base: the CAPSTONE-1 study documented a 2.5-month OS advantage with adebrelizumab-chemotherapy synergy (15.3 vs 12.8 months; HR 0.72, 95% CI 0.58–0.90; p = 0.0017), 8 while ASTRUM-005 revealed unprecedented survival extension with serplulimab (median OS 15.4 vs 13.3 months; HR 0.63, 95% CI 0.49–0.82; p < 0.001). 9 Emerging data from Chinese cohorts further validate this therapeutic advance, with tislelizumab and toripalimab demonstrating comparable clinical benefits.10,11
Critical uncertainties persist regarding the temporal optimization of immunotherapy integration. While concurrent chemoimmunotherapy demonstrates mechanistic synergy through enhanced antigen exposure and T-cell activation,6 –11 emerging preclinical models suggest that sequential administration may exploit chemotherapy-induced immunogenic conditioning effects.12,13
Cytotoxic agents potentially reconfigure tumor immunogenicity via multiple pathways, including the induction of immunogenic cell death phenotypes, dendritic cell maturation potentiation, and depletion of regulatory T-cell populations.14 –17 These microenvironmental modifications theoretically establish an immunological “primed state” conducive to enhanced Immune Checkpoint Inhibitor (ICI) responsiveness.16,17
The clinical translation of these biological insights remains contentious, particularly given the rapid progression kinetics and the predilection of ES-SCLC for cerebral metastases. Our retrospective cohort analysis employed a propensity score matching (PSM) methodology to investigate the temporal immunotherapy sequencing effects. Through rigorous adjustment for baseline prognostic variables, we evaluated the hypothesis that deferred immunotherapy initiation confers survival advantages to clinically defined patient subsets. This study aimed to elucidate chronotherapeutic optimization strategies for immune checkpoint modulation in ES-SCLC management.
Materials and methods
We used the STROBE cohort reporting guidelines. 18
Ethics approval and consent to participate
This retrospective observational study was conducted in accordance with the principles of the Declaration of Helsinki and the 2008 Declaration of Istanbul. Approval was obtained from the Ethics Committee for Clinical Research at Shandong Cancer Hospital and Institute (Approval No. SDTHEC202502020). Given the retrospective nature of the study, the requirement for informed consent was waived.
Data collection
We conducted a review of the medical records of patients diagnosed with ES-SCLC at Shandong Cancer Hospital between January 2020 and December 2022. Patients were included if they met the following criteria: (1) A confirmed diagnosis of ES-SCLC through pathological or radiological methods; (2) Presence of at least one measurable lesion; (3) Availability of imaging reports both before and after immunotherapy; (4) Completion of at least two cycles of standard therapy; and (5) A Karnofsky performance status (KPS) score of 70 or higher. Patients were excluded if they had a KPS score below 70, a history of autoimmune diseases, secondary primary tumors, or disease progression prior to initiating immunotherapy. Data collected included tumor/lymph node/metastasis staging, presence of brain/liver/other metastases, and treatment outcomes. Tumor staging was based on the eighth edition of the American Joint Committee on Cancer (AJCC) Cancer Staging System. The study adhered to the principles of the Declaration of Helsinki and the International Good Clinical Practice Guidelines.
Treatment protocols
Patients received at least one dose of first-line therapy, which consisted of EP (etoposide combined with carboplatin or cisplatin, depending on the investigator’s choice) chemotherapy along with either a PD-L1 inhibitor (atezolizumab or durvalumab) or a PD-1 inhibitor (serplulimab). All medications were administered intravenously every 21 days at the following doses:
Etoposide: 100 mg/m2 on days 1–3;
Carboplatin: Area under the curve of 5 mg/mL/min, or cisplatin: 25–30 mg/m2 on days 1–3;
Atezolizumab: 1200 mg on day 1;
Durvalumab: 1500 mg on day 1;
Serplulimab: 4.5 mg/kg on day 1 of each cycle.
Patients with brain metastases at diagnosis or during treatment were considered for brain radiotherapy based on their cranial metastatic symptoms. For those in remission or with stable disease after initial systemic treatment, chest radiotherapy was administered to the primary tumor and mediastinal lymph node metastases to enhance local control. Other metastatic sites were treated locally, depending on the patient’s clinical condition. Some patients received more than six chemotherapy cycles owing to an observed partial response or stable disease, based on clinical judgment.
Clinical response evaluation
Patients were followed up until the last recorded date (September 11, 2023) or until death. Tumor status was assessed every two cycles during the initial four to six cycles of treatment, every 2 months thereafter during the treatment period, and every 3–6 months subsequently, using pre-treatment imaging as the baseline. Tumor response was evaluated using the Response Evaluation Criteria in Solid Tumors (RECIST), version 1.0. Adverse events (AEs) were monitored throughout the immunotherapy period and graded according to the National Cancer Institute Common Terminology Criteria for AEs (version 5.0). AEs were also assessed within 30 days after completing immunotherapy.
Endpoints
The primary endpoints were PFS and OS. PFS was defined as the time from diagnosis to disease progression in the chest or distant lesions, death from any cause, or the last known follow-up. OS was defined as the time from diagnosis to death from any cause or the last known follow-up. Secondary endpoints included AEs and survival analyses across various subgroups.
