Abstract
Objective:
Neoadjuvant chemotherapy has increased the survival benefit of non-small cell lung cancer (NSCLC) patients. The effects of different neoadjuvant therapies are still controversial. We carried out the study to evaluate the efficacy and safety of neoadjuvant therapy.
Methods:
We performed a search of electronic databases (PubMed, Embase, MEDLINE, Cochrane) for randomized controlled trials (RCTs) comparing neoadjuvant treatment. After literature screening and data extraction, efficacy, and safety were analyzed by the Bayesian network meta-analysis (NMA).
Results:
A total of 19 RCTs were included, covering 3276 patients and six kinds of neoadjuvant therapies, including immunotherapy, targeted therapy, chemotherapy drugs and radiotherapy. Erlotinib, the first-generation epidermal growth factor receptor tyrosine inhibitors (EGFR TKIs), neoadjuvant targeted therapy is best for improving overall survival (OS) and progression-free survival (PFS), which is superior to other neoadjuvant therapy, such as neoadjuvant chemotherapy with platinum drugs [hazard ratio (HR) 0.39, 95% confidence intervals (CIs) 0.16–0.96], neoadjuvant chemoradiotherapy (HR 0.37, 95% CI 0.14–0.96) and neoadjuvant chemotherapy with non-platinum drugs (HR 0.25, 95% CI 0.07–0.90). OS of all neoadjuvant therapies is superior to surgery alone, but only neoadjuvant chemotherapy with platinum drugs showed a significant advantage (HR 0.76, 95% CI 0.59–0.93). Besides, for the stage IIIA N2 NSCLC patients, no significant difference was found between neoadjuvant therapies.
Conclusions:
Targeted neoadjuvant therapy is the best treatment for prolonging PFS. The neoadjuvant chemotherapy with platinum drugs was associated with the better OS benefits for patients with NSCLC, compared with surgery alone. There is no significant difference in the efficacy of neoadjuvant therapy for the stage IIIA N2 NSCLC.
Keywords
Introduction
Lung cancer is one of the malignant tumors with the highest morbidity and mortality around the world, among which non-small cell lung cancer (NSCLC) accounts for about 85%. 1 Radical surgical treatment is the primary treatment for early or partial locally advanced NSCLC. However, the tumor cells in a small number of early NSCLC patients have occult lymph node metastasis, spreading through air spaces and even peripheral blood disseminating. Although radical surgery can achieve local control, the recurrence rate is still relatively high. 2 The goal of a radical cure for all early NSCLC patients cannot be achieved by surgery alone. For patients with advanced NSCLC, the application of surgical treatment is more limited. Therefore, more and more studies have begun to explore the application of perioperative treatment of NSCLC.
At present, perioperative therapies mainly include postoperative adjuvant chemotherapy and preoperative neoadjuvant chemotherapy. Studies have shown that adjuvant chemotherapy, which can reduce the postoperative recurrence rate and extend postoperative survival time, has been recommended for stage II, stage III, and part of stage IB (the maximum diameter of the tumor >4 cm) NSCLC patients. Neoadjuvant therapy is favored by some surgeons because of eliminating micrometastases, improving the rate of complete resection and even reducing tumor stage. A large number of randomized controlled studies have shown that neoadjuvant chemotherapy can improve the 5-year survival rate of NSCLC by nearly 5%, which is the same as the benefit of adjuvant chemotherapy, but this result is still not satisfactory. 3 Meanwhile, breakthroughs have been made in the systemic treatment of advanced NSCLC. With the development of molecular biology technology, tyrosine kinase inhibitors (TKIs) and immune checkpoint inhibitors (ICIs) have successively ushered in the era of lung cancer targeted therapy and immunotherapy. The CheckMate003 study showed a 5-year survival rate of 16% in advanced NSCLC patients treated with novilumab. 4 The long-term follow-up data showed a 5-year survival rate of 15.5% in NSCLC patients treated with pembrolizumab in the KEYNOTE001 study. 5
The rapid development of advanced tumor treatment has also prompted the researchers further to explore perioperative treatment, especially preoperative neoadjuvant therapies. It not only provides more surgical opportunities for some patients with advanced lung cancer but also creates more chances for the better surgical prognosis of patients with early lung cancer. For other types of cancers, such as malignant melanoma and neuroglioma, there have been studies showing that neoadjuvant immunotherapy can achieve better clinical efficacy. 6 In early stage NSCLC patients, nivolumab as neoadjuvant immunotherapy increases the major pathological response (MPR) rate up to 43%. 7 These results make the application prospect of neoadjuvant immunotherapy more and more attractive.
