Abstract
Background:
Transarterial chemoembolization (TACE) is an effective and safe downstaging therapy for hepatocellular carcinoma (HCC). However, the selection of sequential therapeutic modalities is still controversial.
Objectives:
This study compared the effectiveness and safety of surgical resection (SR) and thermal ablation (TA) after patients with HCC underwent TACE downstaging therapy.
Design:
A retrospective, multi-institutional study.
Methods:
From June 2008 to October 2022, a total of 4782 consecutive patients with HCC beyond the initial Milan criteria underwent TACE at 12 hospitals. Among them, 609 patients who received successful downstaging therapy were retrospectively reviewed. Among them, 209 patients underwent an SR, and 390 patients received TA after TACE. The propensity score matching (PSM) method was applied to reduce selection bias between groups. Cumulative overall survival (OS) and progression-free survival (PFS) were compared using the Kaplan–Meier method with the log-rank test.
Results:
After PSM 1:1 (n = 185 in both groups), the cumulative 1-, 3-, 5-, and 10-year OS rates were 98.8%, 89.3%, 82.9%, and 64.4%, respectively, in the SR group and 99.5%, 88.4%, 75.3%, and 53.9%, respectively, in the TA group; these two groups were not significantly different (HR: 1.22; 95% CI: 0.78–1.89; p = 0.381). The cumulative 1-, 3-, 5-, and 10-year PFS rates were 88.5%, 69.2%, 58.8%, and 32.2%, respectively, in the SR group and 90.6%, 71.4%, 53.1%, and 32.0%, respectively, in the TA group, revealing no significant difference between the two groups (HR: 0.97; 95% CI: 0.71–1.32; p = 0.855).
Conclusion:
For HCC patients beyond the Milan criteria who received TACE downstaging therapy, TA might be acceptable as an alternative to SR in the first-line sequential treatment scheme.
Plain language summary
This retrospective, multi-institutional study revealed that thermal ablation (TA) and surgical resection (SR) had comparable long-term survival and safety profiles after transarterial chemoembolization (TACE) downstaging therapy in patients with hepatocellular carcinoma (HCC) beyond the initial Milan criteria. TA is an acceptable first-line alternative to SR for selected patients with higher stages of HCC who receive TACE downstaging therapy, especially those unsuitable for SR.
Keywords
Introduction
Primary liver cancer is the sixth most commonly diagnosed malignancy and the third leading cause of cancer death worldwide; hepatocellular carcinoma (HCC) is the main pathological type and accounts for 90% of liver cancer cases.1,2 Currently, international guidelines recommend first-line curative therapies for HCC based on the Milan criteria (maximum diameter of one lesion ⩽5 cm, two or three lesions ⩽3 cm, without macrovascular invasion or extrahepatic metastasis) that mainly include surgical resection (SR), liver transplantation (LT), and local-region thermal ablation (TA).3–5 Unfortunately, the vast majority of HCC patients are diagnosed at the intermediate-advanced stage at the time of first diagnosis, resulting in a decreased chance of receiving curative therapies.6,7
Notably, downstaging therapy for HCC, which is defined as the reduction of the visible tumor burden to meet the Milan criteria by transarterial or systemic chemotherapy, 8 is a promising option for unresectable HCC (uHCC confirmed by liver surgery expert panels (i.e., R0 resection was technically unachievable or remnant liver volume less than 30% in non-cirrhotic patients or 40% in cirrhotic patients)) patients. 9 The Chinese Expert Consensus on Conversion Therapy for Hepatocellular Carcinoma suggests that conversion therapy is an intermediate goal for the treatment of intermediate-advanced HCC, and long-term survival is the ultimate goal. 10 Transarterial chemoembolization (TACE) is recommended as a potential first-line treatment for intermediate HCC. 11 For those with a single large HCC, TACE can also be considered an alternative to surgery for treatment. 12
The purpose of downstaging therapy is to make a curative procedure applicable in certain HCC patients with a tumor burden beyond the Milan criteria who would otherwise be inappropriate for surgery. 13 In this regard, TACE can occlude the main artery supplying the HCC tumor and minimize heat loss by convection, causing subsequent complete ablation or R0 surgery to become more effective. 14 Therefore, TACE-based downstaging therapy has been regarded as an important treatment strategy for HCC patients with disease that exceeded the Milan criteria.
