Abstract
Background:
Talazoparib plus enzalutamide showed significant improvement of progression-free survival in metastatic castration-resistant prostate cancer (mCRPC) patients with alterations in homologous recombination repair (HRR) genes.
Objectives:
This study aimed to evaluate the cost-effectiveness of HRR status-guided use of talazoparib plus enzalutamide in patients with mCRPC from the U.S. payer perspective.
Design:
A partitioned survival model was used to simulate outcomes of a hypothetical cohort of mCRPC patients.
Methods:
Three treatment strategies were evaluated: (1) talazoparib plus enzalutamide for HRR-deficient patients and enzalutamide for non-HRR-deficient patients (HRR-guided talazoparib plus enzalutamide), (2) talazoparib plus enzalutamide for patients with or without HRR deficiency (empirical talazoparib plus enzalutamide), and (3) enzalutamide for patients with or without HRR deficiency (empirical enzalutamide). Primary outcomes were direct medical costs, life-years (LYs), quality-adjusted life-years (QALYs), and incremental cost per QALY gained (incremental cost-effectiveness ratio, ICER). Sensitivity analyses were performed to examine the robustness of model results.
Results:
In base-case analysis, the empirical enzalutamide strategy had the lowest cost and QALYs gained (US$752,383; 2.4764 QALYs), followed by HRR-guided talazoparib plus enzalutamide (US$878,517; 2.6490 QALYs), and empirical talazoparib plus enzalutamide (US$1,201,221; 2.8418 QALYs). The ICER of the HRR-guided talazoparib plus enzalutamide group (vs empirical enzalutamide) was US$730,671 per QALY gained, and the ICER of empirical talazoparib plus enzalutamide (vs HRR-guided talazoparib plus enzalutamide) was US$1,673,457 per QALY gained. Both ICERs of the HRR-guided strategy and empirical combination therapy were higher than the willingness-to-pay threshold (US$100,000 per QALY) and were not accepted as cost-effective. Sensitivity analyses found drug costs of talazoparib and enzalutamide to be the key influential factors on the cost-effectiveness of HRR-guided therapy.
Conclusion:
HRR-guided talazoparib plus enzalutamide does not appear to be cost-effective for mCRPC patients from the U.S. payer perspective.
Keywords
Introduction
Prostate cancer remains one of the most significant cancer-related health concerns among men globally. In the United States (US), prostate cancer accounts for 29% of all new cancer cases in men, with an estimated 288,300 new cases.1,2 The incidence of homologous recombination repair (HRR) gene alterations is 24.6% in metastatic castration-resistant prostate cancer (mCRPC) patients in the US. 3 mCRPC patients with HRR alterations are associated with improved survival treated with poly (ADP-ribose) polymerase (PARP) inhibitors.4,5
Enzalutamide is an antagonist of the androgen receptor, and is the standard treatment approved for mCRPC. 6 Talazoparib, a PARP inhibitor, works by trapping PARP on single-strand DNA breaks, preventing DNA repair. 7 The TALAPRO-2 is a randomized, double-blind, phase III clinical trial of talazoparib with enzalutamide versus enzalutamide as first-line treatment in 805 mCRPC patients with asymptomatic or mildly symptomatic mCRPC receiving ongoing androgen deprivation therapy. The combination therapy showed a 37% lower risk of radiographic progression or death than enzalutamide alone. 8 The effect of first-line talazoparib with enzalutamide in HRR-deficient mCRPC patients was further analyzed. When compared with enzalutamide alone, the combination therapy significantly improved radiographic progression-free survival (rPFS; hazard ratio (HR) 0.45; 95% confidence interval (CI), 0.33–0.61; p < 0.0001) in the HRR-deficient cohort (n = 399). 9 In order to assist the informed decision-making process on treatment selection for mCRPC patients with and without HRR deficiency, we aim to evaluate the cost-effectiveness of HRR status-guided use of talazoparib plus enzalutamide from the U.S. payer perspective.
Methods
Model design
A partitioned survival model was designed to simulate outcomes of a hypothetical cohort of mCRPC patients with unknown status of HRR deficiency (Figure 1). The partitioned survival model utilizes independent survival curves to derive the proportion of patients in the corresponding predefined health states over time10,11: Progression-free, post-progression, and death. Three treatment strategies were evaluated in the present model: (1) Talazoparib plus enzalutamide for HRR-deficient patients and enzalutamide for non-HRR-deficient patients (HRR-guided talazoparib plus enzalutamide), (2) talazoparib plus enzalutamide for patients with or without HRR deficiency (empirical talazoparib plus enzalutamide), and (3) enzalutamide for patients with or without HRR deficiency (empirical enzalutamide). The model time horizon was 10 years (with monthly cycle) for the estimation of long-term outcomes, including direct medical cost, life-years (LYs), and quality-adjusted life-years (QALYs) gained by each treatment strategy.
