Abstract
Background:
Studies of local therapy (LT) to metastatic foci from nasopharyngeal carcinoma (NPC) are inconsistent and controversial. Here, we aimed to explore the survival benefit of LT directed at metastatic foci from NPC.
Methods:
A retrospective analysis was conducted in NPC patients with liver, lung, and/or bone metastases. The postmetastatic overall survival (OS) rate was analyzed using the Kaplan–Meier method and compared by the log-rank test. Multivariate analysis was performed using the Cox hazard model. Subgroup analyses evaluating the effect of LT were performed for prespecified covariates. Propensity score matching was applied to homogenize the compared arms.
Results:
Overall, 2041 of 2962 patients were eligible for analysis. At a median follow-up of 43.4 months, the 5-year OS improved by an absolute difference of 14.6%, from 46.2% in the LT group versus 31.6% in the non-LT group, which led to a hazard ratio of 0.634 for death (p < 0.001). Matched-pair analyses confirmed that LT was associated with improved OS (p = 0.003), and the survival benefits of LT remained consistent in the subcohorts of liver and lung metastasis (p = 0.009 and p = 0.007, respectively) but not of bone metastasis (BoM; p = 0.614). Radiotherapy was predominantly used for BoM and biological effective dose (BED) >60 Gy was found to yield more survival benefit than that of BED ⩽ 60 Gy.
Conclusions:
The addition of LT directed at metastasis has demonstrated an improvement to OS compared with non-LT group in the present matched-pair study, especially for patients with liver and/or lung metastases.
Introduction
Nasopharyngeal carcinoma (NPC) is rare in Western countries but is prevalent in China and Southeast Asia. NPC is distinct from other head and neck cancers due to its high rates of distant metastasis, including synchronous metastasis (smNPC, metastasis at initial diagnosis) and metachronous metastasis (mmNPC, metastasis over 6 months after radical chemoradiotherapy).1–3 The incidences of smNPC and mmNPC have been reported to be 4%−10% and 15%–30%, respectively.4–6 When metastasis occurs, the survival outcomes of patients are unsatisfactory with a median overall survival (OS) of 12–30 months after multidisciplinary therapies.1,7
Aiming to explore the most effective regimen for metastatic NPC (mNPC) and to improve survival outcome of patients, a variety of strategies, including systemic chemotherapy, immunotherapy, targeted therapy, treatment of in situ primary tumors and local therapy (LT) to metastatic foci, have been utilized over the past decade.8–10 To date, systematic chemotherapy remains the mainstay of treatment for mNPC. 11 Zhang et al. 12 established gemcitabine and cisplatin doublet chemotherapy as the standard first-line treatment for patients with recurrent or metastatic NPC. Immunotherapy based on immune checkpoint inhibitors shows promising efficacy both in first-line and in the following lines of treatment for mNPC.13–18 Studies have reported that the addition of targeted therapy (cetuximab) to chemotherapy is associated with an improvement in survival in patients with recurrent or metastatic NPC.19–22 However, significant toxicities of targeted therapy, such as hemorrhagic events, leucopenia, and hand–foot–skin reactions, cannot be ignored.22–25 Prospective randomized phase III trials will be necessary to validate the efficacy of targeted therapy. In addition to systematic therapy, treatment of in situ primary tumors is also important. The results from a multicenter, randomized phase III clinical trial suggest that definitive radiotherapy (RT) for the primary nasopharynx and cervical lymph nodes as well as systematic chemotherapy significantly improve survival in smNPC patients who are sensitive to induction chemotherapy. 26
As for LT to metastasis, its therapeutic effect in mNPC remains inconsistent and controversial. Several retrospective studies found that LT, such as surgery or radiofrequency ablation (RFA) for liver metastasis (LiM) 27 or lung metastasis (LuM) 28 and RT for bone metastasis (BoM), 29 may provide additional survival benefits for patients. Liang et al. 30 analyzed 448 mmNPC patients and found that LT to metastasis could benefit patients’ survival. In contrast, a multicenter population study of three cohorts containing 977 smNPC patients has demonstrated that LT is not associated with better survival in patients undergoing systemic chemotherapy and radical RT for primary tumors even for patients with oligometastasis and without liver involvement. 31 However, due to limited sample sizes, no propensity score matching was applied or inadequate confounding variables were matched to achieve a balanced exposure groups at baseline in these retrospective studies. Herein, we conducted this study with a large cohort (n = 2041) of mNPC patients (synchronous and metachronous) and long-term follow-up (median: 43.4 months) and adopted a PSM method to strictly homogenize the compared arms, aiming to explore the survival benefit of LT for mNPC patients.