Statistical analysis
Differences in baseline characteristics between subgroups were analyzed using the χ2 test and Fisher’s exact test. PFS and OS were estimated using the Kaplan-Meier method, and survival curves were compared between groups using the log-rank test. Statistical significance was set at p < 0.05. Univariate analysis was performed using the Cox proportional hazards model to identify potential prognostic predictors. Variables with significant correlations or trends (p < 0.10) were included in the multivariate model to determine the relationship between OS and clinical characteristics. All p-values were two-sided, with 95% confidence intervals reported. Statistical analyses were conducted using SPSS version 27.0 (IBM Corporation, Armonk, NY, USA) and Prism version 8.0.2 (GraphPad Software, San Diego, CA, USA).
Results
Patient characteristics
A total of 711 patients were enrolled in the study, of whom 416 received a treatment regimen combining immunotherapy and chemotherapy. The screening flowchart is presented in Figure 1. Patient demographics and treatment data, both before and after PSM, are summarized in Table 1.
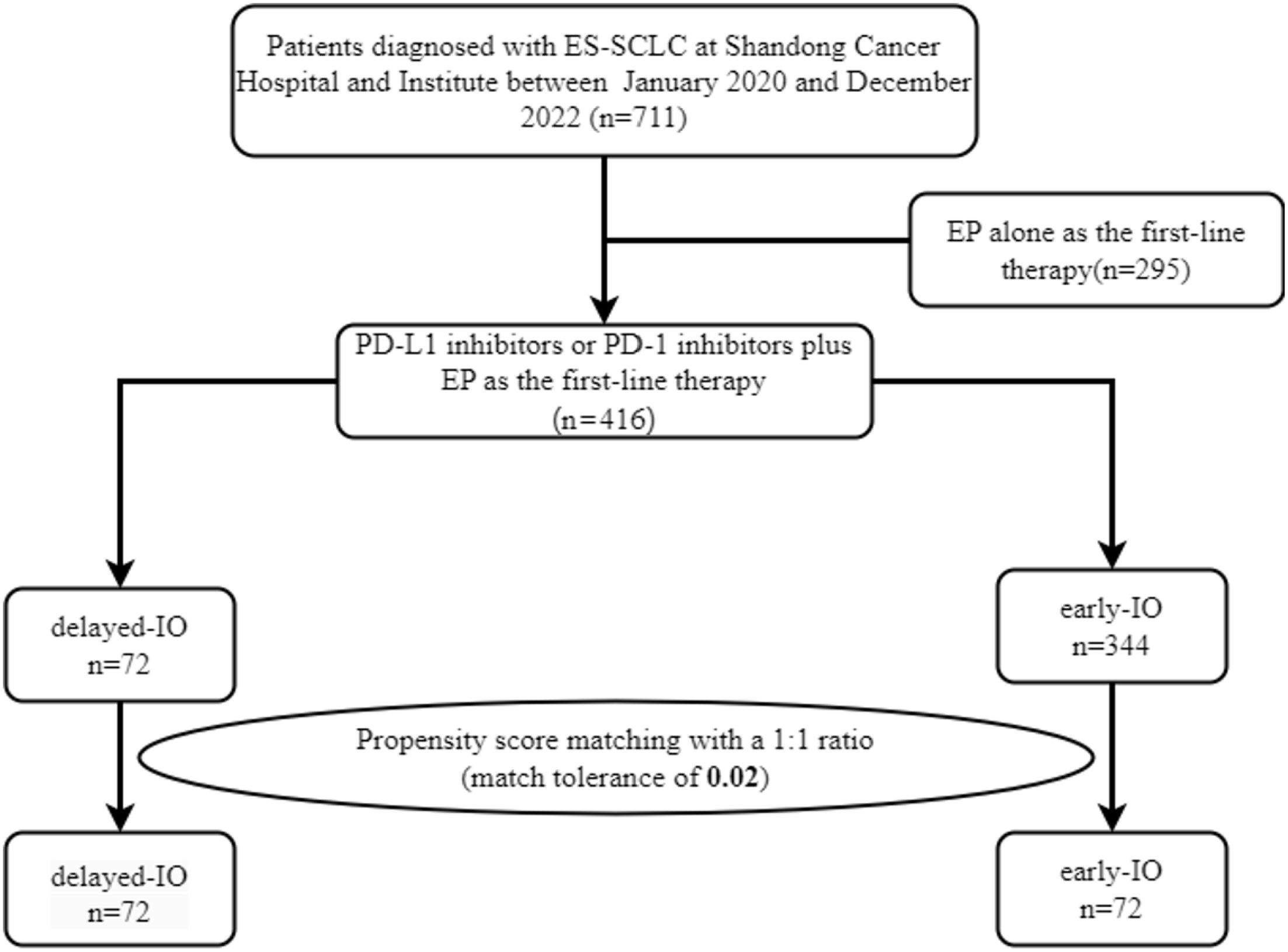
Flowchart of the screening procedure.
Baseline characteristics of all study patients.
