Abstract
Background:
Immune checkpoint inhibitors (ICIs) have demonstrated efficacy in metastatic gastric cancer. Their potential benefits in the neoadjuvant therapy for advanced gastric cancer (AGC) are promising but require real-world evidence.
Objective:
We aimed to assess the real-world impact of adding ICIs to neoadjuvant chemotherapy (NAC) in AGC patients.
Design:
This is a retrospective, propensity score-matched analysis of 580 AGC patients treated with NAC, with or without ICIs, followed by radical gastrectomy.
Methods:
Patients were matched using propensity score matching (PSM) to balance baseline characteristics. Pathological complete response (pCR) rates and toxicity were compared between the ICIs-Chemo and Chemo cohorts.
Results:
After PSM, 188 patients were included: 71 in the ICIs-Chemo cohort and 117 in the Chemo cohort. Chemotherapy regimens in both cohorts included SOX (49.5%, 93/188), FLOT (26.6%, 50/188), XELOX (12.8%, 24/188), and FOLFOX (11.2%, 21/188). The ICIs used were predominantly Sintilimab (67.6%, 48/71), followed by Tislelizumab (21.1%, 15/71), Nivolumab (7%, 5/71), and others (4.2%, 3/71). Adding ICIs to NAC significantly improved the pCR rate (ICIs-Chemo: 29.6% vs. Chemo: 13.7%, p = 0.014). The SOX regimen, with (37.1%, 13/35) or without ICIs (22.4%, 13/58), demonstrated the highest pCR rates. Subgroup analysis indicated that patients older than 60 years (OR = 3.86, p < 0.01) and those with moderately/well-differentiated tumors (OR = 4.27, p = 0.01) may derive greater benefit from adding ICIs. While overall toxicity and surgical complication rates were similar between cohorts, we observed two cases of suspected severe immune-related adverse events (irAEs).
Conclusion:
Adding ICIs to NAC regimens in AGC significantly improves pathological response. While overall toxicity is not increased, close monitoring for irAEs is necessary.
Background
Gastric cancer is the fifth most common malignancy and the fourth leading cause of cancer-related death worldwide.1,2 Many patients, particularly in less developed regions, are diagnosed at advanced stages. 3 While surgical resection offers a potential cure for these patients, the recurrence rate after surgery is high.4,5 Incomplete tumor clearance, which refers to failing to achieve an R0 resection (R1/R2 resection) or the persistence of tumor cell in the residual lymph nodes, is a major contributing factor, 6 even with histologically negative margins (R0) and D2 lymph node dissection, cancer cells may persist in distant lymph nodes or as micrometastases, contributing to recurrence. This highlights the need for effective pre-operative (neoadjuvant) treatment to downstage tumors, enabling more complete surgical removal. Thus, neoadjuvant chemotherapy (NAC) is an important part of gastric cancer management. Studies show NAC can downstage tumors, improve curative resection (R0 resection) rates, and ultimately lead to better survival outcomes.7–11 However, due to the heterogeneity of gastric cancer cells, 12 not all patients respond equally to NAC. 13 In those with poor response, tumors may even progress during NAC, hindering the chance for curative surgery. 14 Since only patients who respond well benefit from NAC, improving response rates is crucial. Therefore, new drugs with novel mechanisms of action are needed.
Immune checkpoint inhibitors (ICIs) have proven to be effective antitumor agents in gastric cancer management. As the most commonly used type of ICIs, programmed cell death protein-1 (PD-1) inhibitors incite a strong antitumor immune response by blocking the interaction of PD-1 with its ligands. 15
In recent years, with the advancement of molecular diagnostic technologies, particularly high-throughput sequencing and gene chip technologies, several new molecular classification strategies for gastric cancer have emerged. In 2014, based on The Cancer Genome Atlas (TCGA) program’s research findings, using six technology platforms including whole-exome sequencing, researchers analyzed fresh frozen tissue and blood samples from 295 treatment-naive patients across multiple global centers. They proposed four molecular subtypes: Epstein-Barr virus-positive (EBV), microsatellite instability (MSI), chromosomal instability (CIN), and genomically stable (GS). Among these, EBV and MSI subtypes are likely to benefit from immunotherapy, while CIN and GS subtypes are less likely to respond to immunotherapy. 12
In clinical practice, ICIs were first used in metastatic cases. In a series of clinical trials, PD-1 inhibitors combined with chemotherapy significantly improved response rates and survival in metastatic gastric cancer patients.16–18 Building on this success, investigators have begun to explore ICIs as neoadjuvant therapy agents. Preliminary trials have demonstrated that ICIs can improve pathological complete response rates, a key indicator of neoadjuvant therapy efficacy.19–25 However, these trials often have small sample sizes, are single-arm, or focus on a single treatment regimen. Robust real-world evidence further demonstrating the value of ICIs in the neoadjuvant setting is still needed.
