Abstract
Background:
Transarterial chemoembolization (TACE) is a potential conversion therapeutic strategy for unresectable hepatocellular carcinoma (uHCC). However, therapeutic options following conversion therapy are still controversial.
Objectives:
This study aimed to compare the efficacy and safety of surgical resection (SR) and microwave ablation (MWA) after TACE conversion therapy for uHCC.
Design:
A retrospective, multi-institutional study.
Methods:
From June 2008 to October 2022, 8842 consecutive uHCC patients underwent initial TACE at 15 hospitals were identified. Among them, 1348 eligible patients who received TACE conversion therapy were included. The propensity score matching (PSM) was applied to reduce selection bias. To explore the effect of age on conversion therapy, a therapeutic factor analysis with age change was performed. The overall survival (OS) and disease-free survival (DFS) were compared using the Kaplan–Meier method with the log-rank test.
Results:
After PSM 1:1, 542 patients in the MWA group were matched with those in the SR group. SR demonstrated better long-term survival outcomes (median OS, 10.6 vs 5.8 years, HR:1.83, 95% CI: 1.48–2.25, p < 0.001 and median DFS, 3.2 vs 2.5 years, HR: 1.27, 95% CI:1.09–1.49, p = 0.003) than MWA. There was an improvement in the 5-year DFS rate for MWA from 17.1% during 2009–2016 to 37.3% during 2017–2022, becoming comparable to the 40.8% of SR (p = 0.129). When the uHCC patients downstage met Milan criteria, the long-term OS and DFS were comparable between two groups (both, p > 0.05). SR presents an OS advantage over MWA at the age (years) of 45–54 (p = 0.036), 55–65 (p = 0.001), and >65 (p < 0.001), except <45(p = 0.140).
Conclusion:
MWA might be acceptable as an alternative to SR in first-line therapeutic scheme after TACE conversion therapy for uHCC, especially, for the aged <45 years cohorts.
Keywords
Introduction
Hepatocellular carcinoma (HCC) is the sixth most common malignancy and the third leading cause of cancer-related death globally.1,2 Over 70% of patients with HCC are diagnosed at the intermediate- or advanced stage, precluding the chance to obtain curative-intent treatment.3,4 For patients with unresectable HCC (uHCC), growing data have suggested the role of transarterial chemoembolization (TACE), alone or in combination with systemic therapies (including antiangiogenic targeted therapies and immune checkpoint inhibitors (ICIs)),5–8 as a safe and effective conversion therapy option to convert a proportion of uHCC into resectable tumors eligible for subsequent curative-intent treatment.
Surgical resection (SR), ablation, and liver transplantation are curative-intent treatment options after successful conversion therapies. Unfortunately, the organ
Therefore, the aim of this nationwide multicenter study was to investigate and compare the long-term survival outcomes as well as safety profiles between SR and MWA following successful TACE-based downstaging for initially uHCC.
Materials and methods
Study design
This retrospective, multi-institutional study was conducted following the principles of the 1975 Helsinki Declaration. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE). 15
From June 2008 to October 2022, a total of 8184 consecutive patients with HCC beyond Milan criteria who received initial TACE were identified at 15 tertiary hospitals. All HCC patients were diagnosed based on the guidelines of American Association for the Study of Liver Disease (AASLD) or pathologic confirmation via preoperative biopsy.16,17 The TACE protocol was introduced in our previous studies and Supplemental Information E1.1. All patients meeting the Chinese Society of Hepatology for hepatitis B virus (HBV) received anti-HBV treatment as clinically indicated. The inclusion criteria were as follows: (a) aged 18–75 years; (b) Eastern Cooperative Oncology Group (ECOG) performance status <2; (c) Child–Pugh class A or B liver function (d) without macrovascular invasion or extrahepatic metastasis (e) and the uHCC patients underwent conversion therapy. The exclusion criteria were as follows: (a) underwent any treatment before TACE; (b) had HCC combined with other malignancies; (c) received other conversion therapies after TACE, such as stereotactic body radiotherapy (SBRT); and (d) lost to follow-up for more than 6 months.
Assessment of TACE for HCC
Two radiologists (K.L., with 10 years of experience, and S.S.W., with 5 years of experience) were assigned to evaluate the response to TACE based on dynamic enhanced images (e.g., CT and MRI), who were blinded to the TACE procedure. The response included complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD) based on the modified Response Evaluation Criteria in Solid Tumors (mRECIST). 18
Follow-up and endpoints
All patients were regularly followed up after SR or MWA at 1 month and every 3–6 months thereafter with serum AFP and imaging techniques (contrast-enhanced ultrasound, CT, or MRI) until death or the last follow-up date (October 31, 2023). Disease-free survival (DFS) was defined as the time from the initiation of conversion therapy to first-documented tumor recurrence (unequivocal radiological and/or histologic identification of HCC, tumor-in-vein, or distant metastasis as per AASLD) or all-cause death. Advanced-stage recurrence was defined as the presence of macrovascular invasion and/or extrahepatic metastasis at recurrence, and advanced-stage disease-free survival (ASDFS) was defined as the time from the initiation of conversion therapy to advanced-stage recurrence or all-cause death. 19 Overall survival (OS) was defined as the time from the initiation of conversion therapy to death of any cause.