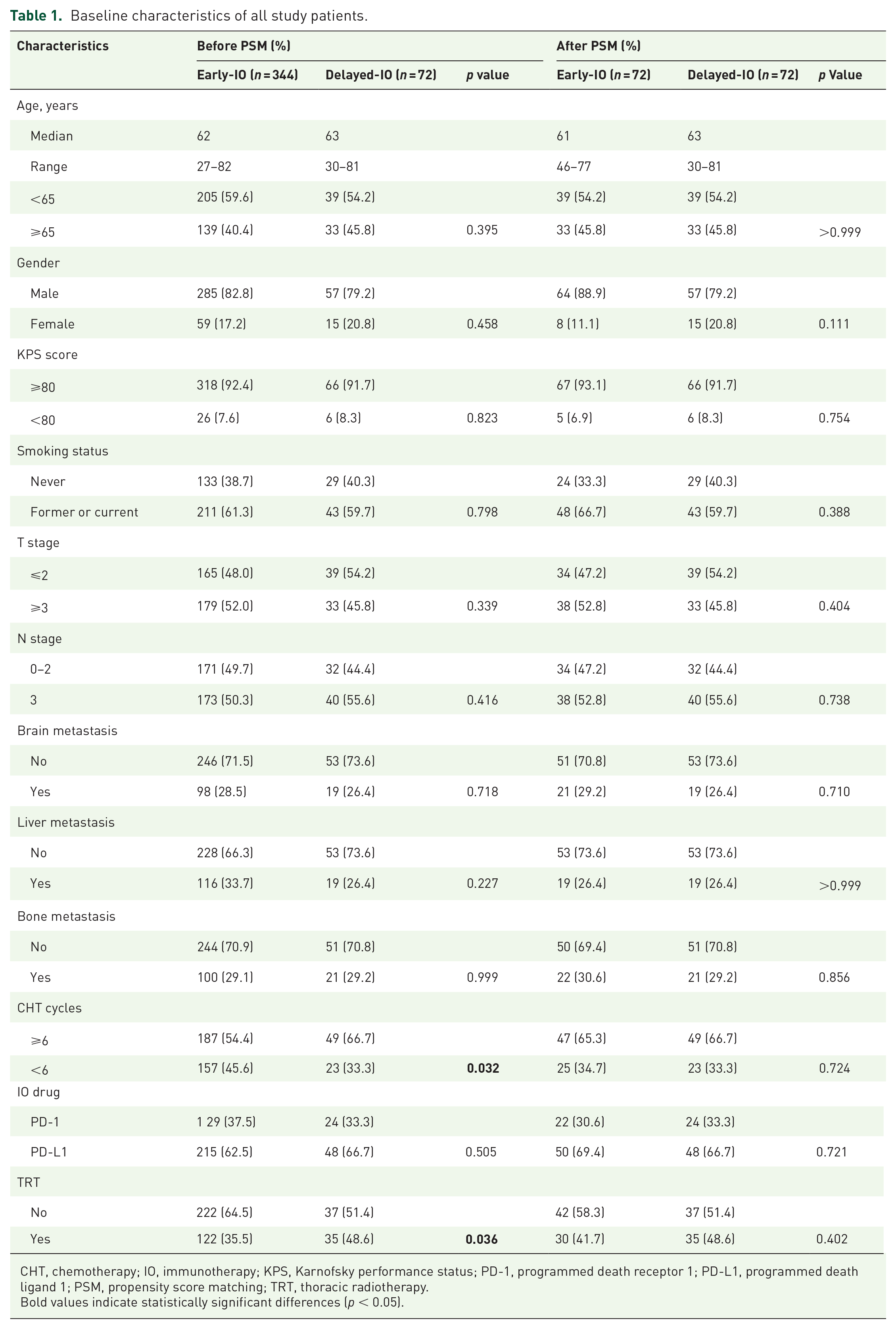
CHT, chemotherapy; IO, immunotherapy; KPS, Karnofsky performance status; PD-1, programmed death receptor 1; PD-L1, programmed death ligand 1; PSM, propensity score matching; TRT, thoracic radiotherapy.
Bold values indicate statistically significant differences (p < 0.05).
Before matching, the delayed-initiation immunotherapy group (delayed-immunotherapy (IO)) included 72 patients, while the early initiation immunotherapy group (early-IO) included 344 patients. Reasons for delayed initiation of immunotherapy included:
Insurance-related issues: (55%),
Poor health status or low treatment tolerance (25%),
High tumor burden, leading patients to opt out of immunotherapy after family discussions (20%).
After PSM, 72 patients were included in both groups. Before PSM adjustment, brain metastases were observed in 98 patients (28.5%) in the early-IO group and 19 patients (26.4%) in the delayed-IO group, with no statistically significant difference (p = 0.718). After PSM matching, brain metastases were present in 21 (29.2%) and 19 patients (26.4%), respectively, with no significant difference between the two groups (p = 0.710).
Before matching, 187 patients (54.4%) in the early-IO group and 49 patients (66.7%) in the delayed-IO group completed six or more cycles of chemotherapy, with a statistically significant difference (p = 0.032). After matching, 47 patients (65.3%) in the early-IO group and 49 patients (66.7%) in the delayed-IO group completed six or more cycles, with the difference no longer significant (p = 0.724).
Before matching, PD-1 inhibitors were administered to 129 patients (37.5%) in the early-IO group and 24 patients (33.3%) in the delayed-IO group (p = 0.505).
After adjustment for matching, PD-1 inhibitors were administered to 22 patients (30.6%) in the early-IO group and 24 patients (33.3%) in the delayed-IO group (p = 0.721).
Before matching, thoracic radiotherapy (TRT) was administered to 122 patients (35.5%) in the early-IO group and 35 patients (48.6%) in the delayed-IO group, with a statistically significant difference (p = 0.036). After PSM, TRT was administered to 30 patients (41.7%) in the early-IO group and 35 patients (48.6%) in the delayed-IO group, with the difference no longer significant (p = 0.402).
