Abstract
Background:
Currently, guidelines prohibit the addition of targeted drugs in neoadjuvant chemotherapy (NAC) for initially resectable colorectal liver metastasis (CRLM).
Objective:
Nevertheless, efficacy data of NAC combined with bevacizumab (Bev) for initially resectable CRLM with risk factors for recurrence (RFR) are lacking.
Designs:
We conducted a multicenter real-world cohort study to retrospectively analyze the efficacy and feasibility of NAC combined with Bev for CRLM with RFR.
Methods:
The patients were divided into the NAC alone group and NAC combined with the Bev group. We designated progression-free survival (PFS), objective response rate (ORR), and overall survival (OS) as the outcomes. Kaplan–Meier, Cox proportional hazards regression models, and subgroup analysis were utilized. RFR was a clinical risk score of 3–5. Subgroup analysis was applied to explore which subgroup was more suitable for NAC combined with Bev.
Results:
Between 2015 and 2020, this multicenter real-world study encompassed 335 CRLM patients from six medical centers who underwent curative hepatectomy following NAC. Two hundred seventeen patients were in the NAC alone group, and 118 received NAC combined with Bev. The NAC alone group exhibited an ORR of 51.15%, compared to 66.95% in the NAC combined with Bev (
Conclusion:
For initially resectable CRLM patients with RFR, NAC combined with Bev exhibited a higher ORR and longer PFS.
Chinese clinical trial registry:
ChiCTR2400082966.
Introduction
The 2024 cancer statistics reveal that colorectal cancer (CRC) maintains elevated incidence and mortality rates. 1 During the entire course of CRC, nearly half of the patients will develop liver metastasis.2,3 Liver metastasis is a major factor affecting the overall prognosis of CRC. 4 The primary objective for colorectal liver metastasis (CRLM) patients is to extend progression-free survival (PFS).5,6
Currently, curative hepatectomy remains the only approach that can provide the possibility of cure. 7 Despite the benefits of curative hepatectomy, the majority of patients will encounter intrahepatic recurrence or distant metastasis, which substantially diminishes their survival duration.8,9 CRLM patients with risk factors for recurrence (RFR) (clinical risk score (CRS): 3–5) generally exhibit a worse prognosis. 8 For initially resectable CRLM with RFR, the European Society for Medical Oncology (ESMO) and the National Comprehensive Cancer Network (NCCN) recommend neoadjuvant chemotherapy (NAC).10,11 However, the use of targeted therapy in NAC for such patients is a matter of considerable debate.10–14 The 2017 NCCN guidelines specify that the concurrent use of targeted therapy, particularly cetuximab (Cet), is prohibited during NAC for initially resectable CRLM patients. 15 This recommendation is based on the randomized controlled trial published in 2014, which showed that combining NAC with Cet worsened the PFS of initially resectable CRLM patients. 16 For initially resectable CRLM patients with RFR, the role of NAC combined with bevacizumab (Bev) remains unclear.10,11
Bev exerts antitumor effects through the inhibition of angiogenesis by binding to vascular endothelial growth factor. 17 The combination therapy with Bev is widely used in initially unresectable CRLM patients, playing a unique role in improving the prognosis of advanced CRLM patients.18–20 Based on current evidence, we designed a multicenter, real-world retrospective cohort study to explore the efficacy of NAC combined with Bev for resectable CRLM patients with RFR.
Materials and methods
Grouping and study population
Our multicenter, real-world retrospective study analyzed the baseline information and prognostic data of initially resectable CRLM patients with RFR (CRS >2) who were treated from December 2015 to December 2020 at six medical centers: Fujian Provincial Hospital, Zhejiang Cancer Hospital, The First Affiliated Hospital of Fujian Medical University, Zhejiang Provincial People’s Hospital, Hangzhou Red Cross Hospital, and The First Hospital of PuTian City. Prior to treatment, initially resectable CRLM patients with RFR were assessed by the multidisciplinary team (MDT). MDT consists of hepatobiliary surgeons, colorectal surgeons, medical oncologists, and radiologists. These patients underwent NAC and curative hepatectomy. According to the guidelines, MDT at the six included tertiary care centers had nearly uniform inclusion criteria (The procedures and criteria for NAC and surgery were included). The decision to combine treatment with Bev was made in consultation between the MDT and the CRLM patients. In brief, the MDT considered the NAC regimen combined with Bev for CRLM with RFR (CRS >2). In addition, the patient’s wishes need to be considered.
