Abstract
Background:
The prognosis for metastatic nasopharyngeal carcinoma (mNPC) remains poor, and effective treatment options are still limited.
Objectives:
This study aims to evaluate the efficacy of stereotactic body radiation therapy (SBRT) and identify prognostic factors in patients with mNPC.
Design:
A retrospective study.
Methods:
From August 2019 to November 2023, this retrospective study included 62 mNPC patients with 84 metastatic lesions treated with SBRT. The primary endpoint was progression-free survival (PFS). PFS1, PFS2, and overall survival (OS) were also assessed, and related prognostic factors were analyzed.
Results:
The median follow-up time from the diagnosis of metastasis was 44 months (range from 12 to 85 months). Combining SBRT with systemic therapy resulted in favorable survival outcomes, with 3-year PFS, PFS1, PFS2, OS, and local control rates of 35.5%, 35.7%, 56.1%, 87.2%, and 86.5%, respectively. The combination of SBRT with first-line systemic therapy, disease control before SBRT, oligometastatic disease, and SBRT to all metastatic lesions exhibited superior PFS, PFS1, and PFS2. Multivariable analysis indicated that the line of SBRT treatment was a predictor of PFS1 and PFS2, and SBRT to all metastatic lesions was a predictor of all PFSs.
Conclusion:
SBRT is an effective and safe local treatment for mNPC patients. Early intervention with SBRT during first-line therapy and SBRT to all metastatic sites for oligometastatic disease may help improve disease control.
Keywords
Introduction
Nasopharyngeal carcinoma (NPC) primarily occurs in Southeast Asia and southern China, with an annual incidence rate of approximately 1.5 per 100,000 individuals. 1 Despite the 5-year survival rate reaching around 80%, NPC still results in an estimated 80,000 deaths annually. 2 Most patients present with localized lesions, with only 4%–15% of NPC cases exhibiting de novo distant metastasis at initial diagnosis, and approximately 15%–30% developing distant metastasis after comprehensive treatment.3–7 For metastatic nasopharyngeal carcinoma (mNPC), the bones, lungs, and liver are the most common sites of metastasis.7,8 The mNPC has a significantly worse prognosis compared to non-metastatic cases, with patients exhibiting substantial heterogeneity. Despite multidisciplinary treatment, the median overall survival (OS) is only 20–30 months, and there remains controversy regarding the optimal treatment strategy.9–12
For mNPC, the primary treatment remains platinum-based systemic chemotherapy with or without immunotherapy, but still with limited efficacy. 13 Local radiotherapy is commonly used to alleviate symptoms and control the progression of local lesions in patients with metastatic tumors, including mNPC.8,14 Currently, local radiotherapy for metastatic disease includes conventional radiotherapy, intensity-modulated radiotherapy (IMRT), and stereotactic body radiotherapy (SBRT). Among these, SBRT has emerged as a key treatment method for oligometastatic disease due to its high dose, precise targeting, and steep dose gradient. The efficacy of SBRT has been demonstrated across multiple metastatic sites in various cancers, with the highest local control (LC) rates exceeding 90%.15,16 However, the optimal radiotherapy approach for metastatic lesions in mNPC patients remains to be determined.
Although SBRT shows promising potential, current evidence for its use in mNPC remains limited. This study aims to evaluate the efficacy and identify prognostic factors of SBRT combined with comprehensive treatment for mNPC, ultimately providing more viable treatment options for these patients.
Materials and methods
Patients
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE). 17
We included all patients with mNPC who received SBRT at the Department of Radiation Oncology, Fudan University Shanghai Cancer Center, between August 2019 and November 2023. The inclusion criteria were as follows: (1) histologically confirmed NPC, (2) radiologically and/or histologically confirmed distant metastases, (3) SBRT treatment to at least one metastatic lesion, (4) at least one cycle of systemic chemotherapy with or without immunotherapy, (5) age >18 years old, (6) Eastern Cooperative Oncology Group (ECOG) performance score ⩽2, and (7) adequate bone marrow, liver, and renal function. The exclusion criteria were as follows: (1) prior surgery, radiofrequency ablation, or radiotherapy to metastatic lesions and (2) prior or concurrent malignancies. All patients were staged according to the TNM staging system by the American Joint Committee on Cancer (AJCC) 8th edition. To provide a clear overview of the patient selection process, a flowchart was shown in Figure S1.
