Abstract
Background:
The poor prognosis of locoregionally advanced nasopharyngeal carcinoma (LANPC) due to the high incidence of metastasis necessitates effective treatment strategies. Synergistic effects have been observed when anti-programmed death-1 (PD-1) inhibitors are combined with chemotherapy or targeted therapy.
Objectives:
To compare the efficacy and safety of induction chemotherapy in combination with nimotuzumab with or without anti-PD-1 inhibitors for LANPC.
Design:
Retrospective study.
Methods:
In total, 319 patients with LANPC were retrospectively enrolled between December 2017 and November 2022. The primary endpoint was progression-free survival (PFS). Propensity score matching was performed to adjust for potential confounders.
Results:
Overall, 150 patients were included after propensity score matching. The immunotherapy + nimotuzumab + chemotherapy (INC) group (n = 50) had a higher 3-year PFS rate (96.6% (95% confidence interval (CI): 93.2–100.0)) than the nimotuzumab + chemotherapy (NC) group (n = 100) (79.8% (95% CI: 75.6–84.0)). The INC group had a hazard ratio of 0.16 (95% CI: 0.02–1.22; p = 0.04). The objective response rates were 100% and 99% for the INC and NC groups, respectively. Grade ⩾3 treatment-related adverse events were reported in eight (5.3%) patients, and hyponatremia (2.0%) was the most common. Grade ⩾3 immune-related adverse events (rash and reactive capillary proliferation) were reported in two (4.0%) patients.
Conclusion:
INC demonstrated remarkable anti-tumor activity with acceptable safety for LANPC.
Introduction
Nasopharyngeal carcinoma (NPC), which is closely associated with the Epstein–Barr virus (EBV) and is highly prevalent in Southeast Asia, is one of the most common head and neck cancers. The incidence of locoregionally advanced NPC (LANPC) in areas where NPC is endemic is approximately 60%–80%. LANPC has a poor prognosis and a high risk of metastasis. 1 The standard treatment for stage III–IVA LANPC is induction chemotherapy (IC), followed by platinum-based concurrent chemoradiotherapy (CCRT).2,3
NPC is characterized by high programmed death-ligand 1 (PD-L1) expression (90%–95% of tumor cells) and abundant non-malignant lymphocyte infiltration (~50% of samples with >70% stromal tumor-infiltrating lymphocytes (TILs) or >10% intratumoral TILs). These make patients amenable to immune checkpoint blockade (ICB).1,4 Several anti-programmed death-1 (PD-1) inhibitors have been approved by the US Food and Drug Administration for first- or later-line treatment of recurrent or metastatic NPC. Immune checkpoint inhibitors are not currently recommended in the LANPC guidelines, but ongoing phase II/III clinical trials (NCT05587374/NCT04907370/NCT05707819/NCT04769076/NCT03984357/NCT03925090/NCT04557020/NCT04447326/NCT04453826/NCT04557020/NCT03427827/NCT04870905) are exploring the timing of PD-1/PD-L1 monoclonal antibody administration and radical chemoradiotherapy using various approaches (induction, concurrent, and adjuvant therapy) for durations exceeding 9–12 months. The CONTINUUM study (NCT03700476), which was presented at the American Society of Clinical Oncology (ASCO) in 2023, showed excellent efficacy with acceptable safety of sintilimab for stage III–IVA NPC.
Increasing evidence suggests that the efficacy of ICB depends on a robust immune response, which is usually compromised in tumors5,6 Chemotherapeutic and targeted agents enhance the immune response by increasing tumor immunogenicity, enhancing CD8+ T-cell infiltration, and inhibiting immunosuppressive cells in the tumor microenvironment (TME).5,6 This immunomodulatory effect provides a rationale for combining chemotherapy, targeted therapy, and ICB. Several preclinical and clinical studies have shown encouraging results.7,8 However, the optimal drug combination, dosage, timing, and administration have not yet been determined for LANPC. Therefore, we aimed to evaluate the anti-tumor activity and safety of anti-PD-1 inhibitors and nimotuzumab combined with IC and CCRT in patients with LANPC.
Materials and methods
Ethics
All procedures were performed in compliance with the relevant laws and institutional guidelines, and the study protocol was approved by the Clinical Research Committee of Sun Yat-sen University Cancer Center (approval number: B2023-705-01; approval date: 29 December 2023). The work described has been carried out in accordance with the Code of Ethics of the World Medical Association (Declaration of Helsinki) for experiments involving humans. The requirement for written informed consent was waived due to the retrospective nature of the study. The reporting of this study conforms to the STROBE-cohort statement (Supplemental Material 1). 9
Study design and participants
Between December 2017 and November 2022, 319 patients with LANPC who received anti-PD-1 inhibitors in combination with nimotuzumab or nimotuzumab alone during IC at our institution were retrospectively enrolled (Figure 1). The inclusion criteria were as follows: (1) newly diagnosed stage III–IVA NPC (Union for International Cancer Control/American Joint Committee on Cancer (eighth edition)); (2) IC and CCRT; (3) two to four cycles of IC; (4) nasopharyngeal and neck magnetic resonance imaging (MRI) before and after IC; and (5) no additional adjuvant chemotherapy. The exclusion criteria were as follows: (1) history of malignancy; (2) immunotherapy (e.g., anti-PD-L1/cytotoxic T-lymphocyte-associated antigen-4 inhibitors); (3) pregnancy or lactation; and (4) significant comorbidities.