Conversion therapy modalities
Conversion therapy modalities were recommended by a multidisciplinary team according to patients’ performance status, liver function, and tumor characteristics, and response to TACE. The indication of conversion therapy included the following criteria: (1) the interval time between TACE and sequential local therapy was more than 1 month at least; (2) the downstage of HCC from BCLC stage B to A; (3) the patients with intrahepatic nodules after TACE were evaluated as CR or PR for at least 8 weeks; (4) SD to TACE of single huge tumor and shrink for at least 8 weeks; (5) normal coagulation function before SR or MWA (i.e., prothrombin time <15 s, prothrombin activity >40%, and platelet count >50 cells × 109/L); (6) no severe or persistent adverse events (AEs) from TACE; (7) no serious comorbidities, including heart, lung, and renal dysfunction; (8) no severe portal hypertension; (9) R0 resection had to be achieved with sufficient remnant liver volume (⩾40% of the standard liver volume for patients with liver cirrhosis or ⩾30% of the standard liver volume for patients without liver cirrhosis); (10) no tumors with bilateral liver lobes or diffuse distribution .
Figure 1 shows the study flow of eligible patients. Finally, a total of 1348 consecutive patients with uHCC received curative-intent SR or MWA following TACE conversion therapy were reviewed. The SR procedure and type, and MWA procedure and imaging guidance modality are described in detail in Supplemental Information E1.2–1.3. The first- and second-line MTAs and ICIs usually were used after TACE according to tumor progression and preventive treatment management, and the combination schemes were shown in Supplemental Information E1.4.

Flowchart of HCC patients beyond Milan criteria who received TACE conversion therapy.
Definition of terminologies
The terminology of MWA was described according to the Society of Interventional Radiology guidelines. 20 Successful downstaging therapy was defined as a viable intrahepatic tumor burden after TACE that met the Milan criteria (a maximum diameter of one lesion ⩽5 cm, two or three lesions ⩽3 cm, without macrovascular invasion or extrahepatic metastasis). These complications were considered highly likely to be SR or MWA related. Major complications were defined as events that caused substantial morbidity and disability, increased the level of care, led to hospital admission, or substantially prolonged the hospital stay based on the Common Terminology Criteria for Adverse Events v4.0. Assessments, Washington, DC.
Statistical analysis
Statistical analysis was performed using SPSS version 23.0 (IBM Corp., NY, USA) and the “RMS package” using R software version 4.3.0 (http://www.r-project.org/). The quantitative variables are presented as the mean ± SD or median with interquartile range (IQR) and were compared by the Kruskal–Wallis test. The qualitative variables are presented as frequencies and were compared using the χ2 test. To adjust for the selective bias of conversion therapy modality and analyze the risk factors associated with survival outcomes, we collected 16 clinicopathologic variables, which are described in the Supplemental Information E1.5.
The cumulative survival was compared using the Kaplan–Meier method with the log-rank test. Univariate and multivariable analyses of independent prognostic factors were evaluated by means of the forward stepwise Cox regression model to calculate hazard ratios (HRs) and 95% confidence intervals (CIs). We used adjusted HRs to compare survival between the two treatment groups, with 95% CIs used to assess the variation around the estimated risk of events. No formal sample size calculation was performed beforehand, but the large number of events compared to that of variables analyzed at the multivariable Cox regression analysis guaranteed the “ten events per variable” rule of thumb, thus implying sufficient accuracy of the regression estimates. Given the number of patients’ death and recurrence were 795 and 1023, respectively, the statistical power of the multivariate analysis might have been adequate for this study. Therefore, we used different analytical methods to compare survival outcomes between groups. To reduce bias and increase reliability, we applied propensity score matching (PSM) using the nearest-neighbor algorithm to adjust for potentially unbalanced variables in both groups. The calculated propensity scores were then used for case-weight estimation (inverse probability treatment weighting, IPTW). Weights applied to patients treated with SR were the inverse of the propensity score, and weights applied to patients treated with MWA were the inverse of 1 minus the propensity score.
Restricted cubic splines (RCS) were used to visualize the correlation between mortality and age in both SR and MWA groups, adjusted for baseline characteristics. Additive interaction between age strata and conversion therapy modality was examined. To quantify the direct and indirect effects of HCC survival and assess if the correlation between conversion therapy modality and survival could be explained by mediation effects or confounders, multiple mediation analysis was applied.
All tests of significance were two-sided, and results for which p < 0.05 were interpreted as statistically significant.
