Abstract
Objective:
To investigate whether Adjunctive PD-1 inhibitors have improved clinical outcomes compared to chemotherapy alone in platinum-pretreated and platinum-naive recurrent or metastatic nasopharyngeal carcinoma (R/M NPCA).
Methods:
The study involved a literature search from PubMed, Cochrane CENTRAL, and Google Scholar for randomized clinical trials (RCTs) on the use of PD-1 inhibitors versus chemotherapy alone in patients with R/M NPCA. Bias was assessed using Cochrane collaboration’s risk of bias tool. Overall Survival (OS) was examined as the primary endpoint. Secondary endpoints were Progression-Free Survival (PFS), Objective Response Rate, Disease Control Rate (DCR), Duration of Response, and Serious/Grade ⩾3 Adverse Events. Outcomes were measured with either Mean Difference, Risk ratio (RR), or Hazard ratios (HRs) at 95% confidence interval.
Results:
Four RCTs were included in the meta-analysis and systematic review. OS for the monotherapy subgroup was a HR of 0.87 [0.67, 1.13] (p = 0.30) while the combination subgroup had 0.64 [0.45, 0.90] (p = 0.01). The monotherapy subgroup exhibited significantly worse outcomes in PFS (HR 1.31 [1.01, 1.68]) (p = 0.04) and DCR (RR 1.52 [1.12, 2.05]) (p = 0.007) but no significant difference in other outcomes. For combination therapy, a statistically significant benefit can be seen in all outcomes except DCR (RR 0.62 [0.38, 1.01]) (p = 0.06) which was a non-significant benefit favoring PD-1 inhibitors.
Conclusion:
Combination PD-1 inhibitor + chemotherapy followed by maintenance PD-1 inhibitor therapy is superior to chemotherapy alone in the first-line treatment of R/M NPCA, implying a potential benefit with the use of PD-1 inhibitors + chemotherapy with maintenance PD-1 inhibitors as first-line in R/M NPCA compared to standard chemotherapy alone.
Introduction
Nasopharyngeal carcinoma (NPCA) is a malignant neoplasm developing from the epithelial lining of the nasopharynx and commonly associated with Epstein-Barr virus (EBV) infection. It is notable as one of the malignancies with a known ethnic predisposition, arising primarily from people of Chinese descent. Aligning with this, its worldwide distribution in 2018 shows China as occupying an age-standardized incidence rate of 60,558 (47.7%) cases out of 100,000. 1 This was closely followed by Southeast Asia (SEA) with an age-standardized incidence rate of 29,317 (29.3%) cases out of 100,000. 1 This could easily be explained by the proximity of SEA to China, leading to a likely Chinese genetic diaspora onto these regions.
It is speculated that the Human Leukocyte Antigen (HLA) gene and the Cytochrome P450 2A6 (CYP2A6) genes are likely culprits for the predisposition of Chinese individuals and Southeast Asians to developing NPCA. The HLA class II gene, HLA-A*0207, was found to be carried by south Chinese populations at a significantly higher rate than in Caucasians.2,3 This relates to EBV as HLA class II receptors are utilized for viral penetration after the viral glycoprotein, gp42, binds to HLA class II on the cell surface of B cells. 4 CYP2A6 polymorphisms were similarly found to be associated with NPCA according to a case-control study done in the National Cancer Institute of Thailand. CYP2A6 polymorphisms cause carcinogen accumulation by activating aflatoxins and nitrosamines into their ultimate form carcinogens that ultimately lead to DNA damage. It is also involved with the metabolization of nicotine, possibly predisposing to a cyclical behavior of addiction that could further expose an individual to nitrosamines from smoking. 5
NPCA is highly sensitive to chemoradiation which changed its 5 years overall survival (OS) from 25 to 40% to approximately 70% in recent years. 6 For those with untreated recurrent or metastatic (R/M) NPCA, current studies support the efficacy of platinum-based doublet chemotherapy as the recommended treatment.7–9 However, current standard of therapy often leads to a median survival rate of no more than 1 year at most in those with distant metastases.10,11 Thus, studies exploring alternative treatment modalities for a possibility of providing better outcomes are warranted.