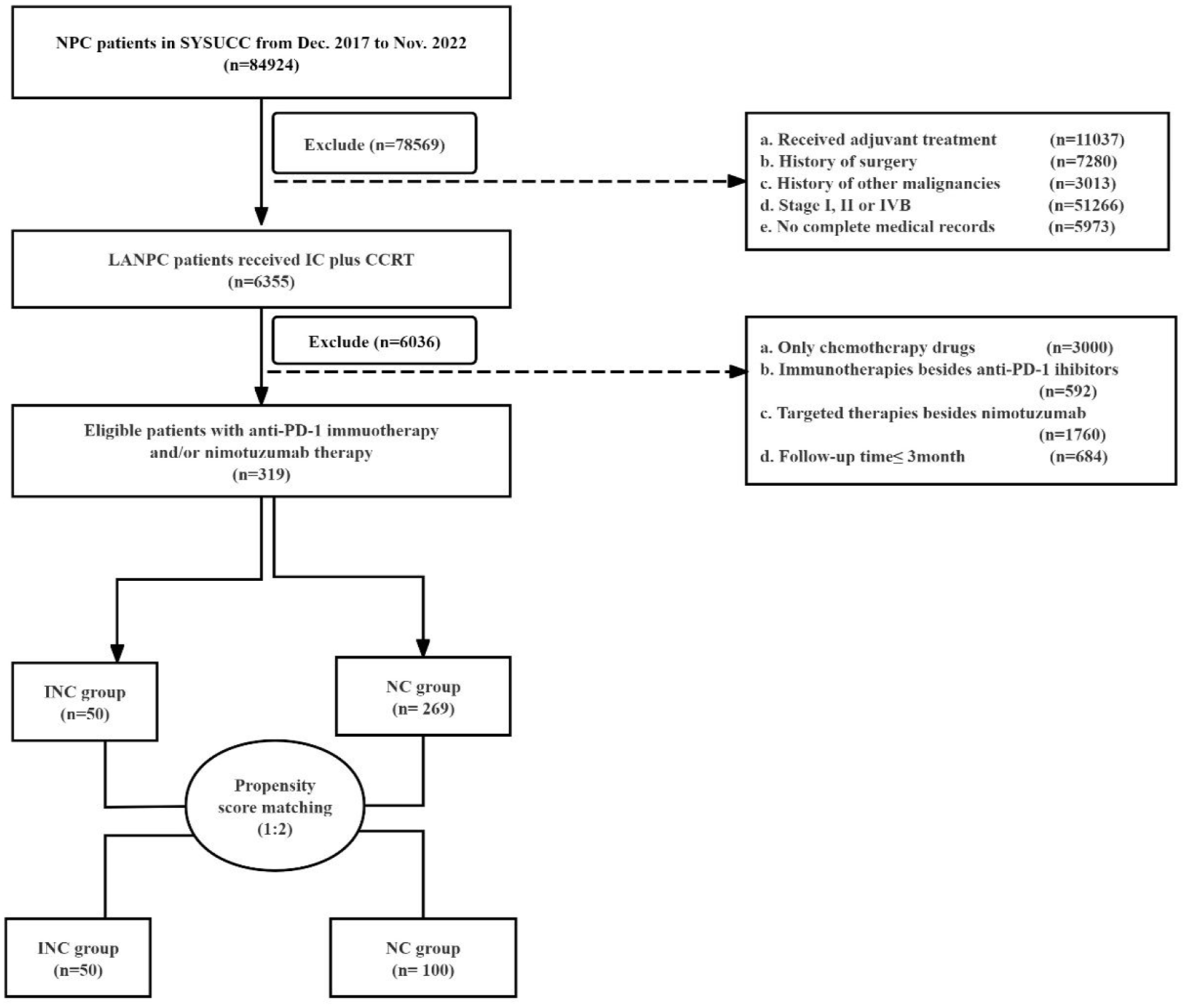
Flowchart for patient selection.
Patients were divided into two groups based on their IC regimen: immunotherapy + nimotuzumab + chemotherapy (INC) and nimotuzumab + chemotherapy (NC).
Plasma EBV–DNA
Plasma EBV–DNA was determined by real-time quantitative PCR (before treatment, after IC, and 3 months after CCRT). The cutoff values of 4000 and 0 copies/mL were used for pre- and post-treatment EBV–DNA, respectively.10,11
Treatment evaluation
Patients underwent IC, followed by CCRT. The IC regimens included TP or TPF, where T = taxane (docetaxel (75 mg/m2) or nab-paclitaxel (260 mg/m2); day 1), P = cisplatin (80–100 mg/m2; day 1), and F = fluorouracil (300–500 mg/m2; continuous intravenous infusion for 3–5 days). Targeted therapy included the anti-epidermal growth factor receptor (EGFR) monoclonal antibody, nimotuzumab (400 mg (first time), 200 mg (thereafter); day 1), and anti-PD-1 inhibitors (camrelizumab (200 mg; day 1), toripalimab (240 mg; day 1), sintilimab (200 mg; day 1) and tislelizumab (200 mg; day 1)). This was administered before chemotherapy. Each regimen consisted of two to four cycles at 3-week intervals. Concurrent cisplatin-based chemotherapy (80–100 mg/m2) on days 1 and 22 of radiotherapy was combined with targeted therapy (nimotuzumab (200 mg/week during radiotherapy)).