Results
Baseline characteristics
Baseline patient characteristics are summarized and stratified by two therapeutic modalities before and after PSM in Table 1. Briefly, 1348 HCC patients (age, 54.0 ± 7.5 years; 1148 men) were included. Among them, 619 were assigned to the SR group, and 729 were assigned to the MWA group, respectively. Chronic HBV infection was the predominant chronic liver disease etiology (94.5%, 1258/1348), and cirrhosis was detected in 1218 (90.4%) patients. Among them, the patients in the MWA group were older (mean, 57.2 ± 8.9 years vs 52.0 ± 11.3 years, p < 0.001), smaller tumor diameter (mean, 6.4 ± 1.2 cm vs 7.6 ± 1.1 cm, p < 0.001), and higher CTP B rate (6.9%, vs 3.4%, p = 0.007) than those in the SR group. After PSM 1:1, 542 patients were assigned to the SR group, and 542 patients were assigned to the MWA group, respectively. The baseline characteristics of these patients were matched using the nearest neighbor method with a caliber of 0.1 (Figure S1A).
Baseline characteristics of the patients with HCC who received TACE downstaging therapy.

Data are number of patients; data in parentheses are percentage unless otherwise indicated. Data in the bracket was percent of patients. The quantitative data with mean ± standard deviation or median with IQR were compared by the Mann–Whitney U test. The qualitative data in two groups were compared by using the Chi square test. The variables matched for PSM included age at diagnosis, and CTP, HCC diameter.
The bold content is P value < 0.05.
Chi square test.
Mean ± standard deviation.
Median with IQR.
p Value < 0.05 suggests statistically significant differences.
AFP, α-fetoprotein; ALB, albumin; ALBI, albumin-bilirubin; ALT, alanine aminotransferase; AST, aspartate aminotransferase; BCLC, Barcelona Clinic Liver Cancer; ECOG, Eastern Cooperative Oncology Group; HBV, hepatitis type B viral; HCC, hepatocellular carcinoma; IQR, interquartile range; TA, thermal ablation; TACE, Transarterial chemoembolization; SR, surgical resection; TBIL, total bilirubin; PSM: propensity score match.
Conversion conditions
In this study, 412 patients received conventional TACE (cTACE) and 207 drug-eluting beads (dTACE), respectively, in the SR group, and 523 patients received cTACE and 206 received dTACE, respectively, in the MWA group. There was a statistically significant difference (p = 0.040). Regarding TACE conversion therapy, the median interval between TACE and SR was 2.5 months (IQR, 1.8–5.2 months) and TACE and MWA was 2.7 months (IQR, 1.2–4.8 months), respectively. There was no statistically significant difference (p = 0.683). Before TACE, 781 patients were in Barcelona Clinic Liver Cancer (BCLC) B, and the mean maximum diameter was 6.9 cm ± 2.8 cm. After conversion therapy, all patients downstage to BCLC A, and the mean maximum diameter was 4.3 cm ± 1.2 cm, showing a significant decrease than those before TACE (both, p < 0.001). Among them, 91.3% (565/619) patients obtained R0 resection in the SR group and 93.0% (678/729) patients obtained complete ablation in the MWA group, showing no statistically significant difference (p = 0.238).
Perioperative parameters
The perioperative parameters of TACE conversion therapy are outlined in Supplemental eTable 1. A total of 22.8% of patients were successfully treated in one session, and 77.2% were treated in no less than two sessions in the MWA group. A total of 97.2% of patients were successfully treated in one session, and 2.8% were successfully treated in two sessions in the SR group.
Comparison of survival outcomes between SR and MWA in overall cohorts
The median follow-up duration in the SR group and MWA group were 3.2 years (IQR, 1.6–7.6 years) and 3.8 months (IQR, 2.2–8.5 years), respectively. A significantly longer median OS, and DFS were observed in SR group than those in MWA group (5-years OS rate: 69.3% vs 57.5%, HR: 1.57; 95% CI, 1.30–1.90; p < 0.001; 5-years DFS rate: 44.2% vs 33.9%, HR: 1.06; 95% CI, 1.01–1.34; p = 0.042) and comparable ASDFS were observed between two groups (5-years ASDFS rate: 79.7% vs 83.9%, HR: 0.79; 95% CI, 0.56–1.05; p = 0.093; Figure 2(a)–(c)). After PSM 1:1, a significantly longer median OS, and DFS were observed in the SR group than those in the MWA group (5-year OS rate: 72.4% vs 54.3%, HR: 1.83; 95% CI, 1.48–2.25; p < 0.001; 5-years DFS rate: 46.2% vs 27.6%, HR: 1.27; 95% CI, 1.09–1.49; p = 0.003) and comparable ASDFS were observed between two groups (5-year ASDFS rate: 80.5% vs 82.4%, HR: 0.96; 95% CI, 0.69–1.34; p = 0.813; Figure 2(d)–(f)). Other statistical analysis results were consistent with the above results and are shown in Supplemental eTable 2. Owing to the application of MWA increased and the sample size was comparable, we categorized all cohorts into 2008–2016 years and 2017–2022 years according to the cutoff of December 31, 2016. Further analysis suggests that OS in the MWA group significantly improved in 2017–2022 cohort but comparable in the SR group to that in 2008–2016 years before and after PSM. DFS and ASDFS were observed similar between 2008–2016 years and 2017–2022 years regardless of SR or MWA before and after PSM.