Materials and methods
Study population
Consecutive mNPC patients at Sun Yat-Sen University Cancer Center (SYSUCC) between January 2006 and December 2020 were recruited. Tumors were restaged according to the eighth edition of the TNM staging system by the Union for International Cancer Control/American Joint Committee on Cancer. 32 The inclusion criteria were as follows: (a) histologically confirmed NPC; (b) Karnofsky performance status (KPS) score ⩾70; (c) smNPC or mmNPC; (d) LiM, LuM, and/or BoM, single, or combined metastasis; (e) treatment-naive for metastatic disease at diagnosis; and (f) with complete medical record. The exclusion criteria were as follows: (a) previously suffering from or concomitant other malignancies; (b) combined with other metastatic sites (spleen, adrenal gland, brain, etc.); (c) combined with tumor recurrence; and (d) follow-up time <1 month.
Data collection and definition
The following data were collected: (a) patient demographics, including sex, age at the initial diagnosis of NPC, KPS score, and Epstein–Barr virus (EBV) DNA count before treatment for metastases (pre-EBV DNA); (b) tumor characteristics, including pathological type, T/N/M stage, and metastatic sequence (synchronous or metachronous); (c) treatment characteristics, including chemotherapy, immunotherapy, targeted therapy, RT for primary tumor, and LT for DM; and (d) survival status and time point. The definition of metastasis has been previously reported. 33 Briefly, suspicious lesions detected by conventional work-up or positron emission tomography/computed tomography (PET/CT) were identified as metastasis only when additional examinations [CT, magnetic resonance imaging (MRI), or biopsy] confirmed the diagnosis.
Treatment
The treatment regimens were extracted directly from the electronic medical records. Chemotherapy was nearly exclusively cisplatin-based with the combination of one or two of the following drugs: fluorouracil, paclitaxel, gemcitabine, navelbine, or others. Targeted therapy included anti-epidermal growth factor receptor agents (cetuximab or nimotuzumab), single target angiogenesis inhibitor (bevacizumab) and multitarget angiogenesis inhibitors (apatinib, famitinib, or anlotinib). Immunotherapy mainly included immune checkpoint inhibitors (antibodies against CTLA-4, PD-1, or PD-L1 inhibitor). LT to metastatic liver included surgery, RT, RFA, transcatheter hepatic artery chemoembolization (TACE), and particle implantation. LT to metastatic lungs included surgery, RT, RFA, and particle implantation. LT to metastatic bones included surgery, RT, RFA, and radionuclide therapy.
Endpoints and statistical analysis
Baseline characteristics were converted into binary variables and are presented as numbers with percentages. The best cutoff value of biological effective dose (BED) was selected from median, receiver operating characteristic (ROC) curve, and minimum p-value method based on R package of ggrisk. The distributional differences of categorical variables were compared using the chi-square test or Fisher’s exact test. OS was defined as the time from the diagnosis of metastasis to the time of death due to any cause or the last follow-up. The cumulative survival rates were estimated using the Kaplan–Meier method and were compared using the log-rank test. The median follow-up duration was calculated by the reverse Kaplan–Meier method. To explore independent prognostic factors, baseline characteristics with statistical significance (p < 0.05) under univariate analyses were included in a Cox proportional hazards model as confounding factors. Matched cohorts were generated via a 1:1 PSM method with a stringent caliper of 0.01. Several rounds of matching were conducted until all parameters were balanced. Statistical analyses were conducted using R 4.0.3 and SPSS version 26.0 (Chicago, IL, USA). Statistical significance was defined as a two-sided p value of less than 0.05.
Results
Patient characteristics
A flow diagram of this study is presented in Figure 1. Totally, 2041 of 2962 patients who met all criteria were enrolled in the final analyses. The median age was 44.0 years (Interquartile range, IQR: 37.0–52.0). Male predominance (81.2%) was found. Patients had a good KPS of ⩾90 in 90.2% of cases. The pathological subtype of nonkeratinizing undifferentiated NPC (type III) accounted for 97.8% of cases. According to the initial diagnosis, patients had a T4 tumor in 30.3% of cases and N3 lymph node involvement in 40.3% of cases. A total of 1102 patients had mmNPC, and 939 patients had smNPC. Half of the patients (50.2%) had a pre-EBV DNA count ⩾ 104 copies/mL (the cutoff value was referred to a previous report 34 ). Bone was the most frequent metastatic site (n = 1341, 65.7%) followed by the liver (n = 908, 44.5%) and lung (n = 833, 40.8%) (Table 1).

