Abstract
Background:
There is still no noninvasive, automated, and accurate model for guiding physicians in the decision-making of transarterial chemoembolization combined with microwave ablation (TACE-MWA) in intermediate-stage hepatocellular carcinoma (HCC).
Objectives:
To develop a prognostic score based on the tumor burden and radiomic features for the prediction of the long-term survival of patients with intermediate-stage HCC after TACE-MWA.
Methods:
From June 2008 to October 2022, a total of 2189 consecutive patients from seven tertiary-care hospitals with intermediate-stage HCC who received initial TACE combined with MWA were enrolled. Among them, 2189 were divided into training cohort (N = 1753), and internal test cohort (N = 436) in a single center, and 316 patients were assigned to external test cohort in another 6 centers. A prognostic scoring system was constructed using tumor burden and radiologic features (TBR) and compared with conventional predicting systems.
Results:
In training cohort, multivariate Cox regression analysis suggested that tumor burden (hazard ratio (HR), 0.693; 95% confidence interval (CI): 0.505, 0.814; 1 point per 1.0 increase, p = 0.024), radiologic features (HR, 0.349; 95% CI: 0.236, 0.517; p < 0.001), and alpha-fetoprotein (HR, 1.629; 95% CI: 1.280, 2.073; p < 0.001) were independent prognostic factors for OS. A prognostic model that comprises TBR was built, which showed significantly higher AUC values than other clinical stagings in all three cohorts. Moreover, the TBR score provided greater net benefit across the range of reasonable threshold probabilities than other models. Based on cutoff values of 32 and 74 centiles of the TBR score, the cohort was divided into low-, middle-, and high-risk strata, which provide consistent performance in survival discrimination across different patient subgroups.
Conclusion:
The TBR score serves as an efficient instrument for risk stratification, guiding the course of adjuvant targeted and immunotherapies for HCC patients undergoing TACE-MWA combined treatment.
Design:
A retrospective, multi-institutional study.
Keywords
Introduction
Hepatocellular carcinoma (HCC) accounts for 75%–85% of the newly diagnosed liver cancer, and is the fourth leading cause of cancer death globally, with a dismal prognosis.1,2 Currently, transarterial chemoembolization (TACE) is the only standard of care for intermediate-stage HCC according to the guidelines of the American Association for the Study of Liver Disease (AASLD) 3 and the European Association for the Study of Liver. 4
Barcelona Clinic Liver Cancer (BCLC) stage B HCC is a heterogeneous group of diseases and there are many studies published in recent years shown TACE combined with microwave ablation (TACE-MWA) can provide superior disease control and long-term survival compared to TACE alone for the selected proportion of patients. 5 However, the best candidates for TACE-MWA remained unknown. A robust scoring system that can provide prognosis prediction and enable precise selection of patients for receiving TACE-MWA treatment is warranted.
Previously, many scholars provided various criteria of risk grade for intermediate-stage HCC receiving TACE, such as 4-and-7 criteria and 6–12 criteria.6,7 Notably, hepatoma arterial-embolization prognostic (HAP) score,8,9 derived from HCC patients receiving TACE, is the current most accurate predictor of survival. However, the value of these models, which were not designed specifically for patients receiving TACE-MWA treatment, remains unknown.
The updated 2022 BCLC guidelines propose that infiltrative HCC, as a special radiologic pattern, should be treated by combining systematic therapy such as targeted therapy, according to the therapeutic scheme of advanced HCC. 10 Huang et al. classified HCC into four subtypes according to the radiologic features. The results of their study showed patients with infiltrative HCC who received TACE have the lowest objective response rate in the four types of HCC. 11 Our previous study showed hepatic arterial infusion chemotherapy is a more suitable treatment method compared to TACE for infiltrative HCCs. Hence, an ideal prognostic model for HCC patients who underwent TACE should also include 12 important radiologic features in addition to the known factors, such as tumor number, tumor diameter, liver function, and physical performance.
Herein, we proposed a hypothesis that supplements the previous tumor burden-based prognostic model with radiological characteristics as a special variable to respond to the updated 2022 BCLC guideline and developed a simple and easy tool for enhancing personalized TACE-MWA management in intermediate-stage HCC patients, thereby improving long-term outcomes.