Our institution began integrating ICIs into neoadjuvant therapy regimens in 2019, and we have observed promising improvements in response rates. This study aims to systematically review these cases, using propensity score matching (PSM), to address the following key questions: Does the addition of ICIs significantly enhance the efficacy of neoadjuvant regimens? Do ICIs increase toxicity or surgical risks? and How do different ICIs-containing chemotherapy regimens compare in terms of efficacy? Our goal is to generate real-world evidence supporting the use of ICIs in gastric cancer management.
Methods
Ethics approval
This study was reviewed and approved by the ethics committee of The Sixth Affiliated Hospital, Sun Yat-Sen University (Approval number: 2022ZSLYEC-020). This study was conducted in accordance with the 1964 Helsinki Declaration.
Study design
This is a retrospective cohort study. Clinical data were retrieved from the Gastric Cancer Database of our institution. Patients were followed up via outpatient clinics and telephone until mortality or loss of follow-up.
Inclusion criteria:
Age 18–80 years old
Histologically confirmed diagnosis of gastric or esophago-gastric junction adenocarcinoma
Received NAC with or without ICIs
Underwent subsequent radical gastrectomy surgery after NAC
Exclusion criteria:
Received concurrent radiotherapy or targeted therapy during NAC
Insufficient clinical staging information or uncertain presence of distant metastasis
Presence of a secondary concurrent malignancy
Irregular chemotherapy or immunotherapy regimens used prior to surgery
These criteria were applied to select the study participants and ensure data quality and relevance.
Primary objective
To evaluate the impact of adding ICIs to neoadjuvant chemotherapy on pathological complete response rate in patients with advanced gastric cancer (AGC).
Primary endpoint: Pathological complete response rate (pCR).
Secondary endpoint: Safety and toxicity profile.
Pre-intervention staging
Before starting the treatments, the clinical stage of the patients was determined by enhanced thoracic-abdominal-pelvic computed tomography and/or endoscopic ultrasonography according to the American Joint Committee on Cancer staging criteria. 26
Multidisciplinary recommendation
NAC recommendations were individualized after evaluation by the gastric cancer multidisciplinary team at our institution. Treatment decisions, including the off-label use of ICIs in the neoadjuvant setting, were based on physician recommendations, and patient informed consent and preference, a practice permitted under Article 29 of the Physicians Law of the People’s Republic of China, 27 which allows off-label drug use with evidence-based medical support.
Consent for participation
All patients included in this retrospective analysis provided written informed consent, via our institution’s standard “Informed Consent for Collection and Use of Sample Data” procedure, for the anonymized collection and use of their medical data for research purposes. This physician-explained paper form, signed by the patient (or their legal representative), permitted the use of their treatment-related anonymized information from our specialized database.
Chemotherapy regimens
The chemotherapy regimens utilized in this study include FLOT, SOX, XELOX, and FOLFOX, as listed below:
FLOT: Docetaxel 40–60 mg/m2, oxaliplatin 85 mg/m2, and fluorouracil 2800 mg/m2; administered every 2 weeks;
SOX: Oxaliplatin 130 mg/m2, tegafur gimeracil oteracil potassium capsule 40–60 mg bid Day 1 to Day 14; administered every 3 weeks;
XELOX: Oxaliplatin 130 mg/m2, capecitabine 1000 mg/m2 bid Day 1 to Day 14; administered every 3 weeks;
FOLFOX: Oxaliplatin 85 mg/m2, fluorouracil 2800 mg/m2; administered every 2 weeks.