Programmed cell death protein 1 (PD-1) inhibitors have been extensively studied in other forms of malignant neoplasms, including other head and neck cancers (HNCs), showing very optimistic outcomes in their use. The phase 3 trial from Ferris et al. 12 on nivolumab use in recurrent and metastatic HNC, except NPCA, showed that OS benefit was significantly longer with nivolumab than with standard therapy [hazard ratio (HR), 0.70; 97.73% confidence interval (CI), 0.51–0.96; p = 0.01]. 12 Similarly, the phase 3 study by Cohen et al. 13 on an identical population showed a significant benefit with pembrolizumab over standard therapy in OS [HR, 0.80 (0.65–0.98); p = 0.016]. 13 PD-1 inhibitors are currently accepted as a standard of treatment in non-small cell lung cancer (NSCLC) with no driver mutations expressing PD-L1 levels of ⩾50% while becoming an essential component of therapy alongside chemotherapy in those with PD-L1 levels of <50%. 14
PD-1 is a surface receptor that has an important part in regulating t-cell function. It is expressed in active T cells, B cells, activated monocytes, and dendritic cells and acts as a negative regulator that helps induce self-tolerance. 15 When stimulated, PD-1 inhibits early activation events of involved cells in the presence of the co-stimulatory signals: CD28 and IL-2. Both CD28 and IL-2 promote an antiapoptotic effect as it affects PD-1. Consequently, withdrawal of these cytokines leads to cell death. 15 With regards to cancers, the most important ligand for PD-1 is the Programmed death-ligand 1 (PD-L1) protein. PD-L1 is usually expressed by tumor-associated antigen presenting cells including dendritic cells, macrophages, fibroblasts, and T cells. 16 It is positively regulated by interferon-γ which is induced in effector t-cells by the presence of tumor cells. 16 When bound to PD-1, PD-L1 inhibits the process of apoptosis that leads to uncontrolled cell growth. Hence the reason why drugs that inhibit the binding of PD-1 to PD-L1 would negatively impact the proliferation of tumor cells. This relationship between PD-1, PD-L1, their chemical regulators, and the association of EBV infection with an increased PD-L1 expression all help explain the role of PD-1/PD-L1 interaction in the growth of NPCA and the rationale behind the use of PD-1 inhibitors as a treatment for malignancy. 17
A meta-analysis would benefit the limited trials currently available by increasing the total power of the studies through pooling together and synthesizing their results. Because of this, we have decided to investigate the efficacy and safety of PD-1 inhibitors as a therapeutic approach for R/M NPCA by doing a meta-analysis.
Methodology
Eligibility criteria
Randomized controlled trials comparing the use of PD-1 inhibitors, with or without chemotherapy, compared to chemotherapy alone in R/M NPCA among adult patients were included in the study. A control group composed of patients diagnosed with NPCA and treated with chemotherapy must be present. The studies included must have a sample population of only adult patients with histologic diagnosis of NPCA. Retrospective studies, case reports, case series, single-armed, and non-randomized studies were excluded from the meta-analysis. Pediatric patients were also excluded given innate differences in physiology. Furthermore, the studies included must also look at the OS, Progression-Free Survival (PFS), Objective Response Rate (ORR), Disease Control Rate (DCR), Duration of Response (DOR), and must provide either the actual counts or HRs. Adverse Effects (AEs) were also included but must be classified as Serious or Grade ⩾3.
Search methods for identification of studies
The search was done according to the PRISMA-P guidelines and wasn’t restricted by language, date, publication status, or any other trial characteristics. The following electronic databases were utilized for the search: PubMed Central as the primary database. Cochrane CENTRAL and Google Scholar were used as secondary database. Specific search keywords and MeSH terms used were: ‘nasopharyngeal carcinoma OR npca’, ‘recurrent OR metastatic’, ‘PD-1’, ‘camrelizumab OR toripalimab OR pembrolizumab OR spartalizumab OR nivolumab OR checkpoint inhibitor OR anti PD’, ‘chemotherapy’ and ‘clinical trial OR RCT OR randomized clinical trial OR randomized controlled trial OR randomized control trial’ to search for relevant studies. The PICO question was ‘Are PD-1 inhibitors, with or without chemotherapy, superior to standard chemotherapy regimens alone in patients with metastatic or recurrent NPCA?’. References within the primary selected studies reviewed in the full text were screened as well as gray literature. Last search was done on 20 September 2022; 2:30:00 pm (Figure 1).