Materials and methods
This analysis was reported according to the Transparent Reporting of a Multivariable Prediction Model for Individual Prognosis or Diagnosis guidelines. 13
Study population
From June 2008 to October 2022, a total of 3494 consecutive patients with unresectable hepatocellular carcinoma (uHCC) who received initial TACE combined with sequential MWA were retrospectively reviewed from seven tertiary-care hospitals. The diagnosis of HCC is established either pathologically or based on the clinical criteria used by the AASLD guideline. 3 The target population met the baseline criteria including good performance status, preserved liver function, and without vascular invasion or extrahepatic spread as “recommended” or “ideal” candidates of TACE-MWA, and the patients with intermediate-stage HCC were identified according to the criteria of BCLC staging systems. The inclusion criteria were as follows: (a) age 18–75 years; (b) Eastern Cooperative Oncology Group (ECOG) performance status <2; (c) Child-Pugh class A or B liver function; (d) tumor number more than three; and (e) with inadequate computer tomography (CT) or magnetic resonance imaging (MRI) quality for assessment in tumor burden, which is defined as the sum of tumor diameter and number, and radiological characteristics. Patients were excluded if they (a) received additional systematic treatment before TACE; (b) had any current or prior malignancies other than HCC; (c) decompensated liver cirrhosis; and (d) had missing image information. Patient inclusion and exclusion are illustrated in Figure 1.

The enrollment pathway of the patients with intermediate-stage hepatocellular carcinoma who received transarterial chemoembolization combined with microwave ablation.
The HCC patients in Sun Yat-sen University Cancer Center were randomly assigned to training or internal test cohorts using computer-generated randomized numbers. During the same period, the HCC patients in other six tertiary-care hospitals were assigned to external test cohorts. The data sources of the seven hospitals are shown in Supplemental Table 1. The largest diameter of tumor nodules (hereinafter measured in centimeters), tumor number, and radiological characteristics were evaluated by two independent radiologists (Q.F.C. and P.W.) using either dynamic contrast-enhanced CT or MRI. 11 Clinical information (e.g., demographics, etiologies of chronic liver diseases) and laboratory results (e.g., alpha-fetoprotein (AFP) and platelet count) were collected at baseline as well as within 2 weeks prior to TACE.
Treatment procedure
The TACE procedure has been described in our previous reports.14–16 Two interventional radiologists (Q.C. and L.S.) with at least 8 years of experience inserted an artery sheath catheter into the femoral artery using the modified Seldinger technique under the guidance of digital subtraction angiography (Philips, type FD 20 1250 mA, Amsterdam, Netherlands). Sequentially, a 5-Fr Yashiro catheter (Terumo, Tokyo, Japan) was advanced into the celiac trunk and superior mesenteric artery to assess the feeding hepatic artery, and the feeding artery was selected or super-selected whenever possible. An emulsion of mixtures of 10–20 ml lipiodol, 30–50 mg platinum drugs, and 20–40 mg epirubicin was injected slowly until the offending vessel occluded. If necessary, embolization using gel foam mixed with contrast medium was injected to reduce the residual blood flow until there was no longer any tumor staining after repeat angiography. A repeat TACE procedure is scheduled at an interval of 1–2 months upon the demonstration of intrahepatic viable residual tumors by dynamic contrast-enhanced CT or MRI in patients with favorable performance status and liver function.
MWA would be recommended if targeted tumors were regarded as achievement of complete ablation (i.e., well-preserved liver function with the anticipation that complete ablation could be achieved successfully) by liver expert panels and objective response to TACE with one or more than one session was achieved for at least 4 weeks. MWA was performed by two interventional radiologists (C.A. and L.J.C.) under moderate sedation and local anesthesia. After the application of local anesthesia with 1% lidocaine (Yiyou, Beijing, China), the microwave antenna was percutaneously inserted into the tumor and placed in the desired location under CT guidance. A power output of 50–60 W for 5–10 min was routinely used during MWA. After the puncture operation, intravenous anesthesia with a combination of propofol (Diprivan; Zeneca Pharmaceuticals, Wilmington, DE, USA) and ketamine (Shuanghe Pharmaceuticals, Beijing, China) was administered via the peripheral vein. If the maximum diameter of residual active tumors is less than 3 cm, one session was permitted. If the maximum diameter of residual active tumors is no less than 3 cm, several sessions are performed repeatedly until the complete margin is confirmed with CT or MRI.
Follow-up and endpoints
All patients were regularly followed up after TACE at 1 month and every 3–6 months thereafter with serum AFP and imaging techniques (contrast-enhanced ultrasound, CT, or MRI) until death or the last follow-up date (October 31, 2023). Overall survival (OS), defined as the time from the initiation of TACE to death of any cause, was the primary endpoint.
Statistical analysis
Inter-rater agreement between the two reviewers was assessed with the intraclass correlation coefficient for tumor size and with Cohen’s kappa value for binary imaging features. No formal sample size calculation was performed beforehand, but the large number of training cohort death events compared to that of variables analyzed at the multivariable Cox regression analysis guaranteed the “10 events per variable” rule of thumb, thus implying sufficient accuracy of the regression estimates.
The multivariable Cox regression analyses with the backward stepwise method were performed to develop the prognostic nomogram.