Previously, downstaging therapy was usually used to achieve successful LT, but the scarcity of organ sources limits its application.8,15 TA is a first-line alternative treatment with many advantages, including cost-effectiveness and minimal invasiveness, and has similar survival outcomes and safety as SR.16–18 However, the appropriate sequential options for treating uHCC after TACE downstaging therapy remain controversial. This study aimed to investigate the long-term outcomes of downstaging therapy for HCC beyond the Milan criteria and compare the safety and efficacy of two treatment modalities (hereafter, SR and TA).
Materials and methods
Study design and patient enrollment
This retrospective, multi-institutional study was conducted following the principles of the 1975 Helsinki Declaration. The protocol obtained approval from the Institutional Review Board of Sun Yat-sen University Cancer Center (Approval No B2022-694) and other participating institutions. The study is reported according to the Strengthening the Reporting of Cohort Studies in Surgery (STROCSS) criteria. 19
From June 2008 to October 2022, a total of 4782 consecutive patients with uHCC whose Barcelona Clinic Liver Cancer (BCLC) stage A or B status exceeded the initial Milan criteria who underwent TACE were reviewed at 12 tertiary hospitals. These HCC patients were diagnosed based on the guidelines of the European Association for the Study of Liver and the American Association for the Study of Liver Disease or via pathologic confirmation via preoperative biopsy.20,21 The data source from different hospitals is shown in Supplemental Table 1. The inclusion criteria were as follows: (a) aged 18–75 years; (b) Eastern Cooperative Oncology Group performance status <2; (c) Child–Pugh class A liver function; (d) complete ablation or R0 resection after TACE downstaging therapy, as a successful curative therapy. The exclusion criteria were as follows: (a) underwent any treatment before TACE; (b) had HCC combined with other malignancies; (c) received other conversion therapies, such as stereotactic body radiation therapy (SBRT); and (d) lost to follow-up for more than 6 months. Figure 1 shows the enrollment pathway of patients who underwent TACE downstaging therapy. The procedures for TACE, protocol discontinuation, SR, and TA are described in the Supplemental Information, E1.1–E1.4. The final downstaging group included 390 patients who received TA and 219 patients who underwent SR.

Enrollment pathway of patients with unresectable HCC who underwent successful TACE downstaging therapy.
Assessment of downstaging of HCC
The Milan criteria were assessed based on the Liver Imaging Reporting and Data System (version 2020). The dynamic enhanced images (e.g., Computed Tomography (CT) and MRI) were evaluated independently by two radiologists (P.H.W. S.S.W., with 10 years of experience and X.X.X., with 5 years of experience) who were blinded to the TACE procedure at the time of data collection to confirm agreement on successful downstaging. The response to TACE, including complete response (CR), partial response (PR), stable disease (SD), and progressive disease (PD), was assessed by dynamic contrast-enhanced imaging based on the modified Response Evaluation Criteria in Solid Tumors. 22
Indication for TACE downstaging therapy
TACE downstaging therapy was initiated according to the recommendations of a multidisciplinary tumor board based on patient performance status, liver function, and tumor characteristics, based on the following criteria: (a) patients with intrahepatic nodules after TACE were evaluated as CR or PR within 8 weeks; (b) R0 resection had to be achieved with sufficient remnant liver volume (⩾40% of the standard liver volume for patients with liver cirrhosis or ⩾30% of the standard liver volume for patients without liver cirrhosis); (c) normal coagulation function (i.e., prothrombin time <15 s, prothrombin activity >40%, and platelet count >50 cells × 109/L); (d) no severe or persistent adverse events (AEs) from TACE; (e) should not be present serious chronic disease, mainly including heart, lung, and renal dysfunction.
Assessments and follow-up
In this study, enrolled patients were censored at the last follow-up date (August 31, 2023). After thorough TACE-based downstaging therapy was completed, the serum alpha-fetoprotein (AFP) concentration and dynamic contrast-enhanced images were examined again at 3- to 6-month intervals and at approximately 3-month intervals in the first year. The two medical records of patients with uHCC who received TACE downstaging therapy are shown in Figures 2 and 3. If recurrent lesions were found after downstaging therapy, local treatment (e.g., TACE, hepatic arterial infusion chemotherapy (HAIC), SR, SBRT, and TA), and targeted immunotherapy (e.g., sorafenib, lenvatinib, and programmed cell death protein-1 (PD-1)) were administered (Supplemental Table 2).
