Abstract
Gastric carcinoma and gastro-oesophageal junction (GC/GEJ) carcinoma remain a significant global problem, with patients presenting with symptoms often found to have advanced or metastatic disease. Treatment options for these patients have broadened in recent years with new chemotherapy agents, agents targeting angiogenic pathways and the development of immune checkpoint inhibitors (ICIs). Most initial advances have occurred in the refractory setting, where it is important to balance treatment benefits versus toxicity and patient quality of life. In the first-line treatment of advanced/metastatic GC/GEJ, platinum- and fluoropyrimidine-based chemotherapy protocols remain the backbone of therapy (with or without HER2-targeted therapy), with the FOLFIRI regimen offering an alternative in patients deemed unsuitable for a platinum agent. Microsatellite instability-high or mismatch repair-deficient cancers have been shown to benefit most from ICIs. In unselected patients previously treated with doublet or triplet platinum- and fluoropyrimidine-based chemotherapy and second-line chemotherapy with irinotecan or taxanes have formed the backbone of therapy with or without the addition of the vascular endothelial growth factor receptor-2 inhibitor ramucirumab in addition to paclitaxel. Beyond this, efficacy has been demonstrated with oral trifluridine/tipiracil and with single-agent nivolumab, in selected refractory patients. In this review, we highlight the positive evidence from key trials that have led to our current practice algorithm, with particular focus on the refractory advanced disease setting, discussing the areas of active research and highlighting the factors, including biomarkers and the influence of ethnicity, that contribute to therapeutic decision-making.
Keywords
Introduction
Gastric cancer currently ranks as the fifth most common cancer (5.6% of all new cases), and the fourth commonest cause of cancer-related deaths worldwide (7.7% of all the deaths).1,2 Curative intent therapy is focused on surgery, with variations in practice internationally around peri-operative approaches used, reflecting the influence of and the declining incidence rates of cardia versus non-cardia gastric carcinoma/gastro-oesophageal junction (GC/GEJ) adenocarcinoma and the pattern of diagnosis, with population screening programmes in Japan and South Korea leading to earlier diagnosis. 2 However, despite progress in early diagnosis and screening up to 50% of patients present with metastatic disease in the west,3,4 and there remain high rates of recurrence after curative intent therapy. Therefore, management of metastatic GC/GEJ remains a substantial contributor to the global cancer health burden. Accumulated evidence from randomized controlled trials and international guidelines have developed to guide the use therapies in the first- and second-line and refractory settings.5–8
Chemotherapy with platinum and fluoropyrimidine (oral or intravenous) is the most common established first-line therapy. The options for patients progressing beyond first-line therapy continue to be based around chemotherapy, thus tending to be limited to patients suitable to receive chemotherapy, with recent evidence extending to third-line chemotherapy and beyond. Advances in the understanding of the genomics of gastric cancer continue but the use of targeted therapy has for the most part been limited to patients with HER2-positive disease.9,10 More recently, the discovery and subsequent blockade of the immune regulatory programmed cell death-1 (PD-1) and programmed cell death ligand 1 (PD-L1) pathway has seen several trials of immune checkpoint inhibitors (ICIs) in patients with advanced/metastatic GC/GEJ, commencing in the refractory setting and progressing to earlier lines of therapy. Tissue-based criteria for patient selection for optimal benefit have also been identified.
This review follows an initial consensus meeting by selected members of the Australian Gastro-Intestinal Trials Group (AGITG) together with a selected invited international panel at ESMO Asia in 2019, reconvened virtually in January 2022. Here we provide an updated review of the evidence for the management of patients with advanced/metastatic GC/GEJ supporting a current treatment algorithm, with a focus on recent data, discussing the areas of active research and highlighting the factors that contribute to therapeutic decision-making. The evidence for current approved therapies is reviewed by treatment line, with more detailed focus on new therapies, with review of biomarkers that may affect treatment decision-making, particularly when deciding on the addition of biologic therapies and immunotherapy.
First-line therapy
Treatment options in GC/GEJ have evolved significantly in the last decade and are fast changing. The evidence that first established the role of chemotherapy in the first-line setting in advanced/metastatic GC/GEJ came from initial trials evaluating chemotherapy efficacy against best supportive care. From these initial studies, chemotherapy was shown to substantially improve time to progression and overall survival (OS), thus establishing it as standard in suitably fit patients. 5 Most studies thereafter looked to build on this by evaluating various combination therapies. Multiple randomized controlled trials have led to broadening treatment options with systematic reviews and international consensus guidelines consistent in their recommendation of first-line platinum (cisplatin or oxaliplatin (P)) and fluoropyrimidine-based chemotherapy (F), based on data confirming superior response rate, delayed time to progression and OS of PF-based regimens compared with single-agent chemotherapy.5–8 Triplet chemotherapy (with the addition of an anthracycline or docetaxel to a PF backbone) has shown superior response rate, delayed tumour progression and survival, but at the expense of greater toxicity, and is thus generally not considered as a routine choice of therapy. 5 Thus, the choice of platinum (cisplatin or oxaliplatin) and fluoropyrimidine [intravenous or oral (capecitabine or S1)] regimen varies by country and institutional practice, and decision-making for a given patient based on disease burden, toxicity differences (triplet versus doublet chemotherapy) and patient tolerance (for oral medications) and patient preferences.5–8,11–14 Platinum-based chemotherapies come with significant toxicities including peripheral neuropathy, oto- and nephro-toxicities.11–14 Where this side effect risk is not considered appropriate for a particular patient, the alternate use of infusional fluorouracil and irinotecan as in the FOLFIRI regimen can be considered a reasonable alternative.5,15
The next evolution in therapy in the first-line setting in GC/GEJ came with the addition of biologic therapies to PF chemotherapy. The first key randomized controlled study, the AVAGAST study evaluated the efficacy of adding the vascular endothelial growth factor (VEGF) monoclonal antibody bevacizumab to a PF backbone. 16 The study failed to meet its primary endpoint for OS, although it did demonstrate superior improvement in progression-free survival (PFS) and overall response rate (ORR) with an acceptable safety profile for the combination of bevacizumab and chemotherapy. 16 Pre-planned subgroup analyses in AVAGAST suggested regional differences in OS with patients from North America and Latin America having a survival benefit with bevacizumab [OS median, 11.5 versus 6.8 months, hazard ratio (HR) 0.63; 95% confidence interval [CI]), 0.43–0.94), whereas patients from Asia appearing to have no benefit. 16 So, while this study did not change practice, it did provide encouraging data for ongoing research with antiangiogenic targeted therapies in patients with advanced GC/GEJ, leading to studies in the second-line setting and beyond of several antiangiogenic therapies (discussed below). However, success in the first-line setting with antiangiogenic therapies has remained elusive, with ramucirumab, a VEGF receptor 2 (VEGFR-2) antagonist monoclonal antibody, failing to show improvement in PFS when added to chemotherapy over chemotherapy alone in the phase III placebo-controlled RAINFALL study. 17
