Abstract
Objectives:
Gamma glutamyl-transpeptidase (GGT) has been shown as a prognostic marker in many cancers. The aim of this study was to explore whether serum GGT could predict tumor recurrence in patients with liver-confined colorectal cancer liver metastases (CRCLM) undergoing R0 resection.
Methods:
We reviewed patients who had underwent liver surgery for CRCLM. Patients with liver-only metastases that underwent R0 resection were included. Pre-operative serum GGT were classified into either high or low using a cut-off value of 33 U/L for female and 51 U/L for male. Relapse-free survival (RFS) was compared in relation to GGT and other clinicopathological factors.
Results:
Of the 350 patients included, 108 (30.9%) had a high serum GGT. Patients with metachronous liver metastases, number of metastases ⩾2, size of the largest metastasis ⩾3 cm, or a history of neoadjuvant chemotherapy had a higher GGT level (p = 0.001, 0.027, 0.001, and 0.002, respectively). In survival analyses, patients with a high GGT had a shorter RFS than those with a low GGT, with a median RFS of 11.8 versus 30.3 months (p < 0.001). RFS was also associated with the number of metastases, size of the largest metastasis and the delivery of neoadjuvant chemotherapy. In multivariate analysis, GGT remained an independent prognostic factor of RFS.
Conclusions:
Our study demonstrates that the serum GGT level before liver surgery is an adverse prognostic factor of RFS for patients with liver-confined CRCLM.
Keywords
Introduction
Colorectal cancer (CRC) is one of the leading causes of cancer death, with an estimate of 1.8 million new cases globally in 2018. 1 Nearly 20% of CRC patients present with liver metastases at diagnosis, and 30% of patients with localized CRC experience tumor recurrence even after curative resection, with the liver being the most common site.2–4 Surgery is considered the only curative therapy for colorectal cancer liver metastases (CRCLM). It helps to prolong patients’ median overall survival to more than 40 months, and cures nearly 20%. 5 Nevertheless, more than 60% of the patients with CRCLM develop new metastases after R0 resection, most of which occurs within 2 years. 6 There is a need for predictors that can identify patients who may not benefit from such an invasive procedure.
The clinical risk scoring (CRS) system proposed by Fong et al. has been widely used in clinical practice as a tool for predicting outcome after hepatectomy for CRCLM. 7 The system stratifies patients’ prognosis based on five clinicopathological factors. Patients with a higher score have worse survival, and therefore are considered ineligible for surgery. However, the system was built before the introduction of modern chemotherapy for CRC, and the factors it used that were once universally available may now become difficult to obtain. For example, the strategy of simultaneous liver resection and “liver first” has rendered the assessment of nodal status before hepatectomy impossible.8–10 In addition, the use of neoadjuvant chemotherapy can significantly reduce the number of involved lymph nodes and the serum level of carcinoembryonic antigen (CEA), which is likely to weaken the efficacy of this scoring system. 11
Gamma-glutamyl transpeptidase (GGT) is an enzyme that can be detected in many human organs, and serum GGT has been used as an index of liver dysfunction for decades. 12 In addition, it has been shown that GGT plays an important role in tumor progression. Elena et al. found that GGT overexpression facilitated metastatic growth of melanoma cells in mouse liver. 13 Hanigan et al. found that GGT accelerated tumor growth and increased resistance to chemotherapy in human prostate tumor cells. 14 In healthy adults, an elevated serum GGT is associated with a higher risk of many cancers, particularly liver cancer.15,16 He et al. found that GGT was an adverse prognostic factor of survival and a predictor of poor response to chemotherapy in patients with metastatic colorectal cancer. 17
However, whether GGT is a prognostic marker for CRCLM patients remains unexplored. Our study aimed to examine whether the pre-operative serum GGT level predicted tumor recurrence after R0 resection for patients with liver-confined CRCLM.
Patients and methods
Patient selection
We reviewed a consecutive series of 442 patients who had undergone liver surgery for CRCLM in Sun Yat-sen University Cancer Center from 1 January 1999 to 31 December 2017. Of them, 51 had extra-liver metastases before surgery and 46 did not received R0 resection; these were excluded from the study. Also excluded were two patients who had not been tested for serum GGT before liver surgery. Finally, 350 patients who had undergone R0 resection for liver-only CRCLM were included in our study.