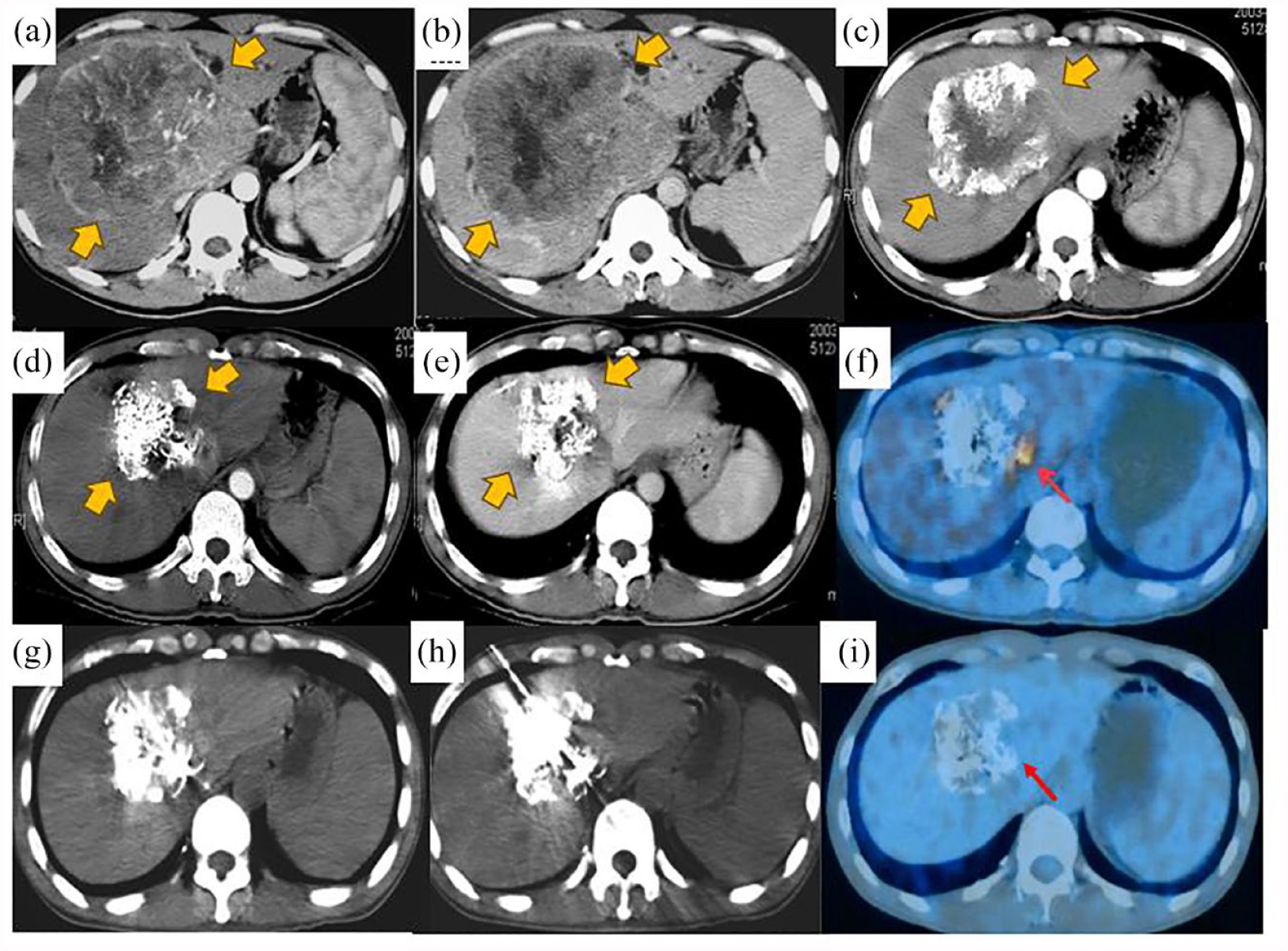
The imageological medical record of uHCC patient underwent thermal ablation after TACE downstaging therapy. A 38-year-old man with a single huge hepatocellular carcinoma (16.2 × 15.9 × 14.5 cm) received TACE combined with sequential ablation. (a, b) Enhanced contrast CT (arterial and portal venous phases) shows a massive tumor with rich blood supply (yellow arrow) and the AFP level was >121,000 ng/mL; (c) Receiving four cycles of TACE, the maximum diameter of the tumor reduced to 10 cm in the arterial phase (yellow arrow) after 1 month and the AFP level was 22,317 ng/mL; (d, e) the maximum diameter of the tumor reduced to 6.6 cm in the arterial phase and portal venous phases after 6 months and (f) PET-CT showed residual lesions with active metabolism (red arrow); (g, h) an electrode was accurately inserted into the residual lesion for RFA treatment; and (i) the residual lesion disappear completely followed by PET-CT after RFA.