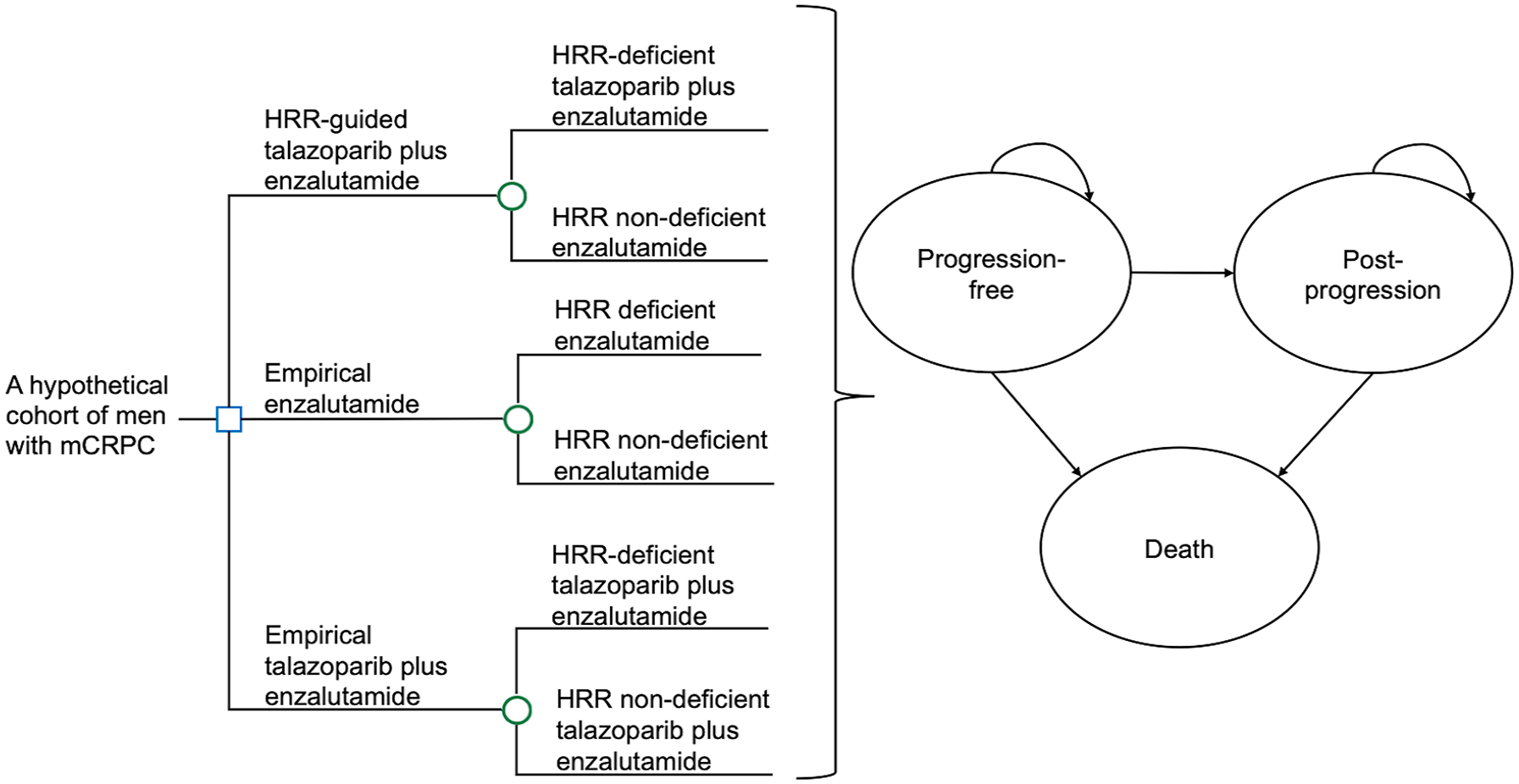
Simplified partitioned survival model of HRR-guided talazoparib plus enzalutamide, empirical enzalutamide, and empirical talazoparib plus enzalutamide for mCRPC treatment.
In the HRR-guided talazoparib plus enzalutamide group, all patients were first tested by the next-generation sequencing (NGS) on the HRR status. Based on the NGS results, the HRR-deficient patients received talazoparib (0.5 mg orally once daily) plus enzalutamide (160 mg orally once daily), and enzalutamide (160 mg orally once daily) for non-HRR-deficient patients. In the empirical treatment groups, all patients received talazoparib plus enzalutamide or enzalutamide alone disregarding the HRR status. All patients received the assigned treatment until progression or death. Docetaxel, abiraterone, and cabazitaxel were options of subsequent therapy provided for symptom control in patients with disease progression. The reporting of this study conforms to the CHEERS statement (detailed in the Supplemental File). 12
Clinical inputs
All model inputs are listed in Table 1. A literature search on MEDLINE over the period 2000–2024 was performed using keywords such as “metastatic castration-resistant prostate cancer,” “mCRPC,” “HRR status,” “overall survival,” “progression-free survival,” “first-line treatment,” “talazoparib,” and “enzalutamide.” The selection criteria of clinical trials were: (1) reports in the English language; (2) adult patients with mCRPC; and (3) PFS, overall survival (OS), or adverse event rates were reported. Preferred studies were meta-analyses and randomized controlled trials. When multiple randomized trials were available for the same model input, the weighted average was used as the base-case value, and the high and low values formed the range for sensitivity analysis.
