Abstract
Therapeutic cancer vaccines aim to generate a robust immune response against tumour-associated antigens (TAAs) or tumour-specific antigens. While their safety is well established, their efficacy as monotherapy remains limited due to factors such as self-tolerance to TAAs and the immunosuppressive tumour microenvironment. Combining cancer vaccines with systemic anticancer therapies (SACTs) offers a promising strategy to improve efficacy. However, the optimal timing and combination with immune checkpoint inhibitors (ICIs) and chemotherapy to enhance immunogenicity are not yet fully understood. This review aims to assess the evidence regarding the immunogenicity of antiviral and anticancer vaccines when combined with SACTs, including chemotherapy and ICIs, with a particular focus on the timing of vaccine administration relative to SACT. Additionally, we evaluate the impact of steroids on immunogenicity. Our findings suggest that the timing of vaccine administration is critical, with improved immunogenic responses observed when vaccines are administered at nadir (15 days post-chemotherapy). Certain chemotherapies, such as low-dose metronomic cyclophosphamide and paclitaxel, demonstrate potential for immunomodulation, enhancing T-cell responses when combined with vaccines. Conversely, steroids may reduce immunogenicity. The combination of ICIs with cancer vaccines shows evidence of a synergistic effect, with concurrent administration generally yielding better outcomes than sequential approaches. Prospective trials exploring various timings and sequences are essential to optimize the efficacy of anticancer vaccines.
Keywords
Background
The introduction of immune checkpoint inhibitors (ICIs) has revolutionized the treatment of solid tumours, achieving durable responses and prolonged survival in several tumour types such as non-small-cell lung cancer (NSCLC). 1 However, most patients still experience primary or secondary resistance, highlighting the need for improved treatments.
Therapeutic cancer vaccines encompass a broad range of strategies that aim to generate a strong immune response against tumour-associated antigens (TAAs) or tumour-specific antigens (TSAs). 2 The platforms used in clinical trials include peptide- or protein-based formulations that deliver defined tumour antigens; dendritic cell (DC) vaccines that employ ex vivo-generated antigen-presenting cells to prime T-cell responses; nucleic acid (DNA or RNA) vaccines that direct in vivo synthesis of tumour antigens; viral and bacterial vector-based platforms that leverage infectious agents as delivery vehicles and whole-tumour-cell preparations that present a broad array of antigens. 3 Regardless of the platform, immunogenicity assessments in clinical trials commonly focus on quantifying vaccine-induced T-cell responses – often through interferon-gamma (IFN-γ) enzyme-linked immunospot (ELISPOT) assays, intracellular cytokine staining and flow cytometric evaluation of T-cell subsets – as well as measuring humoral responses, such as antigen-specific antibody titres. Their safety has been well established in early-phase trials, but their efficacy as monotherapy in solid tumours has been disappointing. 3 There are several major challenges to consider, including self-tolerance to TAAs, the immunosuppressive influence of the tumour microenvironment and the downregulation of human leukocyte antigen (HLA) class I complexes. 3
A strategy to overcome these limitations is the combination of cancer vaccines with systemic anticancer therapies (SACTs). 3 Coadministration with ICIs could help to overcome self-tolerance, enhancing T-cell induction against TAAs. 4 Furthermore, chemotherapy may reduce immunosuppressive myeloid and T regulatory cells, potentially enhancing cytotoxic T-cell activity against tumour cells. 4 Some compounds have been shown to enhance effector T-cell stimulation (e.g., paclitaxel and cyclophosphamide), while others increase tumour immunogenicity (e.g., doxorubicin, 5-fluorouracil and cisplatin).5–8 Additionally, certain compounds reduce tumour-induced immune suppression (e.g., cyclophosphamide, 5-fluorouracil, gemcitabine and carboplatin combined with paclitaxel).9–12 Despite a strong rationale for combination strategies, trials have shown limited benefit. Key questions remain regarding the optimal timing of cancer vaccines relative to chemotherapy and/or ICIs, as well as the effect of different chemotherapy doses and supportive medicines such as steroids on vaccine efficacy. Although the effects of chemotherapy on immunosuppressive cells are established, its impact on vaccine-induced immunogenicity is unclear. In the following sections, we will review data from trials assessing vaccine immunogenicity in patients receiving chemotherapy and/or ICIs. Additionally, we will gather information on the efficacy of cancer vaccines in solid tumours according to their timing and the chemotherapy and/or ICIs agents used.