All initially resectable CRLM patients with RFR were screened according to the following criteria (Figure 1). Inclusion criteria: (1) Age over 18 years; (2) The primary tumor pathologically confirmed as colorectal adenocarcinoma and had been radically resected (Resection of the primary tumor can be performed simultaneously with hepatectomy or in a two-stage procedure); (3) Liver metastasis pathologically confirmed as CRC liver metastases; (4) Liver metastasis deemed resectable by the MDT prior to NAC; (5) No prior local treatment before NAC, including radiotherapy, and interventional therapy; (6) CRS score >2 points; (7) Intact important organ functions. Exclusion criteria: (1) Preoperative use of any medication that could interfere with trial results (including hormones, immunosuppressants, etc.); (2) History of malignancy other than CRC; (3) Metastases to other parts of the body other than liver; (4) Important clinical data missing, including loss to follow-up; (5) Any disease assessed to affect the trial results. Initially resectable CRLM patients with RFR patients who met the inclusion criteria were categorized into the NAC alone group and the NAC combined with the Bev group, depending on whether Bev was combined. To ensure the integrity of the study, we investigated and appropriately addressed all issues. The ethics committees at all six participating medical centers approved this multicenter, real-world retrospective cohort study. Chinese Clinical Trial Registry: ChiCTR2400082966. The reporting of this study conforms to the STROBE statement 21 (Supplemental Material 1).

Flow chart of CRLM patients receiving NAC combined with curative hepatectomy.
Comprehensive treatment procedure
Following admission, initially resectable CRLM patients with RFR underwent individualized treatment after discussion by the MDT. Those who met the inclusion criteria commenced NAC. The choice of regimen was a joint decision made by clinicians and patients. The six medical centers involved in this study employed commonly utilized NAC protocols: CapeOX, mFOLFOX6, and FOLFIRI. Imaging assessments were conducted every two cycles during NAC or whenever the condition of the patient changed (if disease progressed, the next course of treatment was determined by the MDT). Bev was discontinued in the last cycle of NAC to ensure that the final use was at least 5 weeks prior to surgery. 22 Following the last cycle of NAC, imaging was needed, and patients whose liver metastases remained resectable proceeded to surgical treatment. All surgeries were carried out by hepatobiliary surgeons with over 10 years of hepatectomy experience. All liver metastasis, including those responsive to NAC or newly developed tumors, were to be resected. The concurrent use of local treatments such as ablation was not permitted in the treatment plan. Postoperatively, the MDT made decisions regarding the use of adjuvant chemotherapy based on the specific circumstances.
Definitions
For CRLM, PFS is the alternative endpoint of OS.5,6 We designated PFS, objective response rate (ORR), and overall survival (OS) as the outcomes. PFS is defined as the time from the date of NAC to the date of disease recurrence, progression, or death. 23 OS is defined as the time from the date of NAC to the date of death or the last follow-up. 23 Complications were defined in accordance with the Clavien–Dindo (CD) classification. 24 The CRS encompasses the following five criteria, with 1 point for each criterion met (3–5 points constituting the high CRS, indicative of high risk of recurrence): nodal positivity of the primary tumor; synchronous liver metastasis or diagnosis of liver metastasis within 12 months of CRC surgery; more than 1 liver metastases; preoperative serum level of carcinoembryonic antigen (CEA) >200 µg/L; and the largest diameter of liver metastasis >5 cm.10,25 Resectability was determined by MDT, ensuring that all liver metastasis could be completely resected with an R0 margin and that sufficient functional liver tissue remained.10,25,26 The MDT assessed the effectiveness of NAC (Using CT and MRI) in accordance with RECIST 1.1 criteria. 27 The group that received only NAC was defined as the NAC alone group, whereas the group that combined NAC with Bev was defined as the NAC combined with Bev group. R0 resection implied the removal of both the CRC and liver metastasis with all detectable tumor masses on imaging and intraoperative exploration being completely resected. General complications included fever, postoperative bleeding, and wound infection.