Clinical, costs, and utility model inputs in the model.
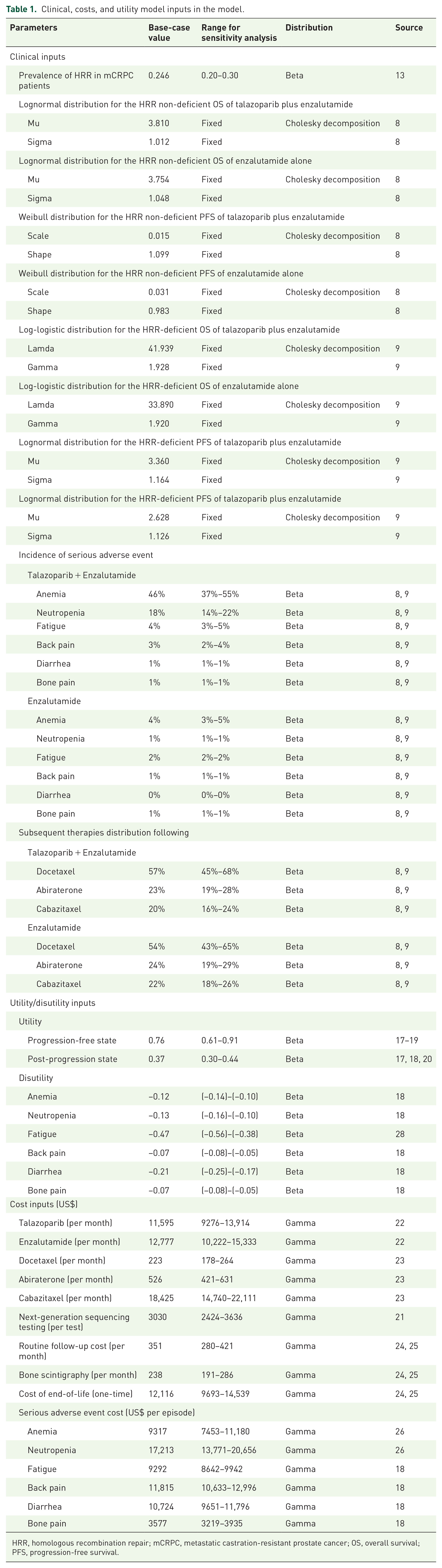
HRR, homologous recombination repair; mCRPC, metastatic castration-resistant prostate cancer; OS, overall survival; PFS, progression-free survival.
The prevalence of HRR-deficient in mCRPC patients in the US (24.9%; range 11%–27% ) was approximated from a cross-sectional analysis using two real-world databases: the Clinico-Genomic Database, and the American Association for Cancer Research Project Genomics Evidence Neoplasia Information Exchange. 3 The PFS and OS in HRR-deficient and non-HRR-deficient patients treated with talazoparib plus enzalutamide and enzalutamide alone were estimated from the Kaplan–Meier survival curves reported by the TALAPRO-2 trial.8,9 Survival data points were extracted from the survival curves by using Engauge digitizer 12.1 to generate patient-level data. 13 The survival rate of PFS and OS was extrapolated by using parametric survival curves. The Akaike information criterion, the Bayesian information criterion, a visual inspection of fit, and the median survival fitting were used to select the appropriate distribution.14,15 The Log-logistic distribution was used for the HRR-deficient OS of talazoparib plus enzalutamide and enzalutamide alone, while the Lognormal distribution was used for the HRR-deficient PFS of talazoparib plus enzalutamide and enzalutamide alone. The Lognormal distribution was used for the HRR non-deficient OS of talazoparib plus enzalutamide and enzalutamide alone, while the Weibull distribution was used for the HRR non-deficient PFS of talazoparib plus enzalutamide and enzalutamide alone.