Survival outcomes
The final follow-up was completed on June 10, 2024, with a median duration of 26 months. Before PSM: PFS was comparable between the delayed-IO and early-IO groups, with medians of 8.75 and 7.57 months, respectively (HR 0.86, 95% CI 0.66–1.13; p = 0.28, Figure 2(a)). OS showed a trend favoring the delayed-IO group, though the difference was not statistically significant (24.00 vs 18.59 months, HR 0.72, 95% CI 0.51–1.01; p = 0.054, Figure 2(b)). After PSM, patients in the delayed-IO group exhibited significant improvements in both PFS (8.75 vs 6.49 months, HR 0.69, 95% CI 0.48–0.99; p = 0.045, Figure 2(c)) and OS (24.00 vs 18.79 months, HR 0.60, 95% CI 0.38–0.97; p = 0.037, Figure 2(d)) compared to those in the early-IO group.

In the overall population, OS and PFS before and after PSM. (a) PFS prior to PSM; (b) OS prior to PSM; (c) PFS following PSM; (d) OS following PSM.
Tumor response evaluation after two and four cycles
In the matched cohort of 144 patients, tumor responses were evaluated in the early-IO and delayed-IO groups after two and four treatment cycles, with the results shown in Figure 3.

Tumor response evaluation after two and four cycles of chemotherapy in the early-IO and delayed-IO groups. (a) early-IO after two cycles; (b) early-IO after four cycles; (c) delayed-IO after two cycles; (d) delayed-IO after four cycles. Tumor responses are categorized as complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD).
In the early-IO group, partial response (PR) was predominant after two cycles, observed in 59% of patients, with 38% showing stable disease (SD). Two patients achieved complete response (CR). No cases of progressive disease (PD) were reported (Figure 3(a)). After four cycles, the CR rate increased to four patients, with a slight increase in the PR rate (60%). Four patients experienced disease progression (Figure 3(b)).
In the delayed-IO group, no CR cases were observed after two cycles, and PR was reported in approximately 45%, which was slightly lower than that in the early-IO group. The remaining patients exhibited SD, with no PD cases (Figure 3(c)). After four cycles, the therapeutic efficacy in this group improved significantly, with four patients achieving CR and most patients showing PR. Two patients experienced disease progression (Figure 3(d)). After more treatment cycles, the delayed-IO group demonstrated superior therapeutic outcomes.
Subgroup analysis
To further clarify the survival benefits between the two groups, we performed a subgroup analysis of PFS and OS in the matched cohort.
For PFS, patients with T stage ⩾3 exhibited a notable benefit from delayed immunotherapy (9.18 vs 6.52 months; HR 0.59, 95% CI 0.35–0.99). Similarly, patients without liver metastasis demonstrated improved PFS in the delayed-IO group (8.95 vs 6.59 months; HR 0.64, 95% CI 0.41–0.99) (Figure 4(a)).

Subgroup analysis of PFS (a) and OS (b) after PSM.
Regarding OS, patients with a KPS ⩾80 experienced significantly longer OS in the delayed-IO group (24.39 vs 18.79 months; HR 0.55, 95% CI 0.33–0.91). In addition, patients with no brain metastases (24.10 vs 18.23 months; HR 0.54, 95% CI 0.31–0.94) and no liver metastases (24.00 vs 20.36 months; HR 0.59, 95% CI 0.33–1.06) in the delayed-IO group was longer than those of the early-IO group (Figure 4(b)).
Brain metastasis population
To evaluate the survival benefits among patients with brain metastases while controlling for confounding factors, we conducted a prognostic analysis on the 144 matched patients.
Among the 40 patients with baseline brain metastases, delayed immunotherapy did not improve PFS (median PFS: 8.75 vs 6.49 months; HR 0.65, 95% CI 0.33–1.26; p = 0.19, Figure 5(a)) nor extend median survival (median OS: 22.13 vs 18.33 months; HR 0.91, 95% CI 0.38–2.18; p = 0.82, Figure 5(b)). However, in the 104 patients without baseline brain metastases, delaying immunotherapy by 2–4 cycles led to more favorable outcomes (median PFS: 8.95 vs 6.95 months; HR 0.71, 95% CI 0.46–1.10; p = 0.13, Figure 5(c); median OS: 24.12 vs 18.23 months; HR 0.56, 95% CI 0.32–0.97; p = 0.03, Figure 5(d)).

In the populations with and without brain metastases, OS and PFS after PSM. (a) PFS following PSM in the population with brain metastases; (b) OS following PSM in the population with brain metastases; (c) PFS following PSM in the population without brain metastases; (d) OS following PSM in the population without brain metastases.