The imageological medical record of the uHCC patient underwent the SR after TACE downstaging therapy. A 58-year-old man with a single large hepatocellular carcinoma (7.2 × 5.2 × 4.8 cm) received TACE combined with sequential SR. (a, b) Dual-phase MRI shows a high signature in T2WI and washout in the venous phase in T1WI (white arrow); (c, d) three cycles of TACE, the maximum diameter of the tumor reduced to 4.6 cm and tumors deposit iodine oil well in the arterial phase and portal venous phases (white arrow) and the AFP level was 48.2 ng/mL; (e) SR of the target tumor under laparoscopy; (f, g) after receiving SR, T2WI, and T1WI showed encapsulated fluid accumulation in the damaged area, and low signature in the venous phase (white arrow); (h) the HCC sample excised from within the liver; (i) hematoxylin and eosin staining. Pathological examination reported that the primary tumor was composed of viable cancer cells. The infiltration of inflammatory cells could be observed (hematoxylin–eosin stain; original magnification, ×200).
Definition of terminologies
Successful downstaging therapy was defined as a viable intrahepatic tumor burden after TACE that met the Milan criteria. Complete ablation was defined as the absence of enhancement of any areas of the mass at a follow-up contrast-enhanced imaging examination performed 1 month after TA. The primary endpoints of this study are OS, PFS, extrahepatic PFS (EPFS), and postoperative progression survival (PPS). The OS is calculated from the date of initial treatment to the date of death or deadline for follow-up. The PFS and EPFS are calculated from the first TACE session to the date of disease progression and extrahepatic progression or the end of the follow-up. The PPS was calculated from the date of PD after TACE treatment to the date of death from any cause or the last follow-up. The second endpoint of this study is safety. Major complications were defined as events that caused substantial morbidity and disability, increased the level of care, led to hospital admission, or substantially prolonged the hospital stay based on the Common Terminology Criteria for Adverse Events v4.0. Assessments. 23 We collected 18 variables, and their definitions are shown in Supplemental Information E1.5.
Statistical analysis
Statistical analysis was performed using SPSS version 23.0 (IBM Corp., NewYork, NY, USA) and the RMS package of R software version 3.5.1 (http://www.r-project.org/). The quantitative variables are presented as the mean ± standard deviation (SD) or median with interquartile range (IQR) and were compared by the Kruskal–Wallis test. The qualitative variables are presented as frequencies and were compared using the χ 2 test. We applied propensity score matching (PSM) using a 1:1 nearest-neighbor algorithm with a caliper distance (0.1 times the SD) to adjust the potential unbalanced variables in the two groups. The cumulative survival was compared using the Kaplan–Meier method with the log-rank test. Univariate and multivariate analyses of independent prognostic factors were evaluated using the forward stepwise Cox regression model to calculate hazard ratios (HRs) and 95% confidence intervals (CIs).
All tests of significance were two-sided, and a p-value <0.05 was interpreted to indicate statistical significance.