Immune checkpoint inhibitors
ICIs were administered intravenously (IV) in combination with chemotherapy. Drugs and standard dosages included the following: Sintilimab (200 mg), Tislelizumab (200 mg), Nivolumab (240 mg), Pembrolizumab (200 mg), Serplulimab (150 mg), and Camrelizumab (200 mg).
Preoperative assessment
After the completion of NAC, the resectability of the primary tumor was re-confirmed by enhanced thoracic-abdominal-pelvic computed tomography.
Surgical resection
All patients underwent curative gastrectomy (total or subtotal) with standard D2 lymphadenectomy. Prior to resection, a thorough abdominal exploration was performed to determine the status of peritoneal metastasis.
Resected specimen examination
VAll resected specimens underwent examination for pathological staging and assessment of tumor regression grade after neoadjuvant therapy.
Tumor regression grading
Tumor regression was graded according to the Ryan standard28,29 as follows:
Grade 0 (pathological complete response): no residual tumor cells
Grade 1 (major response): scattered single tumor cells
Grade 2 (moderate response): clusters of tumor cells with fibrosis
Grade 3 (minor response): extensive residual tumor cells
Toxicity assessment
Toxicity related to neoadjuvant chemotherapy was evaluated using the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE) version 5.0. 30
Post-surgery complication grading
Post-surgical complications were classified using the Clavien-Dindo system. 31 Only Grades IIIa–V complications were documented.
Follow-up
Regular follow-up assessments during the first 2 years included appointments every 3 months followed by subsequent appointments every 6 months. Each follow-up examination consisted of a thorough review of medical history, physical examination, routine blood tests, comprehensive biochemical analyses, and CT scans. The hospital’s follow-up office conducted these assessments through telephone calls or mail correspondence to gather information on the patients’ health status and survival. Overall survival (OS) was defined as the duration from the day of surgery until either the date of death or the final follow-up date.
Propensity score matching
Patients who received neoadjuvant chemotherapy with or without immune checkpoint inhibitors were defined as the Chemo cohort and the ICIs-Chemo cohort, respectively. PSM was used to ensure similar baseline characteristics between the two cohorts. Matching factors included sex, age, tumor location, tumor differentiation, clinical T and N stages, and neoadjuvant therapy regimens. The matching ratio was 1:2 (ICIs-Chemo vs. Chemo), and the caliper was 0.02.
Data analysis
The normality of data was assessed using the Kolmogorov–Smirnov test and normal probability plots. Parameters that were not normally distributed were expressed in the median (upper quartile to lower quartile) and were analyzed using non-parametric tests: Mann–Whitney test or Kruskal–Wallis test, as appropriate. Categorical variables were analyzed by the chi-square test. The survival difference between the ICIs-Chemo and Chemo cohort was compared using the Kaplan–Meier method, and the hazard ratios were calculated in the Cox regression model. A p-value <0.05 was identified as statistically significant. All statistical analyses were performed using the R software version 4.0.2 (The R Foundation for Statistical Computing, Vienna, Austria; www.r-project.org) and Microsoft Excel version 2021 (Microsoft Corporation, Redmond, WA, USA; www.microsoft.com).
The reporting of this study conforms to the STROBE statement. 32 The completed checklist for the STROBE statement is provided as a Supplemental Material.
Results
Patient characteristics
From February 2013 to November 2023, we identified 676 patients who received neoadjuvant therapy followed by D2 radical gastrectomy. Figure 1 illustrates the inclusion and exclusion process of the study, with 580 eligible patients ultimately enrolled. As shown in Table 1, these patients were divided into two cohorts: the Chemo cohort (n = 488) received neoadjuvant chemotherapy alone, while the ICIs-Chemo cohort (n = 92) received neoadjuvant chemotherapy combined with programmed cell death protein 1 (PD-1) inhibitors. Prior to PSM, the ICIs-Chemo cohort exhibited significantly more advanced clinical stages; notably, 81.5% of patients in the ICIs-Chemo cohort presented with clinical N2-3 disease compared to 60.2% in the Chemo cohort. Following PSM, 117 patients remained in the Chemo cohort and 71 in the ICIs-Chemo cohort, with similar baseline characteristics across both cohorts. After matching, patients predominantly consisted of elderly males with tumors located in the upper stomach and clinical stages exceeding T3N+. Among patients receiving ICIs, PD-L1 Combined Positive Score (CPS) results were available for 22/92 (23.9%) in the pre-matching cohort and 18/71 (25.4%) in the post-matching cohort. The median CPS was 5 (full range: 0–50) in both cohorts.