However, there is no consensus on the indications for neoadjuvant therapies and the effects of different neoadjuvant therapies. Therefore, this study included randomized controlled trials (RCTs) that have been published to compare the efficacy of neoadjuvant therapies, and performed a network meta-analysis (NMA) on a variety of NSCLC neoadjuvant therapy to provide some guidance for the clinical application of neoadjuvant therapies and subsequent studies.
Methods
Data sources and searches
The institutional review board of the participating institutions approved our study and waived the need for informed consent due to the retrospective nature of this study (no. K20-032Y). The literature was included in the NMAs following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) extension statement (Supplemental Table 1). 8 The predefined main keywords, such as ‘non-small-cell lung cancer’, ‘neoadjuvant’, and ‘randomized controlled trial’ were searched in the PubMed, Embase, MEDLINE and Cochrane databases. We restricted the scope of the included literature to studies designed according to a RCT precisely to define the study target population and study characteristics, in order to ensure the comparability of all studies and the effectiveness of indirect estimates in the NMAs. We included all relevant articles published before 25 March 2020, based on predetermined search criteria, which was done using Endnote X9. To ensure the integrity of the data as far as possible, we also selected and evaluated the references from the included references. The detailed search strategy is presented in Supplemental Table 2.
Study selection
The inclusion criteria of the literature included: (a) the study was a prospective RCT; (b) The study was carried out in patients with pathological or clinical NSCLC (all stages according to the eighth edition of the American Joint Committee of Cancer staging guidelines); (c) in the treatment plans that have been studied (such as surgery, chemotherapy, radiotherapy, etc.), at least one neoadjuvant treatment plan is included; (d) at least one of the following clinical outcome measures: overall survival, progression-free survival, treatment-related death, objective response rate, and progressive disease was reported in the eligible studies.
According to the inclusion criteria, the titles and abstracts of the relevant studies were screened. The full text and references of the literature were further evaluated. Studies that did not meet the inclusion criteria were excluded. At the same time, any study that only reported the survival data, while through which the relevant hazard ratio (HR) of the survival data could not be evaluated and calculated, was excluded. Any study that only published the conference abstract but no formal paper was also excluded. When data from the same study were released at different stages, we traced it back to the earliest or most recently published findings.
Data extraction
The data information contained in the article that met the inclusion criteria and was not excluded was collected into a spreadsheet. The information extracted included the first author, year of publication, country or region of the study object, study design, number of patients, treatments and outcomes. Outcomes data were collected for the assessment of overall survival (OS), progression-free survival (PFS), treatment-related death (trDeath), objective response rate (ORR), and progressive disease (PD) after neoadjuvant treatment.
Results for data involving event time (such as survival data) are represented by HR and their 95% confidence intervals (CIs). If survival data were analyzed but HR results were not directly reported, they were estimated along with their 95% CIs through Kaplan–Meier curves using the Engauge Digitizer version 4.1, the method introduced by Parmar and colleagues.9,10 For dichotomized variables (such as trDeath and tumor response rate), the Mantel–Haenszel method 11 was used to calculate the odds ratio (OR) based on the number of cases and the number of patients to evaluate the effect of the trial.