Brief methods
For this narrative review, we conducted a comprehensive search of the available literature to explore the immunogenicity and efficacy of therapeutic cancer vaccines and antiviral vaccines when combined with SACT, including chemotherapy and ICIs. We searched databases such as PubMed, EMBASE and Scopus using keywords and combinations such as ‘cancer vaccines’, ‘immunogenicity’, ‘chemotherapy’, ‘immune checkpoint inhibitors’ and ‘steroids’. Studies included in this review covered clinical trials, preclinical studies and meta-analyses focused on vaccine immunogenicity and efficacy in patients receiving chemotherapy and/or ICIs. Additionally, we reviewed studies evaluating the timing of vaccine administration relative to chemotherapy cycles, the impact of steroid use and combinations with ICIs. Our inclusion criteria encompassed the following: peer-reviewed articles published in English; clinical trials (phases I–III) and preclinical studies; and studies assessing vaccine immunogenicity. References were cross-checked for additional relevant articles. Data extracted included study design, patient population, vaccine type, SACT regimen, timing of administration and immunogenicity outcomes.
Immunogenicity of antiviral vaccines in patients receiving SACT
The immunogenicity of antiviral vaccines in patients receiving SACT has been examined in large-scale studies. Since the underlying platforms are similar to those used in anticancer vaccines, the findings from these analyses can provide valuable insights that inform the development and optimization of therapeutic cancer vaccines. As such, in recent years, a large body of evidence regarding the safety and efficacy of COVID-19 and influenza vaccines has emerged. Most of these studies test the rate of seroconversion of patients, which means developing specific antibodies (IgG) against the agent of interest and thus measuring humoral response. 13
The evidence of the safety and efficacy of inactivated multivalent influenza vaccines in patients treated with SACT has been well established. 14 In fact, international guidelines recommend the seasonal vaccination of these patients with an inactivated vaccine. 15 A 2016 study aimed to elucidate the optimal timing of vaccination relative to chemotherapy administration. Patients on a 3-weekly schedule of chemotherapy were randomized to receive the vaccine either concurrently with chemotherapy or on day 11. 16 Although seroconversion rates were numerically higher in the latter group, this difference was not statistically significant except in a subgroup of breast cancer patients, who also had a lower lymphocyte count at nadir. 16 Of note, the trial excluded patients receiving prophylactic granulocyte colony-stimulating factor and/or immunosuppressive agents, aside from those receiving antiemetic steroids. 16 In an older study, patients were randomized to receive a bivalent influenza virus vaccine either concurrently with chemotherapy or at the cycle nadir, determined by daily bloods. 17 Seroconversion was significantly higher in patients who were vaccinated at the nadir (50% vs 93%; p < 0.01), and there was no correlation between lymphocyte count and antibody response. 17 Major limitations of this study include the low number of patients and a heterogeneous population. Another trial randomized colorectal and breast cancer patients to receive an influenza virus vaccine either on day 5 or on day 16 after a chemotherapy cycle. 18 A significantly better antibody response was demonstrated in breast cancer patients vaccinated early. 18 Regarding the combination of ICIs and influenza vaccines, some studies have reported a seroconversion rate of around 70%, without significant differences compared to healthy controls.19,20 An observational study of patients receiving an ICI showed that those who were vaccinated had a lower incidence of influenza-related complications while there were no safety signals. 21
The COVID-19 pandemic disrupted the world in 2020 leading to a race to manufacture effective vaccines. Several products against COVID-19 have been approved since 2021, generally using either mRNA or a viral vector 22 and a growing body of evidence regarding their safety, immunogenicity and efficacy in cancer patients has now emerged.