The probability of survival at the time point of t with the Log-logistic distribution for HRR-deficient OS was estimated by the following formula:

The probability of survival at the time point of t with the Lognormal distribution for HRR-deficient PFS and HRR non-deficient OS was estimated by the following formula:

The probability of survival at the time point of t with the Lognormal distribution for HRR non-deficient PFS was estimated by the following formula:

Utility inputs
The utility inputs are shown in Table 1. The QALYs expected by each subject were estimated from the cumulative time spent in a health state and the health state-specific utility value. The model included three health states: progression-free, post-progression, and death. To account for the impact of treatment-related serious adverse events (SAEs) of Grade 3 or higher, the utility of the progression-free state was adjusted accordingly to include SAE-specific disutility. Utility and disutility estimates were sourced from published reports on quality of life and health economic evaluations of mCRPC.16–19 QALYs were discounted annually at a rate of 3% throughout the model’s time horizon. The incidences of SAEs associated with talazoparib plus enzalutamide and enzalutamide alone were derived from the results reported in the TALAPRO-2 trial.8,9
Cost inputs
The cost analysis was conducted on direct medical costs from the perspective of the U.S. payer. Direct costs included NGS testing for HRR deficiency, costs per month for talazoparib and enzalutamide in a progression-free state, costs per month for docetaxel, abiraterone, and cabazitaxel in a post-progression state, and costs of managing SAEs. The cost of NGS testing was retrieved from the Centers for Medicare & Medicaid Services Clinical Laboratory Fee Schedule 2024. 20 The costs of talazoparib, enzalutamide, abiraterone, and cabazitaxel therapy during PFS were estimated from Medicare Part D Monthly Prescription Drug Plan Formulary and Pharmacy Network Information. 21 The costs of docetaxel therapy were estimated from the Medicare Part B Drug Average Sales Price data. 22 Healthcare resource utilization (including routine follow-up cost, bone scintigraphy cost, end-of-life cost, and management cost of treatment-related SAEs per episode) were obtained from the published health economics analyses.19,23–25 All costs were converted to 2024 US dollars using the CCEMG–EPPI Centre Cost Converter v.1.7. 26 The costs accumulated over the 10-year model timeframe were discounted with an annual rate of 3%.
Cost-effectiveness and sensitivity analyses
The analyses were performed using TreeAge Pro 2024 (TreeAge Software Inc., Williamstown, MA, USA) and Microsoft Excel 16.9 (Microsoft Corporation, Redmond, WA, USA). A treatment option was considered dominated if it resulted in lower QALYs at a higher cost versus an alternative, and the dominated option was excluded from further cost-effectiveness evaluation. When a treatment option yielded higher QALYs at an increased cost versus an alternative, the incremental cost-effectiveness ratio (ICER) of the more effective option was calculated by the equation: Δcost/ΔQALYs. The present analysis adopted US$100,000 per QALY as the willingness-to-pay (WTP) threshold in the base-case analysis.16,17,19,23–25 A treatment option was cost-effective if (1) it gained higher QALYs with cost saving, or (2) it gained higher QALYs at increased cost and the ICER was less than the WTP threshold.
Sensitivity analyses were performed by one-way sensitivity analysis on all model inputs to identify threshold values of influential factors. The probabilistic sensitivity analysis was performed using Monte Carlo simulation. The direct cost and QALYs of each study arm were recalculated 10,000 times by randomly drawing each of the model inputs from the probability distribution specified in Table 1. The incremental costs and incremental QALYs of the 10,000 simulations were presented in scatter plots.
Results
Base-case analysis
Expected direct medical costs and QALYs for the treatment strategies (HRR-guided talazoparib plus enzalutamide, empirical enzalutamide, and empirical talazoparib plus enzalutamide) in the base-case analysis are shown in Table 2. The empirical enzalutamide strategy had the lowest cost and QALYs gained (US$752,383; 2.4764 QALYs), followed by HRR-guided talazoparib plus enzalutamide (US$878,517; 2.6490 QALYs), and empirical talazoparib plus enzalutamide (US$1,201,221; 2.8418 QALYs). The ICER of HRR-guided talazoparib plus enzalutamide group (vs empirical enzalutamide) was US$730,671 per QALY gained, and the ICER of the empirical talazoparib plus enzalutamide (vs HRR-guided talazoparib plus enzalutamide) was US$1,673,457 per QALY gained. Both ICERs of the HRR-guided strategy and empirical combination therapy were higher than the WTP threshold (US$100,000 per QALY) and were not accepted as cost-effective.
Base-case analysis results.

HRR, homologous recombination repair; ICER, incremental cost-effectiveness ratio; QALY, quality-adjusted life year.