CONSORT diagram illustrating patient enrollment, cohort allocation, and key survival outcomes before and after propensity score matching.
Patients’ baseline characteristics before and after PSM.

Expressed in median [upper quartile, lower quartile].
dMMR, mismatch repair deficiency; ICI, immune checkpoint inhibitor; pMMR, proficient mismatch repair; PSM, propensity score matching.
Neoadjuvant therapy
As depicted in Figure 2, four neoadjuvant chemotherapy regimens were utilized: FLOT, SOX, XELOX, and FOLFOX. In the ICIs-Chemo cohort, PD-1 inhibitors were combined with these regimens. Prior to PSM, there were differences in chemotherapy regimen distribution: FLOT predominated in the Chemo cohort (55.9%, 273/488), while SOX was more frequent in the ICIs-Chemo cohort (43.5%, 40/92). After PSM, SOX became the most common regimen in both cohorts (approximately half of the cases), as shown in Table 1. As for ICIs Usage in the ICIs-Chemo Cohort, Sintilimab was the most frequently used ICI (48/71, 67.6%), followed by Tislelizumab (15/71, 21.1%). Nivolumab (5/71, 7.0%), Camrelizumab (2/71, 2.8%), and Serplulimab (1/71, 1.4%) were also administered. The first patient in this cohort began ICI treatment as part of the neoadjuvant regimen in July 2019, and the last patient completed their ICI treatment in November 2023. In both cohorts, patients received a median of four cycles of neoadjuvant therapy prior to surgery. Before starting neoadjuvant therapy, only 5% (29/580) of patients underwent staging laparoscopy prior to neoadjuvant therapy.

Chemotherapy regimens employed in this study include FLOT, SOX, XELOX, and FOLFOX—all commonly used in clinical practice. Before PSM, FLOT was the most frequent regimen in the chemotherapy cohort. SOX was the most common chemotherapy base when combined with ICIs in the ICIs-Chemo cohort. After PSM, chemotherapy regimen proportions were similar across both cohorts.
Toxicity profile