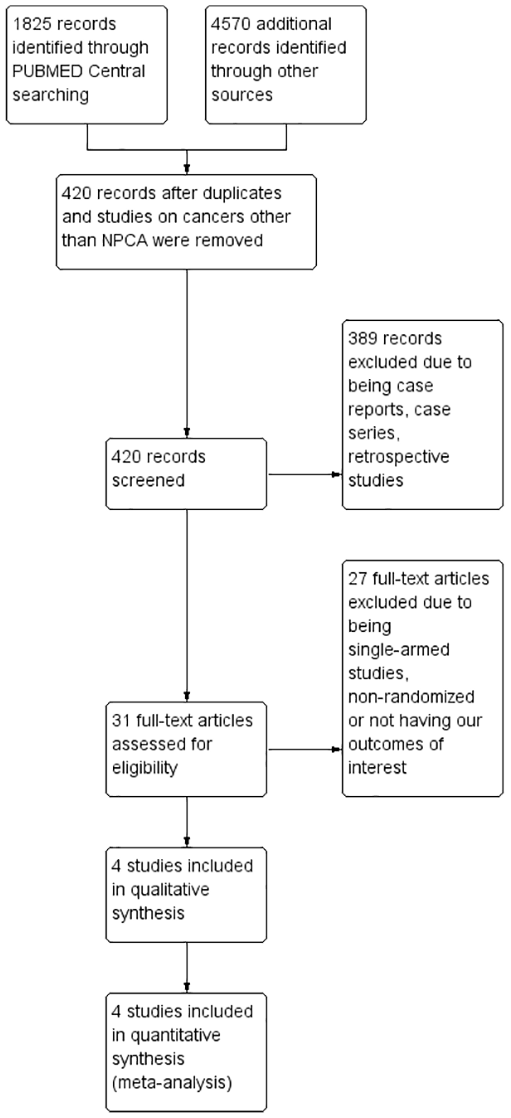
Flow diagram of literature search and study selection based on preferred reporting items for systematic reviews and meta-analyses protocols (PRISMA-P) recommendation.
Selection of studies
Two authors (ETP and JAC) independently screened each title and abstract of each study. For studies with uncertainties evaluated in the title and abstract, the full text was reviewed. All screened studies were assessed for inclusion in accordance with the eligibility criteria. Disagreements were resolved by discussions between the two screening authors. A third author (SC) was consulted when a consensus could not be met.
Data extraction
From the included studies, data were independently extracted such as the type of study design, year of publication, country of origin, sample size and their baseline characteristics, inclusion and exclusion criteria, treatment arms, dosage, length of follow-up, and reported outcomes. From this, the eligibility criteria as well as the nature of intervention were derived.
Outcomes
The primary outcome measure is the OS calculated from both arms and measured in months. We define the OS as the time from randomization until death from any cause. 18 Secondary outcomes include PFS, ORR, DCR, DOR, and Serious or Grade ⩾3 drug-related AEs. ORR is computed by adding complete response and partial response. DCR used the percentage of complete response, partial response, and stable disease. HR was used for OS and PFS measurement. Risk ratio (RR) was used for ORR, DCR, and AEs. Mean difference (MD) was used to measure DOR. All pooled outcomes were assessed with 95% CIs with a random-effects model.
Risk of bias
Two authors (ETP and JAC) independently assessed the risk of systematic errors (bias) in the included studies using the Cochrane collaboration’s risk of bias tool for randomized studies. 19 The criteria to appraise the studies included: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome data, selective reporting, and other biases were assessed (Figure 2).

Risk of bias summary; green: low risk; red: high risk; other bias: presence of sponsorship bias due to financial support from either Merck Sharp & Dohme Corp, Novartis Pharmaceuticals Corporation, Shanghai Junshi Biosciences, or Jiangsu Hengrui Pharmaceuticals and Junshippharma.