Growing evidence that HER2 was an important biomarker and driver of tumorigenesis in gastric cancer, 18 and the efficacy observed with trastuzumab, a HER2 targeting monoclonal antibody, in breast cancer, led to the ToGA trial, an open-label, international, phase III, randomized controlled trial, which evaluated trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced GC/GEJ cancer. 10 Screening for HER2 overexpression for enrolment in this study demonstrated that 22.1% of patients were positive by IHC3+/FISH, which was similar between European and Asian patients but higher in intestinal-type versus diffuse-type GC (31.8% versus 6.1%) and GEJ versus gastric tumours (32.2% versus 21.4%). 19 The ToGA trial established trastuzumab and chemotherapy (cisplatin or fluorouracil/capecitabine) as the first-line standard of care for HER2-positive patients, by demonstrating significantly improved OS (HR: 0.74), PFS (HR: 0.71) and ORR (47% versus 35%) with the addition of trastuzumab to chemotherapy. 10
Ongoing research into new drugs targeting the HER2 pathway following success in HER2-positive breast cancer has led to trials evaluating these drugs in patients with advanced/metastatic GC/GEJ. One example with disappointing results was the JACOB study, a double-blind, placebo-controlled, randomized, multicentre phase III study which evaluated the addition of pertuzumab to first-line trastuzumab and chemotherapy for HER2-positive metastatic GC/GEJ. 20 Pertuzumab, a humanized monoclonal HER2-targeted antibody, binds to a different epitope on the HER2 receptor protein than trastuzumab and was shown to improve survival in HER2-positive early and metastatic breast cancer.21–23 Unfortunately, the JACOB study failed to meet its target primary endpoint for improved OS in patients with HER2-positive metastatic GC/GEJ (OS HR: 0.84; 95% CI: 0.71–1.00; p = 0.057). 20
Other pathways have been targeted in patients with advanced gastric cancer without success despite progression to phase III trial in first and later lines of therapy. These include negative first-line phase III trials evaluating epidermal growth factor receptor monoclonal antibodies, and unsuccessful studies evaluating antibodies or small molecule inhibitors targeting the MET or hepatocyte growth factor axis and inhibitors of the PI3K/AKT/mTOR pathway, which includes the GRANITE-1 study which evaluated everolimus versus placebo. 24
The next class of agents demonstrating efficacy in the first-line setting in patients with advanced or metastatic GC/GEJ adenocarcinoma are the ICIs. Immune evasion was acknowledged as an important emerging hallmark of cancer in 2011. 25 In the decade prior and ensuing, an abundance of research was undertaken exploring cancer’s ability to seize control of immune checkpoints, induced upon T-cell activation. 25 CTLA4 (mainly expressed by activated CD8+ effector T cells) was the first immune checkpoint receptor to be clinically targeted. 25 The PD-1 receptor is an inhibitory T-cell receptor that interacts with its two known ligands, PD-L1 and PD-L2 to limit T-cell activity in peripheral tissues during an inflammatory response and limit autoimmunity, thus creating an immune resistance mechanism within the tumour microenvironment.26,27 Following activity observed in refractory advanced/metastatic GC/GEJ with the PD-1 checkpoint inhibitors nivolumab and pembrolizumab, respectively, clinical trials have been undertaken in earlier clinical settings in combination with standard chemotherapy.28,29
In patients with HER2 amplification or overexpression, the KEYNOTE-811 was undertaken to evaluate the efficacy of adding pembrolizumab or placebo to trastuzumab and chemotherapy for unresectable or metastatic, HER2-positive GC/GEJ adenocarcinoma, in PD-L1 unselected patients. This randomized, double-blind, placebo-controlled phase III study reported on its secondary endpoint ORR from its protocol-specified interim analysis in 2021. 30 ORR in the first 264 patients enrolled was 74.4% in the pembrolizumab group and 51.9% in the placebo group, a significant improvement of 22.7% (p = 0.00006), with 11.3% complete response rate and 96.2% disease control rate (any response or stable disease). 30
In all enrolled participants who received at least one dose of study treatment as of the data cut-off date, with median duration of pembrolizumab treatment 6.2 months (range: 2 days–17.7 months), 57.1% of patients experienced any grade 3 or higher toxicity with pembrolizumab, versus 57.4% in the placebo group. 30 The most common adverse events in both groups were diarrhoea [52.5% (pembrolizumab group) versus 44.4% (placebo group)], nausea (48.8% versus 44.4%) and anaemia (41.0% versus 44.0%). Possibly immune-mediated adverse events and/or infusion reactions in the pembrolizumab group occurred in 33.6%, the most common of these events were infusion-related reactions (18.0% pembrolizumab group versus 13.0% in the placebo group) and pneumonitis (5.1% pembrolizumab group versus 1.4%). 30 Based on this interim data, the US Federal Drug Authority (FDA) granted accelerated approval to this pembrolizumab-based combination as first-line treatment of patients with locally advanced unresectable or metastatic HER2-positive GC/GEJ in 2021, irrespective of PD-L1 status. The study is continuing with final results for the primary endpoints of PFS and OS awaited (https://clinicaltrials.gov, NCT03615326).
In the majority of patients with GC/GEJ that are HER2 negative, two studies have been published reporting superior efficacy with the addition of the ICI nivolumab to standard chemotherapy in the first-line setting.31,32 The International open-label randomized phase III CheckMate 649 study evaluated the efficacy of first-line nivolumab and chemotherapy versus chemotherapy alone for advanced GC/GEJ, and oesophageal adenocarcinoma in HER2-negative, PD-L1 unselected patients. 31 Patients received nivolumab 360 mg 3 weekly or 240 mg 2 weekly and investigator choice chemotherapy (XELOX or FOLFOX) or chemotherapy alone. The dual primary endpoints were OS and PFS by blinded independent central review per RECIST version 1.1. PD-L1 expression on tumour cells and tumour-associated immune cells [combined positive score (CPS)] was used to stratify patients (CPS status ⩾1% versus <1% or indeterminate). Other stratification factors included geographical region (Asia versus USA/Canada versus rest of world), ECOG (Eastern Co-operative Oncology Group) performance status (0 versus 1) and chemotherapy (XELOX versus FOLFOX). 31 Hierarchical testing was used for the OS primary analysis commencing with demonstration of superiority of OS in patients with a PD-L1 CPS of 5 or more. 31
In the enrolled population, approximately 70% had gastric cancer, 18% GEJ with around 12% oesophageal adenocarcinoma, with 14–15% of patients having signet ring carcinoma and 3–4% of patients having microsatellite instability-high (MSI-H) tumours, and 60% of patients had tumours expressing PD-L1 CPS of 5 or more. 31 Both primary endpoints were met in this study. After a minimum follow up of 12.1 months, nivolumab and chemotherapy had superior OS compared with chemotherapy alone (median 14.4 months versus 11.1 months respectively, HR 0.71, p < 0.0001), and superior PFS (median 7.7 months versus 6.0 months, HR: 0.68, p < 0.0001). 31 Further analysis demonstrated significant improvement in OS in patients with PD-L1 CPS ⩾ 1 (HR: 0.77, p < 0.0001) and in all randomly assigned patients (HR: 0.8, p = 0.0002). 30 Interaction analysis of OS by PD-L1 CPS cut-offs showed significant interaction by PD-L1 CPS at the cut-off of five (p = 0.011). 31 Less than 40% of patients received further line therapies in each treatment arm, respectively. 31 No new safety signals were observed for the addition of nivolumab. Based on this study, nivolumab and chemotherapy are now new standard of care first-line options. Global approval for use varies however, with the European Medicines Union (EMA) approving first-line nivolumab only on patients with PD-L1 CPS ⩾5, whereas the US FDA and the Japanese Pharmaceuticals and Medical Devices Agency (PMDA) have approved first-line nivolumab in advanced GC/GEJ without PD-L1 selection.