As shown in Table 2, Grade 3/4 toxicity occurred at similar rates in both groups: ICIs-Chemo cohort (22/71, 31%) and Chemo cohort (38/117, 32.5%). Anemia was the most common hematological toxicity (Chemo 18.8% vs. ICIs-Chemo 18.3%), followed by neutropenia (Chemo 13.7% vs. ICIs-Chemo 8.5%). Overall, severe (Grade 3/4) non-hematological toxicities were uncommon (Chemo 3.4% vs. ICIs-Chemo 4.2%). The most frequent non-hematological toxicities included diarrhea, nausea, and vomiting.
Preoperative treatment-related adverse events before and after propensity score matching according to the CTCAE 5.0.
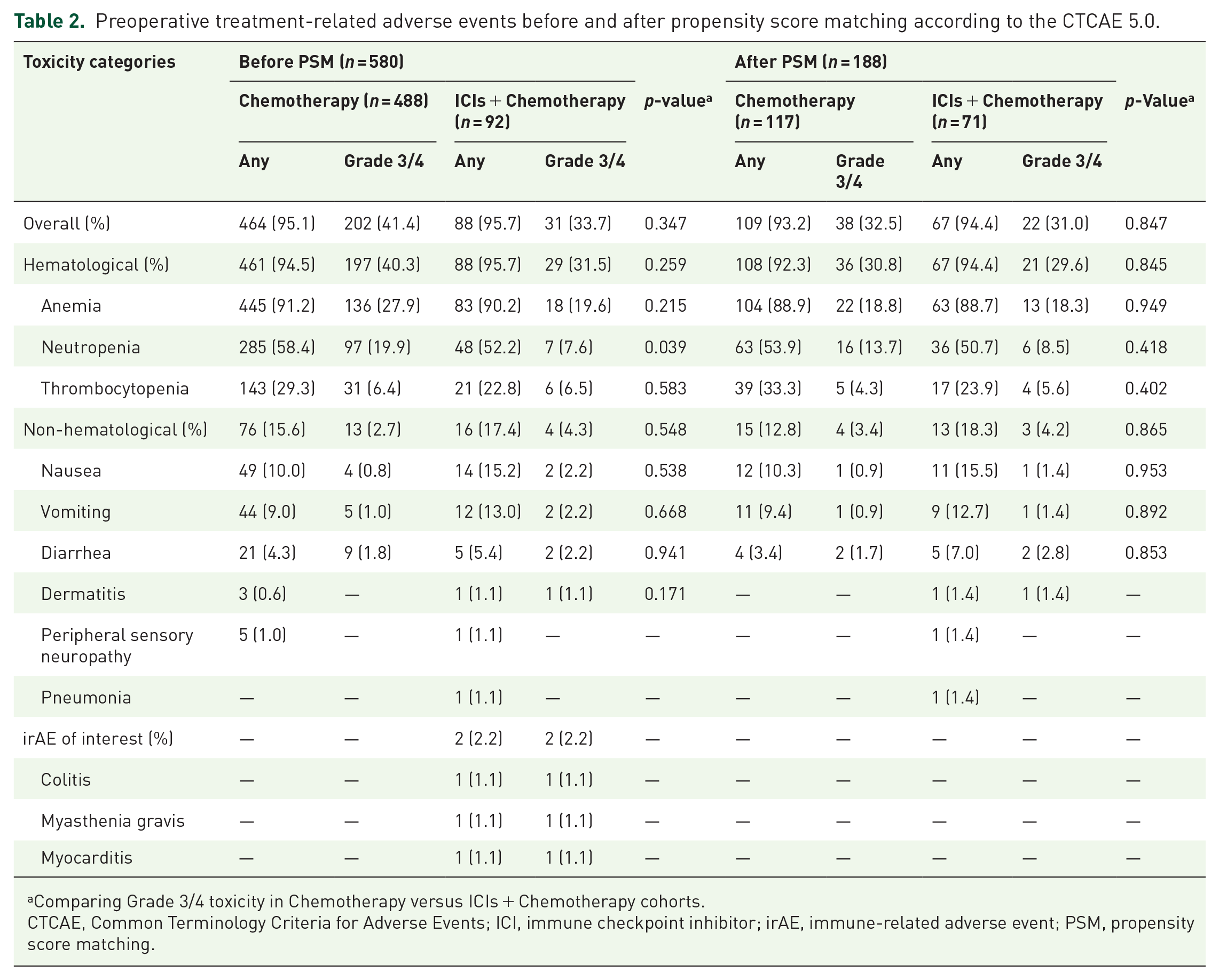
Comparing Grade 3/4 toxicity in Chemotherapy versus ICIs + Chemotherapy cohorts.
CTCAE, Common Terminology Criteria for Adverse Events; ICI, immune checkpoint inhibitor; irAE, immune-related adverse event; PSM, propensity score matching.
Immune-related adverse events
Two cases of suspected severe immune-related adverse events (irAEs) were observed. One patient receiving Sintilimab plus XELOX experienced Grade 3 diarrhea (over 10 stools daily) that resolved after ICI discontinuation and steroid therapy. The other patient receiving Pembrolizumab plus XELOX developed muscle weakness, ptosis, and elevated creatine kinase; the symptoms were consistent with ICI-induced myasthenia gravis and myocarditis, which remitted after steroid administration. These irAEs led to treatment discontinuation and pre-scheduled surgery.
Surgery and complications
As shown in Table 3, all patients underwent curative resection surgery with D2 lymphadenectomy. Over 90% of procedures were performed laparoscopically, and more than half involved total gastrectomy. Following PSM, R0 resection rates were not significantly improved in the ICIs-Chemo cohort (ICIs-Chemo 95.8% vs. Chemo 95.7%, p = 0.987). Similarly, there were no statistical differences in the rates of Grade IIIa–V postoperative complications between cohorts: 11 (15.5%) patients in the ICIs-Chemo cohort and 22 (18.8%) patients in the Chemo cohort (p = 0.563). Anastomotic leakage and abdominal infection were the most frequent complications (ICIs-Chemo 5.6% vs. Chemo 9.4%), followed by pleural effusion (ICIs-Chemo 7% vs. Chemo 6%).
Surgical outcomes, pathological findings before and after PSM.

Complications were graded according to the Clavien-Dindo system.