Follow-up
The six medical centers required patients to undergo disease monitoring every 3 months post-surgery. Tumor markers (CEA, etc.), CT, and endoscopy were collected during the follow-up period. For initially resectable CRLM patients with RFR who showed no tumor progression or distant metastasis within 2 years, the follow-up interval was extended to once every 6 months. For those who remained free of progression or distant metastasis for over 5 years, the follow-up was adjusted to once per year. 23 If any indication of a new tumor was detected during postoperative follow-up examinations, hospitalization for further treatment was necessary. In the event of metastasis or recurrence, the treatment plan needed to be discussed by the MDT, considering options such as surgery, chemotherapy, or local treatment.
Statistical analysis
All data were compiled and subjected to statistical analysis. When continuous variables fulfill the assumptions of normal distribution and variance homogeneity,
Results
Clinicopathological characteristics
From December 2015 to December 2020, 335 initially resectable CRLM patients with RFR were recruited from six medical centers (217 CRLM patients were in the NAC alone group and 118 CRLM patients were in the NAC combined with Bev group). Specifically, the contributions from Fujian Provincial Hospital, Zhejiang Cancer Hospital, The First Affiliated Hospital of Fujian Medical University, Zhejiang Provincial People’s Hospital, Hangzhou Red Cross Hospital, and The First Hospital of PuTian City were 118, 41, 88, 39, 33, and 16 cases, respectively. General clinical data of the initially resectable CRLM patients with RFR are available in Table 1. The decision to combine treatment with Bev was made in consultation between the MDT and the CRLM patients. In brief, the MDT considered the NAC regimen combined with Bev for CRLM with RFR (CRS >2). In addition, the patient’s wishes need to be considered. In the NAC alone group, there were 106 patients under 60 years (48.85%), while in the combined Bev group, there were 58 patients (49.15%). The baseline characteristics of both groups showed no significant deviation (
Baseline characteristics of the included CRLM patients.

Pearson’s χ2 test was used to analyze the basic characteristics.
ASA, American Society of Anesthesiologists; Bev, bevacizumab; CEA, carcinoembryonic antigen; CRLM, colorectal liver metastasis; LN, lymph node; NAC, neoadjuvant chemotherapy.
Chemotherapy and pathological information