Metastasis evaluation involved positron emission tomography/computed tomography (PET/CT) or a combination of chest CT, abdominal ultrasound or CT/MRI, and 99Tcm-MDP bone scintigraphy with single-photon emission computed tomography (SPECT)/CT. Epstein–Barr virus (EBV) deoxyribonucleic acid (DNA) was also tested to help monitor metastasis. Metastases were confirmed by pathological biopsy when available or by radiological diagnosis confirmed by two senior radiologists and a multidisciplinary team. Liver and bone metastases were initially screened using abdominal ultrasound and bone scintigraphy with SPECT/CT, respectively, and suspicious lesions were further evaluated with contrast-enhanced CT, MRI, PET/CT, or biopsy.
This study included both synchronous and metachronous metastases. Synchronous metastasis was defined as distant lesions detected at the time of NPC diagnosis, while metachronous metastases referred to distant lesions that appeared after an interval following the initial diagnosis, typically more than 6 months post-treatment. Each metastatic lesion was recorded as an independent treatment site and included in the total count of metastases. For patients with synchronous metastases who showed a favorable response to comprehensive systemic therapy, radiotherapy to the primary tumor (nasopharynx and neck) was delivered using IMRT techniques, but the treatment of the primary tumor and regional lymph nodes was not classified as part of the treatment sites under investigation in this study.
Oligo-metastasis was defined as the involvement of no more than three metastatic organs and a total of no more than five metastatic lesions, while extensive metastasis was defined as a condition where there were more than three metastatic organs or more than five metastatic lesions.
SBRT technique
During simulation, patients were positioned supine and immobilized with a vacuum cushion, optionally combined with a thermoplastic body mask. CT simulation was performed with a slice thickness of 3 mm. The use of contrast agents was determined by the attending physician and was generally recommended for liver lesions. For certain thoracic and abdominal lesions, observation of diaphragm movement under the simulator was conducted to characterize respiratory motion. A 4D-CT was utilized if respiratory motion was less than 10 mm; if motion exceeded 10 mm, respiratory gating, breath-hold techniques, or abdominal compression were employed to control breathing. MRI fusion was permitted for treatment-planning purposes.
For treatment planning, the gross tumor volume (GTV) included the visible tumor lesions. For thoracic and abdominal lesions, the clinical target volume (CTV) was defined as equivalent to the GTV. The internal tumor volume was delineated using maximum intensity projection from 4D-CT and subsequently expanded 6 mm from CTV to form the planning target volume (PTV). For spine or non-spine bone metastases, CTV was defined according to the previous target volume recommendations.18,19 The PTV was defined as an isotropic 3 mm expansion from the CTV. Organs at risk (OARs) were delineated based on their proximity to the target lesions, generally within 3 cm of the target volume.
Due to the absence of specific SBRT guidelines for mNPC, the selection of SBRT target lesions, timing, and doses in this study was individualized by a multidisciplinary team based on patient-specific characteristics (including ECOG performance status, symptoms, prior treatments, response to treatment, and anticipated survival). For patients with oligometastatic disease, SBRT was delivered to each detectable lesion if feasible. For patients with non-oligometastatic disease, given the extensive number of metastases, irradiating all metastatic sites was impractical. We prioritized the most critical lesions, such as those causing significant symptoms like pain or compression. During systemic treatment, different lesions may respond differently. If most lesions exhibited progressive disease (PD), we generally did not recommend SBRT. However, if most lesions were stable and only a single lesion was progressing, we suggested SBRT for these patients.