The ATTRACTION-04 study, involving multiple Asian countries only, evaluated nivolumab plus chemotherapy versus placebo plus chemotherapy in PD-L1 unselected patients with HER2-negative, untreated, unresectable advanced or recurrent GC/GEJ. 32 Patients received chemotherapy every 3 weeks [oxaliplatin plus oral S-1 or CAPOX], with either nivolumab intravenously every 3 weeks (nivolumab plus chemotherapy group) or placebo (placebo plus chemotherapy group). 31 Randomization was stratified by PD-L1 expression, ECOG performance status, disease status and country. The primary endpoints were centrally assessed PFS and OS. After a median follow-up of 11.6 months for the interim analysis, median PFS was 10.45 months in the nivolumab arm versus 8.34 months in the placebo arm (HR: 0.68, p = 0.0007). 32 However, at final analysis, with median follow-up of 26.6 months, median OS was 17.45 months in the nivolumab arm and 17.2 months in the placebo arm (HR: 0.90, p = 0.26). 31 There were no identifiable differences in any of the subgroups. ORR was numerically higher for nivolumab (57%) versus placebo (48%). 32 One explanation for the lack of survival benefit was the high percentage (66%) of patients that went on to receive subsequent therapies including immunotherapy.
The KEYNOTE-590 study evaluated pembrolizumab plus chemotherapy versus chemotherapy and placebo as first-line treatment in in PD-L1 unselected patients with advanced oesophageal cancer [mainly squamous cell cancer (SCC)] and Siewert type 1 GEJ cancer, including 14.7% of patients with oesophageal adenocarcinoma and 12% with Siewert type 1 GEJ adenocarcinoma. 33 The study was positive for its primary endpoint of OS in patients with oesophageal SCC and PD-L1 CPS of 10 or more [median 13·9 months versus 8·8 months; HR: 0.57 (95% CI: 0·43–0·75); p < 0.0001] and in all randomized patients [median 12·4 months versus 9·8 months; 0.73 (95% CI: 0·62–0·86); p < 0·0001], with HR 0.74 (95% CI: 0.54–1.02) in all adenocarcinomas. 33 PFS was also in favour of pembrolizumab in the adenocarcinoma subgroup [median 6·3 months versus 5·7 months; HR: 0·63 (95% CI: 0.46–0.87)]. 33
The KEYNOTE-062 study was a global study that evaluated the efficacy and safety of first-line pembrolizumab alone or pembrolizumab plus chemotherapy versus chemotherapy alone in patients with advanced GC and GEJ cancer with PD-L1 CPS of 1 or greater. 34 The study’s primary endpoints were OS in patients with PD-L1 CPS of 1 or greater (intention-to-treat population) and PD-L1 CPS of 10 or greater, and PFS by RECIST 1.1 and blinded independent review in PD-L1 CPS of 1 or greater. The study also assessed the non-inferiority of pembrolizumab versus chemotherapy. Pembrolizumab was not superior to chemotherapy alone in patients with PD-L1 CPS of 1 or greater (HR, 0.91; 95% CI, 0.74–1.10). 34 And at final analysis (PD-L1 CPS of 1 or greater), median OS with pembrolizumab and chemotherapy versus chemotherapy alone also did not meet the criteria for superiority (median OS 12.5 months versus 11.1 months respectively, HR: 0.85, p = 0.05). 34 However, at final analysis (PD-L1 CPS of 1 or greater), median OS with pembrolizumab alone did meet the non-inferiority criteria versus chemotherapy alone (median OS 10.6 months versus 11.1 months, respectively, HR: 0.91, 99.2% CI, 0.69–1.18; non-inferiority margin, 1.2). 34
JAVELIN Gastric 100 was a global open-label phase III study that evaluated maintenance avelumab versus continued chemotherapy in non-progressive HER2 negative, PD-L1 unselected patients after 12 weeks of standard first-line oxaliplatin and fluoropyrimidine chemotherapy. 35 Maintenance avelumab was not superior to continued chemotherapy in the primary analysis (median OS 10.4 months versus 10.9 months, respectively, HR: 0.91, p = 0.1779). 35
In summary, chemotherapy has an established place as the backbone of treatment of patients with advanced/metastatic GC/GEJ. New directions have focused on targeting aspects of the disease biology, with the addition of trastuzumab standard in HER2-positive patients and immune therapy with ICIs such as nivolumab given in addition to chemotherapy the latest advance in unselected patients, HER2 negative. The addition of the ICI pembrolizumab to trastuzumab and chemotherapy also looks extremely promising (Table 1).
Key first-line phase III studies evaluating the addition or comparison of biologic therapies or immunotherapy to standard chemotherapy in patients with advanced/metastatic GC/GEJ.