Risk of bias assessment
The Cochrane Risk of Bias Tool was used to assess the risk of bias in each RCT study 12 and to assess the methodological quality of the included studies. Its evaluation index includes seven domains: random sequence generation; allocation concealment; blinding of participants and personnel; blinding of outcome assessment; incomplete outcome data; selective outcome reporting; and other sources of bias. Items were scored as low, high, or unclear risk of bias. All investigators independently conducted study selection and data extraction. Two investigators independently assessed the risk of bias of individual studies. Any discrepancies were resolved by consensus and arbitration by a panel of adjudicators
Statistical analys
For the outcomes related to survival events (such as OS and PFS) and the dichotomous variable (such as trDeath, ORR, and PD), we will calculate and evaluate the HR and OR, respectively, along with their 95% CI calculated as a measure of estimate uncertainty. Using a random-effects model, we achieved the Bayesian NMA for OS and PFS by running the Gemtc package in R (https://cran.r-project.org/web/packages/gemtc/index.html) to call the JAGS software (https://sourceforge.net/projects/mcmc-jags/files). The Gemtc package was run in R to achieve the Bayesian NMA for trDeath, ORR and PD. In the software, according to the area under the cumulative ranking curve (SUCRA), the probability evidence of the ranking of each neoadjuvant therapy is given by comprehensive calculation.13–15 Based on non-informative uniform and normal prior distributions 16 and to fit the model, we set the parameters with four different chains of overdispersed initial values: 400,000 iterations (100,000 per chain) yielded to obtain the posterior distributions of model parameters; 50,000 burn-ins; and a thinning interval of 10 for each chain. We evaluated the convergence of iterations according to Brooks–Gelman–Rubin convergence. 17
Result heterogeneity across studies was evaluated with Cochrane’s Q statistic and quantified with the inconsistency statistic (
The inconsistency model will be used by the Gemtc package in R. Pairwise meta-analyses in a Bayesian framework were also performed with results complemented by those in the frequentist framework to contrast the corresponding pooled hazard or odds ratios from the network meta-analyses for the evaluation of local inconsistency; we evaluated global inconsistency by comparing the fit of consistency and inconsistency models.19,20
To assess the robustness and reliability of the results, we planned the sensitivity analysis. Stage IIIA N2 NSCLC patients were included in the sensitivity analysis.
Results
Eligible studies
A total of 1205 articles were included after repetition was excluded by reviewing the title and abstract. We preliminarily identified 36 studies to read and comprehensively evaluate the full text and references in detail. After layers of screening, 19 RCTs from 21 articles were included (Figure 1) with a total of 3276 patients randomly assigned to receive one of the following six kinds of neoadjuvant therapies: Erlotinib neoadjuvant targeted therapy (Erlotinib), neoadjuvant chemotherapy with platinum drugs (pCT), neoadjuvant chemoradiotherapy (pCT_R), Panitumumab + neoadjuvant chemoradiotherapy (P_pCT_R), neoadjuvant chemotherapy with non-platinum drugs (non-pCT), neoadjuvant radiotherapy alone (R_alone). Besides, three kinds of treatment were included as controls for