Based on the internal and external testing cohort, model discrimination was measured with the concordance index (C-index) and time-dependent area under the curve (td-AUC) from 12 to 60 months. Akaike information criterion (AIC) was also calculated to compare the loss of information for different models. Bootstrapping with 1000 samples was used for model tests in subgroups with sample sizes less than 500. Model calibration was evaluated by the calibration plot and the clinical utility was evaluated with decision curve analysis (DCA).
The cumulative survival outcomes of different risk groups were estimated using the Kaplan–Meier method and compared with the log-rank test, with subgroup analyses performed to adjust for known prognostic factors. To classify patients into high-, middle-, and low-risk groups, the optimal thresholds of the nomograms were determined with X-tile software (version 3.6.1; Yale University School of Medicine, New Haven, CT, USA). The performances of the prognostic nomogram were compared with major prognostic models including 4-and-7 criteria, 17 6–12 criteria, 7 modified HAP-III (mHAP-III) scores, 9 intermediate HCC (BCLC-B sub-classification, 18 AFP, 8 and albumin bilirubin (ALBI) score. 19
Statistical analysis was performed using SPSS version 23.0 (IBM Corp., NewYork, NY, USA) and the RMS package of R software version 3.5.1 (http://www.r-project.org/). All tests of significance were two-sided, and a p value < 0.05 was considered indicative of statistical significance.
Results
Baseline characteristics
In this study, the median follow-up was 28.9 months (interquartile range (IQR) 13.5–45.3 months), 27.5 months (IQR 12.8–39.0 months), and 28.3 months (IQR 16.3–39.5 months) in the training cohort, internal test cohort, and external test cohort, respectively. Finally, a total of 2189 patients from SYSUCC with intermediate-stage HCC who underwent TACE-MWA were included and randomly divided into the training cohort (N = 1753) and internal test cohort (N = 436). Meanwhile, a total of 316 patients with intermediate-stage HCC who underwent TACE-MWA were included in other six hospitals as an external test cohort (N = 316). As shown in Table 1 which details the baseline characteristics of the patients with intermediate-stage HCC and therapeutic parameters, several clinical variables including ECOG PS score, etiology, cirrhosis, ascites, radiological characteristics, DCP, and CRP were significant differences between the three cohorts, while the other variables showed no significant differences in distribution. Chronic hepatitis B virus (HBV) infection stood as the predominant factor in the etiology of HCC (95.6%, 2396/2505).
The baseline characteristics of intermediate-stage HCC patients who underwent TACE in three cohorts.
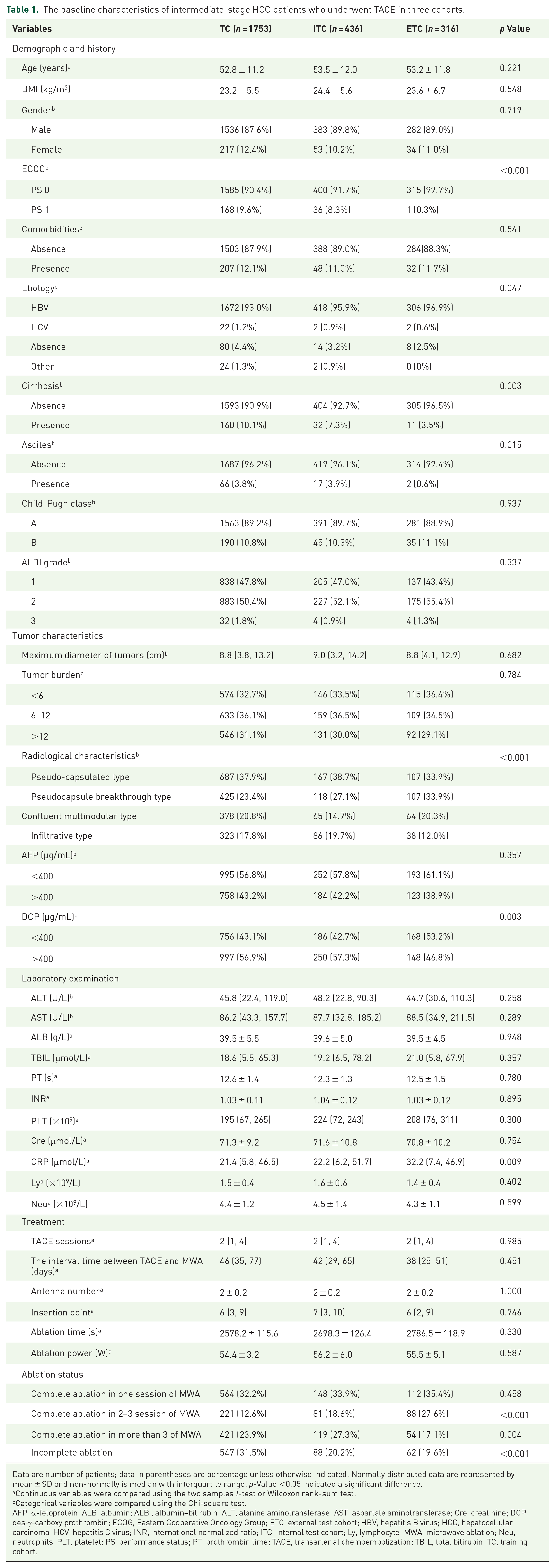
Data are number of patients; data in parentheses are percentage unless otherwise indicated. Normally distributed data are represented by mean ± SD and non-normally is median with interquartile range. p-Value <0.05 indicated a significant difference.