Expressed in median [upper quartile, lower quartile].
ICI, immune checkpoint inhibitor; PSM, propensity score matching.
Notably, two patients in the pre-PSM cohorts experienced fatal complications. One patient in the Chemo cohort died from severe anastomotic leakage leading to abdominal bleeding. In the ICIs-Chemo cohort, one patient died from severe lymphatic leakage and subsequent circulatory failure following aggressive para-aortic lymph node dissection. Twenty patients out of 580 underwent paraaortic lymph node dissection due to preoperative imaging suggesting suspicious paraaortic lymph node enlargement. However, postoperative pathological examination revealed no paraaortic lymph node metastasis in any of these patients.
Pathological response
In terms of treatment efficacy, the addition of ICIs to chemotherapy regimens significantly improved pathological complete response rates in the ICIs-Chemo cohort compared to the Chemo cohort (pre-PSM: 28.3% vs. 11.3%; post-PSM: 29.6% vs. 13.7%). Furthermore, adding ICIs improved the pCR rates of every chemotherapy regimen in our study (Figure 3). Among all regimens evaluated, SOX demonstrated the highest effectiveness, both alone (22.4%, 13/58) and in combination with ICIs (37.1%, 13/35). In addition, the ICIs-Chemo cohort exhibited zero cases of T0N+, compared to seven in the Chemo cohort, suggesting better lymph node remission with ICI addition. Subgroup analysis (Figure 4) indicates that elderly patients (>60 years old) and those with moderately to well-differentiated adenocarcinoma may derive greater benefit from the addition of ICIs.

The pCR rate in the Chemo and ICIs + Chemo cohorts. Overall, adding ICIs to neoadjuvant regimens significantly improved the pCR rate. Moreover, when grouped by specific chemotherapy regimens, ICIs consistently improved pCR rates of each chemotherapy regimen.

Forest plot showing pCR rates across different subgroups. The data suggest that elderly patients and patients with moderately or well-differentiated tumors may benefit more from neoadjuvant immune checkpoint inhibitor therapy.
Survival analysis
The median follow-up time is 25 months for the ICIs-Chemo cohort and 13 months for the Chemo cohort. After PSM, in the Chemo cohort (n = 117), 79 patients survived, with 70 patients achieving disease-free survival and 9 survivors experiencing recurrence. Among the 38 death cases in the Chemo cohort, 34 deaths were attributed to tumor progression, and 4 deaths were due to non-tumor-related causes, which were censored in the survival analysis. In the ICIs-Chemo cohort after PSM (n = 71), 53 patients achieved disease-free survival, and among the survivors, 13 experienced recurrences. There were four deaths related to tumor progression and one non-tumor-related death, which was also censored. As illustrated in Figure 5, the survival analysis revealed that no statistically significant difference in disease-free survival (DFS) or OS was observed between the Chemo and ICIs-Chemo cohorts, either before or after PSM. Figure 1 provides a comprehensive overview of detailed survival outcomes in each cohort before and after matching. The 1-year DFS rates were 82.7% (95% CI 72.8–93.9) in the ICIs-Chemo cohort versus 72.2% (63.7–81.8) in the Chemo cohort after PSM, with a non-significant trend favoring the ICIs-Chemo group (p = 0.17). Regarding OS, due to the limited follow-up duration, the median survival time was not reached in any of the cohorts.

Kaplan–Meier curves illustrating DFS and OS for the Chemo and ICIs-Chemo cohorts. The analysis reveals no statistically significant survival differences between the cohorts before or after PSM. However, a trend indicates that the ICIs-Chemo cohort might exhibit improved disease-free survival outcomes following matching. Extended follow-up is required to validate these observations. (a) DFS before PSM; (b) DFS after PSM; (c) OS before PSM; (d) OS after PSM.
Discussion
In this study, we found that adding ICIs to neoadjuvant chemotherapy regimens significantly improved the pathological response.