Subgroup analysis by immunotherapy drug
In the PSM cohort (n = 144), we conducted a subgroup analysis stratified by immunotherapy drugs (atezolizumab, durvalumab, and serplulimab). The baseline characteristics of the drug subgroups are summarized in
Regarding survival outcomes, only the serplulimab subgroup demonstrated a significantly better PFS in the delayed-IO group compared to the early-IO group (13.18 months, 95% CI 7.89–18.47 vs 6.26 months, 95% CI 4.76–7.77, p = 0.0089, Figure 6(e)). No statistically significant differences were observed in PFS or OS between the other subgroups. Specifically, in the atezolizumab subgroup, PFS was 8.95 months (95% CI 6.28–11.62) in the delayed-IO group and 7.05 months (95% CI 4.396–9.14) in the early-IO group, with OS of 22.42 months (95% CI 19.81–25.05) and 18.78 months (95% CI 12.26–25.32, Figure 6(a) and (b)), respectively. In the durvalumab subgroup, PFS was 7.44 months (95% CI 6.52–8.37) in the delayed-IO group and 6.39 months (95% CI 5.45–7.34) in the early-IO group, with OS of 18.42 months (95% CI 8.01–28.84) and 17.93 months (95% CI 11.15–24.72, Figure 6(c) and (d)), respectively. For serplulimab, OS was 26.36 months (95% CI 18.96–33.76) in the delayed-IO group, while the early-IO group did not reach the median OS (Figure 6(f)).
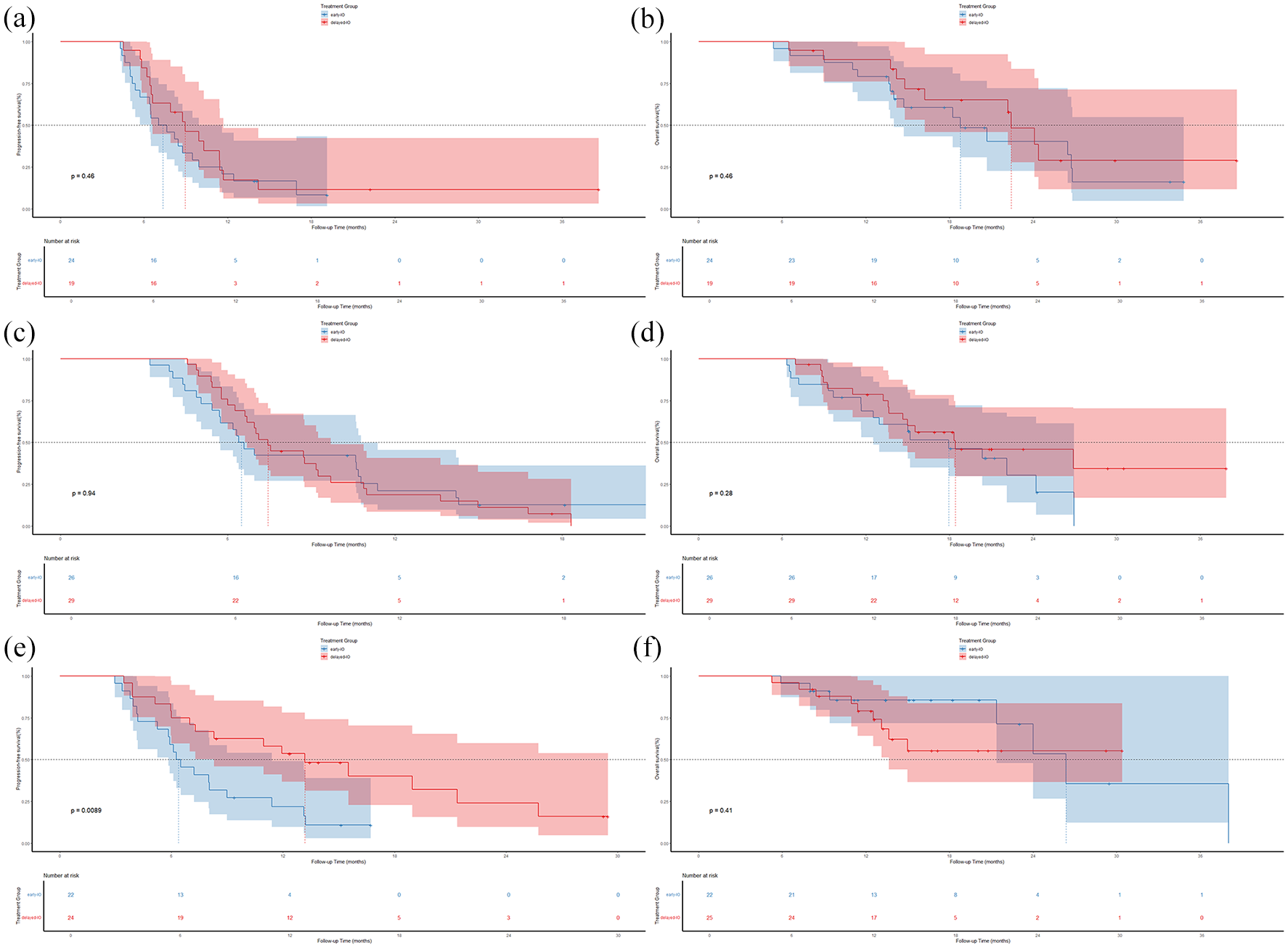
Subgroup survival analysis by immunotherapy drug (post-PSM). (a) PFS of atezolizumab in early-IO versus delayed-IO groups; (b) OS of atezolizumab in early-IO versus delayed-IO groups; (c) PFS of durvalumab in early-IO versus delayed-IO groups; (d) OS of durvalumab in early-IO versus delayed-IO groups; (e) PFS of serplulimab in early-IO versus delayed-IO groups; (f) OS of serplulimab in early-IO versus delayed-IO groups.