Results
Baseline characteristics of the enrolled patients
In this study, a total of 1624 patients received TACE conversion therapy. Among them, 609 treatment-naïve patients (77 females and 532 males; mean age, 54.8 ± 11.2 (SD)) with uHCC after successful TACE downstaging therapy were reviewed. After PSM 1:1, 185 patients were assigned to the SR group, and 185 patients were assigned to the TA group. Before PSM, a higher incidence rate of cirrhosis, ascites, and tumor size <5 cm was found in the TA group than in SR group (p = 0.015, 0.026, and <0.001). The baseline characteristics of these patients were matched using the nearest neighbor method with a caliber of 0.1 (Supplemental Figure 1) and stratified by two downstaging therapeutic modalities after PSM (Table 1).
Baseline characteristics of the patients with HCC who received TACE downstaging therapy.

Data are number of patients; data in parentheses are percentage unless otherwise indicated. Data in the bracket were the percentage of patients. The quantitative data with mean ± standard deviation or median with interquartile range (IQR) were compared by the Mann–Whitney U test. The qualitative data in two groups were compared using the Chi-square test. The variables matched for PSM included age at diagnosis, gender, ECOG, comorbidity, HBV, BCLC stages, ascites, HCC number, HCC diameter, ALBI grade, AFP, tyrosine kinase inhibitors, and immunotherapy.
p-value <0.05 suggests statistically significant differences.
*Respresent the total of number of TACE before SR or TA.
AFP, α-fetoprotein; ALB, albumin; ALBI, albumin–bilirubin; ALT, alanine aminotransferase; AST, aspartate aminotransferase; CTP, Child-Turcotte-Pugh; ECOG, Eastern Cooperative Oncology Group; HBV, hepatitis type B viral; HCC, hepatocellular carcinoma; INR, international normalized ratio; MWA, microwave ablation; PLT: Platelet; PSM, propensity score match; PT, prothrombin time; RFA, radiofrequency ablation; SR, surgical resection; TA, thermal ablation; TACE, transarterial chemoembolization; TBIL, total bilirubin.
Mean ± standard deviation compared by the Mann–Whitney U test.
Median with interquartile range (IQR) compared by the Mann–Whitney U test.
Chi-square test.
Downstaging conditions
In the TA group, the mean number of TACE sessions per patient was 2.9 ± 0.4. The median interval between the first session of TACE and TA was 3.7 months (IQR, 1.2–6.6 months). Before TACE, 102 patients had >3 lesions, and the mean maximum diameter was 6.8 ± 3.3 cm. After downstaging therapy, 8 patients had 2–3 lesions, and the mean maximum diameter was 2.3 cm ± 0.3 cm. In the SR group, the mean number of TACE sessions per patient was 2.2 ± 0.6. The median interval between the first session of TACE and TA was 4.2 months (IQR, 2.5–7.8 months). Before TACE, 94 patients had >3 lesions, and the mean maximum diameter was 6.5 ± 2.4 cm. After downstaging therapy, 10 patients had 2–3 lesions, and the mean maximum diameter was 2.2 ± 0.4 cm. The median intervals between TACE and sequential local treatment in the two groups showed no significant difference (p = 0.478).
Comparison of survival outcomes
The median follow-up durations in the SR group and TA group were 22.4 months (IQR: 15.6–72.6 months) and 24.8 months (IQR: 18.2–68.5 months), respectively. The cumulative 1-, 3-, 5-, and 10-year OS rates were 99.1%, 89.9%, 82.4%, and 65.6%, respectively, in the SR group and 99.5%, 90.2%, 79.1%, and 55.7%, respectively, in the TA group. These two groups were not significantly different (Figure 4(a)) (HR: 1.13; 95% CI: 0.77–1.66; p = 0.543). The cumulative 1-, 3-, 5-, and 10-year PFS rates were 88.8%, 68.4%, 57.3%, and 34.9%, respectively, in the SR group and 89.5%, 70.2%, 52.1%, and 28.5% in the TA group; these two groups were not significantly different (Figure 4(b)) (HR: 1.07; 95% CI: 0.83–1.37; p = 0.614). The cumulative 1-, 3-, 5-, and 10-year EPFS rates were 97.1%, 91.8%, 88.5%, and 83.3%, respectively, in the SR group and 99.5%, 95.5%, 92.1%, and 80.1%, respectively, in the TA group, showing no significant difference between the two groups (Figure 4(c)) (HR: 0.60; 95% CI: 0.33–1.10; p = 0.100). The cumulative 1-, 3-, 5-, and 10-year PPS rates were 94.7%, 85.5%, 78.9%, and 69.0%, respectively, in the SR group and 96.3%, 86.0%, 73.8%, and 60.7%, respectively, in the TA group; these two groups were not significantly different (Figure 4(d)) (HR: 1.07; 95% CI: 0.72–1.57; p = 0.746).