Kaplan–Meier survival curves of overall survival, disease-free survival and advanced-stage disease-free survival before (a–c) and after (d–f) propensity score matching in total cohort.
Comparison of survival outcomes between SR and MWA in 2008–2016 years
The baseline characteristics of uHCC patients stratified by two therapeutic modalities before and after PSM in 2008–2016 years (Supplemental eTable 3). The baseline characteristics of these patients were matched using the nearest neighbor with a caliber of 0.1 (Figure S1B). A significantly longer cumulative OS (5-year OS rate: 66.8% vs 54.6%, HR: 1.61; 95% CI, 1.27–2.06; p < 0.001), and comparable DFS and ASDFS (5-year DFS rate: 41.6% vs 34.1%, HR: 1.17; 95% CI, 0.96–1.42; p = 0.055 and 5-year ASDFS rate: 79.6% vs 84.2%, HR: 0.71; 95% CI, 0.46–1.11; p = 0.132) were observed in the SR group than those in the MWA group (Figure 3(a)–(c)). After PSM 1:1, a significantly longer cumulative OS and DFS were observed in the SR group than those in the MWA group (5-year OS rate: 71.3% vs 52.1%, HR: 1.78; 95% CI, 1.22–2.13; p < 0.001; 5-year DFS rate: 44.6% vs 28.9%, HR: 1.19; 95% CI, 1.12–1.98; p = 0.017) and comparable cumulative ASDFS was observed between the two treatment groups (5-year ASDFS rate: 76.7% vs 76.9%, HR: 0.72; 95% CI, 0.52–1.38; p = 0.860; Figure 3(d)–(f)).Other statistical analysis results were consistent with the above results and are shown in Supplemental eTable 4.

Kaplan–Meier survival curves of overall survival, disease-free survival, and advanced-stage disease-free survival before (a–c) and after (d–f) propensity score matching in 2008–2017 cohort.
Comparison of survival outcomes between SR and MWA in 2017–2022 years
The baseline characteristics of uHCC patients stratified by two therapeutic modalities before and after PSM in 2017–2022 years (Supplemental eTable 5). The baseline characteristics of these patients were matched using the nearest neighbor method with a caliber of 0.1 (Figure S1C). A significantly longer cumulative OS (5-year OS rate: 72.3% vs 60.6%, HR: 1.53; 95% CI, 1.12–2.08; p < 0.007), and comparable cumulative DFS and ASDFS (5-year DFS rate: 43.6% vs 33.1%, HR: 1.16; 95% CI, 0.96–1.43; p = 0.175 and 5-years ASDFS rate: 80.1% vs 85.2%, HR: 0.82; 95% CI, 0.53–1.27; p = 0.100) were observed in the SR group than those in the MWA group (Figure 4(a)–(c)). After PSM 1:1, no significant difference in cumulative OS, DFS, and ASDFS was observed between the two treatment groups (5-year OS rate: 76.8% vs 54.3%, HR: 1.78; 95% CI, 1.43–2.27; p < 0.001; 5-year DFS rate: 48.2% vs 36.5%, HR: 1.21; 95% CI, 0.74-1.95; p = 0.100 and 5-year ASDFS rate: 79.7% vs 83.9%, HR: 0.66; 95% CI, 0.506–1.77; p = 0.900; Figure 4(d)–(f)). Other statistical analysis results were consistent with the above results and are shown in Supplemental eTable 6.

Kaplan–Meier survival curves of overall survival, disease-free survival, and advanced-stage disease-free survival before (a–c) and after (d–f) propensity score matching in 2017–2022 cohort.
Comparison of survival outcomes according to Milan criteria
In the conversion therapy cohort, 42.9% (578/1348) of the uHCC patients achieve successfully downstage met Milan criteria, and the baseline characteristics stratified by two therapeutic modalities before and after PSM are outlined in Supplemental eTable 7. The baseline characteristics of these patients were matched using the nearest neighbor method with a caliber of 0.1 (Figure S1D). Compared with those who fail to downstage met Milan criteria, the patients with HCC met Milan criteria after TACE conversion provided a better survival benefit regarding OS, DFS, and ASDFS before PSM, and similar results were found after PSM (Figure S1E). In a cohort with Milan criteria, no significant difference in cumulative OS, DFS, and ASDFS was observed between the SR group and the MWA group (5-year OS rate: 82.4% vs 79.1%, HR: 1.13; 95% CI, 0.77–0.66; p = 0.543; 5-years DFS rate: 68.4% vs 70.2%, HR: 1.07; 95% CI, 0.83–1.37; p = 0.614 and 5-years ASDFS rate: 91.8% vs 95.5%, HR: 0.60; 95% CI, 0.33–1.10; p = 0.100; Figure 5(a)–(c)). After PSM 1:1, no significant difference in OS, DFS, and ASDFS was observed between the two treatment groups (5-year OS rate: 89.3% vs 84.4%, HR: 1.22; 95% CI: 0.78–1.89; p = 0.381; 5-year DFS rate: 69.2% vs 71.4%, HR: 0.97; 95% CI: 0.71–1.32; p = 0.855 and 5-year ASDFS rate: 92.0% vs 96.9%, HR: 0.46; 95% CI: 0.21–1.01; p = 0.053; Figure 5(d)–(f)). SR yields a better OS and DFS outperforming MWA in the cohort beyond Milan criteria (Figure 5(g)–(l)).