A prospective study conducted in Spain assessed the immunogenicity of mRNA vaccines (BNT162b2 or mRNA-1273) or adenovirus vaccines (ChAdOx1 or Ad26.COV2.S) in lung cancer patients. 23 A total of 1973 subjects were enrolled, and 5.9% received the ChAdOx1 nCoV-19 vaccine. The study showed that 95.3% of the patients were seropositive for SARS-CoV-2 2 weeks after the first dose of vaccination, and 93.6% were seropositive 6 months after the first dose of vaccine. This rate was similar regardless of whether they were receiving SACT or not. However, patients receiving chemotherapy were more likely to be seronegative compared to patients receiving ICIs or oral agents (odds ratio 0.56; p = 0.014). 23
Another prospective study assessing predictive negative factors of seroconversion in cancer patients who received the BNT162b2 vaccine was published in 2021. While among patients on active surveillance, only 1.6% lacked seroconversion, this proportion was 13.9% in patients receiving chemotherapy, and the difference was statistically significant (p = 0.033). 24 Notably, patients receiving ICIs did not show a significant difference compared to patients not receiving SACT. 24 An observational prospective study looked at the differences in seroconversion of patients with or without chemotherapy who were vaccinated with the ChAdOx1 nCoV-19 or CoronaVac vaccine. 25 The median IgG titres in the chemotherapy group were significantly lower (26 vs 237 U/mL; p = 0.001). 25 Patients who received dexamethasone as antiemetic prophylaxis had a significantly lower median anti-SARS-CoV-2 S IgG level and seroconversion rate with the CoronaVac vaccine than patients who did not receive dexamethasone (4 (interquartile range 0.2–28) U/mL (p < 0.001) and 65.6% vs 93.8% (p = 0.002), respectively). 25
A recent meta-analysis of both clinical trials and observational studies included 2477 patients with cancer who received ICIs and were vaccinated against COVID-19. It did not demonstrate a significantly lower rate of seroconversion in patients receiving ICI compared to patients without cancer (risk ratio (RR) 0.97; 95% confidence interval (CI) 0.92–1.03). 26 Among patients diagnosed with cancer and treated with ICIs, the probability of seroconversion was higher than for patients receiving chemotherapy (RR 1.09; 95% CI 1.00–1.18). 26
Overall, these results suggest that seroconversion rates following vaccination against influenza and/or COVID-19 are lower among patients receiving chemotherapy concomitant with vaccination, and steroid medication also has a negative impact on immunogenicity. Conversely, seroconversion rates do not appear to be impacted by concomitant exposure to ICIs.
Combination of chemotherapy and anticancer vaccines
Chemotherapy causes immunosuppression but, in recent decades, it has been shown that some chemotherapy drugs can lead to immunomodulation through an inhibition of specific immune cells subsets. 10 These findings have set the stage for preclinical studies and clinical trials combining vaccines and chemotherapy using various doses and schedules in an attempt to improve immunogenicity.