Target delineation and dose prescription followed the previously published research.19–21 For thoracic and abdominal lesions, 48 Gy in 8-Gy fractions or 50 Gy in 10-Gy fractions were preferred. For spine or non-spine bone metastases, 24 Gy in 8-Gy fractions or 35 Gy in 7-Gy fractions were preferred. The dose prescription was modulated according to tumor size and tumor site. Patients receiving irradiation to a single site were treated 3–5 times/week, with the dose per week depending on the fractionation schemes for thoracic/abdominal or spine/non-spine bone lesions as previously described. Whereas patients receiving irradiation to multiple sites were treated at no more than 2 sites/day, and the daily dose was adjusted according to the relevant fraction sizes for different lesion types.
The dose to OARs was maintained below tolerance limits.
22
The biologically effective dose (BED) was calculated using the following formula:
Optimized treatment plans were delivered using an EDGE linear accelerator, with all SBRT cases utilizing image-guided radiotherapy.
Systemic therapy
All patients in this study received systemic therapy combined with SBRT for metastatic lesions. Most patients underwent platinum-based chemotherapy, including GP (gemcitabine and cisplatin), TPF (docetaxel, cisplatin, and fluorouracil), or TP (docetaxel and cisplatin), with or without immunotherapy. After 4–6 cycles, maintenance therapy with capecitabine, S-1, and/or immunotherapy was administered. Systemic therapy was delayed if severe adverse events occurred. For grade 4 hematologic toxicity or grade 3 or higher non-hematologic toxicity, the chemotherapy dose was reduced by 20% for the next cycle.
Following up
Patients were evaluated 1 month after SBRT, followed by assessments every 3 months. Follow-up included physical examination, nasopharyngoscopy, blood tests (including plasma EBV-DNA), and radiological imaging. Adverse events were assessed using the Common Terminology Criteria for Adverse Events version 4.0, and treatment efficacy was evaluated using the Response Evaluation Criteria in Solid Tumors version 1.1. For bone metastases, we primarily used the Positron Emission Tomography Response Criteria in Solid Tumors and the MD Anderson criteria for efficacy evaluation, with some examples shown in Figure S2, where the bone metastasis of patient 1 was evaluated as a complete response (CR) and that of patient 2 was evaluated as PD. The final follow-up occurred on September 30, 2024.
Endpoints
The primary endpoint of this study was progression-free survival (PFS), defined as the time from SBRT to disease progression or death. As illustrated in Figure S3, PFS1 was defined as the time from the diagnosis of metastasis to disease progression or death, while PFS2 was defined as the time from the diagnosis of metastasis to the second progression or death. The secondary endpoints were PFS1, PFS2, and OS, defined as the time from the diagnosis of metastasis to death. LC was defined as the time from SBRT to local progression.
Statistics
Survival rates were estimated using the Kaplan–Meier method, and intergroup differences were assessed with the log-rank test. Multivariable analysis was conducted using Cox regression. Hazard ratios (HRs) and their corresponding 95% confidence intervals (95% CIs) were calculated using COX regression in the multivariable analysis to assess the strength and precision of the associations between variables and survival outcomes. Statistical analyses were performed using SPSS software, version 29.0 (IBM Corporation, Armonk, NY, USA), and images were generated with GraphPad Prism, version 8.0 (GraphPad Software, San Diego, CA, USA). A two-sided p < 0.05 was considered statistically significant.
Results
Patients
The clinical characteristics of the 62 patients with mNPC are summarized in Table 1. The median age was 49 years, ranging from 21 to 85 years. Most patients were male (83.9%), and the majority had an ECOG performance status of 1 (87.1%). Prior to SBRT, plasma EBV-DNA levels in most patients (74.2%) were ⩽103 copies/mL. Among the patients, 17 cases had synchronous metastases, and 45 cases had metachronous metastases. The most frequently involved metastatic organs were the lung (39.3%), bone (29.8%), and liver (20.2%). Most patients (69.4%) had five or fewer metastatic lesions.
Characteristics of mNPC patients undergoing SBRT treatment (n = 62).

Plasma EBV-DNA detected before SBRT.
DNA, deoxyribonucleic acid; EBV, Epstein–Barr virus; ECOG, Eastern Cooperative Oncology Group; mNPC, metastatic nasopharyngeal carcinoma; No., number; PD, progressive disease; PD-1, programmed cell death protein 1; PR, partial response; SBRT, stereotactic body radiation therapy; SD, stable disease.