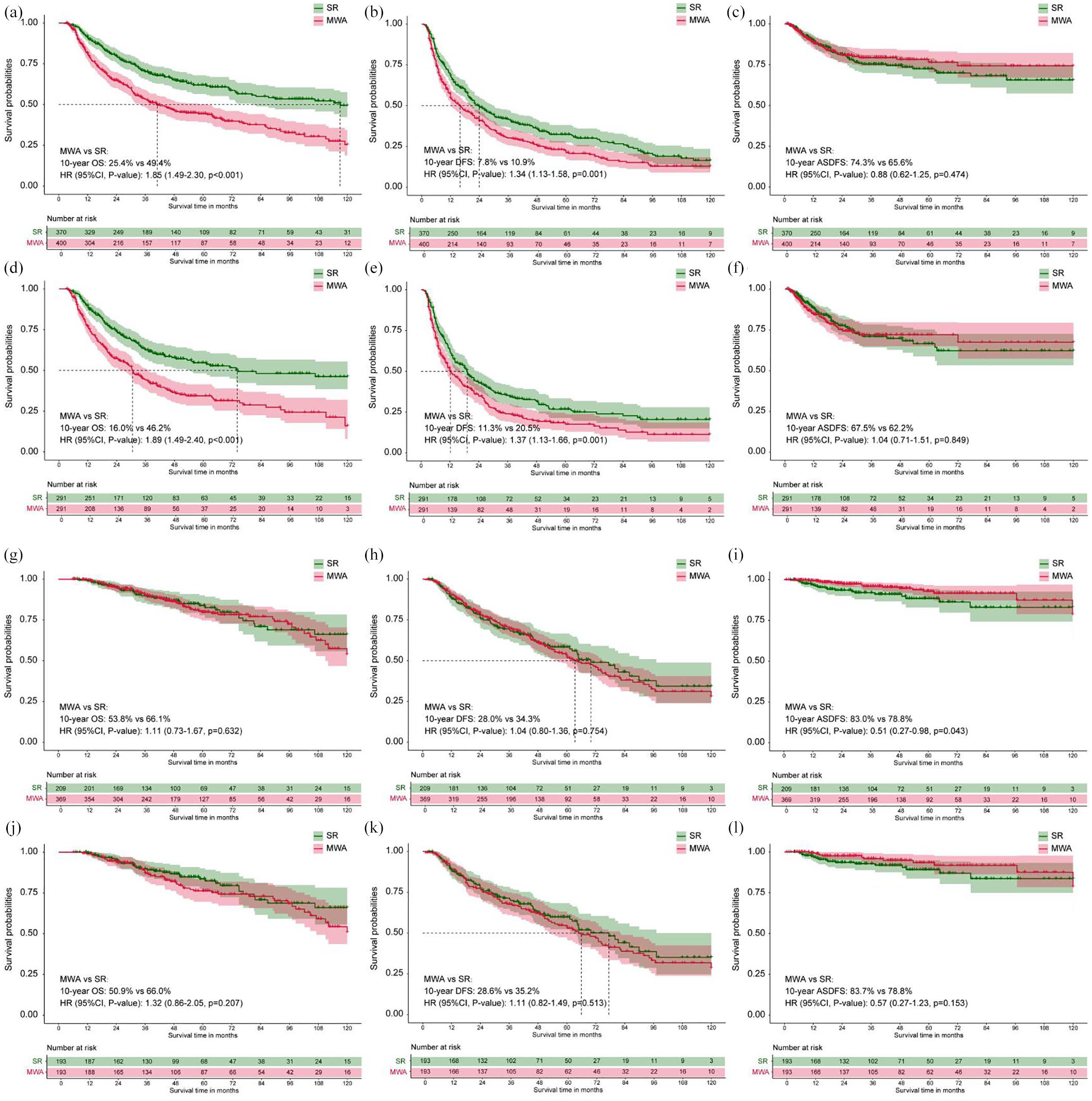
Kaplan–Meier survival curves of overall survival, disease-free survival and advanced-stage disease-free survival before (a–c) and after (d–f) propensity score matching in patients with HCC beyond Milan criteria after TACE conversion therapy. Kaplan–Meier survival curves of overall survival, disease-free survival, and advanced-stage disease-free survival before (g–i) and after (l–k) propensity score matching in patients with HCC met Milan criteria after TACE conversion therapy.