Continuous variables were compared using the two samples t-test or Wilcoxon rank-sum test.
Categorical variables were compared using the Chi-square test.
AFP, α-fetoprotein; ALB, albumin; ALBI, albumin–bilirubin; ALT, alanine aminotransferase; AST, aspartate aminotransferase; Cre, creatinine; DCP, des-γ-carboxy prothrombin; ECOG, Eastern Cooperative Oncology Group; ETC, external test cohort; HBV, hepatitis B virus; HCC, hepatocellular carcinoma; HCV, hepatitis C virus; INR, international normalized ratio; ITC, internal test cohort; Ly, lymphocyte; MWA, microwave ablation; Neu, neutrophils; PLT, platelet; PS, performance status; PT, prothrombin time; TACE, transarterial chemoembolization; TBIL, total bilirubin; TC, training cohort.
Radiologic features assessment
The radiologic features of the four types of HCC are summarized and shown in Figure 2(a). Radiologic features were classified into two types: non-infiltrative type and infiltrative type. Furthermore, the non-infiltrative type was divided into pseudo-capsulated type, pseudocapsule breakthrough type, and confluent multinodular type. Inter-rater disagreements were resolved by a senior radiologist at each participating center who had over 20 years of experience in liver imaging. Cohen’s kappa values for pseudo-capsulated type, pseudocapsule breakthrough type, multinodular fusion type, and infiltrative type were 1.00, 0.95, 0.86, and 0.88, respectively. The inconsistent results were ruled by a senior experienced radiologist. The correlation of tumor burden with radiologic features based on Wang et al. 7 reported is summarized in Supplemental Figure 1.

The development of a nomogram integrates tumor imaging features and tumor burden. (a) Four types of radiologic features for HCC including pseudo-capsulated type, pseudocapsule breakthrough type, multinodular fusion type, and infiltrative type. (b) The nomogram shows the assessment of 1-, 3-, and 5-year OS of patients with intermediate-stage HCC who received transarterial chemoembolization combined with microwave ablation; calibration plot of the nomogram. Calibration curves of the nomogram at 1-, 3-, and 5-year OS show a good correlation between assessed and observed OS. Calibration curves were close to the 45° line. (c) Training cohort. (d) Internal test cohort. (e) External test cohort.
Univariate and multivariate analyses
In the training cohort, univariate analyses of OS are shown in Supplemental Table 2. The multivariate Cox regression analysis suggested that tumor burden (hazard ratio (HR), 0.693; 95% confidence interval (CI): 0.505, 0.814; 1 point per 1.0 increase, p = 0.024), radiologic features (HR, 0.349; 95% CI: 0.236, 0.517; p < 0.001), and AFP (HR, 1.629; 95% CI: 1.280, 2.073; p < 0.001) were independent prognostic factors for OS. The coefficients of variables derived from Cox regression will be considered for model development (Supplemental Table 2).
Development of the prognostic model
Based on the above-mentioned independent risk factors, we developed two possible models as follows: model 1 named the TBR model comprised of Tumor Burden and Radiologic features; model 2, tumor burden, radiologic features, and AFP were included. Two independently associated risk factors were used to develop model 1 as a visual nomogram (Figure 2(b)), described by the formula:

where infiltrative type is the reference group.
On this basis, three independently associated risk factors were used to develop the model 2, described by the formula:

where infiltrative type is the reference group.
The td-AUC value, likelihood ratio χ2, C-index, and AIC of the possible models are shown in Table 2. Compared with model 1, model 2 (with the addition of AFP level) showed only slight improvement regarding td-AUC values and C-indices, with no significant statistical differences (p = 0.534). Given this situation and the purpose of developing an easy-to-use bedside stratification tool, model 1 was selected as the final model. Based on these findings, a nomogram for individual patients’ prognostic risk stratification who received TACE-MWA was developed. The 1-, 3-, and 5-year OS probability and estimated median OS of individual patients with intermediate-stage HCC could be predicted before the TACE procedure with the sum of tumor burden and radiologic features. Calibration curves plotted for 1-, 3-, and 5-year OS show that the nomograms were well calibrated in three cohorts, respectively (Figure 2(c)–(e)).