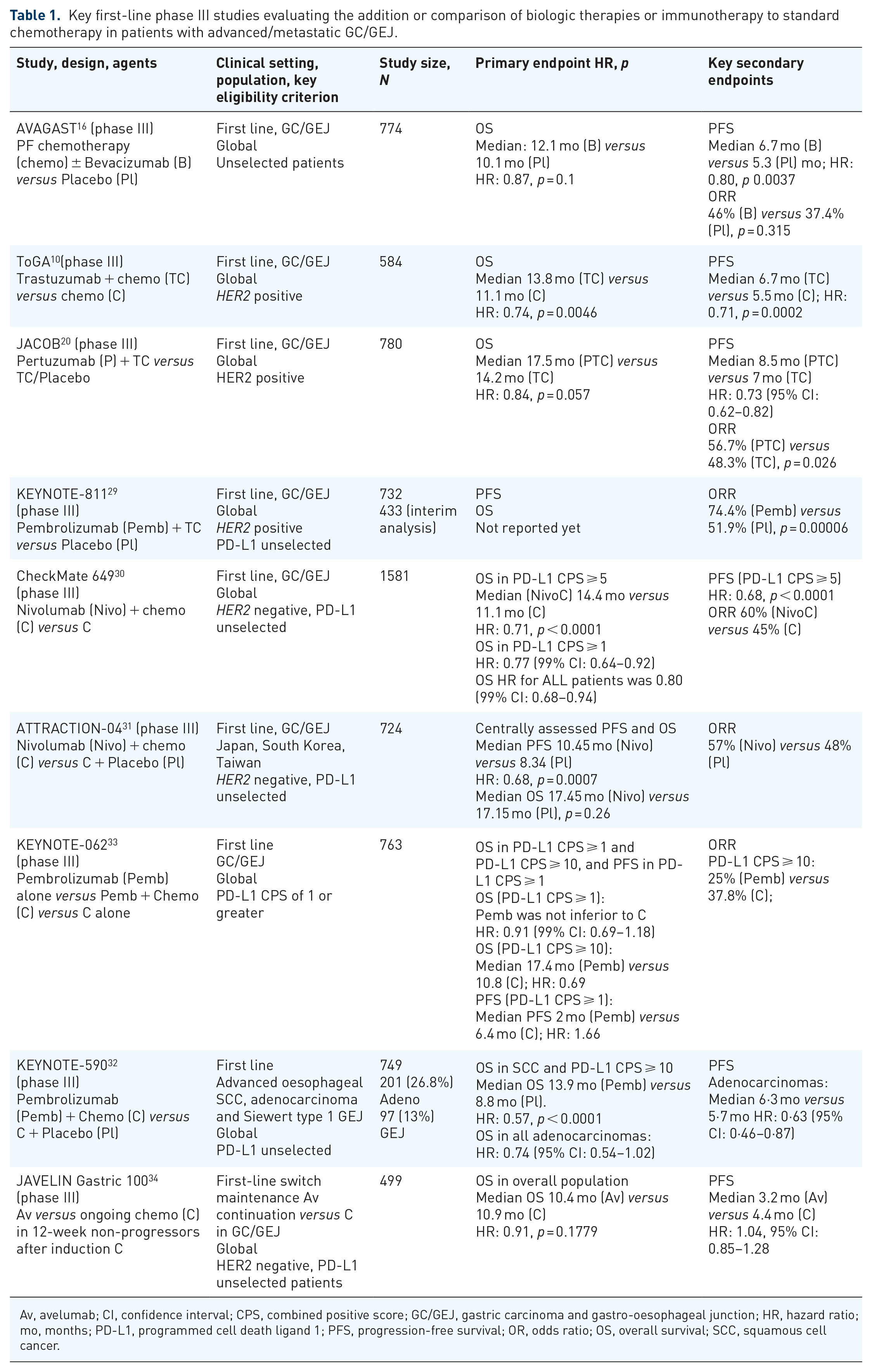
Av, avelumab; CI, confidence interval; CPS, combined positive score; GC/GEJ, gastric carcinoma and gastro-oesophageal junction; HR, hazard ratio; mo, months; PD-L1, programmed cell death ligand 1; PFS, progression-free survival; OR, odds ratio; OS, overall survival; SCC, squamous cell cancer.
Second-line therapy
The next evolution of therapy in unselected patients who had progressed after first-line chemotherapy was with chemotherapy alone, followed by studies evaluating the addition of biologic therapies such as antiangiogenic monoclonal antibodies or ICIs. Three trials evaluated single-agent chemotherapy (docetaxel or irinotecan) versus best supportive care/no chemotherapy with the collective finding of superior survival in patients that received chemotherapy (HR: 0.63, 95% CI: 0.51–0.77, p < 0.0001), thus establishing second-line chemotherapy as a standard of care in suitably fit patients.36–39
The efficacy of ramucirumab, an antiangiogenic VEGFR-2 antagonist monoclonal antibody, was evaluated in two phase III clinical trials, in combination with second-line paclitaxel compared with paclitaxel alone in the RAINBOW study or as monotherapy compared with best supportive care in the REGARD study.40,41 Both studies were in patients with gastric cancer who progressed after standard first-line chemotherapy. In the RAINBOW study, the combination of ramucirumab and paclitaxel improved survival compared with paclitaxel alone in taxanes-naïve patients (median 9.6 months versus 7.4 months, respectively, HR: 0.807, p = 0.017). 40 In the second-line REGARD trial, ramucirumab monotherapy improved OS compared with placebo/best supportive care (median 5.2 versus 3.8 months, respectively, HR: 0.776, p = 0.047). 40 Unique grade 3 hypertension was observed in 14–16% of patients receiving ramucirumab in these studies.40,41
Thus, the standard of care for fit patients considered suitable for active therapy in the second-line setting is chemotherapy with a taxane (docetaxel or paclitaxel) or irinotecan. In countries where ramucirumab is available, paclitaxel and ramucirumab would be considered the standard option in patients fit for chemotherapy, with single-agent ramucirumab an alternative offering modest benefit in patients where chemotherapy is not suitable.
Immunotherapy with single-agent pembrolizumab was evaluated as a second-line treatment in patients with GC/GEJ that progressed on first-line chemotherapy with a platinum and fluoropyrimidine in the KEYNOTE-061study. 42 This global, open-label phase III study evaluated the efficacy of pembrolizumab compared with weekly paclitaxel on the primary endpoints of OS and PFS in patients with PD-L1 CPS of 1 or higher. 42 The initial 489 patients were enrolled irrespective of their PD-L1 status; however, the subsequent 103 patients enrolled had to have a PD-L1 CPS of 1 or higher based on the independent data monitoring committee recommendation following review of outcomes in patients with a PD-L1 CPS less than 1. Of the 592 patients enrolled, 395 had a PD-L1 CPS of 1 or higher. Patients with HER2-positive disease were included. In the primary efficacy analysis for OS, pembrolizumab was not superior to paclitaxel (median 9.1 months versus 8.3 months, respectively, HR: 0.82; 95% CI: 0.66–1.03). 42 Survival rate at 18 months was 26% with pembrolizumab compared with 15% with paclitaxel. 42 PFS was also not improved (HR: 1.27; 95% CI: 1.03–1.57). Post-hoc exploratory analysis indicated pembrolizumab effect was greatest in patients with performance status 0 and high PD-L1 CPS (OS in CPS of 10 or higher: HR: 0.64; 95% 0.41–1.02). 42
In summary, second-line chemotherapy is considered standard in suitably fit patients, with several regimens having established benefit. The antiangiogenic VEGFR-2 antagonist monoclonal antibody ramucirumab has shown efficacy alone or in combination with paclitaxel and is available in selected countries. Single-agent pembrolizumab was not shown to be superior to paclitaxel.
Third-line therapy and beyond
The refractory setting (beyond second-line therapy) has become the hunting ground for new drug exploration in patients with advanced/metastatic GC/GEJ; however, it is a challenging setting in which to conduct clinical trials due to the nature of the disease and its clinical burden, limiting study accrual to the fitter patients with better disease trajectory, often in Asian populations. Nonetheless, several studies have been successfully completed confirming that studies in this setting are feasible and can provide evidence for new treatment options and opportunities to move the field forward as these treatments are then explored in earlier clinical settings.