Patients received intensity-modulated radiotherapy (IMRT; 2.00–2.34 Gy) 5 times/week for 6–7 weeks. The dose delivered to the planning target volume was 68–70 Gy. Other details of the IMRT protocol were consistent with those in previous studies, with doses delivered within the tolerated dose range of the Radiation Therapy Oncology Group 0225 protocol.12,13
Patients underwent nasopharyngeal and neck MRI after IC and 3 months post-CCRT. Images were independently reviewed by two physicians according to the Response Evaluation Criteria in Solid Tumors (version 1.1). Responses were categorized as complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD).
Endpoints
The primary endpoint was progression-free survival (PFS), which was defined as the time from the first day of IC to the date of locoregional relapse, distant metastasis, or death from any cause, whichever occurred first. 14 The secondary endpoints were the objective response rate (ORR; proportion of patients who achieved CR or PR), disease control rate (proportion of patients who achieved CR, PR, or SD), overall survival (OS; time from the first day of IC to death), and safety. Treatment-related adverse events (TRAEs) were assessed using the National Cancer Institute Common Toxicity Criteria for Adverse Events (version 5.0), with dose adjustments based on toxicity. Patients were followed up at 3-monthly intervals for 2 years, 6-monthly intervals for 3–5 years, and annually thereafter until death. Routine evaluation included fiberoptic pharyngoscopy, nasopharyngeal and neck MRI, and EBV serology. Suspected recurrence and distant metastasis were investigated by biopsy or imaging.
Statistical analysis
To minimize assessment bias, propensity score matching (PSM) was adjusted for the N category, overall stage, IC regimen, and IC cycle. Using a logistic regression model, the propensity score was calculated with the IC regimen as the dependent variable. Patients were matched at 1:2 using caliper matching (caliper size, 0.5). The ORR (95% confidence interval (CI)) was calculated using the Clopper–Pearson method. Other clinical outcomes, demographic characteristics, and AEs were summarized using descriptive statistics (n (%) or median (interquartile range, IQR)). Survival curves were plotted using the Kaplan–Meier method and compared using the log-rank test. All statistical analyses were conducted using SPSS (version 25.0) and R (version 4.1.3). Two-sided p-values of <0.05 denoted statistical significance.
Results
The baseline characteristics are presented in Table 1. The median (IQR) age was 44 (12–73) years (male-to-female ratio: 2.7:1). The majority (91.8%) of patients had T3–4 tumors; 30.7% of patients had N3 lymph node involvement. Regarding the IC regimen, 40.8% of patients received TPF chemotherapy plus targeted therapy. Four cycles of IC were administered to 21% of patients.
Patient demographics and baseline tumor characteristics.

CAR, C-reactive protein-to-albumin ratio; EGFR, epidermal growth factor receptor; IC, induction chemotherapy; INC, immunotherapy + nimotuzumab + chemotherapy; MLR, monocyte-to-lymphocyte ratio; NC, nimotuzumab + chemotherapy; LR, neutrophil-to-lymphocyte ratio; NPC, nasopharyngeal carcinoma; PD-1, programmed death-1; PLR, platelet-to-lymphocyte ratio; SUV, standardized uptake value.
To eliminate potential confounders, a balanced cohort was created using PSM. Following 1:2 matching, 150 patients (50 vs 100) were included in the matched cohort: 90.0% had T3–4 tumors, 48.7% had N3 lymph node involvement, and 55.3% had stage IVA disease. For IC, the TPF-to-TP ratio in combination with targeted therapy was 3.29; 53.3% of matched patients received four cycles of IC. In the matched cohort, 86% of patients in the INC group (n = 50) had EGFR+/++ tumors, compared to 81% in the NC group (n = 100). Tislelizumab was the most frequently used anti-PD-1 monoclonal antibody (n = 24), followed by toripalimab (n = 13). Sintilimab was the least used (n = 3).
Short-term efficacy was assessed 3 months after CCRT using nasopharyngeal and neck MRI and plasma EBV–DNA (Tables 2 and 3). After IC, the CR and PR rates for the INC and NC groups were 18% and 80% and 1% and 89%, respectively. At the end of treatment 3 months later, the ORRs (95% CI) of the INC and NC groups were 100% (92.9–100.0) and 99% (94.6–100.0), respectively. The INC group significantly increased CR rates after IC, from 18% to 76%. Among patients with an unsatisfactory response to IC (11/150), one (0.7%) still had SD after CCRT. PD was not assessed. The median (IQR) pre-treatment EBV-DNA copy number was 2970 (0–46,500) copies/mL. The proportions of patients with detectable EBV–DNA after CCRT in the INC and NC groups were 2% (1/50) and 8% (8/100), respectively.
Short-term efficacy.

CI, confidence interval; CR, complete response; DCR, disease control rate; INC, immunotherapy + nimotuzumab + chemotherapy; NC, nimotuzumab + chemotherapy; ORR, objective response rate; PD, progressive disease; PR, partial response; SD, stable disease.
Comparison of EBV-DNA levels between the two groups during treatment.