Comparison of the performance and discriminative ability of two possible models in three cohorts.
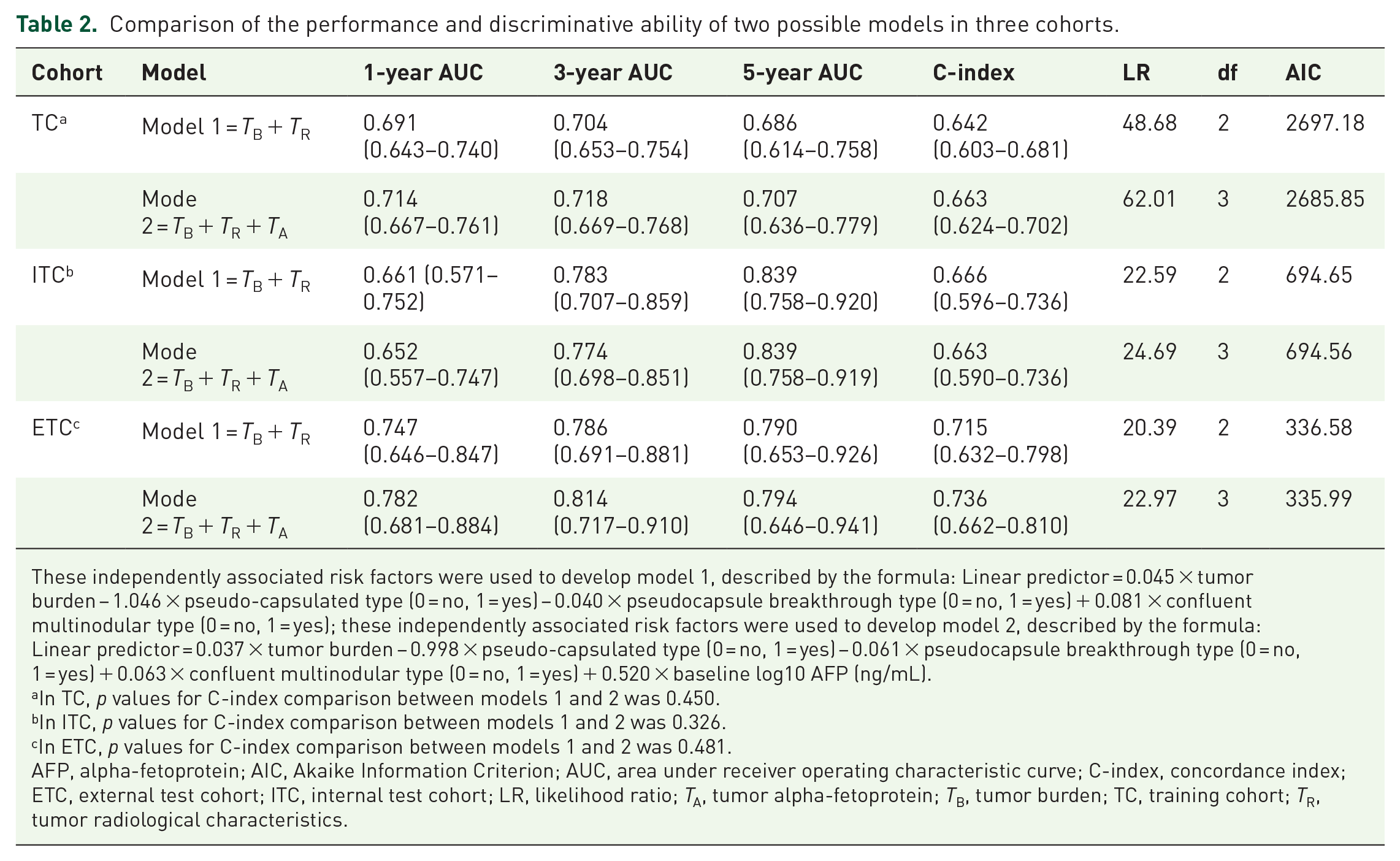
These independently associated risk factors were used to develop model 1, described by the formula: Linear predictor = 0.045 × tumor burden − 1.046 × pseudo-capsulated type (0 = no, 1 = yes) − 0.040 × pseudocapsule breakthrough type (0 = no, 1 = yes) + 0.081 × confluent multinodular type (0 = no, 1 = yes); these independently associated risk factors were used to develop model 2, described by the formula: Linear predictor = 0.037 × tumor burden − 0.998 × pseudo-capsulated type (0 = no, 1 = yes) − 0.061 × pseudocapsule breakthrough type (0 = no, 1 = yes) + 0.063 × confluent multinodular type (0 = no, 1 = yes) + 0.520 × baseline log10 AFP (ng/mL).
In TC, p values for C-index comparison between models 1 and 2 was 0.450.
In ITC, p values for C-index comparison between models 1 and 2 was 0.326.