Prognostic factors
Univariate and multivariate analyses were conducted using the Cox proportional hazards model for matched populations. Univariate analysis indicated that age <65 years, KPS ⩾80, earlier T and N stages, absence of bone metastases, and delayed immunotherapy were favorable prognostic factors for OS. Multivariate analysis confirmed that age <65 years (HR 0.583, 95% CI 0.361–0.941; p = 0.027) and KPS ⩾80 (HR 0.442, 95% CI 0.219–0.892; p = 0.023) were significant clinical factors for improving OS. In contrast, early immunotherapy (HR 1.759, 95% CI 1.087–2.849; p = 0.022) was a disadvantageous factor for OS (Table 2). For PFS, univariate analysis revealed that patients with KPS ⩾80 and those without bone metastases tended to have longer PFS. Multivariate analysis identified early immunotherapy (HR 1.431, 95% CI 1.002–2.054; p = 0.049) as a significant disadvantageous prognostic factor for PFS (Table 3).
Univariate and multivariate analyses of factors influencing overall survival in patients after propensity score matching.

CHT, chemotherapy; IO, immunotherapy; KPS, Karnofsky performance status; PD-1, programmed death receptor 1; PD-L1, programmed death ligand 1; TRT, thoracic radiotherapy.
Bold values indicate statistically significant differences (p < 0.05).
Univariate and multivariate analyses of factors influencing progression-free survival in patients after propensity score matching.

CHT, chemotherapy; IO, immunotherapy; KPS, Karnofsky performance status; PD-1, programmed death receptor 1; PD-L1, programmed death ligand 1; TRT, thoracic radiotherapy.
Bold values indicate statistically significant differences (p < 0.05).
Adverse events
The main treatment-related AEs are shown in Table 4. Hematological toxicity was the most common AE, followed by neutropenia (overall: 77.8%; delayed-IO: 83.3%; early-IO: 86.1%) and lymphopenia (overall: 75.7%; delayed-IO: 70.8%; early-IO: 80.6%). Among grade ⩾3 hematologic toxicities, neutropenia was notably severe, occurring in 39.6% of all patients, with a lower incidence in the delayed-IO group (34.7%) compared to the early-IO group (44.4%). Anemia was another common AE but was generally mild, affecting 86.1% of patients, with only 6.3% experiencing grade ⩾3 anemia. Thrombocytopenia and leukopenia were less frequent, with similar incidences between the two groups.
Treatment-related adverse events in patients after propensity score matching.
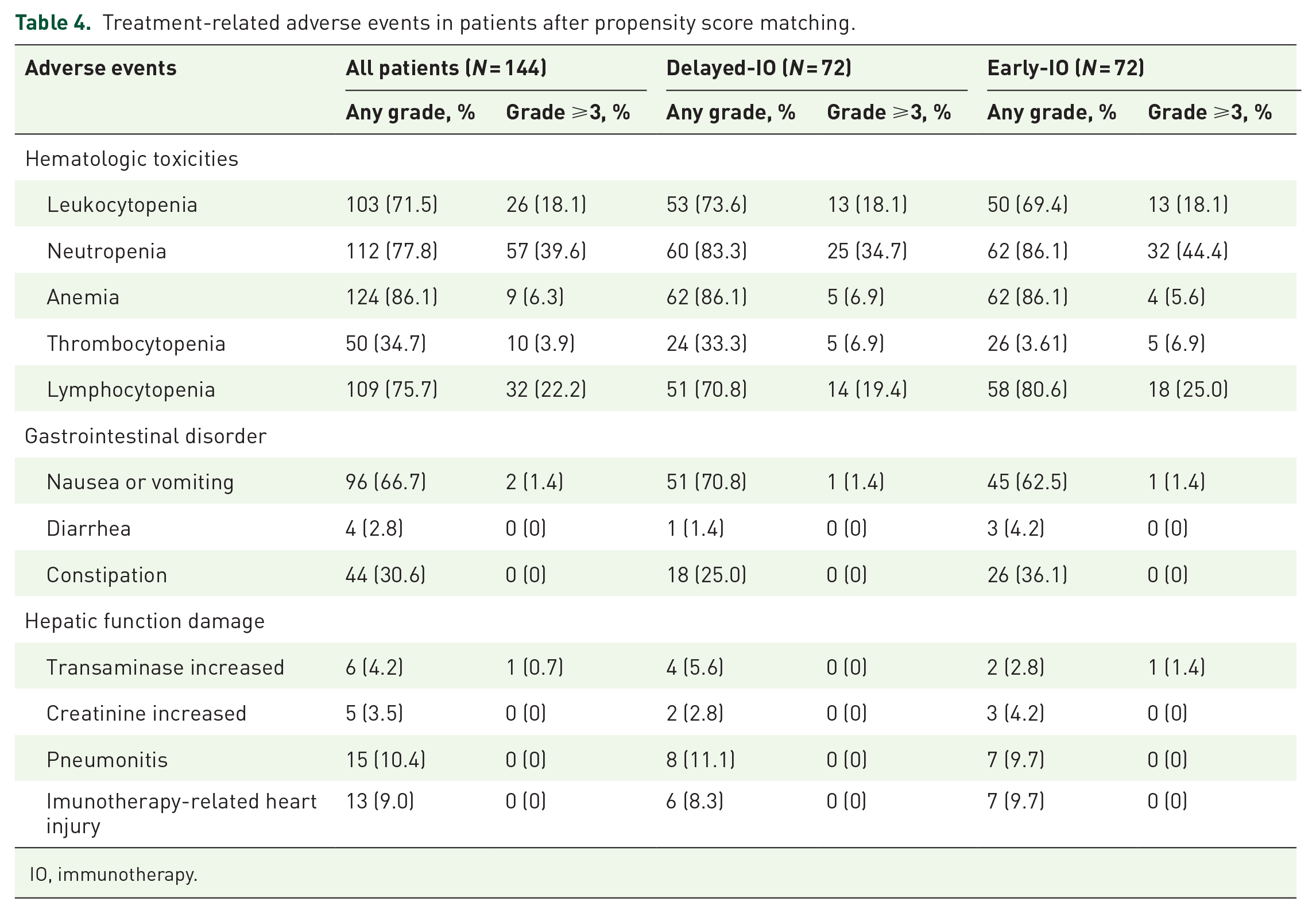
IO, immunotherapy.