The median time from diagnosis to SBRT was 7.5 months, ranging from 0 to 51 months. Most patients (67.7%) received SBRT with disease control, and 58.1% received SBRT concurrently with first-line systemic therapy. In addition, a substantial proportion of patients (59.7%) received immunosuppressants.
Clinical outcome and prognostic factors
Table 2 describes the characteristics related to SBRT treatment, and some confusing key concepts are clearly explained in Figure S4. 23 As shown in Table 2, a minority of patients (40.3%) received SBRT for all metastatic lesions, whereas the majority (74.2%) received treatment for only one metastatic lesion. Details of dose prescription according to the localization of metastases are presented in Table S1. For pulmonary metastases, the most commonly used radiation dose fractionation was 48 Gy in 6 fractions (for 11 pulmonary lesions). For bone metastases, the most frequent dose fraction was 24 Gy in 3 fractions (for 12 bone lesions). For liver metastases, the most common regimen was 50 Gy in five fractions (for five liver lesions). The BED for bone metastases was significantly lower compared to that for pulmonary and liver metastases.
Characteristics of SBRT treatment.

CR, complete response; No., number; PD, progressive disease; PR, partial response; SBRT, stereotactic body radiation therapy; SD, stable disease.
In Table 2, a total of 84 metastatic lesions were treated with SBRT, including 47 in the lungs, 22 in bones, and 15 in the liver. The best observed responses were CR in 67.9%, partial response (PR) in 20.2%, stable disease (SD) in 2.4%, and PD in 2.4%, resulting in an overall response rate of 88.1%. Regrettably, the treatment efficacy of 7.1% of the patients was classified as “unknown” due to the unassessable lesions or the patients’ failure to undergo the examination.
The median follow-up time from the diagnosis of metastasis and the initiation of SBRT was 44 months (range from 12 to 85 months) and 32 months (range from 9 to 58 months). At the time of the last follow-up, eight patients had died (one died from checkpoint inhibitor-associated pneumonitis, the others died from disease progression). As illustrated in Figure S5, the median PFS, PFS1, and PFS2 were 19, 20, and 43 months, respectively. The 1-, 2-, and 3-year PFS rates were 56.0%, 46.4%, and 35.5%, respectively. The 1-, 2-, and 3-year PFS1 rates were 71.0%, 46.8%, and 35.7%, while the 1-, 2-, and 3-year PFS2 rates were 96.8%, 79.0%, and 56.1%, respectively. The 1-, 2-, and 3-year OS rates were 100.0%, 96.3%, and 87.2%. The LC rates of SBRT-treated lesions at 1, 2, and 3 years were 95.1%, 89.2%, and 86.5%, respectively.
As depicted in Figure 1, the combination of SBRT with first-line systemic therapy significantly improved PFS, PFS1, and PFS2 compared to the combination of SBRT with multi-line systemic therapy (p = 0.004, p < 0.001, and p < 0.001, respectively). Similarly, administering SBRT during periods of disease control resulted in significantly enhanced PFS, PFS1, and PFS2 compared to its use during disease progression (p = 0.034, p < 0.001, and p = 0.038, respectively). Patients with oligometastatic disease exhibited superior PFS, PFS1, and PFS2 compared to those with non-oligometastatic disease (p < 0.001, p = 0.005, and p = 0.013, respectively). Furthermore, patients who received SBRT for all metastatic lesions demonstrated improved outcomes for PFS, PFS1, and PFS2 (p < 0.001, p < 0.001, and p = 0.001, respectively).

Kaplan–Meier analysis of survival curves stratified by line of SBRT treatment (a–c), disease control status before SBRT (d–f), oligometastasis (g–i), and SBRT all metastatic sites (j–l).