In ETC, p values for C-index comparison between models 1 and 2 was 0.481.
AFP, alpha-fetoprotein; AIC, Akaike Information Criterion; AUC, area under receiver operating characteristic curve; C-index, concordance index; ETC, external test cohort; ITC, internal test cohort; LR, likelihood ratio; TA, tumor alpha-fetoprotein; TB, tumor burden; TC, training cohort; TR, tumor radiological characteristics.
Assessment and comparison of the performance and discrimination of the current model and other models
The performance and discrimination of the TBR score and other tumor-burden-based models (up-to-7 criteria and 6–12), as well as models involving other parameters (AFP level, mHAP-III score, BCLC-B sub-classification, and ALBI grade), were compared (Table 3). The 1-, 3-, and 5-year AUC values (Figure 3(a)–(c)) and C indices of the TBR score were higher than other models in three cohorts, respectively. Moreover, DCA demonstrated that the TBR score provided greater net benefit across the range of reasonable threshold probabilities than other models in three cohorts (Figure 3(d)–(f)), respectively. Plots of Kaplan-Meier curve (KM) estimates based on up-to-7 criteria, 6–12, mHAP-III score, BCLC-B sub-classification, and ALBI grade are shown in Supplemental Figures 2–6.
Comparison of the performance and discriminative ability between the current model and other models.

ALBI, albumin–bilirubin; AUC, area under receiver operating characteristic curve; BCLC, Barcelona Clinic Liver Cancer; ETC, external test cohort; ITC, internal test cohort; mHAP, modified hepatoma arterial-embolization prognostic; TC, training cohort.

The performance comparison of TBR score and other clinical tumor staging. (a) The time-dependent area under the curve comparison in training cohorts. (b) The time-dependent area under the curve comparison in internal test cohorts. (c) The time-dependent area under the curve comparison in external test cohorts. (d) The DCA comparison in training cohorts. (e) The DCA comparison in internal test cohorts. (f) The DCA comparison in external test cohorts.
Prognostic risk stratification
The cumulative 5-year OS rate of pseudo-capsulated, pseudocapsule breakthrough, confluent multinodular, and infiltrative HCC were 93.2%, 76.7%, 76.8%, and 62.9%, respectively (Figure 4(a)), showing significantly statistical significance (p < 0.001). The similar results were also observed in the other cohorts (p = 0.002, 0.008; Figure 4(b) and (c)). The optimal cutoff values (32 and 74 centiles) based on Xtile were used to divide the cohort into low-, middle-, and high-risk strata. The cumulative 5-year OS rate of low-, middle-, and high-risk strata in the training cohort were 86.3%, 73.7%, and 62.5% (Figure 4(d)), respectively, showing significant statistical significance (p < 0.001). In the internal test cohort, the cumulative 5-year OS rate of low-, middle-, and high-risk strata in the training cohort were 87.5%, 53.4%, and 38.6%, respectively (Figure 4(e)), showing significant statistical significance (p < 0.001). In the external test cohort, the cumulative 5-year OS rate of low-, middle-, and high-risk strata in the training cohort were 93.4%, 80.8%, and 52.3%, respectively (Figure 4(f)), showing significant statistical significance (p < 0.001). The cumulative 1-, 3-, and 5-year OS rates of low-, middle-, and high-risk strata based on model 2 in three cohorts are shown in Supplemental Figure 7. Similarly, our results also provided a favorable performance and discrimination in different subgroups including AFP levels, age, gender, ALBI grade, and etiology, which remained consistent in the aforementioned results (Supplemental Tables 3–7). These results suggest that the TBR score is a prognostic risk stratification tool with strong robustness and resolution for TACE-MWA.

Kaplan–Meier survival curves for patients with four types of radiologic features HCC and three risk strata. (a) Four types of radiologic features HCC comparison in training cohorts. (b) Four types of radiologic features HCC comparison in internal test cohorts. (c) Four types of radiologic features HCC comparison in external test cohorts. (d) Three risk strata comparison in training cohorts. (e) Three risk strata comparison in internal test cohorts. (f) Three risk strata comparison in external test cohorts.
Survival benefit of adjuvant therapies in different TBR risk strata
In the primary cohort, 25.6% (555/2169) of patients with HCC received targeted immunotherapy after TACE-MWA. According to the risk stratification using the nomogram, these patients were grouped into low-, middle-, and high-risk strata. We can observe the cumulative OS of the patients who received adjuvant targeted immunotherapy was significantly higher than that of the patients without targeted immunotherapy (p = 0.013) in the high-risk group. By contrast, the differences in cumulative OS between the targeted immunotherapy group and without targeted immunotherapy group were insignificant in the low-risk and middle-risk groups, respectively (p = 0.449 and 0.113; Figure 5(a)–(c)). A forest plot shows the prognostic hazard risk ratio between targeted immunotherapy and no targeted immunotherapy in three risk groups (Figure 5(d)).

