Abstract
Background:
Geographical variations in the frequency of oncogenic driver alterations in non-small cell lung cancer (NSCLC) are observed worldwide, likely influenced by genetic and environmental factors. Radon, a leading cause of lung cancer in nonsmokers, may influence NSCLC’s molecular profile.
Objectives:
This study aims to explore the potential correlation between estimated indoor radon concentrations and the frequency of driver alterations in NSCLC across Europe.
Design:
Ecological study of European countries with available data on indoor radon exposure (estimated mean levels and the proportion (%) of dwellings with concentrations >200 and >400 Bq/m3), and frequency of NSCLC actionable genomic alterations (EGFR, ALK) from PubMed articles and meeting abstracts with sample size >100 patients.
Methods:
We retrospectively analyzed NSCLC molecular data and radon exposure levels from European countries. Radon data, including the mean and proportion (%) of dwellings with concentrations >200 and >400 Bq/m3, were sourced from the European Commission Report 2005. NSCLC molecular alteration frequency (EGFR, ALK) was extracted from PubMed articles with sample sizes >100 patients. The correlation between molecular alteration frequencies and country-specific radon data were assessed.
Results:
Data on radon exposure and EGFR/ALK frequency were available for 21 European countries. Five countries had mean radon levels >100 Bq/m3, and eight countries had ⩾3% of dwellings with concentrations >400 Bq/m3. The median frequency of EGFR mutations and ALK fusions was 11.3% (range 7%–18.1%) and 4.1% (range 3.4%–12%), respectively. A positive correlation was found between estimated ALK-fusion frequency and the percentage of dwellings with >400 Bq/m3 (r = 0.72, p = 0.001). No correlation was found between EGFR mutation frequency and radon exposure.
Conclusion:
We found a positive correlation between estimated ALK fusion frequency in NSCLC and the proportion of dwellings exceeding 400 Bq/m3 radon exposure in 21 European countries. Further research is needed to explore the potential influence of radon and other environmental factors on NSCLC with driver alterations.
Plain language summary
Lung cancer is one of the most common cancers worldwide. A significant portion of lung cancers are classified as non-small cell lung cancer (NSCLC). Certain genetic changes, known as driver alterations, can promote the growth of these cancers. Two common driver alterations in NSCLC are mutations in the EGFR gene and rearrangements in the ALK gene. Understanding how environmental factors, like radon exposure, relate to these genetic changes is important for prevention and treatment strategies. Radon is a natural radioactive gas that can accumulate indoors, especially in areas with certain geological conditions. Long-term exposure to high levels of radon is a known risk factor for lung cancer, particularly among non-smokers. In Europe, indoor radon levels vary significantly between countries, influenced by factors such as building practices and local geology. This study investigated whether there’s a connection between average indoor radon levels and the frequency of EGFR mutations and ALK rearrangements in patients with NSCLC across 21 European countries. The findings revealed that countries with higher percentages of homes having high radon levels (above 400 Bq/m³) also had higher rates of patients with NSCLC and ALK rearrangements. These results suggest that elevated indoor radon exposure may be linked to specific genetic changes in lung cancer, particularly ALK rearrangements. Further research is needed to confirm this association and to explore how radon and other environmental factors might influence the development of molecularly-driven NSCLC.