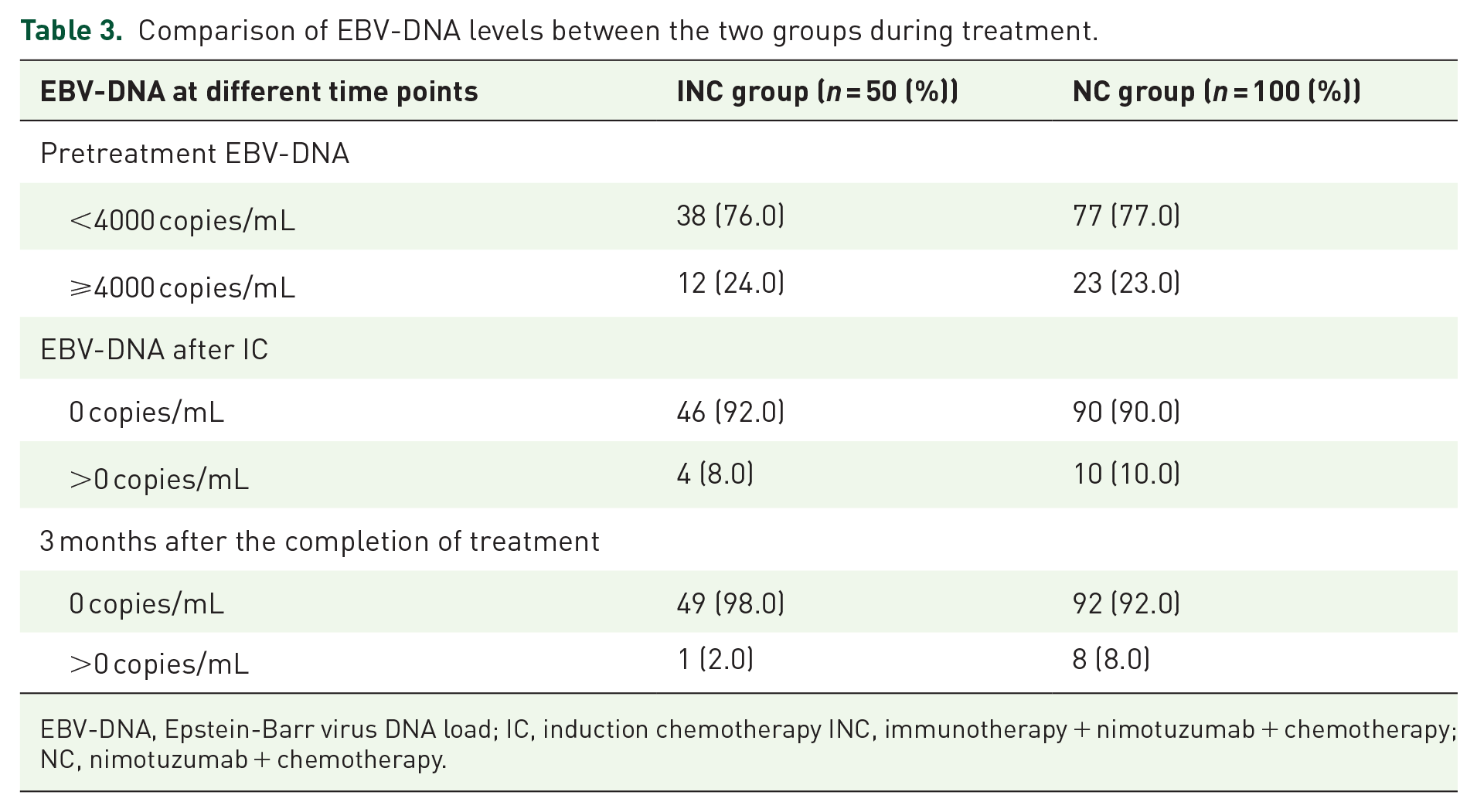
EBV-DNA, Epstein-Barr virus DNA load; IC, induction chemotherapy INC, immunotherapy + nimotuzumab + chemotherapy; NC, nimotuzumab + chemotherapy.
The median (IQR) follow-up was 31 (12–42) months. Ten (6.7%) patients had locoregional recurrence, six (4.0%) had distant metastases, and eight (5.3%) died. The 3-year PFS rate was markedly higher in the INC group compared to the NC group (96.6% (95% CI: 93.2–100.0) vs 79.8% (95% CI: 75.6–84.0), respectively). The hazard ratio (HR; 95% CI) for recurrence, distant metastasis, or death was 0.16 (0.02–1.22; p = 0.043; Figure 2(a)). There was no significant difference between the 3-year OS of the INC and NC groups (93.5% (95% CI: 90.9–96.1) vs 96.6% (95% CI: 93.2–100.0), respectively); HR (95% CI) for death: 0.55 (0.06–4.56; p = 0.57; Figure 2(b)).