Comparing the survival of SR and TA groups for uHCC patients in downstaging cohorts before PSM. Kaplan–Meier curves for the (a) overall survival (OS) and (b) progression-free survival (PFS), (c) extrahepatic progression-free survival (EPFS), and (d) postoperative progression survival (PPS) of uHCC patients between SR group and TA group before PSM.
After PSM 1:1, the cumulative 1-, 3-, 5-, and 10-year OS rates were 98.8%, 89.3%, 82.9%, and 64.4%, respectively, in the SR group and 99.5%, 88.4%, 75.3%, and 53.9%, respectively, in the TA group; these two groups were not significantly different (Figure 5(a)) (HR: 1.22; 95% CI: 0.78–1.89; p = 0.381). The cumulative 1-, 3-, 5-, and 10-year PFS rates were 88.5%, 69.2%, 58.8%, and 32.2%, respectively, in the SR group and 90.6%, 71.4%, 53.1%, and 32.0%, respectively, in the TA group; these two groups were not significantly different (Figure 5(b)) (HR: 0.97; 95% CI: 0.71–1.32; p = 0.855). The cumulative 1-, 3-, 5-, and 10-year EPFS rates were 96.6%, 92.0%, 88.2%, and 82.2%, respectively, in the SR group and 100.0%, 96.9%, 92.0%, and 76.6% in the TA group; these two groups were not significantly different (Figure 5(c)) (HR: 0.46; 95% CI: 0.21–1.01; p = 0.053). The cumulative 1-, 3-, 5-, and 10-year PPS rates were 94.5%, 85.1%, 78.7%, and 67.9%, respectively, in the SR group and 94.4%, 83.5%, 68%, and 65.6% in the TA group; these two groups were not significantly different (Figure 5(d)) (HR: 1.24; 95% CI: 0.80–1.93; p = 0.334).

Comparing the survival of SR and TA groups for uHCC patients in downstaging cohorts after PSM. Kaplan–Meier curves for the (a) overall survival (OS) and (b) progression-free survival (PFS), (c) extrahepatic progression-free survival (EPFS), and (d) postoperative progression survival (PPS) of uHCC patients between SR group and TA group after PSM.
Risk factors for survival outcomes
The risk factors for OS and PFS were assessed by univariate and multivariate analyses (Tables 2 and 3). In the univariate analyses, albumin–bilirubin (ALBI) grade 2–3 (HR: 1.58; 95% CI: 1.10–2.29; p = 0.014) and not receiving targeted immunotherapy (HR: 1.83; 95% CI: 1.11–3.02; p = 0.017) were significant factors for poor OS. Multivariate analyses revealed that the factors that significantly affected OS were ALBI grade 2–3 (HR: 1.63; 95% CI: 1.14–2.53; p = 0.007) and not receiving targeted immunotherapy (HR: 2.02; 95% CI: 1.09–3.67; p = 0.009). According to the univariate analyses, ALBI grade 2–3 (HR: 1.41; 95% CI: 1.11–1.81; p = 0.005), AFP >400 (ng/mL) (HR: 1.37; 95% CI: 1.07–1.75; p = 0.012) and not receiving targeted immunotherapy (HR: 1.51; 95% CI: 1.09–2.08; p = 0.013) were significant factors for poor PFS. Multivariate analyses revealed that the factors that significantly affected PFS were ALBI grade 2–3 (HR: 1.33; 95% CI: 1.12–2.50; p = 0.012), AFP >400 ng/mL (HR: 1.43; 95% CI: 1.11–2.24; p = 0.007), and not receiving targeted immunotherapy (HR: 2.14; 95% CI: 1.11–4.12; p = 0.003). The risk factors for EPFS and PPS are shown in Supplemental Tables 3 and 4.