Flowchart for patient selection, matched cohorts generation and survival comparison.
Baseline patient demographics, tumor characteristics, and treatment characteristics.

BoM, bone metastasis; DM, distant metastasis; KPS, Karnofsky performance status; LiM, liver metastasis; LuM, lung metastasis; LT, local therapy; RFA, radiofrequency ablation; RT, radiotherapy.
Type I: keratinizing; type II: nonkeratinizing differentiated; type III: Nonkeratinizing undifferentiated.
Surgery + RT or surgery + RFA or RT + RFA.
Others included transcatheter hepatic artery chemoembolization (TACE) and particle implantation.
Surgery + RT or surgery + RFA or RT + RFA or RFA + particle implantation;
Others included surgery, radionuclide therapy, and RFA.
Regarding the treatment characteristics, 1873 (91.8%), 490 (24.0%), and 530 (26.0%) patients underwent chemotherapy, targeted therapy, and immunotherapy, respectively. Chemotherapy was nearly exclusively cisplatin-based with 1149 (56.3%) patients who received six cycles or more (median: 6, range: 1–22). 91.7% of patients received RT for primary tumor (median dose: 7000 cGy, range: 5000–7976 cGy). A total of 640 (31.4%) patients received LT to metastatic foci. Specifically, 235, 144, and 305 patients underwent LT for metastatic liver, lung, and bone, respectively. The most frequently used LT method for LiM was RFA followed by RT and surgery. The top three LT methods used for LuM were surgery, RT, and RFA. For BoM, RT was predominantly used as the LT method (Table 1).
Survival benefit of LT in the entire population