The study was approved by the institutional review board of Sun Yat-sen University Cancer Center (approval number: GZR2019-180), and all patients gave written informed consent notifying possible use and publication of clinical data in future studies. The authenticity of this article has been validated by uploading the key raw data onto the Research Data Deposit public platform (www.researchdata.org.cn), with the approval RDD number RDDA2020001464.
Baseline data collection
Demographic and clinicopathological data were collected from hospital records, including sex, age, primary tumor site, TNM stage, and the number and size of liver metastases. Data on serum levels of CEA and some biochemical markers including GGT, total bilirubin (TBIL), alanine aminotransferase (ALT), aspartate aminotransferase (AST), and lactate dehydrogenase (LDH) that were tested within 2 weeks before hepatectomy were also collected.
Patients were divided into synchronous and metachronous groups according to the timing of liver metastasis. Metastases that coexisted with primary tumor at diagnosis were classified as synchronous, while those developed during follow-up as metachronous.
Surgery and peri-operative treatment
All of the patients were discussed by a multiple disciplinary team specialized in the treatment of colorectal cancer to decide whether liver lesions were resectable and whether pre-operative chemotherapy should be prescribed. Chemotherapy regimens were either oxaliplatin-based or irinotecan-based. Treatment response was evaluated every 6–8 weeks using magnetic resonance imaging (MRI) or enhanced computed tomography (CT) scan for the liver. Liver surgery was performed 2 weeks after chemotherapy, or 6 weeks if bevacizumab had been used. R0 resection was defined as complete resection of liver metastases with negative microscopic margins. Adjuvant chemotherapy was recommended for all of the patients, unless there were major complications. Patients were followed every 3 months in the first 2 years, and every 6 months thereafter. A liver MRI or CT scan and serum CEA test were recommended at every visit.
Classification of GGT and other factors
Serum GGT level was divided into high and low groups according to previous reports.18,19 Briefly, for female patients, a GGT level >33 U/L was defined as high, and ⩽33 U/L as low. For male patients the cut-off value was 51 U/L. Some known prognostic factors of tumor recurrence were also analyzed, including the number of liver metastases, size of the largest metastasis, and the use of neoadjuvant chemotherapy. We also analyzed the levels of some liver enzymes and functional indicators including LDH, AST, ALT, and TBIL, and divided them into two groups using their respective upper normal limit as cut-off values, which were 250 U/L, 40 U/L, 40 U/L, and 17 umol/L, respectively. International normalized ratio (INR) and platelet counts were classified as either high or low using the median value (0.96 and 202 × 109/L, respectively). Drinking has been shown to be associated with an elevated GGT and was therefore included in the analysis. 19 Patients who drink casually or daily were classified as drinkers.
Statistics
Relapse-free survival (RFS) was defined as the time from liver surgery to tumor recurrence or death from any cause, whichever occurred first. Overall survival (OS) was defined as the time from liver surgery to death from any cause. Comparisons of factors between different GGT levels were tested using a Chi-square or Wilcoxon test, as appropriate. Correlations of GGT and other liver transferase were displayed using Spearman’s rank correlation coefficients. RFS and OS were estimated by Kaplan–Meier method and examined with log-rank test in univariate analysis. Factors with clinical implications that yielded a p value < 0.05 in univariate analysis were later incorporated into multivariate analysis using Cox regression model. A two-sided p value < 0.05 was considered statistically significant. Statistical analysis was performed in R (version 3.5.1).
Results
Baseline characteristics
Baseline characteristics are listed in Table 1. Of the 350 patients, 116 (33.1%) were female, and the median age was 57.4 years. About two thirds of the disease (237/350) were synchronous, and nearly half of the patients had solitary liver metastasis. A total of 47.4% of the patients received neoadjuvant chemotherapy, and 70% of the patients received adjuvant chemotherapy. RAS testing was performed in 89 patients, and 32 (36.0%) were mutant-type.
Patient characteristics (n = 350).

Chemo, chemotherapy; mets, metastases.