Univariable and multivariable Cox regression analyses of predictors of OS for HCC patients undergoing TACE downstaging therapy.

Cox regression analyses are used to calculate the hazard ratios (HRs) and 95% confidence intervals (CIs) based on overall survival (OS). Covariables that are significant in univariable Cox regression analysis (p < 0.05) are included in the multivariable analysis.
The bold values were < 0.05.
AFP, α-fetoprotein; ALBI, albumin–bilirubin; HBV, hepatitis type B viral; HCC, hepatocellular carcinoma; OS, overall survival; PSM, propensity score match; SR, surgical resection; TA, thermal ablation; TACE, transarterial chemoembolization.
Univariable and multivariable Cox regression analyses of predictors of PFS for HCC patients undergoing TACE downstaging therapy.

Cox regression analyses are used to calculate the hazard ratios (HRs) and 95% confidence intervals (CIs) based on overall survival (OS). Covariables that are significant in univariable Cox regression analysis (p < 0.05) are included in the multivariable analysis.
The bold values were < 0.05.
AFP, α-fetoprotein; HBV, hepatitis type B viral; HCC, hepatocellular carcinoma; PFS, progression-free survival; PSM, propensity score match; SR, surgical resection; TA, thermal ablation; TACE, transarterial chemoembolization.
Subgroup analysis
The results of the subgroup analyses of OS and PFS stratified by important variables are shown in Figure 6. Neither subgroup nor PSM analysis changed our essential finding that SR provided OS and PFS comparable to TA.

Subgroup analyses of SR and TA groups for uHCC patients in downstaged cohorts. Forest plots show the factors associated with overall survival (OS), and progression-free survival (PFS) in the uHCC patients who received SR and TA after transarterial chemoembolization (TACE) downstaging therapy. (a) OS in the downstage cohorts and (b) PFS in the downstage cohorts.
Treatment-related complications
During follow-up, no treatment-related deaths occurred in either group. The incidence of complications after SR or TA is shown in Table 4. Liver dysfunction was the most common minor complication and included the presence of mild ascites and increased transaminase levels. The rate of major complications was 6.8% (15 of 219 patients) in the SR group and 7.7% (30 of 390 patients) in the TA group. After PSM, the incidences of major complications were 6.5% in the SR group and 7.6% in the TA group, and these two groups were not significantly different (p = 0.684).
The complications comparison between SR and TA after TACE downstaging therapy.

Data in the bracket were the percentage of patients. The data in the two groups were compared using the Chi-square test or Fisher’s exact test.
ND, no data; PSM, propensity score match; SR, surgical resection; TA, thermal ablation; TACE, transarterial chemoembolization.
Discussion
In this retrospective study, we utilized a multicenter design, which could result in greater real-world heterogeneity than a single-center design. A total of 12.7% (609/4782) of patients with initial HCC beyond the Milan criteria received successful downstaging therapy that allowed successful R0 resection or complete ablation. Among them, 30.2% (184/609) of patients received TACE combined with targeted immunotherapy (e.g., sorafenib, lenvatinib, and programmed cell death protein 1 (PD-1)). The results suggest that TACE is a safe and feasible downstaging therapy for uHCC with a potential survival benefit. With rigorous PSM analysis, we compared two sequential treatment modalities after TACE-based downstaging therapy and analyzed the long-term survival outcomes over the past 15 years.
After PSM, the long-term survival outcomes of patients who underwent SR or TA after downstaging therapy were comparable (p = 0.381 and 0.855, respectively). Moreover, similar PPS outcomes were observed between the SR and TA groups (p = 0.334), mainly due to sequential repeat local treatment, including TACE, HAIC, TA, or SBRT. Notably, patients in the TA group had a better survival rate after EPFS than those in the SR group (p = 0.053), indicating that TA-induced immune antigen release helps suppress distant metastasis of HCC. Moreover, the incidence of major complications was low, without difference between groups (12 of 185 patients in the SR group and 14 of 185 in the TA group) were also comparable (p = 0.684). Compared with SR, TA was associated with less trauma, faster recovery, shorter hospitalization and lower costs, and a lack of need for anesthesia. These advantages could lead to the use of another reliable treatment option for seniors or patients who are intolerant to SR.