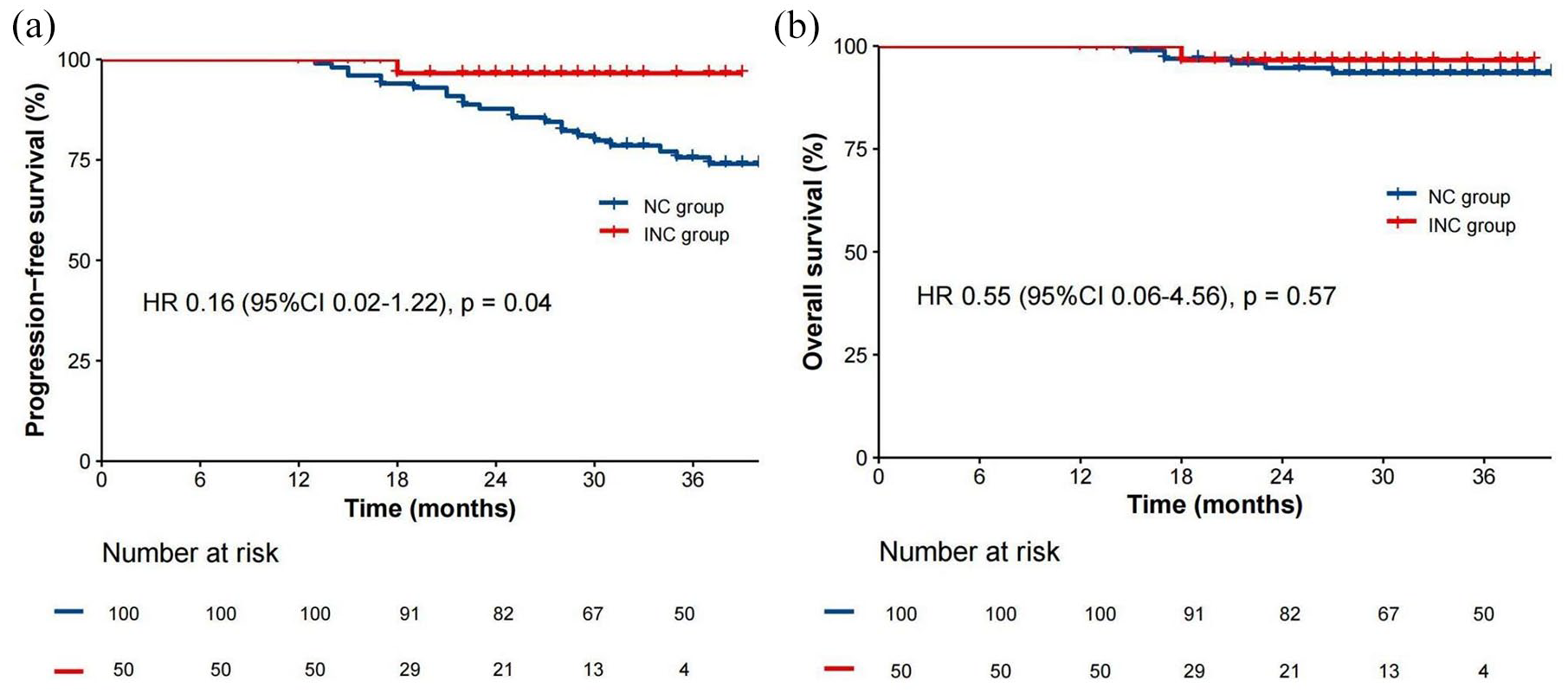
Kaplan–Meier survival curves after propensity score matching: (a) PFS and (b) OS.
Grade 3/4 TRAEs occurred in eight (5.3%) patients in the NC group but none was reported in the INC group (Table 4). No Grade 5 AEs or treatment-related deaths occurred in either group. TRAEs of different grades occurred in all 150 patients at an incidence of >50%: anorexia (n = 144; 96%), hyponatremia (n = 124; 82.7%), hypoalbuminemia (n = 102; 68%), leukopenia (n = 99; 66%), anemia (n = 92; 61.3%), and nausea (n = 79; 52.7%).
Comparison of Grade 1–4 acute toxicities between the two groups during induction chemotherapy.