Comparisons between two GGT levels
Table 2 shows comparisons between patients with different GGT levels. High and low GGT levels were observed in 242 (69.1%) and 108 (30.9%) patients, respectively. Patients with metachronous disease, the number of metastases ⩾2, the size of the largest metastasis ⩾3 cm, or a history of neoadjuvant chemotherapy tended to have a higher GGT level (p = 0.001, 0.027, 0.001, and 0.002, respectively). In the 163 patients who received neoadjuvant chemotherapy, GGT showed an inverse association with treatment response, with 35.4%, 40.4%, and 64.7% high GGT in partial response (PR), stable disease (SD), and progressive disease (PD) patients, respectively. Figure 1 shows the correlation of GGT with TBIL, ALT, AST, and LDH. GGT did not correlate with any of these factors (r = 0.52, 0.574, 0.52, 0.186, respectively).
Comparison of baseline information between two GGT levels.

CRS, clinical risk score proposed by Fong et al. 7 ; GGT, gamma-glutamyl transpeptidase; INR, international normalized ratio; mets, metastases; PD, progressive diseasel; PR, partial response; SD, stable disease.

Correlation of GGT with TBIL, ALT, AST, and LDH, with a coefficient of 0.520, 0.574, 0.520, and 0.186, respectively (Spearman’s rank correlation coefficients).
Follow-up and survival
After a median follow-up of 37.4 month, recurrence and death were observed in 201 and 129 patients, respectively. The most common site of recurrence was the liver, accounting for 57.4% of the events, followed by the lungs (11.7%), and 20.4% involved multiple organs. Re-resection of liver recurrence was performed for 31 patients, of whom nine relapsed.
A higher GGT strongly correlated with worse RFS and OS, with a median survival of 30.3 versus 11.8 months for RFS (p < 0.001) and 94.3 versus 38.0 months for OS (p < 0.001) (Figure 2). Many other factors were found to be associated with RFS and OS, including the number and size of metastases, and the use of neoadjuvant chemotherapy (all p < 0.001), while sex, drinking, and timing of metastasis were not associated with survival (Table 3).