Sensitivity analysis
One-way sensitivity analysis found that the base-case results were robust to variation of all model inputs, and no threshold value in model inputs was identified. The top 10 influential factors of the ICERs of HRR-guided strategy versus empirical enzalutamide (Figure 2(a)) and empirical talazoparib plus enzalutamide versus HRR-guided strategy (Figure 2(b)) are presented in tornado diagrams. The top 5 most influential factors were the utilities of progression-free state and post-progression state, costs per month of talazoparib, enzalutamide, and cabazitaxel. We further conducted a two-way sensitivity analysis to examine the interaction of monthly costs of enzalutamide (ranged from base-case value US$12,777 to US$0) and talazoparib (ranged from base-case value US$11,595–US$0) on the cost-effectiveness outcomes. The results of the two-way sensitivity analysis showed that the HRR-guided combination therapy became accepted as cost-effective if talazoparib was <US$5822 per month (>50% reduction), and enzalutamide was <US$8656 per month (>32% reduction; Figure 3).
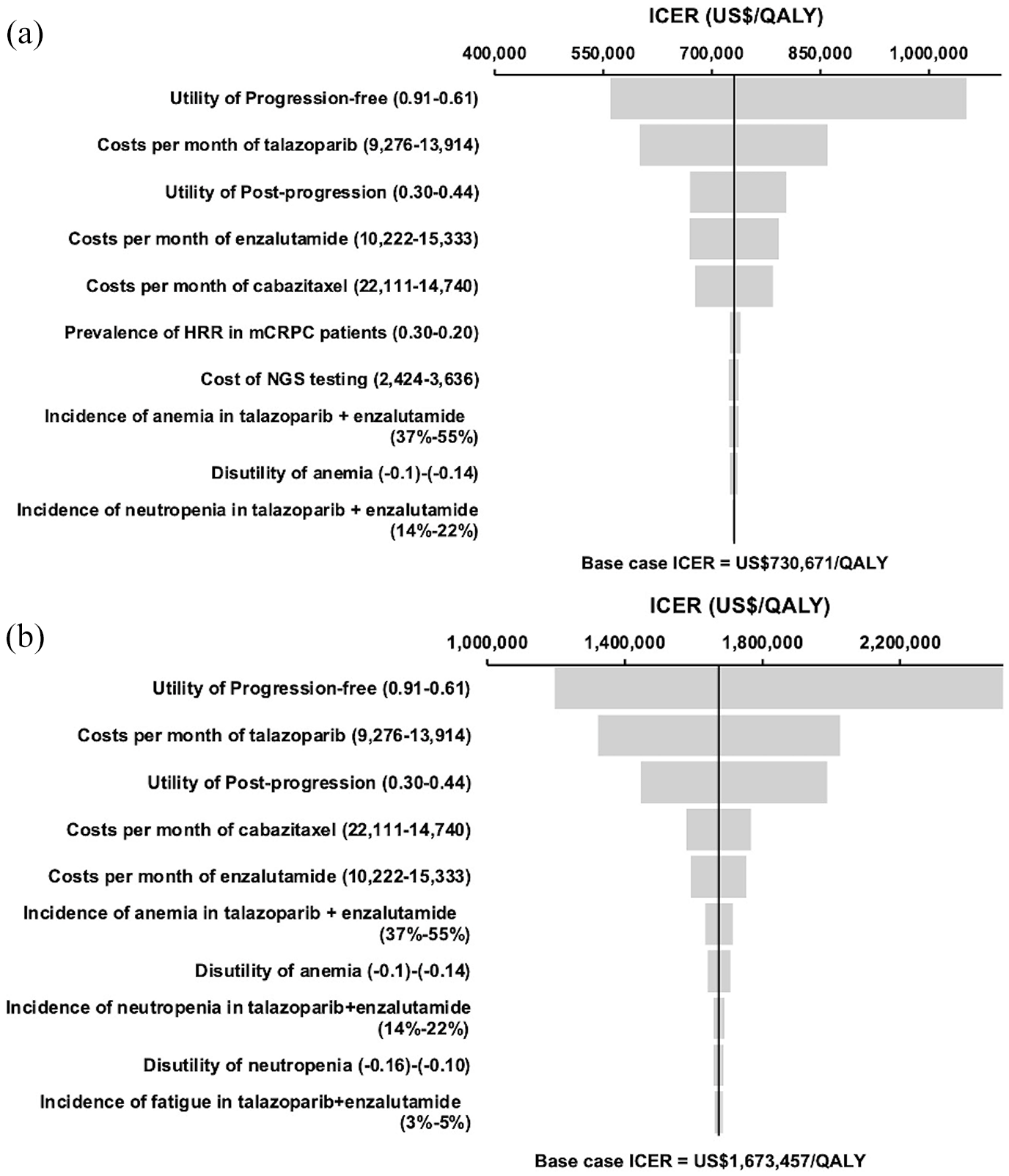
Tornado diagrams of one-way sensitivity analysis of influential parameters on ICER (WTP = US$100,000/QALY) of (a) HRR-guided talazoparib plus enzalutamide versus empirical enzalutamide; (b) empirical talazoparib plus enzalutamide versus HRR-guided strategy.