Introduction
Indoor radon exposure is the leading cause of lung cancer in nonsmokers, and the second major risk factor in ever-smokers after tobacco. 1 Radon-222 (222Rn) is a radioactive gas produced by the decay of uranium-238. Its indoor concentration is linked to the uranium content of the earth’s soil beneath dwellings.2,3 The U.S. Environmental Protection Agency and the International Agency for Research on Cancer classified radon as a human carcinogen in 1987 and 1988, respectively. 4 It is estimated that about 3%–9% of lung cancer deaths in the European Union (EU) are related to indoor radon exposure.5,6 There is no safe threshold for radon exposure and the risk of lung cancer increases by 16% for every 100 Bq/m3 rise in radon concentration.1,7,8 In 2013, the European Directive was revised to include updated basic safety standards, requiring EU member states to implement a national action plan addressing radon exposure. The European Commission (EC) for Atomic Energy set a directive stipulating that radon levels in European households should not exceed 300 Bq/m3 (2013/59/EURATOM).2,8,9 Radon exposure varies significantly across Europe; over 30% of European territory has median radon concentrations >100 Bq/m3. 10
The molecular mechanisms by which radon contributes to lung carcinogenesis remain unclear. 11 Geographical differences in the frequency of driver genomic alterations in NSCLC may be influenced by genetic and environmental factors. 12 Driver alterations like EGFR mutations and ALK fusions are more common in non-smokers and younger patients, yet risk factors remain elusive.13,14 Preliminary evidence suggests a potential role of indoor radon exposure in shaping the molecular profile of NSCLC, particularly in never-smokers.15–17 This ecological study explores the correlation between estimated indoor radon concentrations and the estimated frequency of oncogenic driver alterations in NSCLC across European countries.
Methods
We conducted an ecological study 18 including all European countries with available data on indoor radon exposure (estimated mean levels and the proportion (%) of dwellings with concentrations >200 and >400 Bq/m3), and frequency of NSCLC actionable genomic alterations (EGFR, ALK) from PubMed articles and meeting abstracts with sample size >100 patients (Figure 1). The reporting of this study conforms to the STROBE-ME statement 19 (Supplemental Appendix 4).

Flow chart of study selection. Number of patients comes from the total sample size of all the publications included. Of 27 countries with radon data available, we did not find EGFR/ALK frequency data with >100 patients for six countries.
Indoor radon exposure
In 2005, the Institute for Environment and Sustainability of the EC surveyed 34 European countries to collect data for radon mapping. 5 We extracted estimated annual mean radon levels (Bq/m3), and the percentage of dwellings with radon levels >200 and >400 Bq/m3 from this report. Data for all three variables were available from 27 countries. 5
Frequency of driver genomic alterations in NSCLC by country
We conducted a PubMed search to identify original articles and meeting abstracts reporting the frequency of driver genomic alterations in NSCLC across European countries. Articles published in peer-reviewed journals from January 1, 2013, to December 1, 2023 were included. When original studies for specific countries were not found via Pubmed search, other sources (i.e., abstracts of main international meetings) were used. Search terms were NSCLC, non-small cell lung cancer, EGFR (+country name), or ALK (+country name). Full-text articles and abstracts were identified by two individual reviewers (M.G.-P. and M.G.d.H.). Articles with a sample size <100 patients were excluded. If multiple articles were available for a country, we selected the one with the largest sample size (Figure 1 and Supplemental Appendix 2). Data on EGFR mutations and ALK fusions frequency were found for 21 and 17 countries, respectively. Due to limited data on other alterations (e.g., ROS1 fusions), only EGFR and ALK data were included in the analyses.
Statistical analysis
The correlation between radon exposure and the frequency of genomic alterations was assessed using radon measurement as a continuous variable and according to the percentage of dwellings with a radon exposure >200 and >400 Bq/m3. We used the Pearson’s correlation coefficient and displayed this coefficient as a matrix (Supplemental Appendix 3). All analyses were made with R studio software (Posit team (2025). RStudio: Integrated Development Environment for R. Posit Software, PBC, Boston, MA).
Results
Indoor radon exposure
Data on estimated indoor radon levels were available for 27 European countries, as reported by the Joint Research Centre of the EC 2005. 5 Twenty-one countries had data on the frequency of EGFR mutations and/or ALK fusions available in PubMed-published studies (Figure 1).
The estimated annual average of indoor radon was 77 Bq/m3, ranging from 20 Bq/m3 in United Kingdom to 144 Bq/m3 in Serbia. Five countries had annual averages exceeding 100 Bq/m3. Twelve countries had >5% of dwellings with radon concentrations >200 Bq/m3 and 15 countries had >1% of dwellings >400 Bq/m3 (Figure 2).