As early as 1993, Sitzmann and Abrams were the first to report cases of unresectable cancer that were converted to resectable cancer after radiochemotherapy in 14 patients. 24 In addition, in 1997, Majno et al. proposed downstaging therapy before HT and achieved good survival outcomes 12 ; subsequently, an increasing number of studies have explored the safety and effectiveness of downgrading therapy for uHCC patients.25–29 Recently, the IMbrave-150 study showed encouraging results with the combination of atezolizumab and bevacizumab in terms of the objective response rate (ORR, 27.3% by RECIST 1.1 and 33.2% by mRECIST) of advanced HCC, 30 and it also provided a promising path for further expansion of downstaging therapy. However, their conversion rate remains unsatisfactory, especially among patients with large or large HCCs. Shi Feng et al. demonstrated that patients who underwent microwave ablation or radiofrequency ablation following TACE downstaging of disease beyond the Milan criteria had survival comparable to those who underwent TA alone after meeting the Milan criteria, indicating significant survival benefits.28,29 Min Deng et al. suggested that HAIC was superior to TACE conversion therapy for single enormous HCC patients, providing physicians with a new conversion therapy strategy. 31
In this study, excellent long-term survival outcomes were found after TACE-based downstaging therapy, and the cumulative 10-year OS rate reached 64.4% in the SR group and 53.9% in the TA group after PSM. These results indicate that downstaging therapy contributes to long-term survival, mainly due to the following factors: (a) the application of TACE contributes to eliminating microvascular invasion and satellite lesions and (b) a tumor burden beyond seven criteria has been confirmed as an independent risk factor for poor survival prognosis after TACE in previous studies. In this study, the mean maximum tumor diameter was 6.6 cm, which was reduced to 2.2 cm after TACE. Herein, a low tumor burden is a favorable factor for long-term survival.
According to the multivariate Cox regression analyses that included all patients in the cohort, ALBI, AFP, and TACE combined with targeted immunotherapy were crucial factors for the long-term OS and PFS of HCC patients who received TACE-based downstaging therapy, consistent with the findings of previous studies.32,33 Interestingly, targeted immunotherapy plays an important role in improving survival, possibly through synergistic antitumor effects of chemical agents, antiangiogenic agents, and PD-1 inhibitors. 34 Oxaliplatin can induce immunogenic cell death by releasing tumor antigens, transporting chimeric antigen receptor T cell (CRT) to the cell surface, and secreting HMGB1 and ATP. These molecules are related to cell death and bind to their respective receptors to support the evolution of tumor-specific CD8+ T cells. 35
Although our study has many strengths, including a large cohort and multicenter study design, it also has several limitations. First, this study enrolled patients from several hospitals across our country, with differences in the TACE application scheme and duration of use of chemotherapy drugs across hospitals. These factors may have affected the final outcomes. Second, in this study, we recruited patients in China with mostly large HCC and hepatitis B virus infections as the predominant etiologies. It remains to be elucidated whether the results can be widely applied in Western countries, where the majority of patients have a low tumor burden or alcoholic liver cirrhosis as the predominant etiology. Third, although we collected data from multiple hospitals, the number of recruited patients was limited. In the future, prospective clinical trials should be conducted to further test the reliability and reproducibility of the study results.
Conclusion
In summary, the findings showed that a TACE-based downstaging therapy strategy is feasible and safe for uHCC, and subsequent SR and TA have similar effectiveness and safety given the need for careful preparation and patient assessment. Thus, TA may be an alternative option as a minimally invasive treatment when encountering the technical challenges of implementing SR. However, the retrospective nature of this study and some selective biases may affect the power of our results. Therefore, a prospective clinical trial needs to be established to further validate our findings.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241306648 – Supplemental material for Surgical resection versus thermal ablation: comparison of sequential options after successful TACE downstaging therapy for unresectable hepatocellular carcinoma
Supplemental material, sj-docx-1-tam-10.1177_17588359241306648 for Surgical resection versus thermal ablation: comparison of sequential options after successful TACE downstaging therapy for unresectable hepatocellular carcinoma by Chao An, Songsong Wu, Mengxuan Zuo, Wang Li, Kai Li and Peihong Wu in Therapeutic Advances in Medical Oncology
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