In patients with HER2-positive metastatic GC/GEJ who progress after treatment involving first-line trastuzumab, one recent study evaluating the activity of the novel antibody drug conjugate trastuzumab deruxtecan (T-DXd) in refractory HER2-positive GEJ is notable. The DESTINY-Gastric01 study was an open-label, randomized, phase II trial, evaluating trastuzumab deruxtecan compared with physician’s choice chemotherapy (irinotecan or paclitaxel) in patients with progressive HER2-positive advanced gastric cancer (centrally confirmed) progressing following at least two previous therapies, including trastuzumab. 43 The primary endpoint of the study was ORR by an independent central review. The study population enrolled 188 heavily pre-treated patients from Japan and Korea [median of two prior therapies (range 2–9)] including 31 patients (17%) having received at least four prior therapies, with 72% receiving prior ramucirumab and 86% a prior taxanes. 43 Patients receiving T-DXd had a much higher ORR than patients receiving chemotherapy (51% versus 15% respectively, p < 0.001). 43 Moreover, PFS and OS were superior with T-DXd compared with chemotherapy [PFS HR: 0.47 (95% CI: 0.31–0.71); and OS HR: 0.59 (95% CI: 0.39–0.88), p = 0.01]. 43 Toxicity was mainly haematologic and gastrointestinal (mostly low grade), with grade 3/4 adverse events predominantly haematologic with six patients receiving T-DXd having febrile neutropenia compared with two receiving chemotherapy. 43 However, mainly low-grade drug-related interstitial lung disease or pneumonitis was noted in 12 patients (10%; with 2 grade 3 and one grade 4 event) receiving T-DXd, of which eight had resolved at the time of study publication. Drug-related interstitial lung disease occurred in six (7.6%) patients [grade 1–2 (5 patients) and one grade 5 event]. 43 Based on these results, The Japanese Ministry of Health approved trastuzumab deruxtecan in 2020 and in 2021 the US FDA also approved trastuzumab deruxtecan in patients with locally advanced or metastatic HER2-positive GC or GEJ adenocarcinoma who have received a prior trastuzumab-based regimen.
This was followed by a single-arm study in a Western population (US/EU) – the DESTINY-Gastric02 study (NCT04014075). This study evaluated the activity of T-DXd, measured by ORR by an independent central review in 79 patients with locally advanced or metastatic GC/GEJ who progressed on ⩾1L therapy including trastuzumab. 44 Confirmed ORR was 38% (95% CI: 27.3–49.6) and median PFS was 5.5 months (95% CI: 4.2–7.3). 44
In a general population of patients with treatment refractory advanced/metastatic GC/GEJ, including a small subset of patients with HER2-positive disease, the TAGS study established the efficacy of oral trifluridine/tipiracil as a new standard treatment option in this setting. 44 This global phase III double-blind, placebo-controlled study evaluated the efficacy of trifluridine/tipiracil compared with placebo in reasonably fit patients (ECOG 0-1) who had received at least two previous chemotherapy regimens, with 37% of patients having received three prior regimens, and 63% four lines plus. 45 The study met its primary endpoint for OS, which favoured trifluridine/tipiracil over placebo [median 5.7 months versus 3.6 months, respectively, HR: 0.69 (95% CI: 0.56–0.85), p = 0,00058]. 45 Improvement was also seen in PFS and delay to deterioration of performance status. This benefit was obtained through disease control (44%) rather than tumour response (4%) with trifluridine/tipiracil compared with placebo (disease control 14%). 45 A subsequent post-hoc analysis by line of therapy (third or fourth line) showed numerically greater OS in third-line patients compared to fourth-line patients (median OS 6.8 versus 5.2 months, respectively), with similar corresponding OS HR: 0.68 (p = 0.0318) compared with OS HR: 0.73 (p = 0.0192). 46 The most frequent adverse events ⩾grade 3 were neutropenia (34%) and anaemia (19%) with trifluridine/tipiracil and abdominal pain (9%) and general deterioration of physical health (9%) with placebo. 44 The commonest lower grade (1–2) non-haematologic toxicity in the trifluridine/tipiracil arm included nausea (34%), reduced appetite (26%), vomiting (21%), diarrhoea (20%) and fatigue (20%), compared with 29%, 24%, 18%, 13% and 15% with placebo, respectively, highlighting the common frequency of disease symptoms in this disease in this setting. 45 Quality of life was maintained from baseline in patients treated with trifluridine/tipiracil.45,47 Based on this study’s results, trifluridine/tipiracil has been approved as the standard treatment option for patients with refractory advanced/metastatic GC/GEJ in many parts of the world.
One of the first phase III studies to explore the efficacy of ICIs in patients with refractory advanced GC/GEJ was the ATTRACTION-2 study which evaluated the PD1 checkpoint inhibitor nivolumab versus placebo in patients that were intolerant of, or who had progressed after at least two previous chemotherapy regimens in Japan, South Korea and Taiwan, with 40% each having received 3 and ⩾4 prior lines of therapy, respectively. 28 Patients were enrolled irrespective of their PD-L1 status. The primary endpoint of the study, OS, was met in favour of nivolumab over placebo [median 5.26 months versus 4.14 months. respectively, HR: 0.63 (95% CI: 0.51–0.78), p < 0.0001]. OS was higher in the nivolumab arm at 12 months (27.3% versus 11.6%) and 2 years (10.6% versus 3.2%). 28 Although there was no change in the median PFS, overall PFS was also in favour of nivolumab (HR: 0.60, 95% CI: 0.49–0.75, p < 0.0001), with 11% of patients treated with nivolumab demonstrating objective tumour response. 28 Toxicity was as expected and previously reported with nivolumab. An exploratory analysis was undertaken by PD-L1 expression on immunohistochemistry (IHC; central laboratory using 28-8 pharmDx assay; Dako, Carpinteria, CA, USA), confirming survival benefit independent of PD-L1 expression; however, only 40% of patients had tumours evaluable for PD-L1 testing. 28 The authors acknowledged that more research was required into biomarkers to identify which patients benefit most. The results of the ATTRACTION-2 trial resulted in the approval of nivolumab as a third- and later line treatment independent of PD-1 expression by the US FDA and Japanese PMDA. No EMA approval was granted in this indication.