In the AJCC 9th edition staging of nasopharyngeal cancer, M1 is divided into M1a (⩽3 metastatic lesions) and M1b (>3 metastatic lesions), with significant prognostic differences between them. Therefore, in Figure S6, we grouped patients based on the number of metastatic lesions. The results showed that compared with ⩽3 metastatic lesions, >3 metastatic lesions had significantly better PFS, PFS1, and PFS2 (p = 0.001, p = 0.002, and p = 0.003).
As illustrated in Figure 2, patients with plasma EBV-DNA levels ⩽10³ copies/mL prior to SBRT exhibited significantly better PFS and PFS1 compared to those with levels >103 copies/mL (p = 0.018 and p = 0.042, respectively). Moreover, patients with a median time from diagnosis to SBRT of less than 7.5 months had significantly better PFS1 and PFS2 compared to those with a median time of 7.5 months or more (p = 0.004 and p = 0.034, respectively).
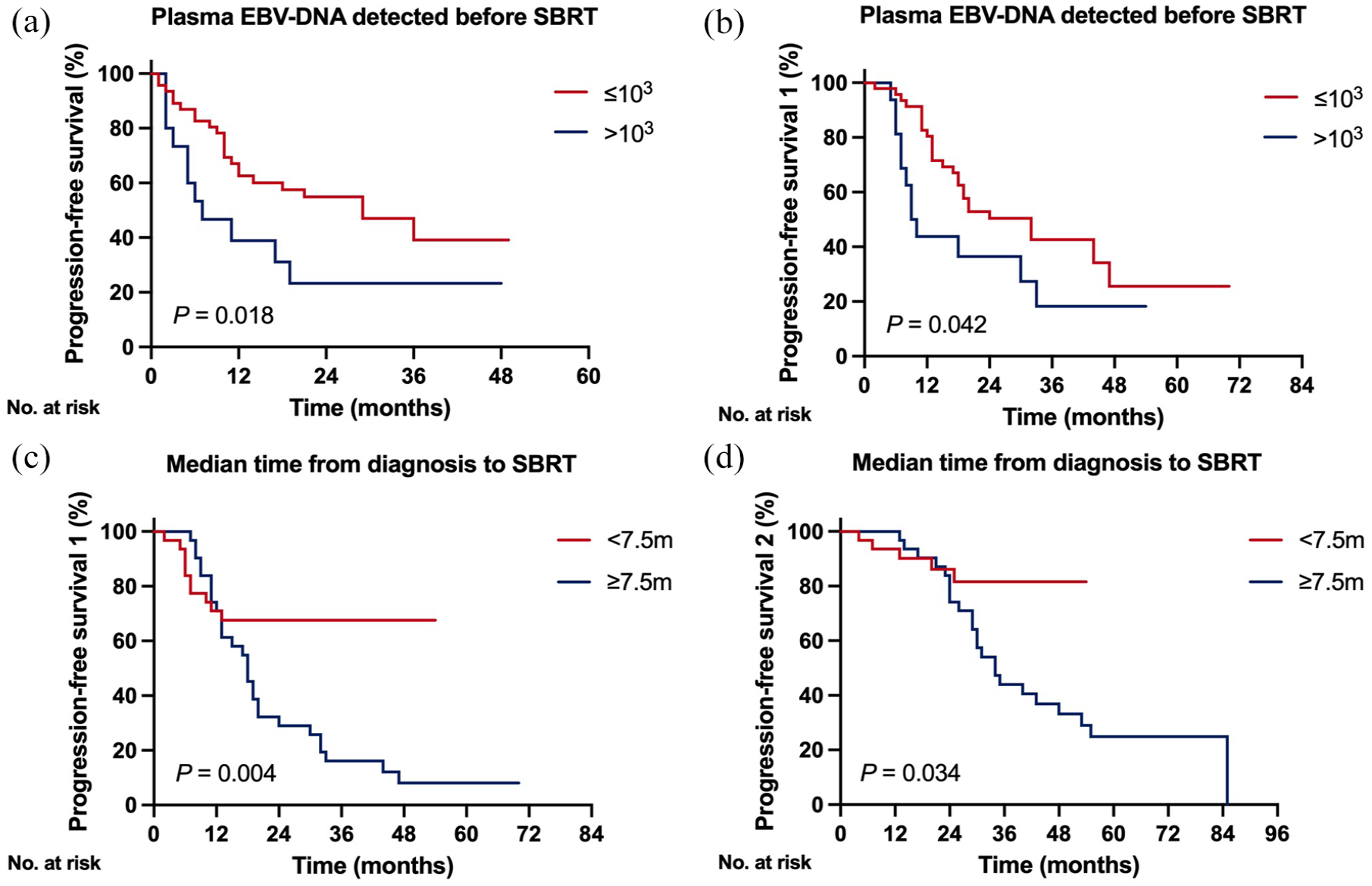
Kaplan–Meier analysis of survival curves stratified by plasma EBV-DNA level detected before SBRT (a, b) and median time from diagnosis to SBRT (c, d).