Data synthesis
Review Manager version 5.4.1 was used for data synthesis of OS, PFS, ORR, DCR, DOR, and Serious or Grade ⩾3 drug-related AEs. Data types for OS and PFS utilized an inverse-variance model with HR as the measure of association. Meanwhile, a dichotomous analysis utilizing RR was used for ORR, DCR, and AEs. However, plotting of results for ORR and DCR was analyzed through non-events analysis to assess the amount of non-responders while events analysis was used for AE. DOR was analyzed with a continuous data model using the mean in months to plot the MD between study arms. All outcomes were assessed for significance using p-values and outliers with the z-score. Where appropriate, the median was used as a surrogate for the mean by conversion using the methods described by Hozo et al. (2005). 20 The standard of error for plotting HR was computed with the formula ln(HR)/z score or [ln(upper limit of CI) − ln(lower limit of CI)]/3.92. 19 For studies with no reported HR, HR was estimated using the hazard rate obtained with the formula h = ln(2)/Mean Survival Time. 21 Random effects were used for all data synthesized.
Assessment of heterogeneity
Heterogeneity was tested by df, I2, Chi2, and Tau2. I2 was interpreted as follows: 0–40%: might not be important; 30–60%: may represent moderate heterogeneity; 50–90%: may represent substantial heterogeneity; 75–100%: considerable heterogeneity. 19 Subgroup analysis was used in an attempt to control for any heterogeneity.
Results
Search results
Around 4 out of 420 studies were included into the meta-analysis. Included studies were required to be two-armed randomized clinical trials (RCTs) measuring the difference between PD-1 inhibitors versus standard chemotherapy on an adult NPCA population. Retrospective studies, case reports or case series, including studies in pediatric patients, were excluded. Single-armed and non-randomized studies were also excluded from the data synthesis. PubMed, Cochrane CENTRAL, and Google Scholar were used to search for eligible studies. Studies must have also measured OS, PFS, ORR, DCR, DOR, and Serious or Grade ⩾3 drug-related AEs as outcomes to be included into the meta-analysis. A flow-diagram of the search strategy can be seen in Figure 1.
All included studies were RCTs comparing the efficacy and safety of PD-1 inhibitors versus chemotherapy published in 2021. The studies by Chan et al., 22 Yang et al., 23 and Mai et al. 24 were all Phase III RCTs whereas the study by Even et al. 25 was a Phase II RCT. All three were published as original articles with the exception of Chan et al. 22 which was published as an abstract. A total of 907 participants were randomized and analyzed according to subgroups with a total of 355 in the monotherapy subgroup and 552 in the combination subgroup. In the analysis, certain distinctions were made for subgroup pairings. The studies by Chan et al. and Even et al. were categorized as the monotherapy subgroup while the studies by Yang et al. and Mai et al. were categorized as the combination therapy subgroup. Characteristics shared within the monotherapy subgroup were the use of single-agent PD-1 inhibitors, an open-label study design, and a population of platinum-pretreated R/M NPCA. Whereas those in the combination subgroup were given combined PD-1 inhibitor + chemotherapy, had a double-blinded design, and a population of R/M NPCA undergoing first-line treatment. A cumulative subgroup analysis was not done due to inherent differences in their populations, namely whether patients were platinum pre-treated or undergoing first-line therapy. A more comprehensive descriptive analysis of the individual studies can be seen in Table 1.
Summary of study characteristics.

Estimated value.
CNS, central nervous system; DCR, Disease Control Rate; DOR, Duration of Response; ECOG, Eastern Cooperative Oncology Group; GP, gemcitabine + cisplatin; HBC, hepatitis B virus; HCV, hepatitis C virus; IQR, interquartile range; irRC, immune-related response criteria; NE, not estimable; NPCA, nasopharyngeal cancer; ORR, Objective Response Rate; OS: Overall Survival; PFS, Progression-Free Survival; RECIST, response evaluation criteria in solid tumors.
Objective response rate
PD-1 monotherapy subgroup analysis showed an RR of 1.12 [0.90, 1.37] (p = 0.31). The combination subgroup showed an RR of 0.67 [0.49, 0.91] (p = 0.01) (Supplemental Figure 1).
Disease control rate
PD-1 monotherapy subgroup showed an RR of 1.52 [1.12, 2.05] (p = 0.007). Combination therapy showed an RR of 0.62 [0.38, 1.01] (p = 0.06) (Supplemental Figure 2).
Duration of response
PD-1 monotherapy showed an MD of 2.43 [−2.86, 7.73] (p = 0.37), revealing a non-significant benefit. The combination therapy subgroup showed a statistically significant benefit with an MD of 3.71 [2.35, 5.06] (p < 0.001) (Supplemental Figure 3).