Gastrointestinal disturbances were less common, with nausea or vomiting being the most frequent (overall incidence 66.7%; delayed-IO: 70.8%; early-IO: 62.5%), but severe events (grade ⩾3) were rare. Hepatic dysfunction and pneumonitis were uncommon, with no significant differences between the two groups. Notably, there were no cases of grade ⩾3 pneumonitis or immune-related cardiac toxicity in either group.
Discussion
This study is the first large-scale retrospective analysis to examine the optimal sequencing of immunotherapy and chemotherapy in patients with ES-SCLC. A total of 416 patients were included, with 72 patients delaying the initiation of immunotherapy due to concerns over AEs and insurance coverage. Most delays occurred after two cycles of chemotherapy. Statistically significant improvements in both PFS and OS were observed in the delayed-IO group after PSM. Given the historically poor prognosis of SCLC and the limited advances in survival outcomes with conventional therapies, these findings are noteworthy.
Our findings are consistent with those of previous phase III trials, such as IMpower133, CASPIAN, ASTRUM-005, and CAPSTONE-1, which have demonstrated the survival benefit of adding immunotherapy to standard treatment regimens for patients with ES-SCLC.6 –9 However, in contrast to the median OS of approximately 12–15 months reported in these clinical trials, our retrospective analysis suggests even greater survival benefits with immunotherapy, likely because of the availability of more extensive treatment options in real-world settings, as previously reported.19,20 Additionally, the early initiation of TRT, administered to approximately 45% of the patients in our study, may have further contributed to the extended OS. 21 Pre-PSM, the higher TRT proportion in the delayed-IO group (48.6% vs 35.5%, p = 0.036) may have influenced survival outcomes, though no significant OS difference was observed (p > 0.05). Post-PSM, this difference was no longer significant (41.7% vs 48.6%, p = 0.402), indicating minimal confounding factors, whereas the significant OS difference underscored the importance of immunotherapy timing. The literature suggests that TRT enhances the synergistic effects of immunotherapy, improving local control and survival through immune activation. 22 The optimal timing for immunotherapy initiation remains inconclusive. Our data indicates that delayed immunotherapy is associated with longer PFS and OS in patients with favorable prognostic factors, such as younger age, higher KPS scores, and absence of brain or liver metastases. A trend toward improved survival with delayed ICIs was observed in patients without liver metastases (OS: 24.00 vs 20.36 months, HR 0.59, 95% CI 0.33–1.06, p = 0.06), possibly due to less aggressive disease biology, as liver involvement often correlates with higher tumor burden and worse prognosis. 23 In addition, PSM balanced baseline characteristics, and a high KPS (⩾70, 91.7–93.1% of patients) likely mitigated the prognostic impact of liver metastases. Conversely, no significant survival benefit was observed in patients with brain metastases (OS: 22.13 vs 18.33 months, HR 0.91, 95% CI 0.38–2.19, p = 0.82), likely due to rapid intracranial progression requiring early concurrent chemotherapy and immunotherapy for effective control. 24 The high incidence of brain metastases (50%–80% during disease progression) and radiotherapy interventions (TRT: early-IO 41.7%, delayed-IO 48.6%) may have normalized their prognostic impact across the cohort. Previous studies identified extra-thoracic metastases (e.g., liver, bone, and adrenal) as significant OS predictors in concurrent chemoimmunotherapy,24,25 though brain metastases do not necessarily indicate poor prognosis.19,26 One possible explanation for the observed benefits of delayed immunotherapy is the immunomodulatory effects of chemotherapy on the tumor microenvironment. Chemotherapy can induce immunogenic cell death, enhance antigen presentation, and reduce tumor burden, creating a more favorable setting for subsequent immune checkpoint blockade.15,16,27 Recent studies in non-small cell lung cancer (NSCLC) have shown that concurrent administration of PD-1 inhibitors with chemotherapy may impair CD8+ T-cell activation, limit expansion, and reduce IFN-γ production, leading to suboptimal outcomes compared with PD-L1 inhibitors alone. Sequential administration, where chemotherapy precedes immunotherapy, may better preserve immune function and represents a potentially more effective treatment strategy. 28 In our study, early immunotherapy demonstrated a higher PR rate after two cycles of treatment. However, delaying the administration of CIs until after two to three cycles of chemotherapy may offer potential advantages, including a lower risk of disease progression. These findings align with observations in metastatic triple-negative breast cancer, in which the highest objective response rates to PD-1 blockade were observed following anthracycline-based induction therapy. 29
A real-world retrospective study of 64 patients with advanced lung cancer treated with chemotherapy combined with PD-1/PD-L1 inhibitors evaluated the timing of immunotherapy administration (e.g., 3–5 days post-chemotherapy). The best outcomes were observed when PD-1 inhibitors were administered 3–5 days after chemotherapy. 30 Although this study focused on later-line treatments, it provided valuable insights that support our findings. Similarly, a prospective randomized trial of resectable esophageal cancer reported significant differences in pathologic complete response (pCR) rates based on immunotherapy timing, with pCR rates of 7.6% when immunotherapy was administered on the same day as chemotherapy and 30.4% when administered 48 h later. 31 In the PACIFIC trial, subgroup analysis showed better outcomes when anti-PD-L1 therapy was administered within 2 weeks of concurrent chemoradiotherapy. 32