Age-dependent analysis and risk factors
To investigate whether survival outcomes of uHCC patients who underwent conversion therapy may change with age, the RCS was plotted and showed that the disparity of two conversion therapy modalities of adjusted long-term OS extended to age <45 years, 45–55 years, 55–65 years, and >65 years. Baseline characteristics of each age strata before and after PSM were shown in Supplemental eTables 8 to 9. The baseline characteristics of these patients were matched using the nearest neighbor method with a caliber of 0.1 in four age subgroups (Figure S1F-I). SR provided OS advantage over MWA started to appear at age <45 (HR: 1.84, 95% CI 1.29–2.62; p = 0.001, 5-year OS 66.3% vs 44.2%, 10-year OS 46.7% vs 34.5%) (Figure 6(a)), persisted at age 55–65 (HR: 1.87, 95% CI, 1.34–2.69, 5-year OS 78.7% vs 61.5%, 10-year OS 32.3% vs 28.3%, p = 0.001; Figure 6(b)) and age >65 (HR: 1.76, 95% CI 1.03–3.01; p = 0.037, 5-year OS 75.9% vs 57.2%,10-year OS 73.7% vs 41.4%; Figure 6(c)). These disappeared at the end of age strata (45–54 age groups, HR: 1.25, 95% CI, 0.86–1.81, 5-year OS 78.7% vs 61.5%, 10-year OS 32.3% vs 28.3%, p = 0.244) (Figure 6(d)). After PSM, the patients with <45 years present comparable OS between two groups (HR: 1.25, 95% CI, 0.86–1.81, 5-year OS 78.7% vs 61.5%, 10-year OS 32.3% vs 28.3%, p = 0.244, Figure 6(e)), but the patients with 45–54, 55–65 and >65 years have a better OS in SR than those in MWA (Figure 6(f)–(h)). Figure 6(i) shows the association between the protective effect of SR (SR-to-MWA: HR of overall mortality) and age-stratified every 10 years from age start to 45 years. SR’s protective effect reached a peak at age >65 years for OS (HR:1.84, 95% CI 1.29–2.62; p = 0.001). Consistent trends were also found in the 2008–2016 cohort and 2017–2022 cohort (Figure 6(j)–(k)). The interaction between age strata and conversion therapy modality is shown in Supplemental eTable 10.

Kaplan–Meier survival curves of overall survival of <45 years, 45–55 years, 56–65 years, and >65 years before (a–d) and after (e–h) propensity score matching in the total cohort. The association between the protective effect of SR-to-MWA and age stratified every 10 years from age start to 45 years (i–k).
The univariate and multivariate analysis demonstrated independent risk factors for OS, DFS and ASDFS were shown in Supplemental eTables 11 to 13 in total cohort. The forest plots of the subgroup analyses for OS, and DFS based on the above-mentioned important variables are shown in Figure S6.
Treatment-related complications, intervention time, and cost
TACE related AEs grade 1–2 and 3–4 were similar after PSM in both groups (p = 0.675, 0.705; Table 2). Liver dysfunction was the most common minor complication, which included the presence of mild ascites and increased transaminase levels. During follow-up, no treatment-related deaths occurred in either group. The incidence of complications after SR or MWA are shown in Table 2. The major complication rate was 7.1% (44 of 619 patients) in the SR group and 6.4% (47 of 729 patients) in the MWA group, showing no significant difference (p = 0.574). The major complication rate was 6.7% in the SR group and 6.4% in the MWA group, and these two groups were not significantly different (p = 0.705) after PSM.
The complications comparison between SR and MWA after TACE conversion therapy.

MWA, microwave ablation; ND, no data; SR, surgical resection.
Data in bracket was percent of patients. The data in two groups were compared by using the Chi square test or Fisher’s exact test.
The mean surgical duration, blood loss, and median hospitalization duration and cost in the SR group were 3.3 h ± 0.5, 24 mL ± 2.2, 11 days, and 42,371 yuan, which were significantly higher than those (1.2 h ± 0.2, 5 mL ± 0.2, 4 days and 23,254 yuan) in the MWA group (all, p < 0.001; Table 3).
Therapeutic parameters comparison of uHCC patients received TACE conversion therapy.

Data are number of patients; data in parentheses are percentage unless otherwise indicated and data in bracket was percent of patients.
The qualitative data using median with IQR in two groups were compared by using the Chi-square test.
p value < 0.05 suggests statistically significant differences.
CR, complete response; HCC, hepatocellular carcinoma; MWA, microwave ablation; IQR, interquartile range; PR, partial response; PSM, propensity score match; SD, stable disease; SR, surgical resection.