Survival benefit of adjuvant therapies in different TBR risk strata. (a) Kaplan–Meier survival curves comparison for patients in high-risk subgroup received adjuvant therapies and no adjuvant therapies. (b) Kaplan–Meier survival curves comparison for patients in the middle-risk subgroup who received adjuvant therapies and no adjuvant therapies. (c) Kaplan–Meier survival curves comparison for patients in the low-risk subgroup who received adjuvant therapies and no adjuvant therapies. (d) The forest plot shows the factors associated with the overall survival of the patients with intermediate-stage hepatocellular carcinoma who received transarterial chemoembolization combined with microwave ablation in three risk strata.
Discussion
In clinical settings, embolization techniques have been advanced through the utilization of innovative embolic agents, including Yttrium-90 microspheres and drug-eluting beads. However, these advancements have not demonstrated substantial superiority over cTACE in terms of OS. 20 In recent years, many studies showed that TACE-MWA treatment can provide superior disease control and long-term survival compared to cTACE for a proportion of patients with intermediate-stage HCC.21–23 However, the criteria for patient selection remained controversial. Herein, our team has proposed a new and simple scoring tool named TBR score based on tumor burden and radiological characteristics after a comprehensive analysis of a multicenter, nationwide cohort comprising 2505 (BCLC-B stage) HCC patients receiving TACE-MWA. The novelty and strength of our study were summarized as follows: (1) The TBR score was developed based on a large treatment-naïve cohort of intermediate-stage HCC patients from multiple tertiary-care hospitals across China with effective internal and external test cohorts; (2) based on tumor imaging, for the first time, we classified BCLC B stage HCCs into four typical radiological subtypes, including pseudo-capsulated type, pseudocapsule breakthrough type, confluent multinodular type, and infiltrative type, with distinct survival times; (3) we demonstrated that the TBR score had superior performance in survival discrimination for HCC patients receiving TACE-MWA combination treatment compared to the well-known “6-and-12” score.
Owing to the heterogeneity of intermediate-stage HCC, for a proportion of patients, TACE alone is not optimal and the strategy of TACE sequentially combined with MWA is widely adopted to achieve the no evidence of disease “NED” status. Therefore, the construction of scoring models that predict patients’ survival was in urgent need. In the past, many prognostic score systems, including ALBI, AFP, HAP based on AFP, tumor diameter, bilirubin, and albumin, were used to predict the prognosis of intermediate-stage HCC.24–26 Campani et al. 9 reported a model (mHAP-III), which used the continue variables, including number of tumors, serum albumin, serum total bilirubin, alpha-fetoprotein, and maximum tumor size, to achieve individual patient prognostication for patients receiving TACE. Campani et al. 9 subsequently validated the mHAP-III model in an independent cohort including 298 patients and proved its superiority over HAP and mHAP-II. The STATE and START scores were also proved to be valuable in identifying candidates for TACE, 27 in this case C-reactive protein was required, which was not routinely measured in the many hospitals. 28 Although these prognostic scores have good performance and discrimination, they are designed specifically for HCC patients receiving TACE. In this study, we demonstrated that the TBR score had superiority in prognosis prediction compared to these prognostic systems in the HCC cohort receiving TACE-MWA combination treatment, suggesting that our model has solid advantages in niche areas.
Tumor burden plays an important role in determining the optimal candidate population for TACE. Han et al. 7 reported that a “6-and-12” score was used to predict OS to stratify recommended TACE candidates, but this viewpoint has always been controversial. Moreover, Ni et al. 29 reported a nomogram that integrated ECOG, ALBI, AFP, tumor size and number, and sessions of TACE and MWA to predict OS of patients receiving TACE-MWA combination treatment. Although the tumor burden plays an important role in prognostic prediction for HCC patients who have undergone TACE-based treatment, it does not fully represent the pathological subtypes and invasiveness of the tumor. Up to now, an increasing number of studies have identified the radiological characteristics of tumors as another key risk factor in addition to tumor burden. In 2022, the BCLC guidelines were revised to recognize infiltrative HCC as a new and independent subtype of HCC. Systematic therapy is now recommended for patients with intermediate-stage infiltrative HCC, starting from the initial treatment phase.30,31
The radiologic subtypes of HCC, including confluent multinodular type, infiltrative type, and extranodular growth type, were recommended to be incorporated into the prediction of response to TACE for intermediate-stage HCC by the recent Asia-Pacific Primary Liver Cancer Expert consensus. 32 Our team has also reported the selection and comparison of intra-arterial therapies for infiltrative HCC, 12 and the results found TACE yields a lower ORR in infiltrative HCC than pseudo-capsulated HCC. The reason may be as follows: (1) the embolization effect of TACE can aggravate liver dysfunction and complications, which result in significantly reduced executable TACE sessions and (2) the collateral circulation caused by the huge size and diffuse distribution of infiltrative HCC makes it difficult for lipiodol or microsphere to completely embolize the targeted tumors. This evidence suggested that TACE alone or TACE-MWA treatment is inadequate to control infiltrative type HCC. In our study, both tumor burden and radiologic subtypes were found of key importance in determining the prognosis of HCC patients receiving TACE-MWA treatment and as mainstays in TBR score. The patients could be subdivided into three strata based on TBR score and the high-risk strata can benefit from adjuvant targeted immunotherapy, which to some extent is consistent with the BCLC guideline’s recommendation on infiltrative HCC.