neoadjuvant therapies: surgery alone (S_alone), chemoradiotherapy alone [pCT_R (non-S)] and radiotherapy alone [R_alone (non-S)]. The characteristics of included RCTs for the NMA are shown in Table 1 (with detailed treatments in each arm shown in Supplemental Table 3). OS was reported in all of the 19 included studies, and PFS was assessed in 14 of the 14 included studies. The trDeath was recorded according to article reports. For those that did not report the trDeath, we believed that there was no treatment-related death.
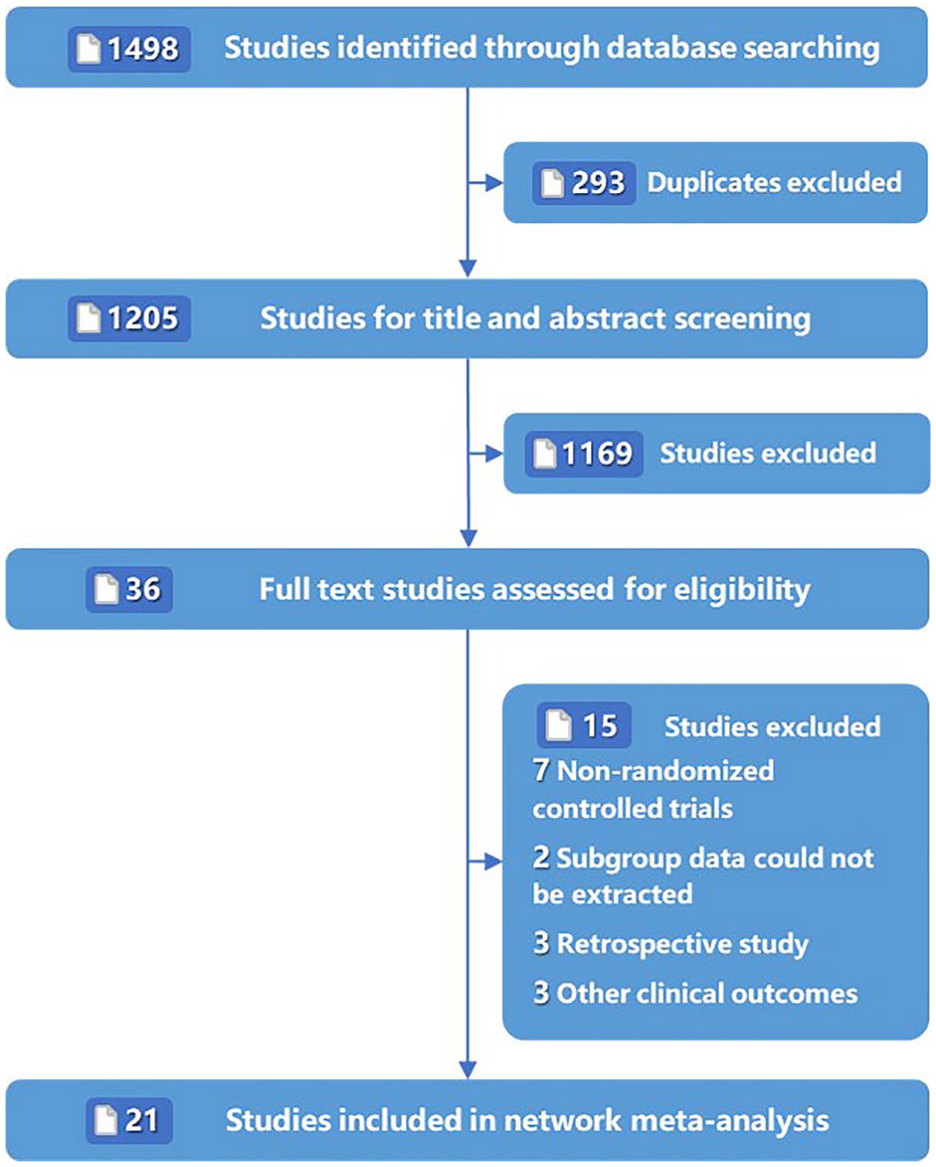
Literature search and selection process.
Characteristics of included randomized controlled trials for the network meta-analysis.

Erlotinib, Erlotinib neoadjuvant targeted therapy; non-pCT, neoadjuvant chemotherapy with non-platinum drugs; pCT, neoadjuvant chemotherapy with platinum drugs; pCT_R, neoadjuvant chemoradiotherapy; pCT_R (non-S), chemoradiotherapy alone (no surgery); P_pCT_R, Panitumumab + neoadjuvant chemoradiotherapy; R_alone, neoadjuvant radiotherapy alone; R_alone (non-S), radiotherapy alone (no surgery); S_alone, surgery alone.
Quality of evidence
The detailed risk of bias assessments was summarised in Supplemental Figure 1. The overall heterogeneity assessment of the results showed that the heterogeneity was low for OS (
The fit of the consistency model was similar to or better than that of the inconsistency model (Supplemental Table 4). The node splitting approach was used to calculate the inconsistency of the model, in which direct and indirect evidence was separately contrasted on a particular comparison. The results show
Network meta-analysis
Efficacy and safety
In STATA (version 16.0), we generated network plots for different outcomes to illustrate the geometries, to clarify which treatments were compared directly or indirectly in the included studies. 42 The network plots are shown in Figure 2. Moreover, the OS and PFS results of NMA using random-effects are summarized in Table 2. According to the accumulative rankings by SUCRA, we found that the possible best treatments prolonging OS are Erlotinib, while the effect is consistent with other treatments. In addition, in the analyses of the OS, the worst treatment was S_alone, and pCT had obvious advantages (HR 0.76, 95% CI 0.59–0.93) and pCT_R had potential advantages (HR 0.77, 95% CI 0.54–1.09) over it. According to the analysis of PFS, the possible best treatments were neoadjuvant targeted therapy (Erlotinib) targeted drugs and neoadjuvant Panitumumab plus chemoradiotherapy, which were ranked first and second, respectively. The two treatments were similar (HR 0.62, 95% CI 0.13–2.99), while Erlotinib was significantly better than other neoadjuvant therapies including pCT (HR 0.39, 95% CI 0.16–0.96), pCT_R (HR 0.37, 95% CI 0.14–0.96), and non-pCT (HR 0.25, 95% CI 0.07–0.90). Erlotinib was also better than non-neoadjuvant therapy, such as pCT_R (non-S) (HR 0.35, 95% CI 0.12–0.97) and S_alone (HR 0.32, 95% CI 0.12–0.80). The ORR and PD results of NMA (Table 3) did not indicate which neoadjuvant therapy could significantly improve ORR and PD. However, compared with non-pCT, the worst treatments for ORR, Erlotinib (OR 0.10, 95% CI 0.02–0.53), pCT_R (OR 0.17, 95% CI 0.04–0.65), pCT_R (non-S) (OR 0.07, 95% CI 0.01–0.43), pCT (OR 0.22, 95% CI 0.06–0.76) have significant advantages in improving ORR. As most of the included study did not report trDeath, it cannot accurately evaluate the OR value of the NMA.