Cyclophosphamide is the most extensively studied cytotoxic agent showing immunomodulation. A single low dose given 1–3 days before antigen exposure enhances T-cell response and antibody production. 27 This regimen, named metronomic cyclophosphamide, has been used in several trials combined with anticancer vaccines. In a trial performed in patients with colorectal cancer, its administration before modified vaccinia Ankara-5T4 was associated with depletion of T regulatory cells, although it did not increase the immune response measured by IFN-γ enzyme-linked immunospot assays. 28
Paclitaxel has also been thoroughly studied as an immunomodulator in solid tumours. 29 A study in tumour-bearing mice tested a connective tissue growth factor/E7 DNA vaccine alone or with different doses of paclitaxel. 29 The 6 mg/kg paclitaxel group had the highest cytotoxic/regulatory T cells ratio in tumour infiltrating lymphocytes compared to the other groups. 29 Another study tested the combination of different doses of paclitaxel and the Granulocyte-Macrophage-Colony-Stimulating Factor-surface-modified tumour-cell vaccine in a prostate cancer mouse model. 30 Each dose was administered either before or after vaccination. It was found that 4 mg/kg was the most effective dose for inducing tumour regression. Interestingly, it also showed that the administration of paclitaxel 2 days before the vaccine injection induced the highest proportion of T-cell infiltration in the tumour. 30 It has been hypothesized that low-dose paclitaxel promotes the TLR4-dependent maturation of DCs and shifts the CD4+ T-helper phenotype from type 2 to type 1, enhancing the priming and lytic activity of CD8+ cells. 31
This differential effect of low-dose, metronomic chemotherapy in tumour-bearing mice has also been shown with other cytotoxic agents. A study combining 5-fluorouracil at a low dose and an adenoviral tumour vaccine showed synergistic benefits on survival in a mouse model. 32 Another study combined cyclophosphamide, doxorubicin and a granulocyte-macrophage colony-stimulating factor-secreting breast tumour vaccine, and showed enhanced response with a 200 mg/m2 cyclophosphamide and 35 mg/m2 doxorubicin doses, although that effect disappeared with higher doses. 33
Some studies have looked at the sequence of the administration of chemotherapy with respect to the vaccines. A phase I/II pilot trial in melanoma patients tested giving dacarbazine 1 day before the administration of a vaccine consisting of HLA-A2 restricted melanoma antigen A (Melan-A/MART-1) and gp100 analogue peptides. 34 It showed a significant improvement in long-lasting memory CD8(+) T-cell response in the group that received dacarbazine compared to those who received the vaccine alone. 34 Notably, since dacarbazine is a highly emetogenic drug, dexamethasone is prescribed as a prophylactic antiemetic, 35 which did not seem to impact the immunogenicity. Another trial conducted in patients with metastatic colorectal cancer tested ALVAC-CEA/B7.1 vaccine followed by chemotherapy or in reverse order. 36 There was a lower proportion of CEA-specific T cells in those who received chemotherapy first. 36
A phase I/II clinical trial tested a MAGE-A3 vaccine, with or without adjuvant chemotherapy, in patients diagnosed with resected stage IB–III NSCLC. 37 Patients who received adjuvant chemotherapy were vaccinated either before or after its administration. The chemotherapy regimen consisted of four doses of cisplatin 80 mg/m2 on day 1 and vinorelbine 30 mg/m2 on day 1 and 8 every 3 weeks. In the cohort of patients who received the vaccine concurrently, it was administered on day 8 (vinorelbine only) and, therefore, without steroids. Both humoral and peripheral cellular immunity were measured. Although seroconversion was increased from 9% to 100%, the cellular responses were more modest (10%–38% for CD4+, 5%–10% for CD8+), with no significant differences observed between patients who received chemotherapy and those who did not. 37 Importantly, the trial did not show significant differences in terms of MAGE-A3-specific CD4+ T-cell responses with regards to the vaccine schedule, highlighting different sensitivities according to the immunogenicity measure used. 37
Carboplatin and paclitaxel, a regimen commonly used in solid tumours such as cervical cancer and NSCLC, have been among the most extensively studied combinations with anticancer vaccines in several trials. In an early phase study performed in advanced cervical cancer patients, this regimen demonstrated a decrease in the count of myeloid cells, reaching the nadir at 1–2 weeks after its administration, with an improvement in T-cell function. 38 That led to the design of a trial in which an HPV16 synthetic long peptide vaccine was administered 2 weeks after the second cycle of carboplatin and paclitaxel. 39 The chemotherapy regimen reduced myeloid-derived suppressor cells while the lymphocyte count remained stable. 39 Carboplatin and pemetrexed, standard treatments for non-squamous NSCLC, have not been as thoroughly researched with anticancer vaccines. A phase II, single-arm trial combined a DC vaccine with the aforementioned regimen. 40 The vaccine was administered on day 15 (at nadir) from cycle 3 onwards. 40 The trial did not show any unexpected safety signal while the efficacy was promising. 40 Taken together, these results highlight the potential for different immunomodulatory effects according to the cytotoxic class, combination, dose and schedule. Figure 1 highlights the primary mechanisms through which chemotherapy can enhance vaccine-induced immunogenicity. Table 1 highlights the main studies concerning the impact of chemotherapy on anticancer vaccine immunogenicity.