During the model development process, we found all patients exhibited good ECOG PS scores at the initial diagnosis; therefore, it was not included in the model. However, liver function and biological features still might be crucial determinants of prognosis and were primarily considered for model development. Interestingly, unlike in previous scores where bilirubin and albumin are major components, the liver function parameters were not identified as independent prognostic factors according to Cox regression analysis. On the other hand, the addition of the biomarker AFP to model 2 (built on radiologic features and tumor burden) only provided marginal, without statistically significant difference regarding model performance. The insignificance of markers of liver function may be caused by the uniqueness of baseline characteristics of HCC patients receiving TACE-MWA treatment. While tumor burden varies in a relatively wide range, according to radiologic features, the liver function of the majority of patients was at a preserved level (Child-Pugh A class), which might lead to the phenomenon that tumor burden plays a key role in the prediction, while other parameters contribute less. This disparity of homogeneity in different parameters may also explain the findings that the TBR score exhibited better performance and discriminative capability than models including up-to-7 criteria, 6–12 criteria, mHAP-III score, BCLC-B sub-classification, and ALBI score.
In this study, the cumulative 5-year OS rate in the three cohorts were all more than 50%, and the cumulative 5-year OS rate of the three different risk strata identified by the TBR score ranged from 62.5% to 86.3%, which was higher than results in previous reports. The phenomenon may be due to the fact that a large proportion of the population with intermediate-stage HCC received downstaging treatment, conversation from BCLC stage B to stage A, which is similar to the results reported results by Shi et al., 33 who found MWA could achieve results similar to surgery after successful downstaging by TACE for intermediate-stage HCC. Patients with pseudo-capsulated HCC had the best OS time, whereas infiltrative HCC had a poor outcome. This further demonstrates that radiologic features to some extent determine the prognosis of HCC patients even when liver function and performance status are preserved, supporting the importance of risk stratification by this radiologic feature in clinical practice and trials.
This study has several limitations. First, this study recruited patients from seven different hospitals across China, with each hospital exhibiting distinct treatment practices. Second, there were variations in the choice of TACE medications, which potentially confounded the ultimate results. Third, this study primarily included HCC patients with HBV infection, which was regarded as the main cause of HCC in China. It is still unclear whether these findings can be broadly applied in Western countries, where alcoholic liver cirrhosis is the main cause of HCC. Finally, since increasing studies showed that radiomics of HCC can predict the prognosis of HCC patients who have undergone TACE, it may be incorporated into our TBR score model to enhance its accuracy in future research.
Conclusion
In conclusion, the TBR score could be easily implemented in clinical practice for risk stratification and individualized management for HCC patients receiving TACE-MWA treatment.
Supplemental Material
sj-docx-1-tam-10.1177_17588359251324052 – Supplemental material for Identification of candidates with hepatocellular carcinoma to receive TACE combined with MWA by assessing tumor burden and radiologic features
Supplemental material, sj-docx-1-tam-10.1177_17588359251324052 for Identification of candidates with hepatocellular carcinoma to receive TACE combined with MWA by assessing tumor burden and radiologic features by Chao An, Lujun Shen, Qifeng Chen, Yiquan Jiang, Chen Li, He Ren, Peihong Wu and Xi Liu in Therapeutic Advances in Medical Oncology
Supplemental Material
sj-docx-2-tam-10.1177_17588359251324052 – Supplemental material for Identification of candidates with hepatocellular carcinoma to receive TACE combined with MWA by assessing tumor burden and radiologic features
Supplemental material, sj-docx-2-tam-10.1177_17588359251324052 for Identification of candidates with hepatocellular carcinoma to receive TACE combined with MWA by assessing tumor burden and radiologic features by Chao An, Lujun Shen, Qifeng Chen, Yiquan Jiang, Chen Li, He Ren, Peihong Wu and Xi Liu in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
The authors acknowledge Wang Li, MD, and Mengxuan Zuo, MD, Department of Minimal invasive intervention, Sun Yat-sen University Cancer Center, for their support in the assessment of tumor burden and radiologic features.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