Network diagrams of comparisons on different outcomes of treatments in patients undergoing different treatments. (A) Comparisons of progression-free survival and overall survival. (B) Comparisons on treatment-related death, objective response rate and progressive disease. Each circular node represents a type of treatment. The node size is proportional to the total number of patients receiving treatment (in brackets). Each line represents a type of head-to-head comparison. The width of lines is proportional to the number of trials comparing the connected treatments.
Estimate results according to the network meta-analysis on overall survival (lower triangle) and progress free survival (upper triangle), along with the treatment ranking distribution (in the below arrow shape).


Erlotinib, Erlotinib neoadjuvant targeted therapy; non-pCT, neoadjuvant chemotherapy with non-platinum drugs; pCT, neoadjuvant chemotherapy with platinum drugs; pCT_R, neoadjuvant chemoradiotherapy; pCT_R (non-S), chemoradiotherapy alone (no surgery); P_pCT_R, Panitumumab + neoadjuvant chemoradiotherapy; R_alone, neoadjuvant radiotherapy alone; R_alone (non-S), radiotherapy alone (no surgery); S_alone, surgery alone.
Estimate results according to the network meta-analysis on the progressive disease (lower triangle) and objective response rate (upper triangle), along with the treatment ranking distribution (in the below arrow shape).


Erlotinib, Erlotinib neoadjuvant targeted therapy; non-pCT, neoadjuvant chemotherapy with non-platinum drugs; pCT, neoadjuvant chemotherapy with platinum drugs; pCT_R, neoadjuvant chemoradiotherapy; pCT_R (non-S), chemoradiotherapy alone (no surgery); R_alone, neoadjuvant radiotherapy alone; R_alone (non-S), radiotherapy alone (no surgery).
Rank probabilities
Figures 3 and 4 show the ranking probabilities for all treatments included (with detail ranking results summarized in Supplemental Table 6). For the treatment effect of OS prolongation, P_pCT_R, Erlotinib and R_alone ranked first with the highest probability (44.0%, 23.0% and 21.0%, respectively), while R_alone (non-S) and S_alone ranked last with the highest probability (31.0% and 28.0%, respectively). For the effect of PFS prolongation, Erlotinib and P_pCT_R ranked first with the highest probability (71.0% and 26.0%, respectively), while non-pCT and R_alone ranked last with the highest probability (48.0% and 25.0%, respectively). The trDeath was used to assess the safety of treatments included. We found that the R_alone had the highest probability of ranking first in the incidence of less trDeath (41%), while the P_pCT_R had the highest probability of ranking last in the incidence of more trDeath (56%). According to the analysis of ORR and PD, pCT_R (non-S) (56.0%) and pCT_R (42.0%) were the treatments with the highest probability of achieving a good prognosis. In contrast, the treatments with the lowest probability were both non-pCT, with a probability of 71.0% and 42.0%, respectively.

Ranking curves of treatments according to the results of the network meta-analysis on overall survival (solid line) and progression-free survival (dotted line).