RFS and OS after hepatectomy as related to serum GGT levels. Median RFS were 11.8 and 30.3 months for patients with high and low GGT, respectively. Median OS were 38.0 and 94.3 months for them, respectively.
Univariate analysis of predictors of RFS.

CEA, carcinoembryonic antigen; GGT, gamma-glutamyl transpeptidase; INR, international normalized ratio; mets, metastases; RAS, KRAS or NRAS; RFS, relapse-free survival.
Subsets analysis
In subsets analysis, GGT remained a prognostic factor of RFS regardless of the number of metastases and the timing of metastasis. In patients with liver metastases ⩾3 cm or with a history of neoadjuvant chemotherapy, GGT remained a significant prognostic factor (p = 0.0015 and 0.012, respectively, Figure 3).
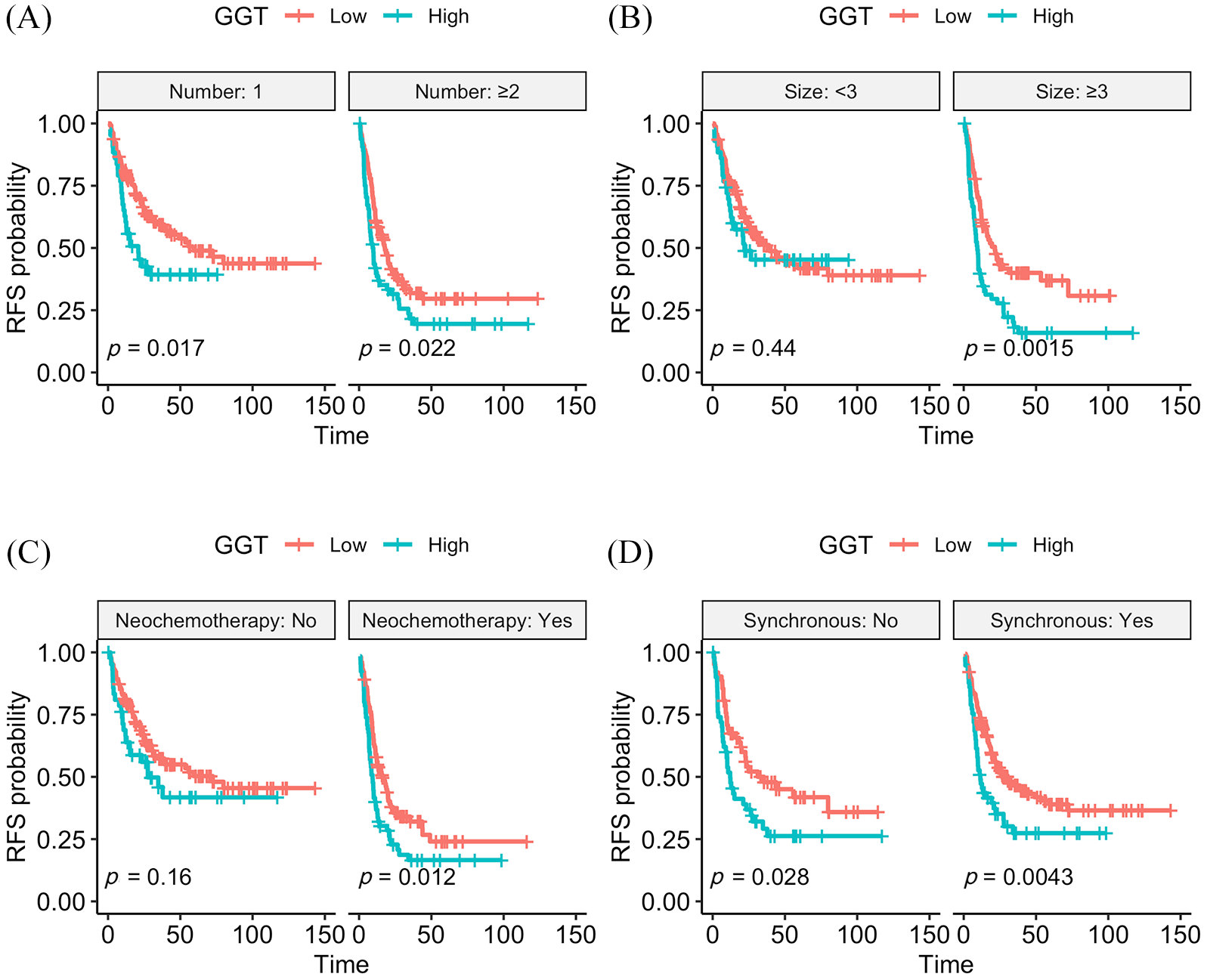
Subsets analysis of GGT stratified by the number of metastases (A), size of the largest metastasis (B), history of neochemotherapy (C), and timing of metastasis (D). GGT remained prognostic of RFS regardless of the number and timing of metastasis. However, with respect to size and neochemotherapy, GGT were only prognostic in patients with size⩾ 3cm or with a history of neochemotherapy, but not in their counterparts.
Interestingly, GGT was prognostic in male patients but not in female patients (Figure 4A). Since alcohol intake is a known factor of an increased GGT, we then looked into the impact of drinking on GGT. In 234 male patients, 84 drank casually or daily, and 150 were abstainers. No correlation was observed between drinking and GGT level, though drinking accounted for a higher proportion of patients with GGT > 80 U/L (31.3% versus 17.4%, Figure 4C). In multivariate analysis, GGT remained an independent prognostic factor of RFS (HR = 1.40, p = 0.038) (Figure 5).
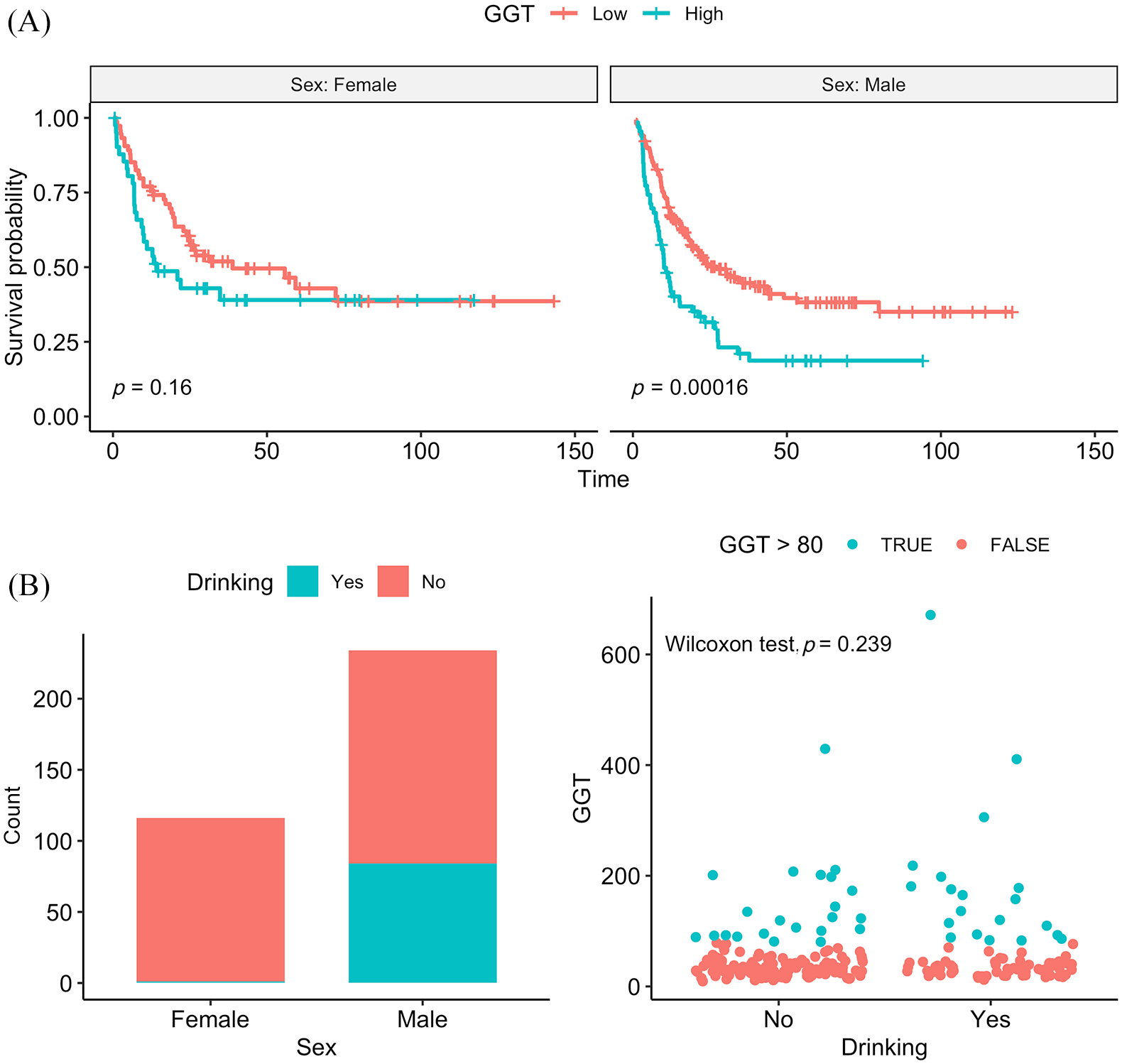
Stratified by sex, GGT was prognostic of RFS only in male patients (A). Male patients were more likely to drink than female patients (B). In male patients, no correlation was observed of GGT with drinking, although drinking seemed to account for a higher proportion of patients with GGT > 80U/L (31.3% versus 17.4%, p = 0.239).