In the entire cohort, 640 (31.4%) patients received LT. Before matching, significant differences were detected for age, pre-EBV DNA and other variables (p < 0.05 for all) (Table 2). As shown in Figure 2(a), a statistically associated difference was observed at 1, 3, and 5 years for OS rates (91.7 versus 85.6%, 61.2 versus 47.8% and 46.2 versus 31.6%, respectively; all p < 0.001) between patients with and without LT. After PSM, 364 patients who underwent LT were matched with 364 patients who lacked LT (all p > 0.1; Table 2). Compared to patients who did not receive LT, LT did improve the survival of mNPC patients (p = 0.003; Figure 2(b)). Multivariate analyses confirmed LT as an independent favorable predictor of OS (hazard ratio, HR = 0.634, and 95% CI = 0.531–0.757% CI; p < 0.001) (Table 3).
Baseline characteristics between patients with and without LT before and after matching in the entire cohort.
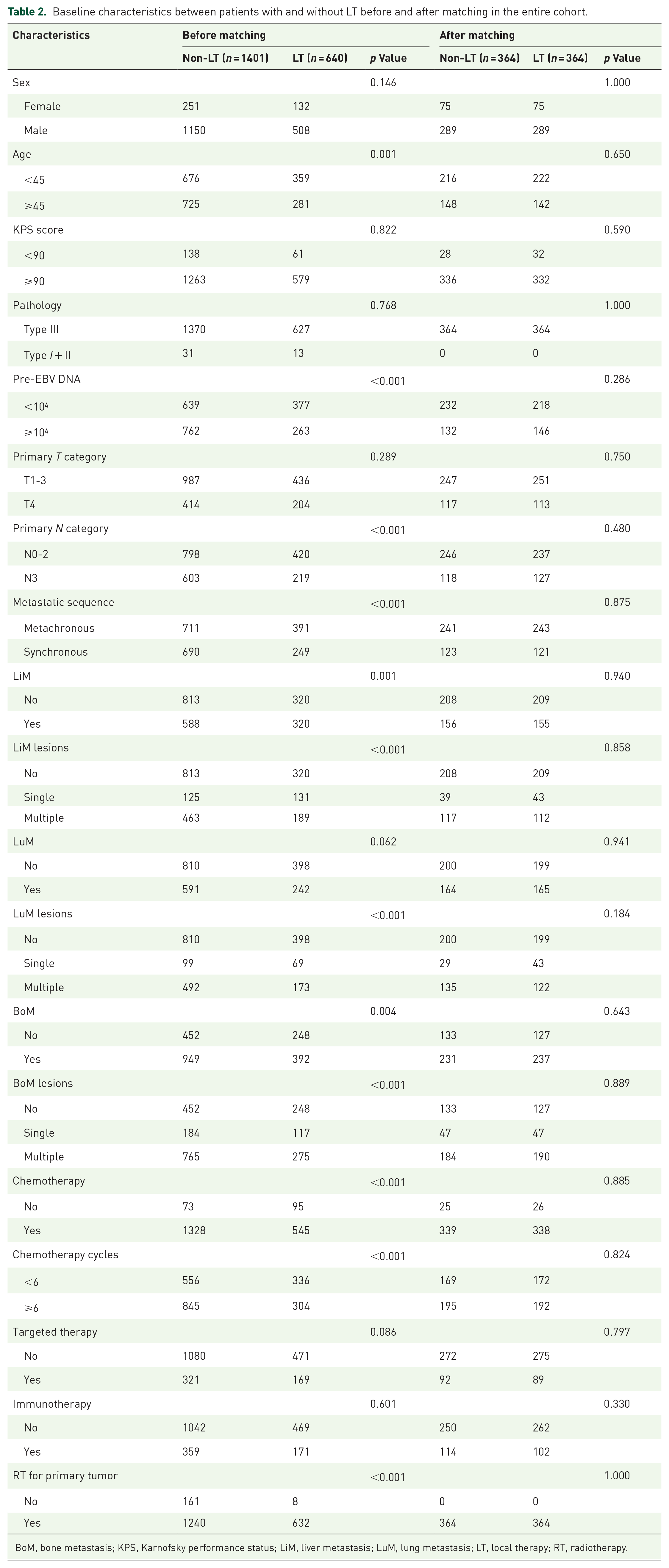
BoM, bone metastasis; KPS, Karnofsky performance status; LiM, liver metastasis; LuM, lung metastasis; LT, local therapy; RT, radiotherapy.
Prognostic factors in the unselected population by univariate and multivariate analyses.
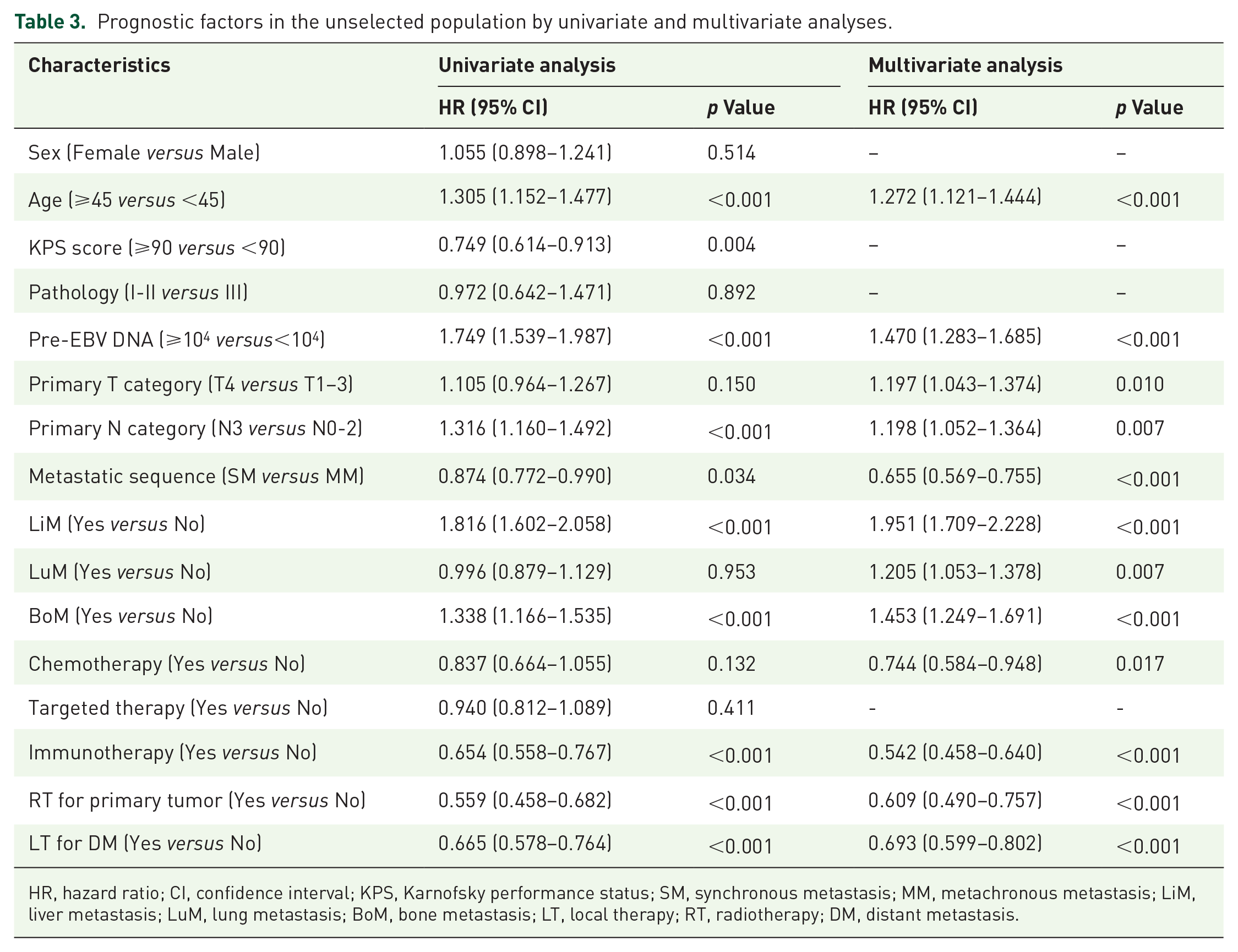
HR, hazard ratio; CI, confidence interval; KPS, Karnofsky performance status; SM, synchronous metastasis; MM, metachronous metastasis; LiM, liver metastasis; LuM, lung metastasis; BoM, bone metastasis; LT, local therapy; RT, radiotherapy; DM, distant metastasis.

Overall survival rates of mNPC patients who received either LT or did not receive LT for metastatic sites before (a) and after (b) matching. LT: local therapy.
Association between LT and OS by subgroup analyses
Subgroup comparisons for OS of mNPC patients with or without LT demonstrated that LT was associated with improved OS in nearly all subgroups, except for the subgroup of patients without chemotherapy (HR = 0.646, p = 0.108). Significant interactions were detected for metastatic sequence and LT, which indicates that patients with mmNPC may benefit more from LT than those with smNPC (HR: 0.639 versus 0.777, p = 0.045) (Figure 3).