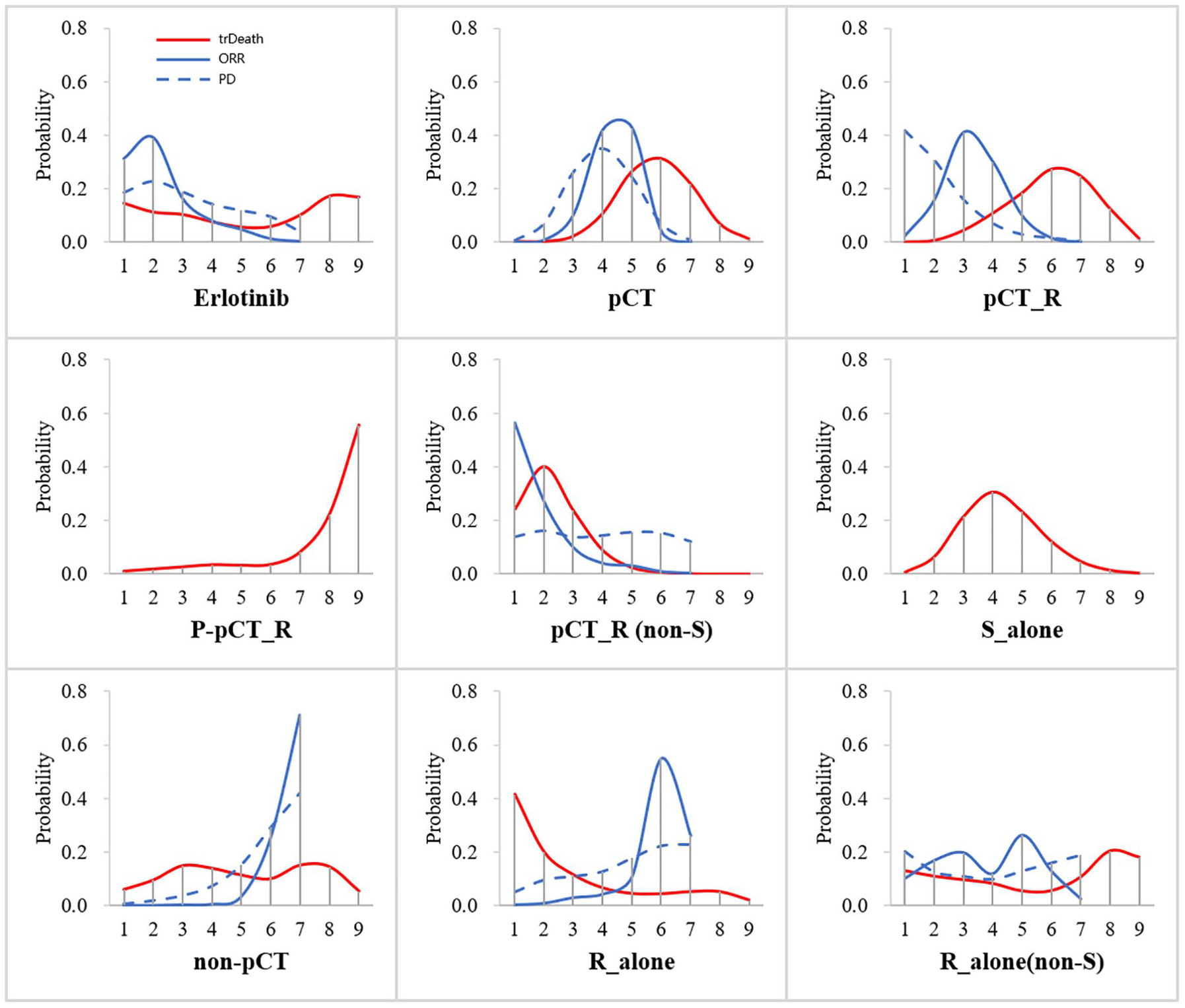
Ranking curves of treatments according to the results of the network meta-analysis on treatment-related death (solid red line), objective response rate (solid blue line) and progressive disease (blue dotted line).
Sensitivity analysis
With a total of 1382 patients, 11 studies21–25,29,31,33,37–40 on stage IIIA N2 NSCLC patients were included in the first sensitivity analysis. Results did not show relevant deviations compared with the original NMA (Supplemental Table 7). For the effect of OS and PFS prolongation, P_pCT_R (44.0%) and Erlotinib (65.0%) ranked first with the highest probability, respectively. The OS, PFS, ORR and PD results of NMA using random-effects are summarized in Supplemental Figure 4. From the analysis, we found that P_pCT_R and Erlotinib showed a tendency to be superior to other neoadjuvant therapies, and S_alone was at a disadvantage compared with neoadjuvant therapies for the stage IIIA N2 NSCLC patients. However, there was no significant statistical difference between different neoadjuvant therapies or between neoadjuvant therapies and S_alone for the stage IIIA N2 NSCLC patients.