Potential mechanisms by which chemotherapy may enhance vaccine-induced immunogenicity.
Impact of chemotherapy on anticancer vaccine immunogenicity.
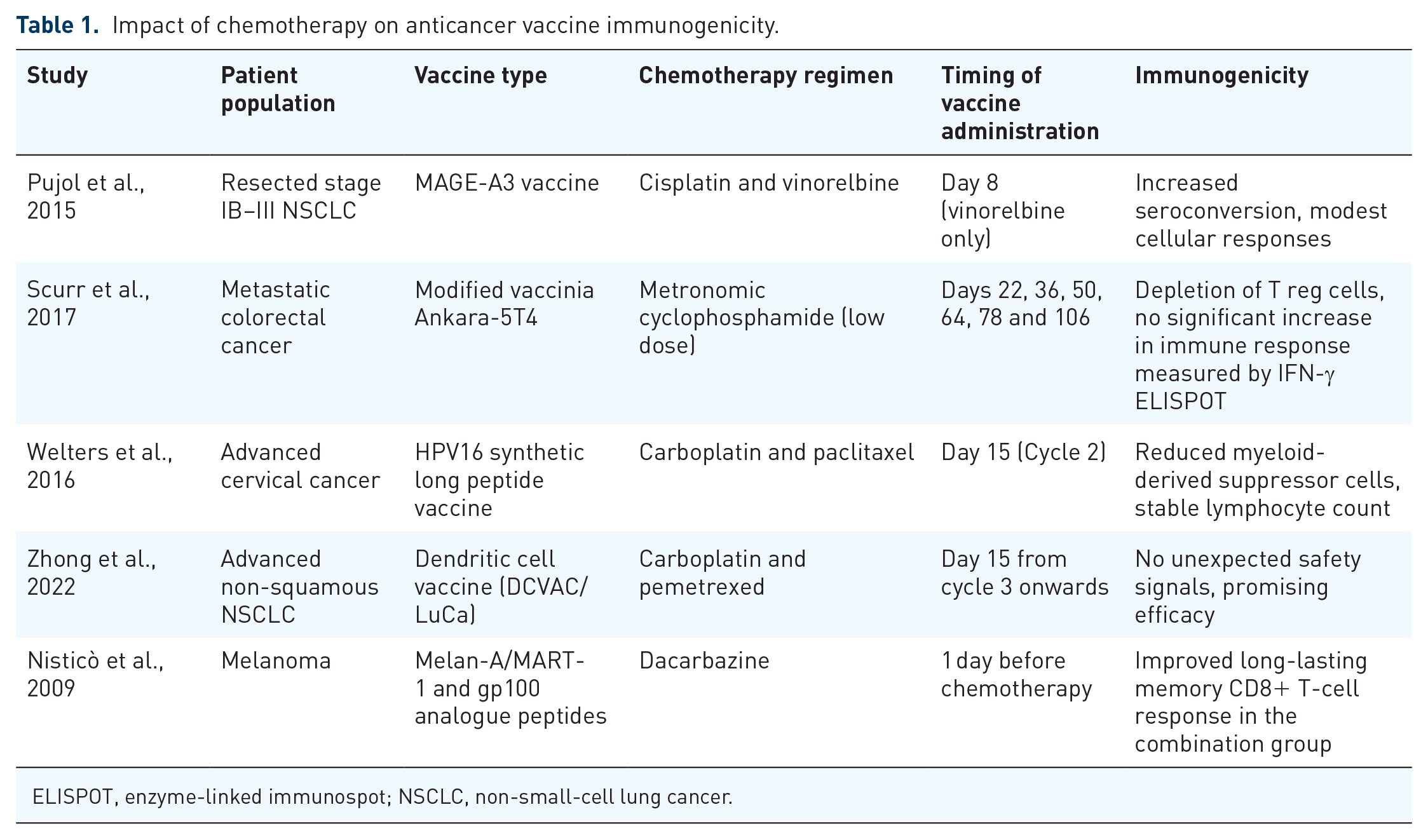
ELISPOT, enzyme-linked immunospot; NSCLC, non-small-cell lung cancer.