Forest plot of the association between LT and overall survival by subgroup based on multivariate analyses.
The efficacy of LT for patients with liver-only, lung-only, and bone-only metastasis
As LT efficacy was confirmed in the unselected patients with mNPC, we further conducted subcohort analysis in patient with liver-only (n = 253), lung-only (n = 340), and bone-only (n = 636) metastasis to evaluate the survival benefit of LT. Baseline characteristics between patients with or without LT before and after matching were listed in Supplemental Tables 1–3. Based on matched-pair analyses, LT to LiM or LuM was shown to improve survival of patients with liver-only and lung-only metastasis (p = 0.009 and p = 0.007, respectively; Figure 4(a) and (b)) and were confirmed by multivariate analyses in the unmatched cohorts (Figure 4(d)). However, LT to BoM failed to benefit patients with bone-only metastasis (p = 0.614; Figure 4(c) and (d)).

Comparison of overall survival (OS) in mNPC patients in the local therapy (LT) for liver-only (a), lung-only (b), or bone-only (c) metastasis and non-LT groups after matching. (d) Adjusted hazard ratios (HRs) for OS stratified by LT in subcohorts of liver-only, lung-only, and bone-only metastasis. The group with non-LT was the reference group (HR = 1). The adjusted variables were age, pre-EBV DNA, metastatic sequence, LiM lesions, chemotherapy, targeted therapy, immunotherapy, and RT for primary tumor for liver-only metastasis subcohort. The adjusted variables were age, KPS score, pre-EBV DNA, LuM lesions, chemotherapy, immunotherapy, and RT for primary tumor for lung-only metastasis subcohort. The adjusted variables were age, KPS score, pre-EBV DNA, primary N category, metastatic sequence, BoM lesions, and RT for primary tumor for bone-only metastasis subcohort.