The JAVELIN Gastric 300 study outcomes were reported not long after this study and the results were disappointing. This global randomized phase III study evaluated avelumab, a human anti-PD-L1 IgG1 monoclonal antibody, versus physician’s choice chemotherapy (weekly paclitaxel or irinotecan) as third-line treatment in PD-L1 unselected patients with advanced GC/GEJ. 48 The study failed to meet its primary endpoint of improving OS (HR: 1.1, 95% CI: 0.9–1.4, p = 0.81), and did not demonstrate any advantage of avelumab over chemotherapy for PFS or ORR. 48 PD-L1 expression was not shown to be associated with prognosis. 48
Targeting angiogenesis as a therapeutic strategy in patients with advanced/metastatic GC/GEJ continued with the development of apatinib, a small-molecule tyrosine kinase inhibitor (TKI) of VEGFR-2. In a Chinese placebo-controlled phase III study (The ANGEL study), apatinib was evaluated for efficacy compared with placebo in patients with advanced/metastatic GC/GEJ who had previously received at least two prior lines of chemotherapy. 49 With 35% of patients having received three or more prior lines of therapy both primary endpoints of OS and PFS were met, with apatinib improving OS compared with placebo (median 6.5 months versus 4.7 months respectively, HR: 0.709, p = 0.0156), and prolonging PFS compared with placebo (2.6 months versus 1.8 months respectively, HR: 0.44; p < 0.001). 49 The ORR was 6.9% in the apatanib arm compared to no responses in the placebo arm (p = 0.002). 49 The commonest grade ¾ non-hematologic adverse events were hand–foot syndrome, proteinuria and hypertension. 49
The AGITG evaluated regorafenib in the INTEGRATE study, a placebo-controlled randomized phase II study in patients with advanced/metastatic GC/GEJ having received up to two prior lines of therapy (58% two lines), in Australia, New Zealand, Canada and South Korea. 50 Regorafenib is a broadly acting oral multi-kinase inhibitor targeting angiogenic (VEGF-R1, VEGF-R2 and TIE-2), stromal (platelet-derived growth factor receptor beta) and oncogenic (RAF, RET and KIT) receptor tyrosine kinases. Promising activity was seen with patients receiving regorafenib having prolonged PFS (HR: 0.4, 95% CI: 0.28–0.59), with benefit observed across all patient subgroups, but particularly in Korean patients (region-by-treatment interaction p < 0.001). 50 The AGITG has since followed this with the International phase III placebo-controlled INTEGRATE IIa study (NCT02773524), evaluating the effect of regorafenib versus placebo on OS. This study has completed accrual.
In summary, third-line therapy and beyond is a challenging area in patients with refractory advanced/metastatic GC/GEJ, due to high disease burden and less fit patients. Yet, many of the latest advances have been evaluated here with several now accepted as proven therapeutic options such as oral trifluridine/tipiracil chemotherapy, nivolumab in chemotherapy refractory patients and trastuzumab deruxtecan emerging as an option in HER2-positive patients. Small molecule antiangiogenic TKIs have also shown activity with apatinib showing efficacy in a phase III trial in China and the regorafenib in phase III development (Table 2).
Key randomized controlled trials in refractory advanced GC/GEJ.
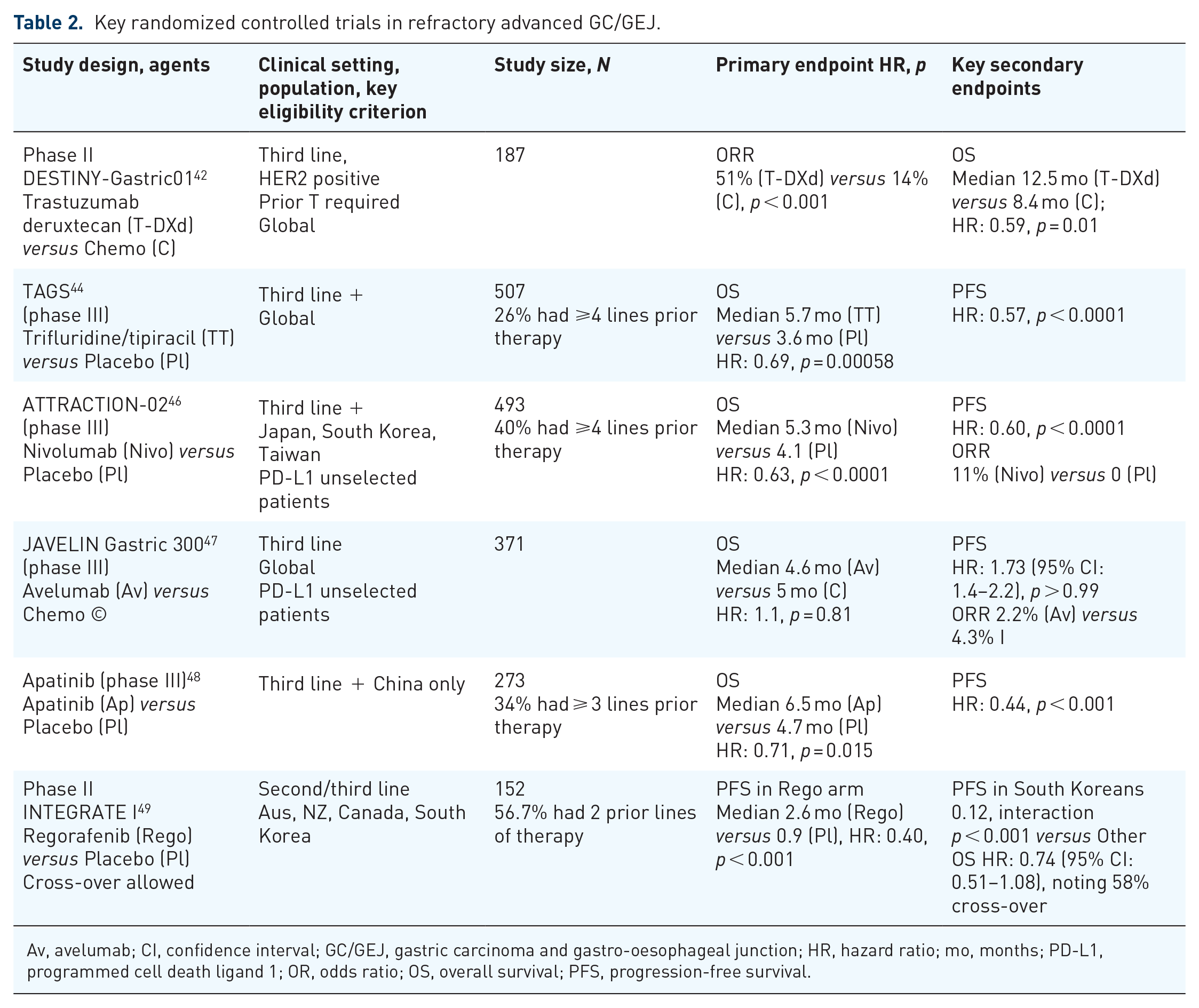
Av, avelumab; CI, confidence interval; GC/GEJ, gastric carcinoma and gastro-oesophageal junction; HR, hazard ratio; mo, months; PD-L1, programmed cell death ligand 1; OR, odds ratio; OS, overall survival; PFS, progression-free survival.