Information related to NAC and postoperative pathology can be found in Table 2. All initially resectable CRLM patients with RFR completed at least two cycles of NAC. The NAC alone group and the NAC combined with the Bev group used an average of 4.46 ± 1.22 and 4.57 ± 1.24 cycles of NAC before surgery, respectively (
Chemotherapy and pathological outcomes of CRLM patients.
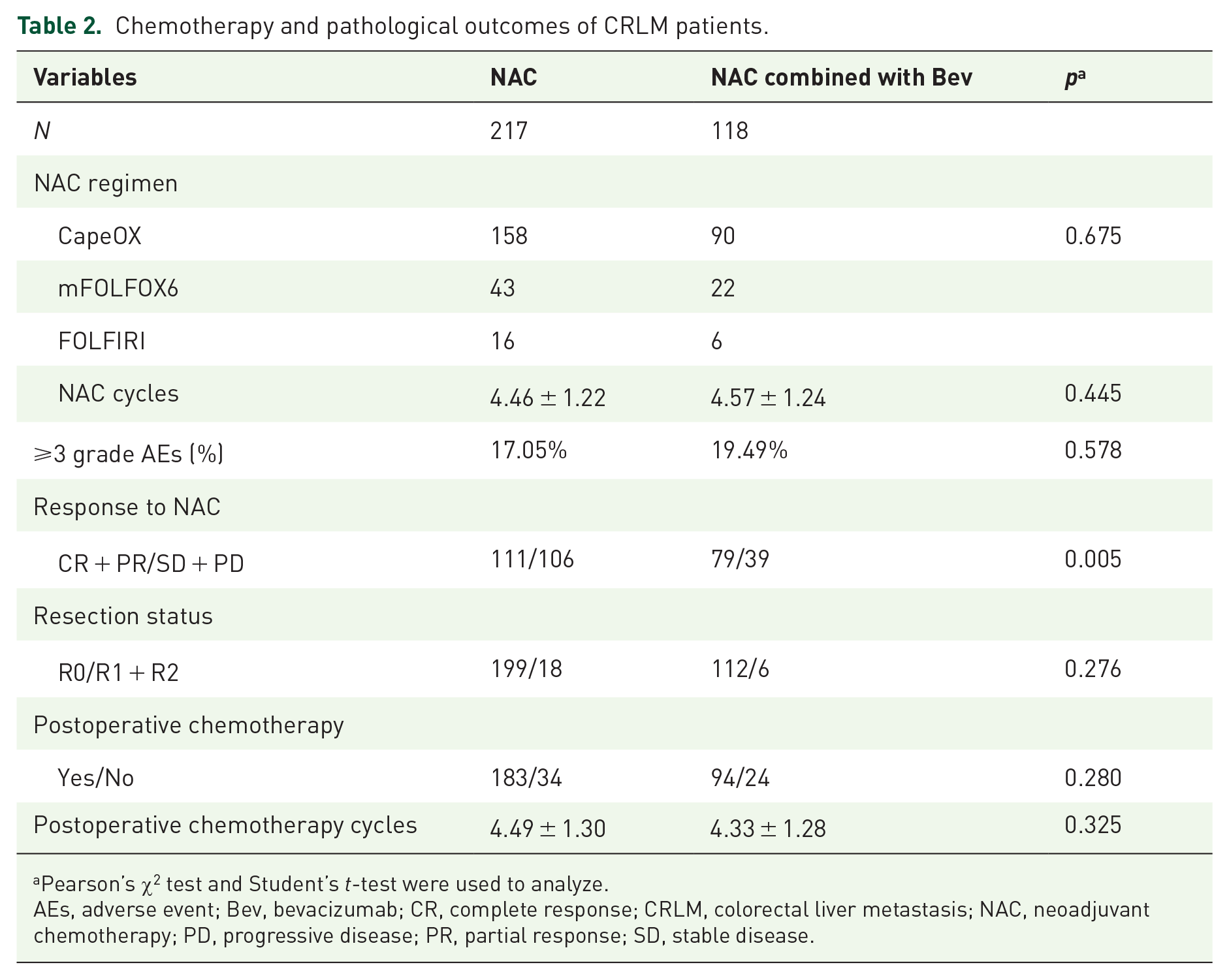
Pearson’s χ2 test and Student’s
AEs, adverse event; Bev, bevacizumab; CR, complete response; CRLM, colorectal liver metastasis; NAC, neoadjuvant chemotherapy; PD, progressive disease; PR, partial response; SD, stable disease.
Perioperative information
Information related to operative details can be found in Table 3. In the NAC alone group, 11.98% (26/217) of patients underwent synchronous CRC curative surgery combined with curative hepatectomy, compared with 10.17% (12/118) in the combined Bev group (
Perioperative outcomes of CRLM patients.

Continuity correction, Pearson’s χ2 test, or Student’s
According to the Clavien–Dindo classification.
Bev, bevacizumab; CRC, colorectal cancer; CRLM, colorectal liver metastasis; LOS, length of hospital stay; NAC, neoadjuvant chemotherapy.
Survival analysis
As of January 2024, the median follow-up time of the 335 resectable CRLM patients with RFR was 35.0 (28.0, 44.0) months. The median PFS was 27.0 (17.0, 37.0) months for the NAC alone and 31.0 (21.0, 40.0) months for the NAC combined with Bev. The NAC combined with Bev had a significantly longer PFS than the NAC alone (log-rank;

Kaplan–Meier survival curve of PFS and OS. (a) PFS, and (b) OS.
Analysis of prognostic factors associated with PFS.

Cox univariate analysis included group (NAC combined with Bev), Gender, Age (years), Primary tumor, ASA, T stage of primary tumor, LN metastasis, CEA at diagnosis (ng/mL), Timing of metastasis, Number of liver metastases, Size of largest lesion (cm), Response to NAC, Resection status, Post-operation complications, and Postoperative chemotherapy. Variables with a significance level of
Bev, bevacizumab; CR, complete response; HR, hazard ratio; NAC, neoadjuvant chemotherapy; PD, progressive disease; PFS, progression-free survival; PR, partial response; SD, stable disease.
The median OS was 33.0 (26.0, 39.0) months for the NAC alone and 38.0 (29.8, 49.0) for the NAC combined with the Bev group. There was no significant difference in OS between the NAC combined with the Bev group and the NAC alone group (log-rank;
Analysis of prognostic factors associated with OS.