Treatment outcomes of different radiation dose prescriptions and no RT in patient with BoM
Since RT was the primarily used LT to BoM, we further compared the survival outcomes brought by different radiation doses among patients with BoM. Dose prescriptions were converted to BEDs, and a BED of 60 Gy was selected as the best cutoff value for separating patient outcomes (Figure 5(a)). In the subcohort of bone-only metastasis, 150 patients who have detailed RT record and 451 patients who did not receive RT for BoM were enrolled for analysis. Pairwise comparisons demonstrated that the OS rate was significantly higher in high BED (>60 Gy) group than those without RT (p = 0.033), whereas there was no survival benefit observed in low BED (⩽60 Gy) group when compared to non-RT group (p = 0.893) (Figure 5(b), Supplemental Table 4). We further conducted matched-pair analyses between different BED groups and non-RT group to confirm the above results. After matching (Supplemental Table 5), high BED group (>60 Gy), but not low BED group (⩽60 Gy) was beneficial as LT to BoM for survival when compared to patients without RT (p = 0.004 and p = 0.716, respectively; Figure 5(c) and (d)).
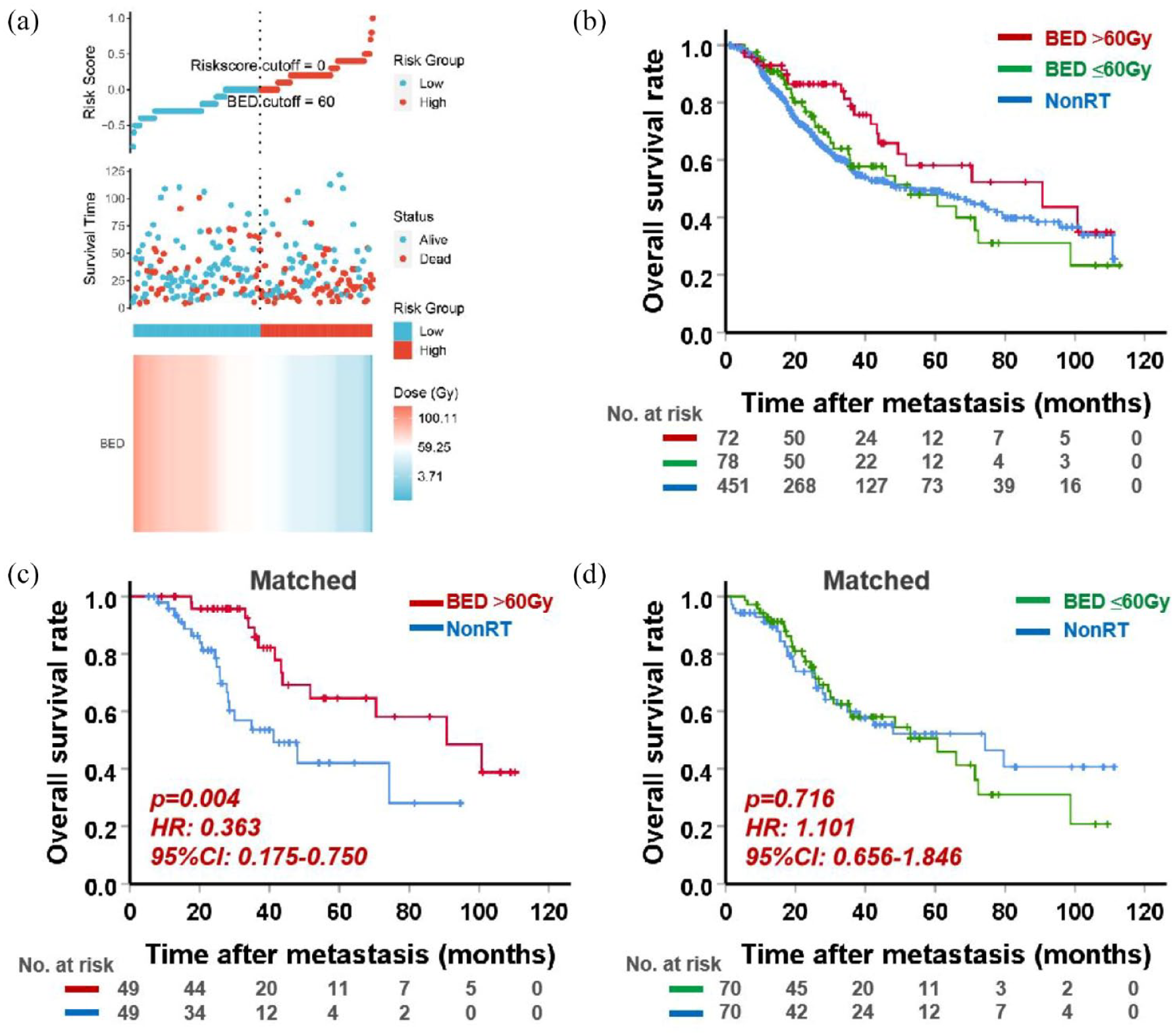
Comparison of survival outcomes brought by different radiation doses and non-RT. (a) The best cutoff value of BED was selected. (b) Survival comparison of patients who received BED > 60 Gy, BED ⩽ 60 Gy or non-RT for bone metastasis. (c and d) Survival outcomes were compared in BED > 60Gy and BED ⩽ 60 Gy groups with non-RT group after matching.