Key ongoing phase III studies in advanced/metastatic GC/GEJ
With the successes of ICIs seen in many cancers, and its failures in others, an increasing focus on research has been on better understanding the complex biology of the tumour immune microenvironment (TIME) and its influence on treatment response. 51 The TIME is a complex ecosystem involving cancer cells, tumour vasculature and immune cells that can both facilitate and act as a barrier to many therapies. 51 One way of enhancing the effect of ICIs has been to use antiangiogenic therapies to reduce the immunosuppressive effect of angiogenesis in the TIME in combination with ICIs. 52 Promising activity was seen with the combination of regorafenib and nivolumab (REGONIVO) in a chemo- and immunotherapy refractory patient population in a phase Ib trial in Japan. 53 The AGITG has since commenced a global phase III study evaluating REGONIVO versus standard of care chemotherapy in patients with refractory advanced/metastatic GC/GEJ (INTEGRATE IIb, NCT04879368). Other approaches in new treatment development in gastric cancer include novel agents with broader or novel mechanisms of action, for example, SHR1701, a bifunctional fusion protein targeting PD-L1 and TGF-β, which is currently being evaluated in combination with first-line chemotherapy in a phase III trial in previously untreated, advanced/metastatic GC/GEJ (NCT04950322).
Current and future biomarkers for treatment selection in advanced/metastatic GC/GEJ
There are few biomarkers that are used to direct therapy in advanced/metastatic GC/GEJ. In newly diagnosed patients upfront HER2 testing for selection of HER2-targeted therapy is well established in the treatment algorithm (Figure 1). Despite promising results with HER2-targeted therapy, resistance develops in many patients, with several different mechanisms identified such loss of or mutations in the HER2 receptor and upregulation of alternative receptors such as MET, HER3 and FGFRs. 54 This raises the role of re-biopsy in HER2-positive patients to identify if persisting with a HER2-targeted approach is worthwhile in these patients.
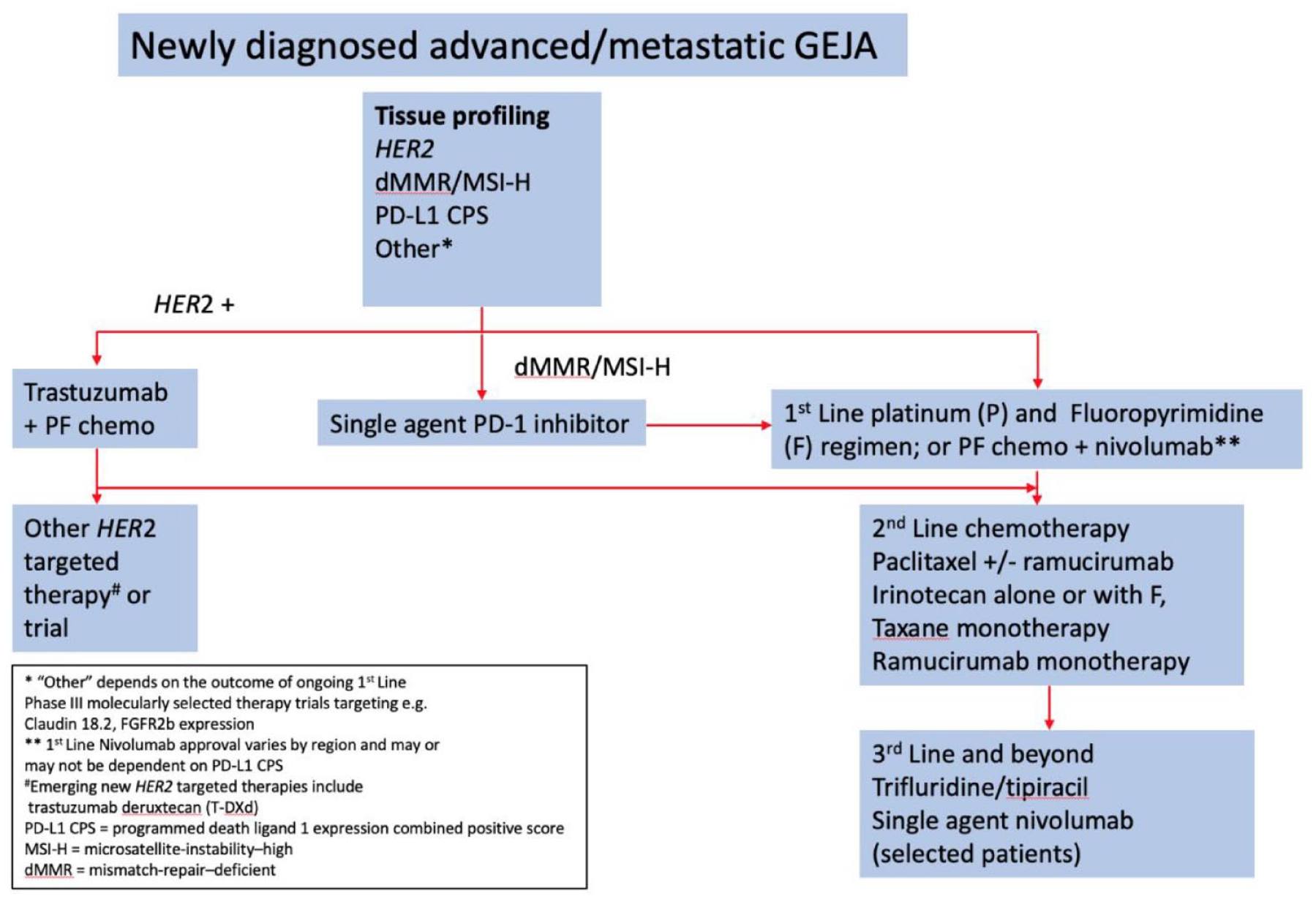
Treatment algorithm describing treatment options for newly diagnosed patients with advanced/metastatic GC/GEJ.