Cox univariate analysis included group (NAC combined with Bev), Gender, Age (years), Primary tumor, ASA, T stage of primary tumor, LN metastasis, CEA at diagnosis (ng/mL), Timing of metastasis, Number of liver metastases, Size of largest lesion (cm), Response to NAC, Resection status, Post-operation complications, and Postoperative chemotherapy. Variables with a significance level of
ASA, American Society of Anesthesiologists; Bev, bevacizumab; HR, hazard ratio; NAC, neoadjuvant chemotherapy; OS, overall survival.
Subgroup analysis
Subgroup analysis was conducted to identify which patients would most likely benefit from NAC combined with Bev. Our forest plot reaffirmed previous results (Figures 3 and 4). In various subgroups, NAC combined with Bev still demonstrated superiority over NAC alone in terms of PFS (Figure 3). Interestingly, the advantage of NAC combined with Bev in PFS was particularly pronounced in certain populations, such as those aged ⩽60 years, males, primary colon cancer, T stage (I–II), lymph node metastasis from CRC, synchronous liver metastases, a single liver metastasis, and the largest liver metastasis diameter being ⩾5 cm. Regarding OS, the benefits of NAC combined with Bev were especially significant in males with primary colon cancer and those with an ASA score of I–II.

Forest plot evaluating the impact of NAC combined with curative hepatectomy on PFS.

Forest plot evaluating the impact of NAC combined with curative hepatectomy on OS.
Discussion
This is the first large-sample, multicenter, real-world cohort study comparing the effects of NAC combined with Bev for resectable CRLM patients with RFR. Surprisingly, the comprehensive statistical results of this study revealed that for resectable CRLM patients with RFR, NAC combined with Bev significantly improved PFS and ORR. NAC combined with Bev also exhibited a potential trend toward improvement in OS. As for the concern of increased postoperative complications associated with curative hepatectomy, NAC combined with Bev did not result in an increase. Cox regression and subgroup analysis further validated our findings.
Maximizing the survival span of CRLM patients with RFR stands as a focal point of contemporary research efforts. Administering NAC prior to curative hepatectomy in CRLM patients affords multiple benefits.28,29 Currently, guidelines including ESMO, NCCN, and the Chinese Society of Clinical Oncology recommend that resectable CRLM patients with RFR (CRS >2) receive NAC prior to curative hepatectomy.12,15,26,30,31 However, the prognosis for CRLM patients with RFR remains pessimistic. Targeted drugs have become standard in combination therapy for conversion and palliative treatment of CRLM.12,13,25,26,30–32 Nevertheless, there is controversy regarding the combination of targeted drugs with NAC in resectable CRLM.12,13,15,25,26 Currently, clinical studies on NAC combined with Bev for resectable CRLM are limited, with only some prospective single-arm trials suggesting a good pathological response.33,34 The 2017 NCCN guidelines prohibit the addition of targeted therapies during NAC for initially resectable CRLM patients, 15 which has led to few cases of NAC combined with Bev. We increased the sample size by including more medical centers. In addition, considering the cost-effectiveness and ethical issues associated with prospective randomized trials, we designed this multicenter, real-world cohort study to explore this controversial and highly debated topic. 35
The safety of NAC combined with Bev is fundamental to advancing the application of Bev in resectable CRLM. Surgeons are concerned about the impact of combining Bev on postoperative safety. Adverse events associated with chemotherapy combined with Bev have been previously discussed,
36
which was consistent with our findings. Our research found that NAC combined with Bev did not increase the probability of any postoperative complications. The safety of Bev in post-hepatectomy has been preliminarily confirmed, the result was also validated in a retrospective study in 2014.