Safety
The monotherapy subgroup revealed an RR of 0.64 [0.30, 1.39] (p = 0.26) for Total Adverse Events (Supplemental Figure 4) While the combination subgroup showed an RR of 1.01 [0.96, 1.07] (p = 0.65) for Total Adverse Events (Supplemental Figure 5). Analysis of specific AEs showed no significant difference between treatment and control arms with the exceptions of monotherapy anemia (RR 0.13 [0.02, 0.71]; p = 0.02), monotherapy neutropenia (RR 0.08 [0.01, 1.01]; p = 0.05), and monotherapy diarrhea (RR 0.10 [0.01, 0.83]; p = 0.03). Information on the AEs included in the data synthesis can be seen on Supplemental Table 1.
Progression-free survival
The PFS for PD-1 monotherapy showed an HR of 1.31 [1.01, 1.68] (p = 0.04). Whereas, the combination subgroup showed an HR of 0.53 [0.41, 0.68] (p < 0.001) (Figure 3).

Progresion-free survival forest plot.
Overall survival
PD-1 monotherapy revealed an HR of 0.87 [0.67, 1.13] (p = 0.30). The combination subgroup revealed an HR of 0.64 [0.45, 0.90] (p = 0.01) (Figure 4).

Overall survival forest plot.
Discussion
Included PD-1 inhibitors
The studies included in our data synthesis utilized four drugs within the category of PD-1 inhibitors: pembrolizumab, spartalizumab, toripalimab, and camrelizumab. PD-1 inhibitors are a form of targeted therapy that blocks PD-1/PD-L1 interaction using humanized monoclonal IgG4 kappa anti-PD1 antibodies. Because of this, they have no cytotoxic effects unlike conventional chemotherapy. Pembrolizumab is the first FDA approved PD-1 inhibitor in the US on 4 September 2014. 26 It is often used in the treatment of metastatic or advanced unresectable melanoma and PD-1 positive NSCLC.27–29 Spartalizumab is a drug that is currently not approved by the FDA for use outside of clinical trials with a phase 1 dose escalation study finding spartalizumab being well tolerated in patients with advanced solid tumors. 30 However unlike pembrolizumab, spartalizumab is usually associated with only a non-significant benefit in unresectable metastatic melanoma. 31 Toripalimab was approved for use in China in December 2018 for use in the treatment of unresectable or metastatic melanoma that has failed previous systemic therapy and is generally well tolerated in Chinese patients with advanced malignancies.32,33 Camrelizumab was a drug conditionally approved for the treatment of relapsed or refractory classical Hodgkin lymphoma who have received at least two previous systemic chemotherapies in May 2019 by China. 34 A more detailed comparison of the included drugs could be seen in supplemental Table 2.
Monotherapy as second-line
Results from our data synthesis of the monotherapy subgroup showed that PD-1 inhibitors exhibit no significant benefits with OS, DOR, and total AE. Significantly worse outcomes were seen on PFS and DCR while a non-significant result was seen in ORR. However, PD-1 monotherapy had significantly less rates of anemia, neutropenia, and diarrhea compared to chemotherapy alone. Which is congruent with the less cytotoxic effects of PD-1 inhibitors in contrast to platinum-based chemotherapy. When compared to the phase III trial by Cohen et al. on platinum pre-treated R/M squamous cell HNC samples, those with Combined Positive Score (CPS) ⩾1 showed a significant OS benefit (0.74 [0.58, 0.93]; p = 0.0049). 13 While PFS, ORR, and AEs showed no significant difference between treatment and control arms. The difference in results between the pooled NPCA and HNC outcomes with PD-1 monotherapy tells us that the physiologic and genetic differences driving HNC do not equate to that of the NPCA population as the study by Cohen et al. analyzed R/M squamous cell HNC of the oropharynx, hypopharynx, and larynx. Supporting this is the fact that there is a notable predominance of Caucasians in the HNC samples by Cohen et al. which is in contrast to the majority Asian population from the monotherapy subgroup’s NPCA samples. This highlights the necessity of differentiating NPCA from other HNCs. Additionally, Cohen et al. analyzed those with CPS ⩾1 separately from those with CPS <1 whereas Chan et al. and Even et al. did not separate their analysis by CPS score. There is also a difference in the number of participants analyzed as Cohen et al. (n = 387) outnumbers the monotherapy subgroup (n = 276) in CPS ⩾1 participants. These differences could be the reason as to why NPCA appears to be responding worse compared to other HNC. Overall, it appears that PD-1 inhibitor monotherapy is not superior to chemotherapy in the platinum pre-treated R/M NPCA in an Asian population.