Multivariate analysis of RFS. Serum GGT, size of the largest metastases and history of neochemotherapy were independent factors.
Discussion
Although surgery is thought to be the only curative therapy for CRCLM, less than 20% of the patients can be cured—most of them develop new metastases after hepatectomy. 5 For patients who experience a quick relapse, such an invasive procedure seems unnecessary because it delays chemotherapy and exposes patients to surgical complications. Our study shows that a higher serum GGT before hepatectomy is strongly associated with poor RFS and OS in patients with CRCLM, providing a readily available biochemical marker for identifying patients who may not benefit from the surgery. To our knowledge, it is the first time serum GGT has been used to predict tumor recurrence in this setting.
GGT has long been used as an index of liver dysfunction, especially when caused by obstructive liver disease. 20 Recently, it has been demonstrated both in vivo and in humans that GGT assists in the development and progression of many tumors.13,21,22 In line with previous studies, we found that GGT was an adverse prognostic marker for patients with CRCLM undergoing R0 resection. Patients with a high serum GGT had shorter RFS and OS after the surgery. To eliminate possible interaction of liver dysfunction with survival, we compared GGT with other liver function indicators, including TBIL, AST, ALT, and LDH, and found no significant correlations. It follows that liver dysfunction is not the only contributor to a high GGT; the liver tumor and its activity may also be an important source. This hypothesis is supported by a previous report in which tumor tissue was proven a source of serum GGT in melanoma-bearing mice. 23 In human colorectal cancer, serum GGT was shown to be a predictor of tumor response to chemotherapy, suggesting that GGT might be able to reflect tumor activity. 17 Miyata et al. found that GGT activities in resected liver tumor tissues (including CRCLM) were associated with a risk of postoperative recurrence. 24 In the present study, GGT was strongly associated with tumor size and the number of metastases.