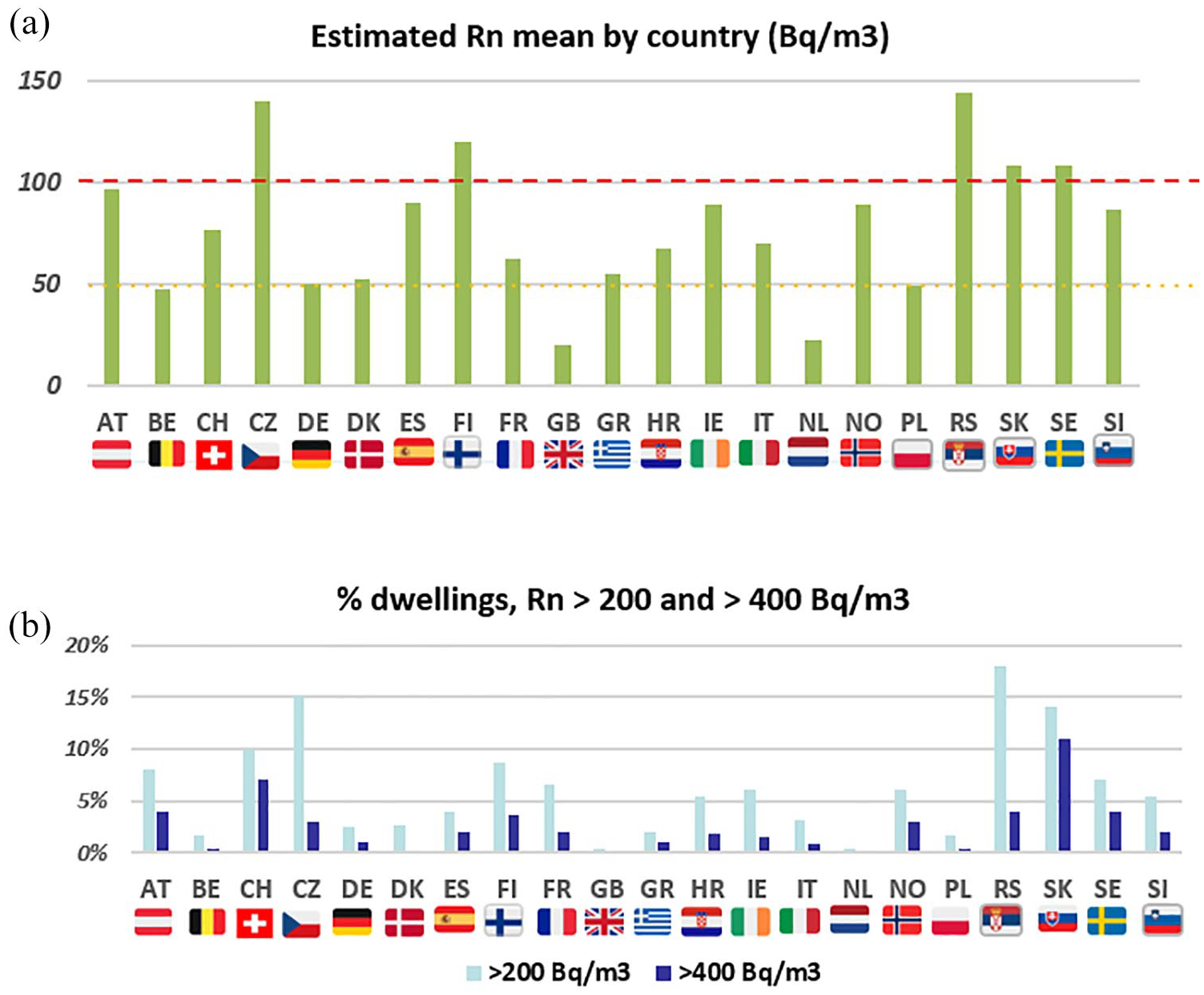
Indoor radon exposure by country. (a) Estimated annual radon mean levels (Bq/m3). (b) Proportion of dwellings above 200 and 400 Bq/m3 by country.
Frequency of driver genomic alterations in NSCLC
We identified data on EGFR mutations and ALK fusions in NSCLC from 21 and 17 studies, respectively, covering the same number of European countries (Figure 1 and Supplemental Appendix 2). A total of 150,165 NSCLC cases were evaluated for EGFR mutations and 103,706 cases for ALK fusions. The median frequency of EGFR mutations was 11.3% (n = 17,292; range 7%–18.1%) and 4.1% for ALK fusions (n = 3430; range 3.4%–12%).