Multivariate analysis highlighted the importance of patient selection. Younger patients, those with higher KPS scores, and those without brain metastases were more likely to experience improved OS with delayed immunotherapy. In the PSM cohort (n = 144), only serplulimab showed significantly better PFS in the delayed-IO group (13.18 vs 6.26 months, p = 0.0089), with no significant differences in PFS for atezolizumab and durvalumab, or in OS across subgroups. Serplulimab’s higher PD-1 binding affinity may enhance its antitumor effect in the delayed-IO setting, 9 potentially amplified by prolonged chemotherapy induction, reshaping the immune microenvironment. 7 The lack of significant differences in other subgroups may reflect variations in drug mechanisms, patient characteristics (e.g., metastasis status), and treatment heterogeneity. 19 The TRT proportion difference in the durvalumab subgroup (p = 0.073) may further complicate its survival outcomes. 21 Regarding AEs, the overall safety profile was consistent with previous reports.6 –11 Hematological toxicities, particularly neutropenia and lymphopenia, were the most common AEs, with a slightly lower incidence of severe neutropenia in the delayed immunotherapy group. Gastrointestinal AEs, such as nausea and vomiting, were also frequent but generally mild. Importantly, there was no significant difference in the incidence of severe AEs, including immune-related pneumonitis and cardiac toxicity, between the early and delayed immunotherapy groups. This suggests that the timing of immunotherapy initiation does not significantly affect the safety profile.
This study has limitations. As a retrospective analysis from a single institution, it may be subject to selection bias, confounding, and limited generalizability. Excluding patients who progressed before ICIs may introduce selection bias, favoring those with SD; future studies should include these patients in sensitivity analyses. The small sample of brain metastasis patients (n = 40, early-IO 21, delayed-IO 19) restricted stratification by metastasis number [26]; future research should collect such data via standardized imaging (e.g., MRI/CT (Computed Tomography) and expand sample sizes (recommended n > 100) for detailed subgroup analyses. Despite these limitations, PSM mitigated baseline imbalances, providing preliminary evidence for delayed immunotherapy’s survival benefits in ES-SCLC. Prospective trials are needed to validate optimal sequencing and explore underlying mechanisms and biomarkers for personalized treatment strategies.
Conclusion
Our findings suggest that administering PD-1/PD-L1 inhibitors during the 2nd or 3rd cycle of chemotherapy may offer superior survival benefits compared to concurrent initiation of immunotherapy with the first cycle of chemotherapy in patients with ES-SCLC, while maintaining a similar safety profile. Notably, delayed immunotherapy was associated with improved survival outcomes in patients without baseline brain metastases. Conversely, in patients with baseline brain metastases, early initiation of immunotherapy appeared to offer a more significant survival benefit. These results highlight the potential for tailoring immunotherapy timing based on the presence of brain metastases and provide valuable guidance for optimizing treatment strategies in ES-SCLC. Further prospective studies are warranted to validate these findings and refine patient stratification.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251356919 – Supplemental material for Impact of delayed addition of PD-1/PD-L1 inhibitors to chemotherapy on outcomes in patients with extensive-stage small cell lung cancer
Supplemental material, sj-docx-1-tam-10.1177_17588359251356919 for Impact of delayed addition of PD-1/PD-L1 inhibitors to chemotherapy on outcomes in patients with extensive-stage small cell lung cancer by Shuangqing Lu, Chao Wang, Xiaoyang Zhai, Zhuoran Sun, Ke Zhao, Dawei Chen and Hui Zhu in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251356919 – Supplemental material for Impact of delayed addition of PD-1/PD-L1 inhibitors to chemotherapy on outcomes in patients with extensive-stage small cell lung cancer
Supplemental material, sj-docx-2-tam-10.1177_17588359251356919 for Impact of delayed addition of PD-1/PD-L1 inhibitors to chemotherapy on outcomes in patients with extensive-stage small cell lung cancer by Shuangqing Lu, Chao Wang, Xiaoyang Zhai, Zhuoran Sun, Ke Zhao, Dawei Chen and Hui Zhu in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
Part of this study was presented as a poster at the 2024 ASCO Breakthrough Summit (Poster Identifier: ![]() ). We appreciate the recognition extended by the Organizing Committee and the academic exchange opportunities facilitated for researchers. The authors thank all participating subjects for their kind cooperation in this study.
). We appreciate the recognition extended by the Organizing Committee and the academic exchange opportunities facilitated for researchers. The authors thank all participating subjects for their kind cooperation in this study.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