Multivariable Cox regression analysis was conducted to identify potential prognostic factors, including age (<49 vs ⩾49 years), sex (male vs female), ECOG performance status (1 vs 2), plasma EBV-DNA levels prior to SBRT (⩽103 vs >10³ copies/mL), type of metastasis (metachronous vs synchronous), oligometastasis (yes vs no), median time from diagnosis to SBRT (<7.5 vs ⩾7.5 months), line of SBRT treatment (first-line vs multi-line), disease control status before SBRT (PR/SD vs PD), use of Programmed Cell Death Protein 1 (PD-1) inhibitors (yes vs no), SBRT to all metastatic sites (yes vs no), dose per fraction (<8 vs ⩾8 Gy), total dose (<48 vs ⩾48 Gy), and BED (<77 vs ⩾77 Gy), as shown in Table 3. The analysis indicated that SBRT to all metastatic sites (HR 6.335; 95% CI: 2.399–16.726; p < 0.001), plasma EBV-DNA levels prior to SBRT (HR 2.347; 95% CI: 1.129–4.880; p = 0.022), and use of PD-1 inhibitors (HR 2.072; 95% CI: 1.016–4.225; p = 0.045) were predictors of PFS. Line of SBRT treatment (HR 3.222; 95% CI: 1.449–7.161; p = 0.004), SBRT to all metastatic sites (HR 2.900; 95% CI: 1.063–7.912; p = 0.038), and type of metastasis (HR 2.322; 95% CI: 1.113–4.846; p = 0.025) were predictors of PFS1. In addition, the line of SBRT treatment (HR 2.785; 95% CI: 1.062–7.300; p = 0.037) and SBRT to all metastatic sites (HR 4.466; 95% CI: 0.973–20.495; p = 0.054) were predictors of PFS2. None of these factors was predictive of OS.
Cox multivariable regression analysis of potential prognostic factors for PFS, OS, PFS1, and PFS2.

Plasma EBV-DNA detected before SBRT.
95% CI, 95% confidence interval; EBV, Epstein–Barr virus; HR, hazard ratio; OS, overall survival; PFS, progression-free survival; SBRT, stereotactic body radiation therapy.
Toxicities
In this study, the majority of adverse events following SBRT were Grade 1 or 2, predominantly including radiation pneumonitis in 17 patients (27.4%), cough in 3 patients (4.8%), asymptomatic radiation-induced hepatic fibrosis in 2 patients (3.2%), and chest pain in 1 patient (1.6%). No patients experienced spinal cord injury, brachial plexopathy, or gastrointestinal symptoms such as abdominal pain or distension. Grade 3 radiation pneumonitis was observed in only two patients (3.2%), which recovered with corticosteroid pulse therapy. No patients experienced death related to SBRT.
Discussion
Advancements in treatment modalities have led to improved prognosis for mNPC, with increasing interest in SBRT for metastatic lesions. This study demonstrated that mNPC patients treated with combined systemic therapy and SBRT for metastatic lesions had favorable survival outcomes, with 3-year PFS, 3-year PFS1, 3-year PFS2, 3-year OS, and 3-year LC rates of 35.5%, 35.7%, 56.1%, 87.2%, and 86.5%, respectively. In addition, combining SBRT with first-line therapy and administering SBRT with disease control were associated with better outcomes.