Two-way analysis on the cost of talazoparib per month versus the cost of enzalutamide per month at the willingness-to-pay threshold (US$100,000/QALY). Yellow area: combinations of monthly costs of enzalutamide and talazoparib for “empirical enzalutamide” to be cost-effective; blue area: combinations of monthly costs of enzalutamide and talazoparib for “HRR-guided talazoparib plus enzalutamide” to be cost-effective; red area: combinations of monthly costs of enzalutamide and talazoparib for “empirical talazoparib plus enzalutamide” to be cost-effective.
The probabilistic sensitivity analysis was performed by 10,000 Monte Carlo simulations. Figure 4(a) shows the scatter plots of the increment costs and QALYs gained by the HRR-guided strategy versus the empirical enzalutamide. HRR-guided strategy gained higher QALYs at higher cost versus empirical enzalutamide by 0.1726 QALYs (95% CI, 0.1722–0.1730; p < 0.01) and US$126,251 (95% CI, US$126,224–126,278; p < 0.01), respectively. Figure 4(b) shows the scatter plots of the incremental cost and QALYs gained by empirical talazoparib plus enzalutamide versus HRR-guided strategy. When compared with the HRR-guided strategy, empirical talazoparib plus enzalutamide gained higher QALYs by 0.1928 QALYs (95% CI, 0.1921–0.1934; p < 0.01) with an incremental cost of US$322,725 (95% CI, US$322,822–322,628; p < 0.01).
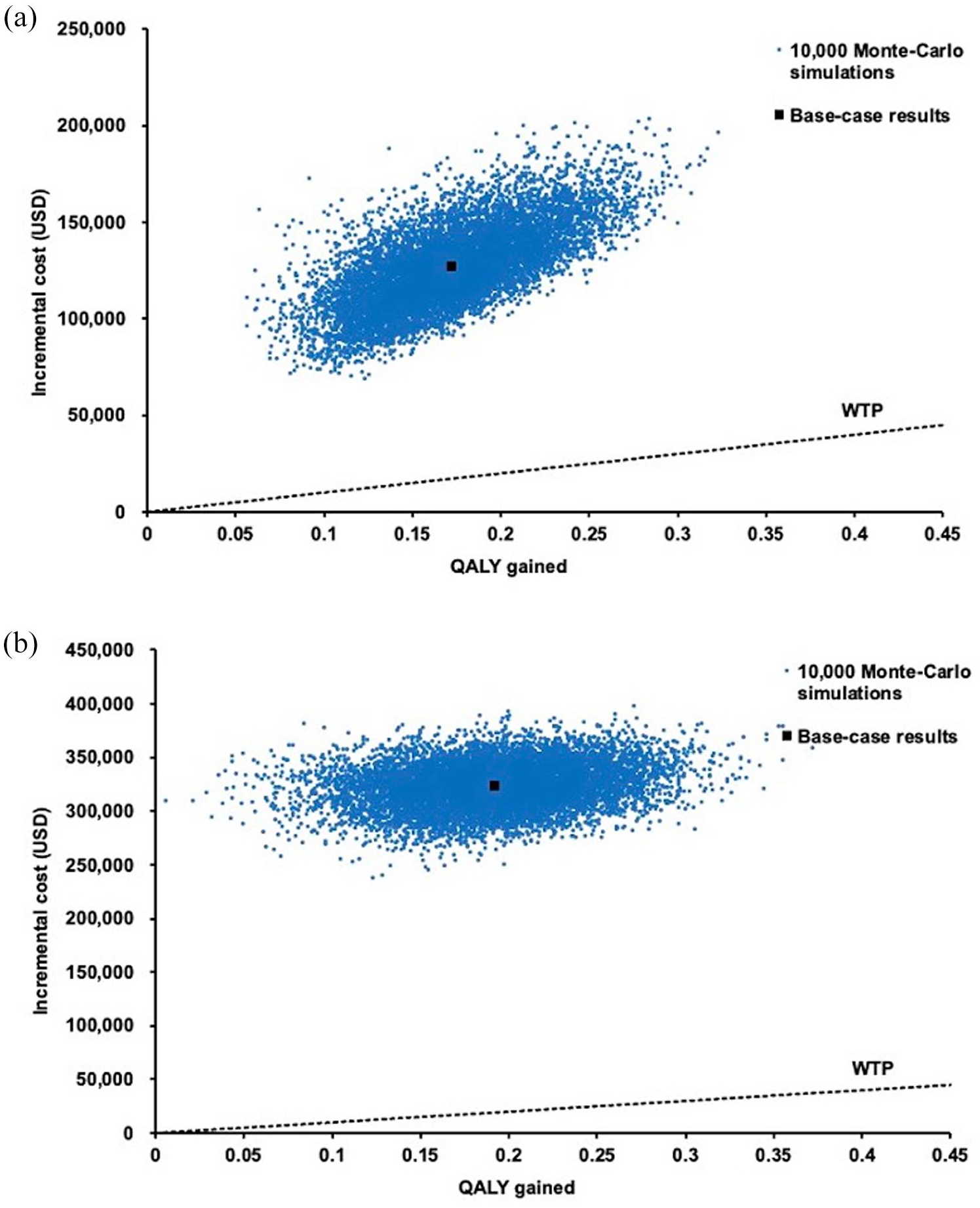
Scatter plot of incremental cost against QALY gained by (a) HRR-guided talazoparib plus enzalutamide versus empirical enzalutamide; (b) empirical talazoparib plus enzalutamide versus HRR-guided strategy.