Discussion
To the best of our knowledge, this retrospective study evaluating the efficacy of LT for LiM/LuM/ BoM in mNPC patients had the largest sample size to date. Our study substantiated several noteworthy findings. First, LT resulted in a significant OS advantage for mNPC patients based on strict matched-pair analysis. Second, subgroup analyses revealed that LT remained a significant favorable prognosticator in nearly all subgroups. Third, in subcohort analyses, the addition of LT could benefit patients with liver and/or lung metastases but not with BoM. Last, RT with high BED (>60 Gy) for BoM was found to yield more survival benefit for patients with BoM than that of low BED (⩽60 Gy).
The optimal strategy for mNPC is to eradicate all detectable and invisible tumors through the combination of systemic and local treatment. Systemic therapies are effective in controlling microscopic diseases, but their impact on macroscopic diseases may be limited. In contrast, local therapies may provide ideal local control for macroscopic lesions. Curative LT to metastatic foci has been reported to improve survival among patients with lung, liver, breast, and prostate cancer.35,36 For NPC, success of LT has been confirmed in patients with limited metastases. 37 In the present study, we demonstrated that LT was associated with better survival of mNPC patients, owing to its potential effect on reducing tumor burden and preventing further dissemination of metastases. Subgroup analysis demonstrated that LT did not benefit patients without chemotherapy, which indicates that LT may not confer survival benefit when the microscopic metastases are not eliminated simultaneously. Additionally, our data showed that patients with mmNPC benefited more from LT than those with smNPC. One potential explanation is that for mmNPC, LT of metastatic sites is mostly implemented after the treatment of primary tumors (⩾6 months). The patient’s physical condition has recovered and has good tolerance to LT. As for smNPC (metastasis at initial diagnosis), patient may receive chemotherapy and LT for primary tumors first. On this basis, adding LT to metastatic sites may increase the risk of severe treatment toxicities, thus to reduce survival benefit. Currently, there is no relevant literature report, which is worthy of further exploration and verification.
LiM are commonly regarded as an incurable disease and portend an inferior prognosis compared to bone or LuM, and the treatment has largely been palliative.38,39 With the advancements in imaging methods, systemic treatment and local therapies (including surgery, RT, and locally ablative techniques) over the past decades, evidence has been provided for a more aggressive treatment for LiM. Successful treatment experience has been achieved with hepatectomy in colorectal cancer with LiM, and previous studies on surgical resection for patients with noncolorectal hepatic metastases have shown encouraging results with median postoperative follow-up or OS periods of 23.5–49 months.40–42 For NPC, literature regarding surgical treatment of LiM is rare. Delis et al. 43 reported a patient with a solitary LiM from NPC who underwent segmentectomy and no recurrence was noted during the 6-month follow-up after resection. Another case study reported two patients with metachronous LiM from NPC who underwent metastasectomy had a mean postoperative survival period of 58.0 months. 44 Hung raises the point that LiMs present an invasive outgrowth pattern due to the deficiency of hepatic capsule and blood supply. 41 Thus, metastasectomy has a role in curbing the further spread of tumor.
Apart from surgical management, nonsurgical LT options, such as RFA, RT, and TACE, are available for LiM. CT-guided RFA can be administered with high technical efficiency and may prolong survival in selected NPC patients.28,45,46 RFA for treating NPC patients with 1–3 LiMs prolongs survival compared to those who only receive chemotherapy (median OS: 48.1 versus 25.9 months). 47 Peng et al. 48 reported the effectiveness of TACE for NPC patients with LiM, and they suggested that it can be optional for suitable patients because it may control intrahepatic lesions and prolong survival. The advantage of RT, especially stereotactic body radiation therapy, is that its effectiveness is not impaired by intravascular blood flow or bile accumulation in the gallbladder. Furthermore, external irradiation can reach any tumor locations of liver, such as the subphrenic area. NPC patients with LuM are historically regarded as a distinct group with favorable prognosis.39,49 In a case–control study that matched 55 and 22 patients who underwent surgery and nonsurgery for LuM, patients who underwent pulmonary surgery had a better postmetastatic survival (PMS) than those without surgery (5-year PMS: 75.53 versus 47.81%, p = 0.005). 50 A study by Ma et al. 51 included 105 patients with solitary LuM from NPC who received surgery ± chemotherapy, RT ± chemotherapy or chemotherapy alone, surgery ± chemotherapy, and RT ± chemotherapy showed better OS than chemotherapy alone (82.4 versus 49.6 versus 29 months, p < 0.001). Surgical resection has also been shown to be superior to chemotherapy for LuM from NPC in selected series. Cheng et al. 52 reported a 2-year OS of 80% in 12 patients who underwent surgical resection and only 24.1% in 65 patients who underwent only chemotherapy.