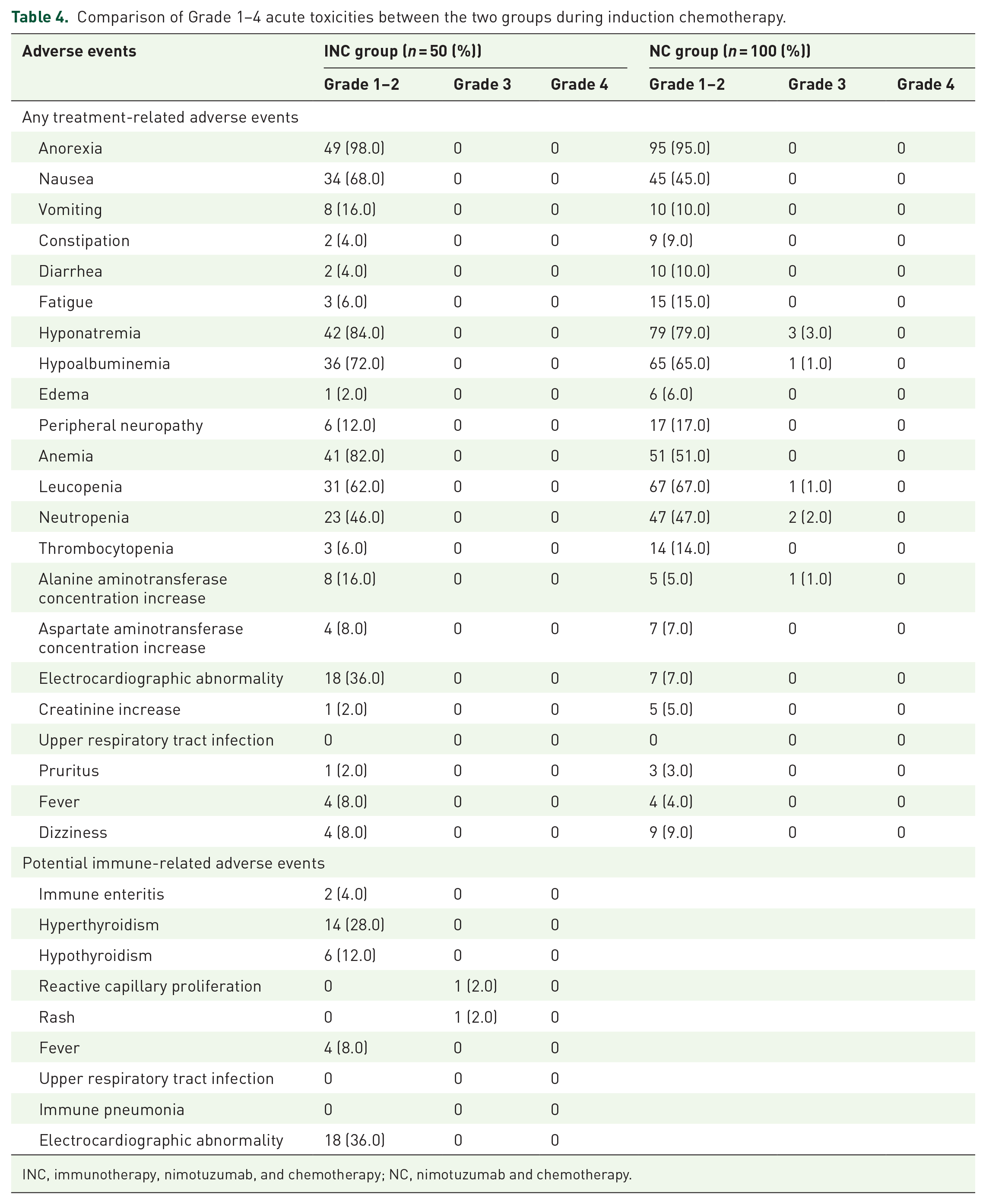
INC, immunotherapy, nimotuzumab, and chemotherapy; NC, nimotuzumab and chemotherapy.
There were no Grade ⩾4 potential immune-related AEs (irAEs). The most common irAEs included thyroid dysfunction (hyperthyroidism (n = 14) and hypothyroidism (n = 6)). Grade 3 reactive cutaneous capillary endothelial cell proliferation (RCCEP) and immune-related rash were observed in two patients. Chest CT and cardiac enzymes revealed no immune-related pneumonia or myocarditis.
Discussion
This is the first study to evaluate the anti-tumor activity and safety of immunotherapy combined with anti-EGFR therapy in LANPC. Our results demonstrate promising anti-tumor activity and acceptable toxicity profiles in combination with anti-PD-1 inhibitors and nimotuzumab, providing preliminary evidence to guide the design and implementation of future immunotherapy trials in LANPC.
IC followed by CCRT is recommended in the ASCO and Chinese Society of Clinical Oncology guidelines as first-line treatment for stage III–IVA NPC. IC improves outcomes by eliminating distant metastases and has played an important role in the IMRT era. Two phase III randomized controlled trials evaluated the TPF (docetaxel (60 mg/m2), cisplatin (60 mg/m2), and 5-FU (600 mg/m2); daily intravenous infusion for 120 h once every 3 weeks for 3 cycles) and GP (gemcitabine (1000 mg/m2 on days 1 and 8) and cisplatin (80 mg/m2) once every 3 weeks for 3 cycles) regimens in patients with LANPC. In both trials, the IC group had a better 5-year OS rate.3,15,16 However, not all patients respond well to IC. Approximately 30% of patients had a suboptimal response (SD or PD), leading to treatment failure and a poor prognosis. 17 EBV-associated NPC is a prime example of an “immune-hot” tumor, characterized by a stroma heavily infiltrated by immune cells. 18 Research through whole-exome and single-cell sequencing has revealed a suppressive TME filled with dysfunctional CD8+ T cells and effector T cells that overexpress inhibitory immune checkpoints such as PD-L1, LAG3, galectin 9-TIM3, TIGIT, and CTLA4. In addition, the presence of regulatory T cells, M2 macrophages, and myeloid-derived suppressor cells, along with various chemokines and cytokines, exacerbate this immunosuppressive environment.18–24 Recognizing these immunological and genomic properties of NPC not only highlights the complexity of the disease but also highlights the potential for effective immunotherapy. Implementing immunotherapies that enhance tumor immunogenicity, boost CD8+ T-cell infiltration, and reduce immunosuppressive cells are essential to address the challenges of the immune-compromised microenvironment of LANPC. Large numbers of lymphocytes infiltrate the tumor tissue, suggesting that NPC may benefit from anti-PD-1/PD-L1 immunotherapy.25–27 In recurrent or metastatic NPC, three Chinese phase III trials (CAPTAIN-1st (NCT03707509), 28 JUPITER-02 (NCT03581786) 29 , and RATIONALE 309 (NCT03924986) 30 ) reported that anti-PD-1 antibodies in combination with GP improved efficacy. The ORR ranged from 69.5% to 87.3%; the median PFS ranged from 9.2 to 11.4 months. However, data on anti-PD-1 immunotherapy in LANPC are lacking. The CONTINUUM trial (NCT03700476), reported at ASCO 2023, is a multicenter, randomized, controlled phase III trial of 425 patients with LANPC, in which the study group received immunotherapy in combination with sintilimab (induction, concurrent, and adjuvant phases). Event-free survival (EFS) increased from 76% to 86% in the study group relative to the control group. The incidence of grade 3/4 irAEs was <10%. The results are encouraging and further demonstrate the clinical utility of immunotherapy in patients with LANPC.