Overall, nine studies (1990–2003), including five treatments for 945 patients and 10 studies (2007–2019), including seven treatments for 2331 patients, were included in the second sensitivity analysis by published year. Owing to the low number of involved studies, only comparisons for overall survival were accessible in the subgroup published range from 1990 to 2003 (Supplemental Figure 5.A, B). In the subgroup published range from 2007 to 2019, each neoadjuvant therapy and S_alone seemed to show the same OS, ORR and PD benefit, and Erlotinib had the greatest probability to provide the best progression-free survival (Supplemental Figure 5.C). In the subgroup published range from 1990 to 2003, pCT is significantly superior to S_alone; there was no significant statistical difference among other treatments for the OS.
Discussion
In this NMA, we comprehensively summarized and analyzed the efficacy and safety of various neoadjuvant therapies among NSCLC patients. The results showed that Erlotinib had the highest cumulative probability of ranking first in the effect of prolonging OS and PFS. There was no significant statistical difference for OS among most treatments except that pCT was significantly superior to S_alone. However, the analysis of PFS data showed that Erlotinib was significantly superior to pCT, pCT_R, non-pCT, as well as non-neoadjuvant therapies such as pCT_R (non-S) and S_alone. In terms of trDeath, R_alone had the greatest probability of ranking first (the lowest mortality), and P_pCT_R had the greatest probability of ranking last (the highest mortality). According to the evaluation of ORR and PD among NSCLC patients, we found that non-pCT showed a significant disadvantage in ORR, and other neoadjuvant therapies had a similar effect on tumor remission or progression.
Perioperative adjuvant therapy has been widely used. Neoadjuvant therapies have been gradually promoted in clinical practice because of their significant advantages: 43 (a) Neoadjuvant therapy can eliminate the early micrometastases of the tumor; (b) Observation indicator such as MPR after neoadjuvant treatment can be used as an effective alternative for prognosis evaluation besides survival data; (c) In terms of clinical research, the pathological response indicator of neoadjuvant therapies can shorten the research time and improve the efficiency of drug development and marketing, etc. It is not difficult to find through the article included in this study that large randomized controlled studies in the early years have shown that the treatment of neoadjuvant chemotherapy combined with surgery can benefit the survival of NSCLC patients better than surgery alone.35,39 With the development of molecular biology, a series of therapeutic targets and immune checkpoints for lung cancer has been discovered, and the number of studies on NSCLC targeted drugs and immunotherapy drugs have increased dramatically, which also enables researchers focusing on targeted therapy and immunotherapy to explore new types of neoadjuvant treatment options.
Unlike traditional neoadjuvant chemotherapy, which seeks to shrink tumor lesions and delay the advancement of the tumor in order to create surgical opportunities, neoadjuvant targeted therapy or immunotherapy focuses more on the control of tumor cells at the molecular level or controls immune cells by inducing the immune effect of the body on the tumor to achieve long-term benefits, respectively. The response rate (RR) of EGFR TKI targeted therapy (58–74.9%) was significantly higher than the RR of traditional neoadjuvant chemotherapy (25–63%).44,45 Fored
In the field of immunotherapy, the NADIM trial from Spain evaluated the efficacy of neoadjuvant chemotherapy plus nivolumab immunotherapy in patients with resectable stage IIIA NSCLC. The results reported that 85.4% of patients achieved MPR, and 71.4% of patients achieved pathological complete response (pCR) after neoadjuvant therapy. However, most of the neoadjuvant immunotherapy that has been reported so far is in a single-arm study, and only the pathological response rate has been reported. There are many phase III clinical trials that are ongoing (such as NCT02998528, NCT03456063, etc.), so there is no high-level evidence for the efficacy of neoadjuvant immunotherapy. Our study provides a reference for the following development of this field.