Combination therapy as first-line
The combination subgroup showed a consistent benefit in all five outcomes with no significant difference noted in the risk of total AEs. In comparison, a recently published RCT abstract by Zhang et al. comparing tislelizumab + chemotherapy versus placebo + chemotherapy on 263 R/M NPCA patients undergoing first-line therapy is similar to our combination subgroup’s population. Zhang et al. 35 found that an HR of 0.60 [0.35, 1.01] for OS, 0.50 [0.37, 0.68] for PFS, and 0.38 [0.25, 0.58] for PFS post-tislelizumab monotherapy after noted disease progression. The results from this study supports our findings despite the non-significant benefit seen in OS for the tislelizumab arm. Similarly, a meta-analysis using nine RCTs by Petrelli et al. on the usage of immune checkpoint inhibitors (ICI) on NSCLC shows similar results to our findings where OS, PFS, and ORR were found to be significantly in favor of ICI + chemotherapy compared to chemotherapy alone. 36 In contrast to our results however, Grade 3–5 AEs were noted to be significantly more frequent in the ICI arm. This could be explained by the usage of CTLA-4 and PD-L1 inhibitors where it was found that those patients given ipilimumab, a CTLA-4 inhibitor, or atezolizumab, a PD-L1 inhibitor, expressed significantly higher rates of AEs. It is also notable that Petrelli et al. showed a moderate to high amount of heterogeneity on all parameters measured (I2 = 48–82%) which implies inconsistency in the effects of the pooled studies. Interestingly, Petrelli et al. also found that the magnitude of benefit was low in those with squamous histology, PD-L1 expression <50%, liver metastases, female sex, and never-smoking history. Making investigations into these subpopulations of patients is worth looking into for NPCA as well. Nonetheless, our findings suggest a general trend of improved outcomes with PD-1 inhibitor + chemotherapy over chemotherapy alone as a treatment modality in the first-line setting of R/M NPCA patients.
Recommendations and caveats
Although other published studies with PD-1 inhibitors in the HNC population show promising results, there is currently a severe lack of PD-1 or PD-L1 checkpoint inhibitor RCT studies on the NPCA population. Our meta-analysis suggests a very optimistic outcome with the use of combined PD-1 inhibitor + chemotherapy as a treatment modality in R/M NPCA as first-line treatment. Thus, we recommend more clinical trials comparing this modality with standard of care to definitively conclude the existence of superiority which could potentially change the future management of this disease. Currently, there are three phase 3 trials (NCT04458909, NCT03924986, and NCT04974398) and one phase 1 trial (NCT04282070) ongoing that compares combined chemotherapy and PD-1 inhibitors with standard chemotherapy alone in R/M NPCA patients. 37 Another meta-analysis could be done once more RCTs on this subject are available for pooling.
According to a cost-effectiveness analysis by Zhu et al. toripalimab + chemotherapy associated with an incremental cost-effectiveness ratios (ICERs) of $19,726 per quality-adjusted life-years (QALY) while camrelizumab + chemotherapy was associated with $20,438 per QALY. 38 In comparison, standard chemotherapy is estimated to have a total cost of $26,680 compared to $48,525 and $46,293 for toripalimab and camrelizumab + chemotherapy respectively. Despite this substantial increase in cost, the same study goes on to say that willingness-to-pay (WTP) for Chinese citizens was found to be at $35,673 per QALY using a Markov model. Therefore, due to ICER being lower than WTP, it would appear that cost-effectiveness for PD-1 inhibitor therapy is still met despite the higher total cost of a PD-1 inhibitor + chemotherapy regimen for at least the Chinese population.