Evidence suggests that EGFR is highly expressed in ~85% of patients with NPC and is associated with a more aggressive phenotype, greater drug resistance, and a poorer prognosis.31–33 Meta-analyses show that EGFR over-expression significantly predicts poorer OS and disease-free survival (DFS).34–36 In 2018, a retrospective analysis of 596 patients reported that anti-EGFR therapy in combination with IC has promising anti-tumor activity and a favorable safety profile for LANPC. 37 A multicenter randomized controlled trial showed that nimotuzumab plus cisplatin-5-fluorouracil (PF) achieved a higher cervical lymph node response rate than docetaxel plus PF (81% vs 60%, respectively; p = 0.036). 38 The combination of nimotuzumab and IC in LANPC shows encouraging efficacy and is well tolerated.
Conventional chemotherapy (paclitaxel and cisplatin)39,40 and targeted therapies (anti-EGFR monoclonal antibodies) 41 have immunomodulatory effects on the TME. They promote anti-tumor immune responses by enhancing tumor cell immunogenicity, increasing CD8+ T-cell infiltration, and suppressing immunosuppressive cells. 5 Encouraging results have been obtained in preclinical and clinical studies for several solid tumors5,42,43 The Impower150 (NCT02366143) trial showed that combining atezolizumab with bevacizumab and chemotherapy as first-line treatment for metastatic non-squamous non-small-cell lung cancer improved PFS and OS. 44 The median PFS was longer for the combined group relative to the control group (8.3 vs 6.8 months, respectively). 44 Camrelizumab plus famitinib and chemotherapy were evaluated in 48 patients with triple-negative breast cancer (NCT04129996). 45 The objective remission rate was 81.3% (95% CI: 0.70–0.92) with a median PFS of 13.6 months. When toripalimab and apatinib were combined with gemcitabine as first-line therapy for advanced NPC, the median PFS was >25.8 months, and the ORR was 90.2%. 46 Triple therapy shows promising anti-tumor activity with manageable toxicity. However, whether anti-PD-1 immunotherapy, targeted therapy, and chemotherapy are safer and more effective than chemotherapy alone as IC for LANPC has not been established. In the field of head and neck squamous cell cancer (HNSCC), pivotal trials such as JAVELIN Head and Neck 100 and KEYNOTE-412 demonstrated that combining immunotherapy with CCRT did not yield significant EFS benefits in patients with locally advanced disease.47,48 The JAVELIN Head and Neck 100 trial reported an HR for PFS of 1.21 (95% CI: 0.93–1.57), favoring the placebo group consistently across subgroups. 47 In KEYNOTE-412, the median EFS in the pembrolizumab group was not reached, and the placebo group achieved an EFS of 46.6 months (HR: 0.83; 95% CI: 0.68–1.03; p = 0.043), superiority threshold p ⩽ 0.024). 48 In addition, the inclusion of both p16-positive and p16-negative patients likely affected the outcomes, with the majority having HPV-negative disease (66% vs 34% and 74% vs 26%).47,48 The INC group in this study did not significantly enhance 3-year OS compared to the NC group, reinforcing the conclusions drawn from the two prior trials. This concordance suggests that the optimal drug combination, dosage, timing, and administration are yet to be determined.
In this study, the CR rates of the INC and NC groups after IC and 3 months after treatment were 18.0% and 1.0% and 76.0% and 19.0%, with ORRs of 98% and 90% and 100% and 99%, respectively. A phase II trial of 50 patients with TanyN3M0 NPC, reported at ASCO 2022, demonstrated the clinical activity of camrelizumab and apatinib in combination with IC against LANPC. The ORR was 97.9%, the 1-year distant metastasis-free survival rate was 98%, and 3–5 AEs were reported with a higher incidence of 64%. The IMplus trial, evaluating camrelizumab and apatinib in combination with IC for locally advanced HNSCC, reported an ORR of 83%, with no Grade 4/5 AEs in phase I. A follow-up trial is ongoing. The ORR in the current study aligns more closely with NPC patterns; this may be attributed to the unique presentation of NPC relative to other HNSCCs. This underscores the promising anti-tumor activity and acceptable safety of anti-PD-1 immunotherapy combined with targeted chemotherapy and establishes it as an effective systemic treatment. The optimal combined modality of immunotherapy, targeted therapy, and chemotherapy for LANPC was further explored at ASCO 2023. The CONTINUUM (NCT03700476) and NEOSPACE (NCT03734809) trials, which combined anti-PD-1 antibodies (sintilimab and pembrolizumab) with standard treatment in induction, concurrent, and adjuvant settings, deserve mention. The 3-year EFS rate for the CONTINUUM trial was 86.1% and 76.0% compared to the standard group (p = 0.019). Five phase II trials have shown that anti-PD-1 antibody combination therapy with platinum-based chemotherapy for locally advanced HNSCC is effective and safe with ORRs between 54.5% and 92% (toripalimab plus nab-paclitaxel and cisplatin). They have also reported safety profiles comparable to that reported by the CONTINUUM trial with <10% Grade 3/4 irAEs.