Discussion
Previously, conversion therapy was defined as the conversion of uHCCs into resectable HCCs, but uHCCs also can be converted to HCCs that are acceptable for ablation in current with the continuous progress of imaging-guided technology. 21 In China, the Chinese Expert Consensus on Conversion Therapy for Hepatocellular Carcinoma suggests that conversion therapy is an intermediate goal for the treatment of uHCC, while long-term survival is the ultimate goal. 22 SR and MWA were the first-line curative therapies for early HCC according to international guidelines. However, SR remains a primary therapeutic modality following conversion therapy, which lacks powerful evidence for alternative treatments. In this retrospective study, we utilized a multi-center design, resulting in greater real-world heterogeneity than a single-center design. This study aimed to provide evidence-based medicine for the minimally invasive treatment after TACE conversion therapy of uHCC.
As early as 1993, Sitzmann and Abrams were the first to report cases of unresectable cancer that were converted to resectable cancer after radiochemotherapy in 14 patients. 23 In addition, Majno et al. proposed conversion therapy before LT and achieved good survival outcomes in 1997. 24 Subsequently, an increasing number of studies have explored the safety and effectiveness of conversion therapy for patients with unresectable carcinoma.6,14,25–27 Recently, the IMbrave-150 study showed encouraging results with the combination of atezolizumab and bevacizumab in terms of the ORR (27.3% by RECIST 1.1 and 33.2% by mRECIST) of advanced HCC, providing a promising path for further expansion of conversion therapy. 28 However, this conversion rate remains unsatisfactory, especially among patients with high tumor burden. Over the past decade, intra-arterial therapies become promising options for conversion therapy of uHCC. Shi Feng et al. demonstrated that patients with HCC beyond Milan criteria underwent MWA following TACE downstaging therapy had a comparable survival with those who underwent MWA alone for HCC met the Milan criteria.14,27 Deng et al. showed that HAIC and TACE as conversion therapies for single huge HCC, providing physicians a new strategy.
In this study, a total of 16.5% (1348/8184) of patients with uHCC beyond Milan criteria who received initial TACE conversion therapy were included. Furthermore, 40.4% (545/1349) uHCC patients obtained a successful downstaging therapy that met Milan criteria in all received conversion therapy. The mean maximum tumor diameter was 6.9 cm before TACE, which was reduced to 4.3 cm after multi-cycle TACE (median 2 TACE sessions). Herein, a low tumor burden was regarded as a favorable factor for long-term survival in previous studies. The CR and PR rates were comparable between two groups, but the number of patients who achieved successfully downstage met Milan criteria in the MWA group was more significant than that in the SR group. The reason may be the mean maximum tumor diameter in the MWA group was smaller significantly than that in the SR group (6.3 cm vs 7.6 cm). MWA was associated with less trauma, faster recovery, shorter hospitalization, and lower costs compared with SR. These advantages could lead to the use of another reliable treatment option for seniors or patients who are intolerant to SR. In regarding safety profile, the TACE-induced AEs (3–4 grade, 87 of 542 patients in the SR group and 82 of 542 in the MWA group, p = 0.675) and SR/MWA-related complications were comparable (major complications, 47 of 542 patients in the SR group and 47 of 542 in the MWA group, p = 0.705), suggesting comparable safety of the two local-region treatment after TACE.
With more than 3 years of median follow-up time, we compared long-term survival outcomes between two therapeutic modalities (MWA vs SR) after TACE conversion therapy in the total cohort, 2008–2016 years cohort and 2017–2022 years cohort, respectively. For different cohorts, we now summarize the results and analyze the reasons as follows: (1) better significantly long-term OS and DFS were found in the SR group than those in the MWA group before and after PSM, indicating SR following conversion therapy for uHCC is a better choice outperforming MWA in those who beyond Milan criteria; (2) a better 5-year cumulative OS rate was found in the MWA group in 2017–2022 years than before. The reason may be the number of applications of ICI and MAT treatments has significantly increased in the past 5 years, and previous studies have confirmed that MWA combined with ICIs and MATs for treatment in HCC can improve significantly than MWA alone29–31; (3) the similar results were observed in 2008–2016 years cohort with those in total cohort, but a comparable DFS and ASDFS were observed between SR and MWA group in 2017–2022 years cohort before and after PSM, indicating that MWA-induced coagulation necrosis is more precise and reasonable than before, mainly due to the continuous emergence of auxiliary technologies, 32 including artificial fluid infusion, three-dimensional ablation planning systems, and multi-model image fusion navigation; (4) The comparable long-term OS and DFS were observed between two groups before and PSM in the cohorts met Milan criteria, but a better cumulative OS and DFS were observed in the SR group than those in the MWA group in the cohorts beyond Milan criteria. The reason may be the resection boundary obtained by the liver segment or hemihepatectomy should far exceed the safe margin of MWA. The results also indicated that the tumor burden after TACE can be reduced to within Milan criteria determines the choice of SR and MWA following conversion therapy. We also used IPTW and multivariate analysis adjusted statistical methodologies to further confirm the above results that reinforce the robustness of our findings.