An interesting result from Even et al. 25 is their finding of a negative correlation between IFN-gamma, TIM-3, and LAG-3 status to spartalizumab response. Additionally, Huang et al. found that PD-1 inhibitors and PD-L1 inhibitors have differing effectiveness in HNC patients, as only a non-statistically significant result in PD-L1 inhibitor + chemotherapy combination was seen [PD-1 inhibitors (RR 1.91 [1.42, 2.56]); PD-L1 inhibitors (RR of 1.54 [0.81, 2.94])]. 39 In comparison, a statistically significant benefit with PD-1 inhibitor + chemotherapy was seen from the clinical trials included in this meta-analysis as well as the subgroup analysis done by Huang et al. themselves. Therefore, the difference in efficacy between PD-L1 versus PD-1 inhibitors as well as further studies on IFN-gamma, TIM-3, and LAG-3 positive patients’ response to PD-1 inhibitors would be worthwhile avenues to further investigate.
Limitations
Only four eligible studies were included in our pooled data synthesis. The addition of a phase II trial with a smaller sample size decreases the total power of our pooled analysis. For similar reasons, the subjects may have also been given suboptimal drug dosing which could have led to the non-significant results seen. 40 The two studies included in the monotherapy subgroup were open-label, which predisposed both studies to performance and detection bias, and did not meet their primary endpoints. Of the studies included in the combination subgroup, the CAPTAIN-1st trial by Yang et al. and the JUPITER-02 trial by Mai et al. had the additional limitation of comparing their maintenance phase PD-1 inhibitor therapy with placebo instead of maintenance chemotherapy.23,24 Notably however, this is still in line with current guidelines as maintenance chemotherapy is currently not recommended due to being associated with higher risks for AEs. 41 Approximately, only 60% are able to finish their planned maintenance chemotherapy and nearly 50% require dose reduction. 41 It is also worth noting that all studies were financed by pharmaceutical companies, risking sponsorship bias especially for those under an open-label design. However, third-party review committees were utilized by all included studies in an attempt to mitigate this effect.
In three out of four studies, PD-L1 was the only biomarker measured while Yang et al. didn’t measure for PD-L1 status at all. There was also no measurement of outcomes by PD-L1 status, resulting in our data synthesis being unable to do a subgroup analysis response per PD-L1 status. Similarly, Even et al. was the only one who measured for IFN-gamma, TIM-3, and LAG-3 status which appeared to have shown a negative correlation for spartalizumab response in platinum-pretreated patients. However, their small sample size limits the validity of this finding and more studies are needed to verify this result. Another issue concerns the study by Chan et al. where the abstract and results have been published but has not yet undergone full peer-review at the time of this paper’s writing. Additionally, all of the included studies had a majority EBV-positive patient sample. This indicates that the findings from our analysis is only applicable to EBV-positive patients. Lacking representation of EBV-negative patients, a relevant population of NPCA patients in non-endemic regions outside of Asia.
Finally, all studies included were RCTs which represent the best level of evidence available at the time of this article’s writing. However, the presence of confounders leads to a downrating in validity based on the issues raised by all the aforementioned biases. Despite this, the consistent benefit seen in combination PD-1 inhibitors + chemotherapy subgroup in all five outcomes and lack of significant toxicity even after prolonged exposure in the PD-1 inhibitor arm’s maintenance phase leads us to believe our present evidence warrants at least a GRADE certainty rating of moderate for all outcomes measured. 42 Meanwhile the monotherapy subgroup’s outcomes could be given a low to moderate GRADE certainty rating due to the inherent limitations mentioned before. 42
Conclusion
In R/M NPCA, both PD-1 inhibitor monotherapy and combination therapy are comparable in safety to chemotherapy alone. However, PD-1 inhibitor monotherapy in platinum pre-treated R/M NPCA is not superior to chemotherapy alone as second-line treatment. Conversely, combination PD-1 inhibitor + chemotherapy as first-line treatment of R/M NPCA appears to be superior to chemotherapy alone, thereby implying a potential benefit with the use of PD-1 inhibitors + chemotherapy with maintenance PD-1 inhibitors as first-line in R/M NPCA as opposed to standard chemotherapy alone. More evidence from RCTs are needed to further validate the role of PD-1 inhibitors in the management of NPCA.