Discussion
Prostate cancer that progressed despite ongoing androgen deprivation therapy with evidence of distant metastasis is classified as mCRPC. 27 Based on the U.S. National Comprehensive Cancer Network Guideline, and the European Association of Urology–European Association of Nuclear Medicine–European Society for Radiotherapy and Oncology–European Society of Urogenital Radiology–International Society of Urological Pathology–International Society of Geriatric Oncology Guidelines on mCRPC, the preferred options include abiraterone, docetaxel, or enzalutamide. In HRR mutation-positive mCRPC patients, targeted therapies such as talazoparib plus enzalutamide are recommended (category 1).28,29 The clinical effectiveness of talazoparib plus enzalutamide for mCRPC is supported by evidence from phase III trials TALAPRO-2 trial.8,9 The combination therapy significantly improved rPFS when compared to enzalutamide alone, with a HR of 0.63 (95% CI, 0.51–0.78). The median rPFS was not reached for the talazoparib plus enzalutamide group (95% CI, 27.5 months to not reached), compared with 21.9 months (95% CI, 16.6–25.1) for the enzalutamide group. In subgroup analysis of HRR-deficient patients, the benefit was more pronounced as rPFS was significantly prolonged (median not reached at the time of the analysis for the talazoparib group vs 13.8 months for the placebo group; HR, 0.45; 95% CI, 0.33–0.61; p < 0.0001).8,9 Secondary outcomes also favored the combination group, including higher confirmed objective response rate in measurable disease (67% vs 40%), prolonged time to prostate-specific antigen progression (HR, 0.41), and delayed initiation of cytotoxic chemotherapy (HR, 0.46).8,9 A significant benefit was also seen in PFS on subsequent therapy, with HR 0.57 (95% CI, 0.39–0.85).8,9 Despite the guidelines’ recommendations of testing for alterations in HRR genes in patients with metastatic prostate cancer, most mCRPC patients in real-world settings (62.3%) do not receive HRR testing, limiting the application of biomarker-guided treatment. 30 The three treatment strategies analyzed in this study are consistent with current clinical guidelines and regulatory approvals in major healthcare settings, including the U.S. Food and Drug Administration and European Medicines Agency.31,32 While specific recommendations and indications vary across regions, these strategies represent clinically relevant approaches in the management of mCRPC.
This is the first study to evaluate the cost-effectiveness of HRR-guided strategy for mCRPC in the US The current findings demonstrated that the empirical combination strategy was the most costly strategy with the highest QALYs gained, followed by HRR-guided talazoparib plus enzalutamide and empirical enzalutamide. The base-case ICERs indicated that the HRR-guided strategy was not cost-effective from the U.S. payer’s perspective. The one-way and probabilistic sensitivity analyses supported the robustness of the base-case findings. The one-way sensitivity analysis identified that the costs per month of talazoparib, enzalutamide, and cabazitaxel, and the utilities of progression-free state and post-progression state were the most influential factors. Among these, the utility of progression-free state and costs per month of talazoparib showed the greatest impact. A higher utility value of progression-free state would increase the incremental QALYs gained and reduce the ICER of the HRR-guided strategy. In contrast, a higher cost of talazoparib per month would increase the incremental cost and increase the ICER of the HRR-guided strategy. The two-way sensitivity analysis identified the potential drug price thresholds of talazoparib (price reduction by >50%) and enzalutamide (price reduction by >32%) for the HRR-guided strategy to be cost-effective.
Beside HRR-guided therapy, there are treatments targeting other genetic mutations (such as BRCA, AR pathway, and PI3K/AKT alterations) which are also known to contribute to mCRPC progression. 33 A study in Australia found BRCA-guided olaparib treatment not cost-effective, with an ICER (AU$143,613 per QALY) higher than the WTP threshold (AU$100,000 per QALY). 34 Similar to our study, the cost of olaparib and the utility in the progression-free state were key factors influencing the results of this cost-effectiveness analysis. The present findings, together with results of the prior study, implied the challenges faced by gene mutation-guided treatments in mCRPC to achieve acceptable cost-effectiveness, highlighting the need for identifying the modifiable influential factors (such as drug price) and corresponding thresholds to improve the cost-effectiveness of gene mutation-guided therapy. Furthermore, the combination of multiple targeted therapies, such as the use of PARP inhibitors with immune checkpoint inhibitors or androgen receptor signaling inhibitors, could offer additional clinical benefits.35,36 Future research on the cost-effectiveness of combination therapies for different genetic alterations in mCRPC is warranted.