NAC plays a crucial role in the management of AGC. By downstaging tumors, eliminating micro-metastases, and improving R0 resection rates, NAC offers potential survival benefits. The landmark MAGIC trial 8 (2006) demonstrated the superiority of NAC with the ECF regimen (epirubicin, cisplatin, and fluorouracil) over surgery alone. Subsequent RCTs, including the FNCLCC 9 and FLOT4 10 studies, further solidified the value of NAC, and the RESOLVE 11 study established the advantage of a “sandwich modality” (NAC-surgery-adjuvant chemotherapy) over direct surgery followed by adjuvant treatment. As a result, NAC is recommended in most guidelines for AGC patients.33–35
Despite the use of NAC, OS outcomes in AGC remain unsatisfactory. The considerable heterogeneity of gastric cancer cells leads to variations in patient response and treatment outcomes after NAC. Previous studies demonstrated that the benefit AGC patients derives from NAC closely correlates with pathological response, as measured by residual tumor cells.14,36 pCR, defined as the absence of residual tumor cells, is a strong predictor of prolonged survival. 37 Unfortunately, in the aforementioned RCTs, pCR rates were limited. This emphasizes the crucial need to incorporate novel drugs with distinct mechanisms to improve the pCR rate and overall patient outcomes.
ICIs are a novel class of antitumor drugs that differ from traditional cytotoxic chemotherapy in two key ways. First, they directly activate tumor-specific cytotoxic T cells in the tumor microenvironment by blocking the PD-1/PD-L1 pathway, which normally suppresses immune responses. Second, ICIs enhance tumor antigen presentation by dendritic cells in tumor-draining lymph nodes, promoting further T-cell activation and migration to tumor sites.
Administering ICIs in the neoadjuvant setting offers a unique advantage: the primary tumor provides abundant endogenous tumor antigens, allowing for more effective T-cell activation and expansion. These activated T cells could potentially persist after surgery, providing long-term immunological surveillance against micrometastases and reducing recurrence risk.
The efficacy of ICIs in treating metastatic gastric cancer has been validated by multiple RCTs.18,38–41 Currently, it is recommended as the first-line treatment for unresectable locally advanced, recurrent, or metastatic gastric cancers by major guidelines.
Inspired by the potential of ICIs to enhance tumor response, our institution began integrating ICIs into NAC regimens in 2019, under ethical approval. The addition of ICIs to neoadjuvant therapy significantly improved the pCR rate, a key indicator of successful neoadjuvant treatment. This finding is consistent with previous studies demonstrating the benefits of adding ICIs to neoadjuvant therapy. For example, Yuan et al. 24 reported that adding toripalimab to SOX/XELOX regimens improved the pCR rate to 22.2% and the major pathological response rate to 44.4%. Similarly, Yin et al. 19 found a 25% pCR rate when utilizing tislelizumab with SOX. Karukonda et al. 22 (PROCEED Trial) demonstrated even further improvement when combining ICIs with neoadjuvant chemoradiotherapy, achieving a 35.7% pCR rate. Preliminary results from recent phase III trials20,25 (KEYNOTE-585, MATTERHORN) further validate this trend, showing a 2- to 6-fold increase in pCR rates in cohorts receiving ICIs compared to those with chemotherapy alone. The observed pCR rate in our study appears higher than those reported in other studies. This discrepancy is likely due to selection bias, as our data come exclusively from our institution’s surgical database, meaning it only includes patients who successfully underwent resection. Patients who received neoadjuvant therapy with the intent of surgery but did not undergo the procedure due to poor response were excluded, potentially leading to an over-estimation of the true pCR rate. However, this over-estimation bias exists in both cohorts, making the observed improvement in the ICIs-Chemo cohort compared to the Chemo cohort still valid. In addition, in the ICIs-Chemo cohort, we observed zero cases of T0N+, compared to seven cases in the Chemo cohort, suggesting that ICIs may promote better lymph node tumor remission. This finding aligns with previous research demonstrating that the PD-1/PD-L1 checkpoint can restrain T-cell immunity in tumor-draining lymph nodes. 42
Regarding safety, findings from the MATTERHORN and KEYNOTE-585 trials, along with our results, indicate that adding ICIs to neoadjuvant chemotherapy does not significantly increase toxicity or post-surgical complications. However, it is crucial to remain cautious for potential irAEs, which likely result from the disruption of immune regulation. While irAEs predominantly affect the gastrointestinal system, skin, and liver and are often mild, they can also target endocrine organs, lungs, heart, and the nervous system. Their insidious onset can lead to delayed diagnosis and high mortality risk. For example, a report by Aldrich et al. 43 (MD Anderson Cancer Center) found a considerably high mortality rate (22%) in patients with ICI-induced myositis. In our study, we observed two irAEs (myasthenia gravis and colitis) requiring steroid therapy and neoadjuvant treatment discontinuation. As ICI indications expand, clinicians may encounter more unprecedented toxicities with a higher risk of misdiagnosis. Research on irAE mechanisms, diagnosis, and management is crucial for improved patient monitoring and treatment.