Supplemental Material
sj-docx-6-tam-10.1177_17588359221137429 – Supplemental material for Adjunctive PD-1 inhibitor versus standard chemotherapy in recurrent or metastatic nasopharyngeal carcinoma: a systematic review and meta-analysis
Supplemental material, sj-docx-6-tam-10.1177_17588359221137429 for Adjunctive PD-1 inhibitor versus standard chemotherapy in recurrent or metastatic nasopharyngeal carcinoma: a systematic review and meta-analysis by Edgar Theodore Polintan, Stephanie Krystel Canicula, Jesus Alfonso Catahay, Kevin Bryan Lo, Miguel Villalona-Calero and Herbert Ho-fung Loong in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-7-tam-10.1177_17588359221137429 – Supplemental material for Adjunctive PD-1 inhibitor versus standard chemotherapy in recurrent or metastatic nasopharyngeal carcinoma: a systematic review and meta-analysis
Supplemental material, sj-docx-7-tam-10.1177_17588359221137429 for Adjunctive PD-1 inhibitor versus standard chemotherapy in recurrent or metastatic nasopharyngeal carcinoma: a systematic review and meta-analysis by Edgar Theodore Polintan, Stephanie Krystel Canicula, Jesus Alfonso Catahay, Kevin Bryan Lo, Miguel Villalona-Calero and Herbert Ho-fung Loong in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-eps-1-tam-10.1177_17588359221137429 – Supplemental material for Adjunctive PD-1 inhibitor versus standard chemotherapy in recurrent or metastatic nasopharyngeal carcinoma: a systematic review and meta-analysis
Supplemental material, sj-eps-1-tam-10.1177_17588359221137429 for Adjunctive PD-1 inhibitor versus standard chemotherapy in recurrent or metastatic nasopharyngeal carcinoma: a systematic review and meta-analysis by Edgar Theodore Polintan, Stephanie Krystel Canicula, Jesus Alfonso Catahay, Kevin Bryan Lo, Miguel Villalona-Calero and Herbert Ho-fung Loong in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-eps-2-tam-10.1177_17588359221137429 – Supplemental material for Adjunctive PD-1 inhibitor versus standard chemotherapy in recurrent or metastatic nasopharyngeal carcinoma: a systematic review and meta-analysis
Supplemental material, sj-eps-2-tam-10.1177_17588359221137429 for Adjunctive PD-1 inhibitor versus standard chemotherapy in recurrent or metastatic nasopharyngeal carcinoma: a systematic review and meta-analysis by Edgar Theodore Polintan, Stephanie Krystel Canicula, Jesus Alfonso Catahay, Kevin Bryan Lo, Miguel Villalona-Calero and Herbert Ho-fung Loong in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-eps-3-tam-10.1177_17588359221137429 – Supplemental material for Adjunctive PD-1 inhibitor versus standard chemotherapy in recurrent or metastatic nasopharyngeal carcinoma: a systematic review and meta-analysis
Supplemental material, sj-eps-3-tam-10.1177_17588359221137429 for Adjunctive PD-1 inhibitor versus standard chemotherapy in recurrent or metastatic nasopharyngeal carcinoma: a systematic review and meta-analysis by Edgar Theodore Polintan, Stephanie Krystel Canicula, Jesus Alfonso Catahay, Kevin Bryan Lo, Miguel Villalona-Calero and Herbert Ho-fung Loong in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-eps-4-tam-10.1177_17588359221137429 – Supplemental material for Adjunctive PD-1 inhibitor versus standard chemotherapy in recurrent or metastatic nasopharyngeal carcinoma: a systematic review and meta-analysis
Supplemental material, sj-eps-4-tam-10.1177_17588359221137429 for Adjunctive PD-1 inhibitor versus standard chemotherapy in recurrent or metastatic nasopharyngeal carcinoma: a systematic review and meta-analysis by Edgar Theodore Polintan, Stephanie Krystel Canicula, Jesus Alfonso Catahay, Kevin Bryan Lo, Miguel Villalona-Calero and Herbert Ho-fung Loong in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-eps-5-tam-10.1177_17588359221137429 – Supplemental material for Adjunctive PD-1 inhibitor versus standard chemotherapy in recurrent or metastatic nasopharyngeal carcinoma: a systematic review and meta-analysis
Supplemental material, sj-eps-5-tam-10.1177_17588359221137429 for Adjunctive PD-1 inhibitor versus standard chemotherapy in recurrent or metastatic nasopharyngeal carcinoma: a systematic review and meta-analysis by Edgar Theodore Polintan, Stephanie Krystel Canicula, Jesus Alfonso Catahay, Kevin Bryan Lo, Miguel Villalona-Calero and Herbert Ho-fung Loong in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