Correlation between estimated indoor radon and frequency of driver genomic alterations in NSCLC
No correlation was found between EGFR mutation frequency and radon exposure (Supplemental Appendix 1). In countries with an estimated annual indoor radon mean >100 Bq/m3, median EGFR frequency was 11.7% compared to 11.9% in countries with <100 Bq/m3. Similarly, in countries with ⩾1% of dwellings with >400 Bq/m3, median EGFR frequency was 12.5% versus 11.2% in those countries with <1% of dwelling above 400 Bq/m3.
For ALK fusions, no significant differences were observed in frequency based on the annual average radon level (5.6% with >100 Bq/m3 vs 6.2% with <100 Bq/m3). A higher ALK frequency was noted in countries with ⩾1% of dwellings >400 Bq/m3 (7%), compared to those with <1% (3.2%), not statistically significant. A positive correlation was observed between ALK-fusion frequency and the percentage of dwellings with >400 Bq/m3 (r = 0.72, p = 0.001), statistically significant, with a trend, not clinically significant for dwellings >200 Bq/m3 (r = 0.39, p = 0.1; Figure 3).

Correlation between ALK-fusion frequency in NSCLC by country and indoor radon exposure. The size of each bubble represents the sample size for each country.
Discussion
This study is the first to explore the potential association between estimated indoor radon exposure and the frequency of driver alterations in NSCLC across Europe. We identified a significant correlation between the frequency of ALK fusions in NSCLC and areas with higher radon exposure (i.e., regions where a higher percentage of dwellings have radon levels exceeding 400 Bq/m3).
Previous studies have suggested a similar correlation, noting that patients with ALK-rearranged NSCLC were more likely to live in areas with higher radon exposure.15,16 In a retrospective study conducted in Galicia (northwest region in Spain), indoor radon exposure was higher in patients with NSCLC harboring EGFR mutations and ALK-fusions. 15 Similarly, a prospective study in Madrid (Spain) found higher radon exposure in patients with ALK-rearranged and BRAFV600E mutated NSCLC. 20 However, these studies were limited by small sample sizes. The Radon France ecological study also suggested that the frequency of certain driver alterations in NSCLC is higher in regions of France with elevated radon exposure. 16 This association was further supported by the BioRadon France Study, which found that patients with NSCLC born in high-radon areas were more likely to harbor driver alterations, with higher radon exposure reported in patients with ALK-rearranged NSCLC. 21
Radon-induced carcinogenesis involves complex and not yet fully understood molecular mechanisms. Radon can cause significant cytotoxic and genotoxic effects, including DNA double-strand breaks, mutations, and chromosomal aberrations. 22 This genomic instability may be critical for the development of driver alterations, highlighting the importance of further research into the molecular pathways induced by radon exposure.
Our findings contribute to the growing evidence that environmental exposures may influence the molecular epidemiology of lung cancer. While we focused on radon exposure and its correlation with EGFR mutation and ALK fusion frequency at the population level, recent work from Hill et al. 23 has provided important biological insights into how air pollution—another environmental factor—may promote oncogenesis. In a large analysis involving over 32,000 EGFR-mutated lung cancer cases, exposure to fine particulate matter (PM2.5) was associated with increased lung cancer incidence in never-smokers. Using functional models, the authors demonstrated that PM2.5 promotes a pro-inflammatory microenvironment that facilitates tumor growth from cells harboring preexisting oncogenic mutations, rather than inducing new mutations. In line with this, ultra-deep sequencing of histologically normal lung tissue showed that EGFR and KRAS mutations were detectable in a significant proportion of healthy individuals. These findings support the idea that environmental factors such as radon and air pollution may act as tumor promoters, contributing to the development of oncogene-addicted NSCLC through distinct but potentially complementary mechanisms. Further studies integrating ecological, clinical, and molecular data are needed to better understand these complex relationships.