We examined the toxicity profiles of different combination regimens. Grade ⩾3 TRAEs were infrequent (5.3%), with no Grade 5 events or treatment-related deaths. Notably, no grade 3–4 diarrhea was reported in either group. However, the NC group had a higher incidence of grade 1–2 diarrhea, with 6 in 10 patients receiving TPF chemotherapy. This contrasts with the approximately 15% rate of grade 3–4 diarrhea seen in previous studies. 49 In addition, proper monitoring and documentation of diarrhea symptoms were hampered by the early discharge of patients. irAEs were mild and manageable, including thyroid dysfunction and skin rash. The overall incidence of irAEs was 92% (46/50; all Grade 1/2), with the highest incidence being for thyroid dysfunction (n = 20; 40%). The incidence of thyroid dysfunction with anti-PD-1 monotherapy has previously been reported to be approximately 10%, which is lower than that in the current study. 50 One of the two patients who experienced immune skin reactions and was treated with camrelizumab developed Grade 3 RCCEP 2 weeks after the first cycle of IC, which resolved spontaneously. The incidence of RCCEP with camrelizumab monotherapy (66.8%) was reduced to approximately 30% with combination chemotherapy or apatinib.51–53 In another patient, a Grade 3 rash developed after toripalizumab treatment, which improved upon discontinuation of anti-PD-1 immunotherapy and administration of symptomatic hormone therapy. The combined results of 10 clinical trials of anti-PD-1 monotherapy with IC for locally advanced HNSCC reported at ASCO over the past 2 years showed that the incidence of Grade 3–5 AEs ranged from 0% (toripalizumab + TP) to 74.2% (sintilizumab + GP). The incidence of irAEs was within 2.9%–20.0%. The discrepant results of the current study may be attributed to varying sample sizes (10–425) and the use of different anti-PD-1 antibodies. Second, the Memorial Sloan Kettering Cancer Center (NCT04722523) trial and the present study share a similar design, with the exception that the trial used ciplizumab and cetuximab in combination with TP. Two of the 10 patients who received IC for HNSCC developed Grade 3 immune-related transaminase elevation and Grade 4 myocarditis, which resolved, presumably due to differences in drug toxicity profiles.
Our study has several limitations. Patient selection may have been biased due to the retrospective nature of the study. The well-balanced treatment-related factors post-PSM may not be representative of the entire cohort due to the removal of unmatched data and should be interpreted with caution. Patients in the INC group received different immunotherapy drugs (there is no evidence to suggest that different anti-PD-1 inhibitors have different effects). Lastly, the short observation period restricts our understanding of long-term effects and complicates the analysis of key efficacy measures like PFS, OS, and DFS. Longer follow-up is needed to assess long-term survival and validate our results.
Future research should consider the diversity and complexity of combination therapies and explore potential biomarkers. Clinical trials (NCT03925090, NCT05341193, and NCT04907370) may offer insight into the optimal regimen, timing, and duration of anti-PD-1 immunotherapy.
Conclusion
Neoadjuvant therapy with anti-PD-1 inhibitors and nimotuzumab combined with chemotherapy demonstrates promising anti-tumor activity with acceptable safety for LANPC. More well-designed randomized trials with larger patient cohorts are needed to confirm long-term efficacy, including OS, local recurrence-free survival, and distant metastasis-free survival.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251316094 – Supplemental material for Anti-programmed death-1 inhibitors and nimotuzumab in combination with induction chemotherapy for locoregionally advanced nasopharyngeal carcinoma: a propensity score-matched analysis
Supplemental material, sj-docx-1-tam-10.1177_17588359251316094 for Anti-programmed death-1 inhibitors and nimotuzumab in combination with induction chemotherapy for locoregionally advanced nasopharyngeal carcinoma: a propensity score-matched analysis by Ya-Ni Zhang, Yu-Pei Chen, Pu-Yun OuYang, Tai-Xiang Lu, Fang-Yun Xie, Fei Han and Chun-Yan Chen in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251316094 – Supplemental material for Anti-programmed death-1 inhibitors and nimotuzumab in combination with induction chemotherapy for locoregionally advanced nasopharyngeal carcinoma: a propensity score-matched analysis
Supplemental material, sj-docx-2-tam-10.1177_17588359251316094 for Anti-programmed death-1 inhibitors and nimotuzumab in combination with induction chemotherapy for locoregionally advanced nasopharyngeal carcinoma: a propensity score-matched analysis by Ya-Ni Zhang, Yu-Pei Chen, Pu-Yun OuYang, Tai-Xiang Lu, Fang-Yun Xie, Fei Han and Chun-Yan Chen in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