Impact of steroids on anticancer vaccine immunogenicity
Corticosteroids are widely used as antiemetic agents concomitantly with some of the most common chemotherapy regimens. As they are immunosuppressive medications, there are concerns regarding their effect on vaccine response. It has been shown that they can impair upstream B-cell receptor signalling and reduce transcriptional output from the three immunoglobulin loci, among other detrimental effects. 42 A preclinical study in mice showed that the administration of dexamethasone concomitantly with a vaccine based on a liposomal formulation of tumour-antigen-encoding RNA impaired antigen-specific T-cell expansion, therefore reducing the vaccine response. 43 Notably, the effect was less profound when dexamethasone was administered post-vaccination. 43
There is conflicting clinical evidence regarding the use of steroids in patients who receive anticancer vaccines. A small pilot trial investigated DCs mobilized with Flt3 ligand and loaded with multiple antigens. 44 Remarkably, five out of six patients developed a T-cell response to at least one of the tested antigens. 44 The sixth patient, however, required corticosteroid treatment at the time of DC administration, which may have compromised their immune response. 44 A phase I/Ib trial tested a personalized neoantigen vaccine in patients diagnosed with glioblastoma multiforme (GBM). 45 While all the patients receiving dexamethasone during vaccine priming failed to generate interferon-γ responses, those who did not receive steroids had a stronger immune response. 45 By contrast, a phase I study in patients diagnosed with GBM tested an immunotherapeutic multiple-peptide vaccine 46 and there was no significant impact of concomitant dexamethasone on the immune response. 46 This might be explained by the exclusion of patients on a dexamethasone dose higher than 4 mg/day such that the median dose was 2 mg daily. 46 Another phase I/II trial in patients diagnosed with GBM tested a multipeptide vaccine but did not demonstrate a significant difference in vaccine-specific CD4 and/or CD8 T-cell responses when dexamethasone was administered after vaccination. 47 Importantly, most studies have focused on the influence of chronic corticosteroid use, rather than episodic administration for antiemetic purposes, which may have a different impact on immunogenicity (see Table 2).
Impact of steroids on anticancer vaccine immunogenicity.