By 2030, China is about to enter an aging society, therefore, the choice of treatment for HCC in different age subgroups is also one of the clinical issues that we focus on. Given the MWA with minimally invasive characteristics, the elderly HCC population seems to be more willing to accept this therapeutic modality. There have been many previous reports on MWA treatment for HCC in the elderly population, including a well-designed population-based study and a prospective study. Our results revealed a typical age-dependent patients with uHCC received SR yield OS advantage appearing at 45–54, 55–65, and >65 age years, especially, peaking at >65 years. However, the patients aged <45 years are a special population because SR provided a comparable OS compared with MWA. The reason may be that MWA is a minimally invasive interventional technique that can be performed repeatedly. Younger populations have better physical tolerance and can receive more MWA treatments. Even if the tumor recurs, eradication treatment can be carried out through MWA. These results suggest that MWA may be a therapeutic modality that can replace SR following TACE conversion therapy for uHCC patients aged 45–54 years, considering its minimally invasive characteristics. According to the multivariate Cox regression analyses, tumor number, AFP and TACE combined with targeted immunotherapy were crucial risk factors for the long-term OS, consistent with the findings of previous studies. Interestingly, targeted immunotherapy plays an important role in improving long-term survival after conversion therapy of uHCC. TACE embolization causes tumor hypoxia necrosis, upregulates the expression of PDL1, and exposes tumor cells to extreme hypoxia or even anaerobic environments, releasing free VEGF. ICI enhances the anti-tumor immune response of T cells by blocking the PD-L1/PD-1 pathway, while MAT is a humanized monoclonal antibody that specifically binds to VEGF, clearing free VEGF and promoting the transformation of the tumor microenvironment from immunosuppression to immune permissiveness. 33 Therefore, the combination of the three has a potential synergistic effect.
Although our study has many strengths, including a large cohort and multi-center study design, it also has several limitations. First, although the PSM and IPTW were used to balance the variables, there were still inevitable biases due to the retrospective study nature and sample loss after PSM. Second, this study enrolled patients from several hospitals across our country, with differences in the TACE application scheme and duration of use of chemotherapy drugs across hospitals. These factors may have affected the final survival outcomes. Third, there was no information about the location of tumors in the hilar or subcapsular regions, so a direct comparison of tumors in these locations could not be performed between the two groups. Fourth, in this study, we recruited patients in China with mostly large HCC and HBV infections as the predominant etiologies. It remains to be elucidated whether the results can be widely applied in Western countries, where the majority of patients have a low tumor burden or alcoholic liver cirrhosis as the predominant etiology. In the future, prospective clinical trials should be conducted to further test the reliability and reproducibility of the study results. The role of immunotherapy in enhancing survival post-conversion therapy for HCC, along with anti-VEGF treatment deserves further exploration and forward-looking research in the future.
Conclusion
In summary, we reported the long-term outcomes, efficacy, and safety of patients with HCC beyond Milan criteria who received TACE conversion therapy. The findings indicated that the TACE conversion therapy strategy is feasible and safe for uHCC. SR provided better long-term survival than MWA after TACE, but comparable long-term survival was observed for uHCC patients after TACE who met the Milan criteria between the two groups. Notably, MWA technology has made significant progress and is now comparable to SR in terms of DFS over the past 5 years, which may present an alternative option as a minimally invasive treatment when facing technical challenges with SR implementation. Age is a crucial factor that deserves special attention, as it is closely related to the selection of subsequent therapeutic modalities for TACE conversion therapy. Our study results found that patients under 45 years old who underwent SR had better long-term OS than those who underwent MWA, suggesting that this age group should be considered a priority before TACE conversion therapy for uHCC.
Supplemental Material
sj-doc-1-tam-10.1177_17588359251316665 – Supplemental material for Local-region treatment comparison following conversion therapy of hepatocellular carcinoma: a period and age-dependent analysis
Supplemental material, sj-doc-1-tam-10.1177_17588359251316665 for Local-region treatment comparison following conversion therapy of hepatocellular carcinoma: a period and age-dependent analysis by Jiahui Hu, Shu Zhao, Mengxuan Zuo, Chun Hui Li, Wang Yao, Xinyu Yang, WeiWei Xing and Peng Song in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251316665 – Supplemental material for Local-region treatment comparison following conversion therapy of hepatocellular carcinoma: a period and age-dependent analysis
Supplemental material, sj-docx-2-tam-10.1177_17588359251316665 for Local-region treatment comparison following conversion therapy of hepatocellular carcinoma: a period and age-dependent analysis by Jiahui Hu, Shu Zhao, Mengxuan Zuo, Chun Hui Li, Wang Yao, Xinyu Yang, WeiWei Xing and Peng Song in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