Despite the significant advances in neoadjuvant therapy for lung cancer, we are well aware that many problems remain unsolved. For example, survival indicators such as OS and PFS play an irreplaceable role in the evaluation of tumor therapies, but the clinical trials of NSCLC with OS as the primary research endpoint often take 10 years or more, which dramatically increases the cost of drug development and the difficulty of innovation. Could the endpoint of neoadjuvant therapies be simplified? Some studies show that MPR is a clinically proved surrogate of efficacy. When used as the primary endpoint, it can allow for a more efficient evaluation of drugs in the neoadjuvant setting. Pataer
In addition, the patients’ tumor beds shrank after neoadjuvant immunotherapy. However, the receding tumor bed is filled with a large number of lymphocytes, intact lymphoid tissue and necrotic tumor cells. There are even numerous new blood vessels and fibrotic hyperplasia around the tumor. All these make the traditional pathological assessment criteria represented by MPR challenging. Therefore, many new pathological evaluation indicators for neoadjuvant therapies have begun to appear. For example, Cottrell
Of course, there are still some limitations to this study. First, the data source of NMA is based on the collection of published clinical studies. Similar to observational studies, there are inevitable confounding factors in the included data. Direct evidence for clinical problems comes from direct comparisons of treatment regimens, especially randomized controlled clinical trials and meta-analyses with the highest level of evidence. However, for the NMA, the conclusion mainly depends on the indirect comparison between the studies. Although all the included articles in this study are officially published RCTs, the consistency and transmissibility of the data should still be paid attention to in the estimation and interpretation of the results. Second, for targeted neoadjuvant therapies and neoadjuvant immunotherapies, most of the studies published so far are single-arm clinical trials, and the phase II clinical trials included in the study contain a few subjects and are very vague in the description of the sample randomization method. Therefore, there may be potential publication and selection bias in this study. Third, for the NMA, the higher the probability of Bayesian cumulative ranking, the greater the significance of its ranking. However, when the probability of ranking of each treatment shows similar results, it is difficult for us to define its ranking clearly. However, the uncertainty caused by the confounding factors is also difficult to calculate. Therefore, in order to evaluate accurately the efficacy differences among treatments, besides the cumulative ranking, we also need to consider the consistency and transmissibility of the NMA data. In the analysis results, HR or OR and their 95% CIs are all used to determine whether the differences are significant, or if there are only potential differences. Fourth, as far as this study is concerned, most of the studies reported OS and PFS, while only a few studies reported trDeath, ORR and PD, which may be one of the reasons why the results of the NMA data of the latter did not show significant differences. Therefore, we believe that in studies on neoadjuvant therapies in the future, not only patient survival data should be evaluated, but also tumor progression and treatment safety after neoadjuvant therapies should be evaluated more accurately.
Erlotinib (the first-generation EGFR TKI) neoadjuvant targeted therapy was the highest priority treatment for OS and PFS and showed significant advantages in PFS. Neoadjuvant chemotherapy with platinum drugs was significantly superior to surgery alone. In summary, the effect of most neoadjuvant therapies is similar. Further studies are needed to evaluate neoadjuvant therapies for the tumor evaluation index (such as ORR and PD) and the safety of neoadjuvant therapies, especially for stage IIIA N2 NSCLC.
Supplemental Material
supplementary_materials_tamR1 – Supplemental material for Bayesian network meta-analysis of efficacy and safety of neoadjuvant therapy for non-small-cell lung cancer
Supplemental material, supplementary_materials_tamR1 for Bayesian network meta-analysis of efficacy and safety of neoadjuvant therapy for non-small-cell lung cancer by Yijiu Ren, Hai Tang, Jie Zhang, Yunlang She, Xiaoting Sun, Dong Xie and Chang Chen in Therapeutic Advances in Medical Oncology
Footnotes
Author contributions
YR and HT contributed equally to this paper and are joint first authors. All corresponding and first authors contributed to the study concept and design. All authors selected the articles and extracted the data. YR, HT, XS and YS analyzed and interpreted the data. YR and HT wrote the first draft of the report. All authors approved the final version of the report. YR was responsible for the integrity and accuracy of the data and is the guarantor. The corresponding authors attest that all listed authors meet authorship criteria and that no others meeting the criteria have been omitted.
Conflict of interest statement
All author(s) have completed the ICMJE uniform disclosure form at ![]() and declare no support from any organization for the submitted work; no financial relationships with any organizations that might have an interest in the submitted work in the previous 3 years; no other relationships or activities that could appear to have influenced the submitted work.
and declare no support from any organization for the submitted work; no financial relationships with any organizations that might have an interest in the submitted work in the previous 3 years; no other relationships or activities that could appear to have influenced the submitted work.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: It was supported by projects from the Shanghai Hospital Development Center (SHDC12017114), Shanghai Pulmonary Hospital Innovation Team (FKCX1906, FKXY1902), and the Shanghai Science and Technology Committee (20YF1441100, 20XD1403000, 18DZ2293400).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