Regarding survival, while it is clear that ICIs improve pathological responses in locally advanced gastric cancer (LAGC), their impact on long-term survival outcomes like OS and DFS remains uncertain. Lessons from other cancers are mixed. For example, in triple-negative breast cancer 44 and non-small-cell lung cancer, 45 adding ICIs to neoadjuvant chemotherapy improved pCR rates, which translated into longer event-free survival. However, a meta-analysis on esophageal cancer found that while neoadjuvant chemoradiotherapy resulted in higher pCR and R0 resection rates, along with lower recurrence, it did not improve 5-year survival. 46 This highlights the need for longer follow-up to assess the Regarding safety true impact of ICI-induced pCR on long-term survival in LAGC.
Regarding adjuvant therapy, the crucial question is whether adding ICIs provides benefit. The recent Attraction-5 47 trial showed no overall DFS improvement with ICIs compared to traditional adjuvant chemotherapy. However, an important insight emerged from the subgroup analysis: patients with tumors expressing PD-L1 ⩾1% seemed to derive a DFS benefit. This suggests that sensitivity to ICIs is a potential indicator of the effectiveness in the adjuvant therapy. Using ICIs in the neoadjuvant setting allows for assessing this sensitivity, potentially helping to determine whether a patient could benefit from adjuvant ICIs therapy.
Regarding biomarkers, several recent studies aim to identify robust ICIs efficacy indicators in AGC. Promising candidates include PD-L1 expression, mismatch repair (MMR) status, and EBV status. However, the relationship between these biomarkers and treatment outcomes requires further clarification. While PD-L1 expression is currently the most common biomarker in GC, 48 the optimal CPS cutoff value is uncertain. The Orient-16 study found greater ICI benefit in patients with CPS ⩾5, 41 yet similar benefits were absent for CPS ⩾1 patients in KEYNOTE-062. 17 This highlights the need for more research to determine an optimal threshold. Mismatch repair deficiency (dMMR) tumors exhibit high immune checkpoint expression, suggesting sensitivity for anti-PD-1 therapy. Studies on dMMR colorectal and gastric cancers demonstrated promising pCR rates with neoadjuvant immunotherapy.49,50 However, due to the rarity of dMMR in AGC, most data come from small-scale phase II trials. Larger studies are needed to confirm this correlation. EBV-associated gastric cancer (EBVaGC), with its amplified PD-L1/PD-L2, is another promising immunotherapy target. However, conflicting results from two phase II trials (100% response rate with pembrolizumab 51 vs. none with camrelizumab 52 ) indicate significant heterogeneity within EBVaGC, warranting further investigation.
Our study has certain limitations. First, the retrospective nature of the analysis introduces potential selection bias, as only patients who completed surgery were included in the analysis. Second, the incomplete availability of biomarker data (e.g., PD-L1 expression, MMR status) limits our ability to identify predictive factors for treatment response. Third, the relatively immature follow-up for the ICI cohort likely contributed to the rapid decline in at-risk patients beyond 10 months in survival curves.
Conclusions
Adding ICIs to neoadjuvant therapy regimens substantially improves pathological response in AGC patients, without increasing toxicity or post-surgical complications. However, well-designed randomized clinical trials with longer follow-up are needed to confirm a definitive long-term survival benefit.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251346423 – Supplemental material for Immune checkpoint inhibitors improve the efficacy of neoadjuvant chemotherapy in advanced gastric cancer: a retrospective cohort study
Supplemental material, sj-docx-1-tam-10.1177_17588359251346423 for Immune checkpoint inhibitors improve the efficacy of neoadjuvant chemotherapy in advanced gastric cancer: a retrospective cohort study by Yonghe Chen, Yi Lin, Yingying Xu, Jianwei Zhang, Shi Chen, Shuai Ye, Gang Liu, Lei Lian and Junsheng Peng in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