Previous studies have demonstrated that combining systemic chemotherapy with radiotherapy significantly improves survival in patients with mNPC compared to chemotherapy alone.8,24,25 However, most of these studies focused on conventional radiotherapy. Due to its high-dose delivery, precise targeting, and steep dose gradients, SBRT has emerged as a promising approach for treating oligometastatic lesions. Previous research has shown that SBRT not only achieves effective LC but also offers long-term prognostic benefits for patients. 26 Although evidence for SBRT in mNPC metastases is limited, a recent study reported favorable outcomes with metastasis-directed SBRT, showing 3-year LC, 3-year PFS, and 3-year OS rates of 89.1%, 29.4%, and 57.9%, respectively, which are consistent with our findings. 27 Notably, the 3-year OS in our study was superior to that in the previous study, which may be attributed to differences in metastatic organ distribution, proportion of oligometastatic status, usage of maintenance therapy, and other factors.
Currently, there is a lack of evidence regarding the optimal timing for initiating local radiotherapy. Due to the high heterogeneity among mNPC patients, including differences in metastatic sites, treatment lines, and disease status, the ideal timing for local radiotherapy intervention remains unclear. Although PFS is commonly used to compare disease progression after SBRT, the limited systemic treatment options for mNPC patients and the significant prognostic differences between first-line, second-line, and multi-line treatments make this challenging. Therefore, in this study, we also used PFS1 and PFS2 to further evaluate prognosis. Our findings suggest that prognosis is improved when SBRT is administered as early as possible after diagnosis, during first-line systemic therapy before the disease progresses. Thus, we recommend early initiation of SBRT following disease response or stabilization after 4–6 cycles of first-line chemotherapy, followed by maintenance therapy.
Previous studies have demonstrated that BED is a strong prognostic factor.27,28 However, in our study, BED was not a predictor of survival. One possible explanation is the significant variation in BED across different metastatic sites. 29 Typically, for lung and liver metastases, BED greater than 80 Gy is used, while for bone metastases, BED below 60 Gy is applied. Therefore, further research is required to elucidate the relationship between prescribed SBRT doses and treatment efficacy across different metastatic sites.
The toxicity profile of SBRT in treating metastases exhibits site-dependent variations. In lung metastases, SBRT demonstrates a favorable safety profile across multiple clinical series.30–32 However, some research suggests centrally located tumors are associated with elevated risks of pulmonary and esophageal toxicity, emphasizing the importance of stringent patient selection criteria.33–35 For liver metastases, clinical evidence predominantly supports the safety of SBRT, 36 with one study using liver SBRT with active motion—compensation reporting only mild acute toxicities. 37 Regarding bone metastases, SBRT has been widely adopted as one of the standard treatment options in clinical practice, due to its safety, remarkable pain—relieving effect, and high LC rate.38–42 The OligoCare Study, being the largest prospective research on oligometastatic disease to date, evaluated as many as 1468 oligometastatic patients. 43 It reported that only 0.5% of patients experienced acute grade ⩾3 SBRT-related adverse events, including 0.1% fatal ones, further validating SBRT’s safety for such lesions. 43 Consistent with the literature, our study also showed mild SBRT-related toxicities with only two patients (3.2%) developing Grade 3 radiation pneumonitis and recovered after appropriate therapy. While SBRT shows promise in treating metastases, continuous evaluation of toxicity, considering tumor location, and patient factors is crucial for optimal use.
In the era of PD-1 inhibitors for mNPC, the integration of SBRT remains investigational due to the absence of prospective randomized controlled trials establishing its universal efficacy across all metastatic sites. Until high-level evidence emerges, SBRT deployment in this clinical context is suggested to be restricted to high-volume centers with multidisciplinary tumor boards. The optimal dose, fractionation, patient selection, and sequencing with immunotherapy remain to be determined through further trials. Future research should focus on identifying predictive biomarkers and conducting randomized studies to better understand the synergistic effects and potential toxicities of combining SBRT with PD-1 inhibitors in mNPC.