However, it should be noted that a number of factors were not balanced between the two GGT groups, including tumor size, the number of metastases, and the use of neoadjuvant chemotherapy. These factors also showed strong correlations with RFS and OS, and therefore might confound the results. Nevertheless, in subset analysis, GGT remained prognostic regardless of the timing and the number of metastases. As for neoadjuvant chemotherapy, GGT was prognostic among patients who received this treatment, but not among patients who underwent direct surgery. One possible reason is that patients receiving neoadjuvant chemotherapy had a larger tumor burden and a worse prognosis, which allowed for easier discrimination between the two groups. In addition, studies have shown that GGT is associated with sinusoidal obstruction syndrome (SOS), a complication caused by oxaliplatin-based chemotherapy that can impair long-term survival.25,26 Therefore, the prognostic ability of GGT might be just a bystander effect of SOS, though platelet counts and INR (markers of sinusoidal obstruction) were not associated with survival.
Serum GGT level can also be affected by some non-tumoral factors, including sex, hepatitis, alcohol intake, and dietary pattern.19,27 Interestingly, although these factors were well balanced between high and low GGT groups, in subset analysis stratified by gender GGT remained prognostic only in male patients. This intrigued us to look into the relation between GGT and gender, but no difference was observed between them (p = 0.161). Because alcohol intake was more common among male patients (35.9% versus 0.9%), we compared GGT levels between drinkers and abstainers in this group, but there was no difference (p = 0.24, Wilcoxon test), though drinking seemed to account for a higher proportion of patients with GGT > 80 U/L (31.3% versus 17.2%, Figure 4C).
In multivariate analysis, serum GGT remained an independent prognostic factor of RFS. However, it should be noted that in this study the CRS score was excluded from the analysis due to changes in treatment strategy. Neoadjuvant chemotherapy was delivered to nearly one third of the patients, making a CEA > 200 ug/L uncommon (18/350 in our study). It also increased the proportion of negative lymph nodes (131/314 negative). Patients remaining node-positive or CEA > 200 ug/L after neoadjuvant chemotherapy were considered insensitive to chemotherapy and had unfavorable outcomes. If the CRS score was incorporated into the model, all other factors would be eliminated. In previous research, 7 patients with synchronous CRCLM had a worse outcome than those with metachronous CRCLM, but in our study they did not differ in both RFS and OS. One reason may be that patients in the synchronous group had smaller liver lesions than patients in the metachronous group, with the proportion of CRCLM < 3 cm being 63.1% versus 47.3%.
Due to its retrospective nature, our study is subject to several limitations. First, since baseline information were taken within 2 weeks before surgery, for those receiving chemotherapy, the original tumor burden was not able to be evaluated. Second, the use of targeted drugs has been shown to be associated with a better long-term survival, 28 but due to the scarcity of cases (n = 42) it was not analyzed in this study. Third, although post-operative adjuvant chemotherapy was routinely recommended, one third of patients did not follow the advice. Also, some factors were dispensed within the study due to a lack of samples, including the state of RAS mutation, the timing of liver surgery, and the performance of repeat surgery. Lastly, we did not perform pathological evaluation for biliary obstruction; therefore it remained to be validated whether biliary obstruction contributed to an elevated GGT.
Conclusion
Taken together, our study demonstrates that serum GGT level before liver surgery is an adverse prognostic factor of RFS for patients with liver-confined CRCLM.
Footnotes
Acknowledgements
We thank professor Gong Chen, Liren Li, and Zhenhai Lu for their generosity in providing cases; special thanks are due to Sherry Young, without whose support this work could never have been completed.
Author contributions
Conception and design: Binyi Xiao, Jianhong Peng and Jinghua Tang. Collection and assembly of data: Jianhong Peng, Yuxiang Deng, and Yujie Zhao. Data analysis and interpretation: Binyi Xiao, Jianhong Peng, and Jinghua Tang. Manuscript writing: Binyi Xiao. All authors read and approved the final manuscript.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by National Natural Science Foundation of China (No. 81502459, 81772959) and Science and Technology Project in Guangdong Province (No. 2013B021800146, 2017A030310204).