37
Compared to NAC alone, no significant difference was observed in the overall complication of NAC combined with Bev (
The pathological response of NAC serves as a potent predictor of long-term prognosis after surgery for CRLM patients.40,41 The multivariable analysis of 305 CRLM patients who received NAC followed by surgical resection suggested that pathological response (
For initially resectable CRLM patients with RFR (CRS >2), the ultimate goal of NAC is to improve PFS and OS. The final outcomes of this study are encouraging. NAC combined with Bev compared to NAC alone showed a clear advantage in PFS and a potential OS benefit for initially resectable CRLM patients with RFR. Multivariable and subgroup analyses further confirm the role of NAC combined with Bev in extending PFS in initially resectable CRLM patients with RFR. Although there was no statistical difference in OS, there still appears to be a potential protective effect of combined Bev on OS. There are several possible explanations for better PFS prognostic advantages of NAC combined with Bev in initially resectable CRLM patients with RFR: (1) Combined Bev achieves higher ORR; (2) NAC combined with Bev may better eliminate local and distant micro-metastases, thereby improving PFS; (3) CRLM patients with RFR experience high postoperative relapse rates and poor prognosis. They are more likely to benefit from combined Bev therapy; (4) NAC combined with Bev can reduce complications such as SOS associated with oxaliplatin. 43 Lower postoperative complications are related to better survival prognosis. 44 The 2016 ESMO guidelines consider the combination with Bev in selected patients. 26 The existing study on NAC combined with Bev for the prognosis of initially resectable CRLM is a retrospective study in 2014. 37 A retrospective cohort study included 501 CRLM patients, comparing those who received FOLFOX chemotherapy alone with those combined with Bev, and suggested that NAC combined with Bev did not improve the 3-year OS and PFS. 37 However, in that study, about one-quarter of CRLM patients were initially unresectable. 37 Moreover, there were significant baseline differences between the two groups in that 2014 study. 37 In contrast to that 2014 study, the two groups in our study had balanced baselines, and the target population was initially resectable CRLM patients with RFR. The fact that NAC combined with Bev only showed benefits in terms of PFS, and not a significant increase in OS, might be attributable to the variance in treatment choices after recurrence. This needs to be further validated in future studies.
Personalized treatment is of great significance for initially resectable CRLM with RFR. Subgroup analysis in this study demonstrated that NAC combined with Bev exhibits significant efficacy in specific patients, including those with primary colon cancer, male, synchronous liver metastases, single liver metastases, and tumor diameters >5 cm. These characteristics may be associated with the clinical efficacy of this treatment strategy. Biological differences between colon and rectal cancers influence therapeutic responses, with patients having colon cancer liver metastases more likely to benefit from antiangiogenic therapy, potentially due to their greater dependence on angiogenesis. 45 Gender differences in responses to anticancer therapy have also garnered attention. Some studies suggest that male patients exhibit a more pronounced response to anti-angiogenic therapy than females, possibly related to the regulatory effects of sex hormones on angiogenesis. 46 In addition, CRLM patients with synchronous liver metastases and tumor diameters >5 cm often exhibit greater tumor burden and more rapid metastatic progression, necessitating antiangiogenic therapy to inhibit disease progression. For patients with single liver metastases, the mechanisms underlying the superior efficacy of NAC combined with Bev require further investigation. It is evident that NAC combined with Bev provides promising efficacy in certain CRLM subgroups, and these subgroup characteristics may serve as references for clinical treatment strategy selection. However, further studies are required to validate the predictive value of these features in guiding CRLM treatment.
These findings have practical clinical significance for initially resectable CRLM patients with RFR. Nonetheless, our study has certain limitations. First, the results are based on a small and specific patient population from six medical centers, which may limit the generalizability of the findings to broader, more diverse populations. Second, the study did not include patients whose tumors progressed to unresectability or those who achieved complete remission without hepatectomy. Lastly, the mutational status (including RAS, RAF, and MMR) is an important target for assessing the prognosis of CRLM patients. 47 Due to the retrospective nature of the study, the KRAS mutation status was not recorded in the cases. These issues need to be addressed by further multicenter, large-sample, and prospective randomized trials.
Conclusion
This large-scale, multicenter, real-world cohort study has yielded promising results. NAC combined with Bev is safe and controllable, providing better pathological remission, and is associated with better PFS. Large-sample prospective studies are needed to further validate the safety and feasibility of NAC combined with Bev for initially resectable CRLM patients with RFR.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251328457 – Supplemental material for Neoadjuvant chemotherapy combined with bevacizumab for resectable colorectal liver metastasis with risk factors for recurrence: a multicenter real-world study
Supplemental material, sj-docx-1-tam-10.1177_17588359251328457 for Neoadjuvant chemotherapy combined with bevacizumab for resectable colorectal liver metastasis with risk factors for recurrence: a multicenter real-world study by Yizhen Chen, Yuanyuan Zheng, Jia Wu, Rong Ye, Hangdong Jia, Zhenyuan Zhou, Weijie Chen, Linwei Xu, Yuhua Zhang and Ming Zheng in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