This study has several limitations. First, as a retrospective analysis, it is subject to inherent selection bias. Second, being a single-center study, it is constrained by a small sample size and potential bias from patient selection and limited follow-up duration. Third, the lack of standardized treatment guidelines led to inconsistency in SBRT dose prescriptions for metastatic lesions, potentially affecting patient outcomes.
Conclusion
In conclusion, this study demonstrates that SBRT is an effective and safe local treatment for patients with mNPC. Our findings also indicate that early intervention with SBRT for metastatic lesions during first-line therapy and SBRT to all metastatic sites for oligometastatic disease may help improve disease control, but it should be cautioned when applied clinically. Further prospective, randomized clinical trials are urgently needed to evaluate the feasibility.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251351226 – Supplemental material for Clinical outcomes and prognostic factors of stereotactic body radiation therapy in metastatic nasopharyngeal carcinoma
Supplemental material, sj-docx-1-tam-10.1177_17588359251351226 for Clinical outcomes and prognostic factors of stereotactic body radiation therapy in metastatic nasopharyngeal carcinoma by Ying Zhu and Fen Xue in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251351226 – Supplemental material for Clinical outcomes and prognostic factors of stereotactic body radiation therapy in metastatic nasopharyngeal carcinoma
Supplemental material, sj-docx-2-tam-10.1177_17588359251351226 for Clinical outcomes and prognostic factors of stereotactic body radiation therapy in metastatic nasopharyngeal carcinoma by Ying Zhu and Fen Xue in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-3-tam-10.1177_17588359251351226 – Supplemental material for Clinical outcomes and prognostic factors of stereotactic body radiation therapy in metastatic nasopharyngeal carcinoma
Supplemental material, sj-jpg-3-tam-10.1177_17588359251351226 for Clinical outcomes and prognostic factors of stereotactic body radiation therapy in metastatic nasopharyngeal carcinoma by Ying Zhu and Fen Xue in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-4-tam-10.1177_17588359251351226 – Supplemental material for Clinical outcomes and prognostic factors of stereotactic body radiation therapy in metastatic nasopharyngeal carcinoma
Supplemental material, sj-jpg-4-tam-10.1177_17588359251351226 for Clinical outcomes and prognostic factors of stereotactic body radiation therapy in metastatic nasopharyngeal carcinoma by Ying Zhu and Fen Xue in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-6-tam-10.1177_17588359251351226 – Supplemental material for Clinical outcomes and prognostic factors of stereotactic body radiation therapy in metastatic nasopharyngeal carcinoma
Supplemental material, sj-jpg-6-tam-10.1177_17588359251351226 for Clinical outcomes and prognostic factors of stereotactic body radiation therapy in metastatic nasopharyngeal carcinoma by Ying Zhu and Fen Xue in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-7-tam-10.1177_17588359251351226 – Supplemental material for Clinical outcomes and prognostic factors of stereotactic body radiation therapy in metastatic nasopharyngeal carcinoma
Supplemental material, sj-jpg-7-tam-10.1177_17588359251351226 for Clinical outcomes and prognostic factors of stereotactic body radiation therapy in metastatic nasopharyngeal carcinoma by Ying Zhu and Fen Xue in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-jpg-8-tam-10.1177_17588359251351226 – Supplemental material for Clinical outcomes and prognostic factors of stereotactic body radiation therapy in metastatic nasopharyngeal carcinoma
Supplemental material, sj-jpg-8-tam-10.1177_17588359251351226 for Clinical outcomes and prognostic factors of stereotactic body radiation therapy in metastatic nasopharyngeal carcinoma by Ying Zhu and Fen Xue in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-png-5-tam-10.1177_17588359251351226 – Supplemental material for Clinical outcomes and prognostic factors of stereotactic body radiation therapy in metastatic nasopharyngeal carcinoma
Supplemental material, sj-png-5-tam-10.1177_17588359251351226 for Clinical outcomes and prognostic factors of stereotactic body radiation therapy in metastatic nasopharyngeal carcinoma by Ying Zhu and Fen Xue in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