Combination of ICIs and anticancer vaccines
The combination of ICIs with anticancer vaccines is an attractive strategy due to the potential to overcome self-tolerance to TAAs and the immunosuppressive tumour microenvironment. 4
A phase I/II trial in patients with advanced melanoma demonstrated the safety of combining the PD-1 inhibitor pembrolizumab with a helper peptide vaccine. 48 It also showed increased intratumoural T and B cells, as well as Th1 cells. 48 A further phase I trial in melanoma patients whose tumours expressed NY-ESO-1, of a NY-ESO-1 vaccine and the anti-CTLA4 antibody ipilimumab were evaluated. An increase in proliferating intratumoural CD8+ T cells and T-cell responses to NY-ESO-1 was detected in patients 21 days after vaccination. 49 Another trial combining a NY-ESO-1 vaccine and the PDL-1/PD-1 inhibitor atezolizumab in soft-tissue sarcoma patients showed some improvement of an anti-NY-ESO-1 response compared to receiving atezolizumab alone, demonstrated by increased NY-ESO-1-specific T cells (p = 0.01) and NY-ESO-1-specific antibody responses (p < 0.001), although it did not translate to a survival benefit. 50
Recently, results from a phase I trial testing a melanoma mRNA vaccine targeting four TAAs (NY-ESO-1, MAGE-A3, Tyrosinase and TPTE) alone or in combination with an ICI in patients with stage IIIB, IIIC or IV melanoma have been reported. 51 A high magnitude of T-helper-1 phenotype of FixVac-induced T cells was demonstrated in the latter group. 51 Radiological responses were observed in patients who received the vaccine alone and in combination with ICI in individuals who had previously progressed on ICI treatment. 51 An open-label, randomized, phase II trial of an mRNA neoantigen vaccine administered with pembrolizumab also showed improved recurrence-free survival (RFS) over pembrolizumab alone in patients with resected high-risk melanoma. 52
A phase I trial in patients with resected pancreatic ductal adenocarcinoma tested the administration of an individualized mRNA cancer vaccine after one atezolizumab dose. 53 Chemotherapy was administered after the vaccination. Induced de novo high-magnitude neoantigen-specific T cells were demonstrated in 50% of patients, and it was associated with longer RFS. 53
Overall, these results point to the feasibility and possible synergy of the combination of an ICI with a vaccine to one or more TAAs (Table 3). However, there is scarce evidence regarding the optimal sequence and timing of immunotherapy with anticancer vaccines. In a mouse model, synergy in tumour growth inhibition was observed only on simultaneous administration of vaccine and ICI, whereas the blockade of PD-1 before antigenic stimulation fully abrogated the anti-tumoural effects. 54 Concurrent vaccine and ICI administration is supported by the results of a clinical trial conducted in patients with prostate cancer that tested the combination of a DNA vaccine and pembrolizumab, either sequentially or concurrently. When combined with concurrent PD-1 blockade, patients with evidence of Th1 immunity experienced PSA declines, objective tumour responses and CD8+ T-cell infiltration into metastatic lesions. 55
Immunogenicity of vaccines combined with ICIs.

ICI, immune checkpoint inhibitors; TAA, tumour-associated antigens.
Conclusions and future directions
Anticancer therapeutic vaccines represent a promising modality to elicit an immunogenic response. Unfortunately, clinical trial results of vaccines as monotherapy have been disappointing. Results from early phase trials where anticancer vaccines are combined with SACT suggest feasibility, with the demonstration of immunogenicity. However, the variation in immunogenicity assays, their uncertain correlation with anti-tumour activity and the timing of their application between studies complicate the interpretation of results. Standardizing and validating the immunogenicity assays would facilitate the clinical development of anticancer vaccines. Furthermore, the heterogeneous anticancer vaccine platforms used difficult the interpretation of the evidence available.
Factors to consider in the choice of combination therapy and areas where further data are required include the choice of chemotherapeutic agent, timing of vaccine administration and avoidance of concomitant steroid use. There is evidence that carboplatin and paclitaxel can reduce immunosuppressive myeloid cells, thereby enhancing T-cell responses. This effect is notably more pronounced 2 weeks after treatment, suggesting this period is an optimal window for vaccine administration.
Some prospective trials are assessing the immunogenicity of antiviral vaccines in patients receiving SACT (e.g. NCT06605625), which may help validate the previously discussed findings. Other trials are comparing the anti-tumour activity of ICIs alone or in combination with anticancer vaccines (e.g. NCT05344209), as well as vaccines administered alone or in combination with ICI (e.g. NCT05232851). These studies will be essential for determining whether the potential synergistic effect translates into clinically significant benefits. To the best of our knowledge, no ongoing trials explicitly address the optimal timing and sequencing of anticancer vaccines in relation to SACT. Future prospective studies examining these factors will be crucial to identifying the most effective strategies for enhancing vaccine efficacy.