In addition to mCRPC, genetic mutation-guided therapies have also been evaluated in other cancers, such as non-small-cell lung cancer (NSCLC). Various studies have assessed the cost-effectiveness of EGFR-guided therapies for NSCLC, and these findings underscore the potential for genetic testing to improve treatment outcomes in NSCLC by targeting specific mutations.37–40 While genetic mutation-guided therapies in lung cancer demonstrated favorable cost-effectiveness, challenges remain in other cancer types to identify a cost-effective precision medicine-based treatment strategy. The differences in cancer type, mutation prevalence, treatment costs, and comparator choice would result in substantial variability in economic outcomes. Future research should explore the cost-effectiveness of genetic mutation-guided treatments across various cancer types to provide more comprehensive insights into the economic viability of precision medicine.
Recent studies have increasingly explored the role of radiotherapy (RT), particularly stereotactic body radiotherapy (SBRT) in managing mCRPC. In terms of limiting disease progression, the phase II ARTO trial showed that combining SBRT with abiraterone acetate significantly improved biochemical response and PFS in oligometastatic mCRPC patients compared to systemic treatment alone. 41 A subsequent subgroup analysis suggested that applying SBRT after first progression might extend time to further progression, although the difference was not statistically significant (p = 0.489). 42 Regarding symptom control and prevention, a prospective cohort study reported that prostate SBRT was associated with a statistically significant improvement in time to symptomatic progression (p = 0.019), but no significant difference in PFS (p = 0.249) or OS (p = 0.230) compared to the control group. 43 Retrospective analyses showed that radiotherapy in oligoprogressive mCRPC was safe and seemed to prolong the efficacy of androgen receptor-target therapy in patients who otherwise would have switched to systemic treatment. The findings were yet limited by the retrospective study design.44,45 The rationale of metastasis-directed therapy for controlling resistant clones has been discussed by experts, and the lack of standardized definitions, biomarkers, or randomized evidence was emphasized. 46 These clinical effects may have potential cost-effectiveness implications. A health economic modeling study indicated that RT is a cost-effective palliative treatment for metastatic prostate cancer compared to chemotherapy. 47 Deferring use of high-cost systemic agents through metastasis-directed RT could potentially reduce total treatment costs. However, it must be emphasized that there is no level 1 evidence supporting the use of RT to metastases in mCRPC for delaying systemic treatments with proven survival benefit. Thus, while RT holds clinical promise in this setting, its integration into standard care and cost-effectiveness evaluation requires confirmation in prospective randomized trials.
There were some limitations in the present study. The model simplified real-life events of mCRPC therapy. The model included SAEs of talazoparib and enzalutamide with high incidence or with severe outcomes requiring medical therapy. The less SAEs were not included, and the impact of adverse drug events was therefore not fully represented. The clinical model inputs were mostly retrieved from findings reported by the TALAPRO-2 study, which might not fully reflect treatment outcomes or adverse event rates in routine clinical practice. The present model examined talazoparib and enzalutamide for the first-line treatment of HRR-deficient mCRPC, and the use of chemotherapy was not included due to the lack of efficacy evidence of chemotherapy in mCRPC patients with HRR mutation. The use of chemotherapy for HRR-deficient patients warrants further cost-effectiveness evaluation. The model assumed universal implementation of HRR testing in the HRR-guided strategy, whereas a substantial proportion of patients might not receive multigene tumor testing in clinical practice. Also, the payer perspective did not include the patients’ loss of productivity and might therefore underestimate the impact of treatment on indirect costs.
Conclusion
HRR-guided talazoparib plus enzalutamide does not appear to be cost-effective for mCRPC patients from the U.S. payer perspective. Drug costs of talazoparib and enzalutamide were the key influential factors on the cost-effectiveness of HRR-guided therapy. The combination therapy (with or without HRR guidance) gained higher QALYs at a higher cost than the monotherapy, and the ICERs of the combination therapy exceeded the WTP threshold in both base-case and sensitivity analyses. Of the three examined treatment options, monotherapy with enzalutamide was therefore the preferred cost-effective treatment.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251356109 – Supplemental material for HRR deficiency-guided use of talazoparib plus enzalutamide in patients with metastatic castration-resistant prostate cancer: a cost-effectiveness analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359251356109 for HRR deficiency-guided use of talazoparib plus enzalutamide in patients with metastatic castration-resistant prostate cancer: a cost-effectiveness analysis by Mingjun Rui, Yingcheng Wang, Qiran Wei and Joyce H. S. You in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