The recent activity seen with ICIs in selected patients with gastrointestinal malignancies has led to increasing interest in identifying reliable biomarker predictors of efficacy. Mismatch repair-deficiency (dMMR) was first identified as a possible predictor of clinical benefit from immune checkpoint blockade, with pembrolizumab first demonstrating this in a phase II study in patients with advanced gastrointestinal cancers. 55 A subsequent phase III study has confirmed superior efficacy with first-line pembrolizumab compared with chemotherapy in MSI-H or dMMR metastatic colorectal cancer. 56 The activity of the PD-1 inhibitor pembrolizumab in 84 patients with MSI-H advanced GC/GEJ was evaluated by Chao et al across three studies: the single-arm third-line or greater KEYNOTE-059 (pembrolizumab monotherapy, N = 174), the phase III second-line KEYNOTE-061 (pembrolizumab versus chemotherapy, N = 514) and the first-line KEYNOTE-062 study (pembrolizumab alone or with chemotherapy versus chemotherapy, N = 682). 57 The overall incidence of MSI-H cancers across the three studies was 6% (84/1370). 57 The objective response rate for pembrolizumab alone was 57% in both KEYNOTE-059 and KETNOTE-062 and 46.7% for KEYNOTE-061, with median OS not reached for pembrolizumab alone in KEYNOTE-059, KEYNOTE-061 and KEYNOTE-062, or for pembrolizumab and chemotherapy in KEYNOTE-062. 57 In the CheckMate 649 study, the unstratified HR for OS for nivolumab and chemotherapy versus chemotherapy alone was 0·33 (95% CI: 0.12–0.87) in patients with MSI-high tumours. 31
PD-L1 scoring using IHC has been examined across a variety of tumours and with various antibodies both as a selection marker for enrolment into clinical trials and as a predictor for efficacy with ICIs. Out of 45 USA FDA approvals for ICIs from 2011 to 2019, nine approvals were linked to a specific PD-L1 threshold and companion diagnostic, and one in GC/GEJ cancers. 58 This approval was first made in 2017 for pembrolizumab, using the pharmDx 22C3 PD-L1 IHC assay (Agilent Technologies, Santa Clara, CA, USA), based on ORR with pembrolizumab being greater in PD-L1 positive as opposed to negative patients in the Keynote-059 study. 58 The PD-L1 CPS, defined in the Keynote-059 study, is the number of PD-L1-positive cells (tumour cells, macrophages, lymphocytes) divided by the total number of tumour cells, multiplied by 100, with a positive PD-L1 CPS defined as 1 or greater. 29 But there have been many other studies with different ICIs undertaken in GC/GEJ, with different assays and different PD-L1 cut-offs selected for efficacy, with the optimal cut-off for maximum benefit not defined in GC/GEJ cancer patients.
The Cancer Genome Atlas has categorized gastric cancer into four molecular subtypes: Epstein-Barr virus positive, (EBV) MSI-H, genomically stable and chromosomally instable. 9 Of these subtypes, it is expected that EBV-positive tumours, which have been associated with high PD-L1 tumour expression and tumours with MSI-H may confer greater sensitivity to immune checkpoint inhibition.55,59 Through design and/or lack of accessible tissue the key randomized phase III studies evaluating ICIs in advanced/metastatic GC/GEJ to date have evaluated this.28,32–35,48 In a study looking at prospective genomic profiling to identify predictive biomarkers of drug response in 295 patients with stage IV oesophagogastric adenocarcinoma, MSI-H tumours were shown to be chemotherapy resistant but more likely to respond to immunotherapy. 59 And a single EBV-positive patient had a durable complete response to immunotherapy. 59
The VIKTORY Umbrella Trial in Korea used genetic sequencing to identify then classify patients with metastatic gastric cancer into eight different biomarker defined groups to assign patients to one of ten associated phase II clinical trials in second-line treatment. The study enrolled 772 patients, profiling 715 patients with 14.7% of patients receiving biomarker-assigned treatment, confirming feasibility of this approach in the clinic. 60 The application of genetic sequencing to gastric cancer has provided greater insights into the molecular heterogeneity of this disease in addition to highlighting targets for drug therapy, opening the door to the paradigm of biomarker directed therapy.61,62 Diffuse-type gastric cancer has gained particular attention due to its aggressive nature and poor prognosis. Promising results with a biomarker directed approach have recently been seen in two separate phase II trials with the anti-claudin 18.2 (CLDN18.2) antibody, zolbetuximab (FAST study), and the FGFR2-IIIb antibody, bemarituzumab (FIGHT study) in patients with CLDN18.2-positive and FGFR2-IIIb-positive gastric cancer, respectively. 62 Other areas of exploration include evaluation of antibody–drug conjugates, chimeric antigen receptor T-cell technology and targeting of bispecific T-cell engager cells. 62
The VICTORY trial also examined circulating tumour (ct) DNA, using various assays and baseline and at CT evaluation of treatment effect. This was based on previous observations confirming genomic heterogeneity between primary tumours and untreated metastases and high concordance between alterations seen in metastatic tissue and ctDNA. 63 The VICTORY trial found high concordance (89.5%) between tumour and ctDNA for MET amplification (using Guardant360 ctDNA assay) and with KRAS status (76.9%, using an Astra Zeneca assay). 61 Others have shown that ctDNA could be used to monitor and identify mechanisms of trastuzumab resistance in patients with advanced HER2-positive GC/GEJ undergoing trastuzumab treatment. 64 Taken together, these examples highlight the potential future role of ctDNA for identifying patients for targeted therapies and in monitoring for therapy resistance mechanisms.
Building on this promise phase II data, key ongoing phase III trials include the FORTITUDE101 study, evaluating bemarituzumab or placebo and chemotherapy in patients with previously untreated advanced GA/GEJ and Fibroblast Growth Factor Receptor 2b (FGFR2b) overexpression (NCT05052801); and the Spotlight study, evaluating zolbetuximab and mFOLFOX6 chemotherapy compared to placebo and mFOLFOX6 chemotherapy in patients with claudin 18.2 positive, HER2-negative previously untreated advanced GA/GEJ (NCT03504397). The GLOW phase III study evaluating first-line zolbetuximab and CAPOX compared with placebo and CAPOX in patients with advanced GA/GEJ and CLDN18.2-positive, HER2-negative disease has completed accrual (NCT03653507).
In summary, HER2 was the first biologic subgroup for treatment selection and new directions here have focused on understanding and detecting resistance in HER2-positive patients. Advances in the molecular understanding of advanced/metastatic GC/GEJ with the application of next generation sequencing and ctDNA have identified novel targets, such as FGFR2 and CLDN18.2 with targeted therapies in phase III development. Selection of patients for benefit from ICIs is an ongoing area of research. PD-L1 CPS has been the leading biomarker, but the optimal cut-off for treatment benefit remains to be defined across all patients.
Current treatment algorithm and patient selection for treatment
The approaches described with genomic profiling provide renewed hope for patients with advanced/metastatic GC/GEJ to obtain meaningful and durable disease control and prolonged survival with modern treatments. However, in the meantime, outside of HER2 positive and MSI-H disease, an empiric approach to treatment selection by line of therapy is the best on offer to the majority of patients (Figure 1). Chemotherapy continues to hold an important place in the treatment algorithm from first- to third-line settings in suitably fit patients. Empiric treatment selection is driven by clinical factors such as race (Asian versus rest of world based on evidence), performance status, disease burden and patient age and co-morbidity, and country-specific drug access. Antiangiogenic therapy with ramucirumab has proven its place in the second-line setting and continues to be explored in refractory disease with antiangiogenic TKIs, alone or in combination with ICIs. Immunotherapy with the PD-1 inhibitor nivolumab has shown to be effective in the first-line setting in combination with platinum/fluoropyrimidine chemotherapy. ICIs have not shown superior benefit in the second-line or third-line settings when compared with chemotherapy. In Asian patients having received two or more prior lines of therapy, nivolumab is effective compared with placebo. Identifying the patient most likely to benefit from ICIs is currently an area of ongoing research. Although PD-L1 CPS has been used to stratify patients in the CheckMate 649 study of nivolumab and chemotherapy, 31 the optimal cut-off for maximum benefit with ICIs and the implementation of CPS reporting is still evolving in the clinic.