For unresectable LuM, RFA and RT are two alternative therapies for patients with well-controlled primary disease. In a study by Pan et al., 28 there were no significant differences between the survival probability of patients with RFA (n = 10) and surgery for LuM (n = 27). Therefore, patients who have unresectable tumors, have contraindications to surgery or are unwilling to receive surgery could be recommended to receive RFA treatment. Regarding RT for LuM, Wu et al. 53 reported that stereotactic RT is an effective and safe treatment for LuM and that BED ⩾ 75 Gy has a better local control on LuM.
Although improved survival was detected in patients who received LT for LiM and/or LuM, this survival benefit was not observed in patients with BoM. In a study by Shen et al., 29 local RT combined with chemotherapy was associated with longer survival of NPC patients with solitary BoM than those who received chemotherapy alone (5-year OS: 57.3 versus 11.2%). Nevertheless, this impact was not significant for patients with multiple bone metastases. 29 Although RT is the primary treatment method for BoM, consensus regarding the optimal dose prescription is lacking. Most patients were reported to receive 30–60 Gy doses for BoM.54–58 A previous study has noted that aggressive RT (Equivalent Dose in 2 Gy/fractions, EQD2 ⩾ 60 Gy) does not present survival benefits for patients with synchronous BoM based on systemic chemotherapy and radical RT for primary tumor. 31 Conversely, a study showed that patients who received intensive RT (60–75 Gy/30–35 fractions) to bone metastatic sites had improved survival; the study concluded that for patients with a long-term predicted life expectancy and treatment goals, radical RT to BoM should be considered. 59 Fukushima et al. 60 reported that intensive LT is beneficial not only for patients with solitary BoM but also for those with multiple bone metastases. Our results demonstrated that patients who received BED > 60 Gy, but not BED ⩽ 60 Gy, resulted in better survival outcomes than those who did not receive RT to BoM. In our subcohort, more than half of patients received BED ⩽ 60 Gy, which may be a potential explanation for the negative results obtained by LT for BoM.
Collectively, we demonstrated that LT could benefit patients with mNPC, especially for liver and/or LuM. Aggressive RT may be beneficial in the management of BoM. Further prospective randomized trials are warranted to confirm the results. What remain to be determined is the optimal candidates for LT to metastatic foci and which treatment paradigm is the most suitable and effective for specific metastasis.
Limitations
First, the patient selection may be biased due to the retrospective nature of this study. Nevertheless, matched-pair analysis was performed based on a large-scale population. Second, we failed to include more potential prognostic factors, such as metastatic lesion site/size, posttreatment EBV-DNA, and others, due to the lack of medical records or the difficulty of unifying data collection standards. Third, this was a single-center study derived from a high prevalence area.
Supplemental Material
sj-docx-1-tam-10.1177_17588359221112486 – Supplemental material for Efficacy of local therapy to metastatic foci in nasopharyngeal carcinoma: large-cohort strictly-matched retrospective study
Supplemental material, sj-docx-1-tam-10.1177_17588359221112486 for Efficacy of local therapy to metastatic foci in nasopharyngeal carcinoma: large-cohort strictly-matched retrospective study by Meng-Xia Zhang, Ting Liu, Rui You, Xiong Zou, Yong-Long Liu, Xi Ding, Chong-Yang Duan, Han-Shi Xu, You-Ping Liu, Rou Jiang, Zhi-Qiang Wang, Chao Lin, Yu-Long Xie, Si-Yuan Chen, Yan-Feng Ouyang, Ruo-Qi Xie, Yi-Jun Hua, Rui Sun, Pei-Yu Huang, Shun-Lan Wang and Ming-Yuan Chen in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
None.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