Our study has certain limitations. One limitation of previous studies is that the risk associated with radon exposure is typically based on regional estimates of mean indoor radon levels. Given the high degree of variability and heterogeneity in radon concentrations, using regional mean estimates may not accurately reflect individual radon exposure. The use of the percentage of dwellings exceeding radon levels >400 Bq/m3 may better capture the population-level radon risk. Our findings align with this, showing a higher frequency of ALK-rearranged NSCLC in countries with a greater proportion of high-radon dwellings. However, it is important to emphasize that these associations are correlative and do not imply causality. The number of high-radon dwellings may not, in itself, be the reason for increased frequencies of oncogene-addicted NSCLC. Additionally, the molecular frequency data may not accurately reflect the true prevalence of molecular alterations in each country. As with all ecological studies, the lack of individual patient-level is a key limitation, and our analysis cannot control for known confounding factors. 18 In particular, ethnicity and gender—which are well-established determinants of molecular alteration prevalence in NSCLC—were not available in the datasets analyzed. It is possible that differences in immigration patterns or demographic structure may partly explain the observed variation. In addition, oncogene-addicted lung cancers are more frequently diagnosed in younger individuals. If radon exposure contributed significantly to their development, one might expect a clear temporal increase in their frequency over time, which has not been uniformly observed. Age-related trends in mutation prevalence were not captured in our analysis and warrant further investigation.
Despite these limitations, our study is the first to explore the correlation between country-level estimated indoor radon exposure, and NSCLC molecular profile in Europe. The findings are consistent with previous research, suggesting that environmental factors, such as radon may contribute to the geographical variation observed in molecular subtypes of NSCLC. 12 This study may serve as a proof of concept for a novel approach to ecological cancer research, placing molecular epidemiology within the framework of precision medicine. A deeper understanding of radon’s role in lung carcinogenesis is necessary. Importantly, future research should adopt an exposome-based perspective, investigating not only individual risk factors but also their complex interactions, to better understand the multifactorial etiology of lung cancer in populations with low smoking prevalence. Our results may help guide and expedite such efforts, aligning with current public health priorities and supporting the goals outlined in the European Council Directive 2013/59/Euratom. 9
The ongoing EORTC 1920 LCC–BIORADON study aims to prospectively explore the correlation between NSCLC molecular profiles and indoor radon exposure, including patient-level data and translational research. 24 As environmental factors gain prominence, raising awareness is essential for enhancing research and improving cancer prevention strategies.1,23
Conclusion
In conclusion, this ecological study suggests that environmental factors, such as radon exposure, may contribute to the geographical variation in NSCLC molecular profiles. Specifically, we observed a positive correlation between ALK fusion frequency in NSCLC and the proportion of dwellings exceeding radon levels of 400 Bq/m3 in European countries. While causality cannot be inferred from ecological data, this association highlights the need for further research into the role of radon and other environmental exposures in the development of driver alterations in NSCLC. High-quality data and multidisciplinary collaboration will be essential to advance our understanding of lung carcinogenesis, support prevention, and early diagnosis strategies, and ultimately improve patient outcomes.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251351449 – Supplemental material for Frequency of driver oncogenic alterations in NSCLC and estimated indoor radon exposure in Europe (RADON EUROPE study)
Supplemental material, sj-docx-1-tam-10.1177_17588359251351449 for Frequency of driver oncogenic alterations in NSCLC and estimated indoor radon exposure in Europe (RADON EUROPE study) by Miguel García-Pardo, Marta García de Herreros, Edouard Auclin, Miquel Ferriol-Galmes, Juan Carlos Laguna, Teresa Gorría, Gerard Caravaca, Joan Sart, Víctor Albarrán-Artahona, Rafael López-Castro, Cristina Teixidó, Giancarlo Castellano, Alex Martínez Bedmar, Ainara Arcocha, Laura Alcolea, Manuel Jiménez, Mario Bernabeu, David Delgado-Cano, Alejandro Navarro, Aleix Prat, Noemí Reguart, Javier Elio, Pilar Garrido, Natasha B. Leighl, Benjamin Besse and Laura Mezquita in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
