Abstract
Background:
The most common loadable chemotherapeutic drugs in drug-eluting bead transarterial chemoembolization (DEB-TACE) include doxorubicin, epirubicin, etc. CalliSpheres® beads have exhibited efficient loadability and eluting characteristics for raltitrexed as well as in vitro and animal experiments. However, the efficacy and safety of raltitrexed-loaded DEB-TACE in patients with intermediate-stage hepatocellular carcinoma (HCC) remain unclear.
Objectives:
To assess the efficacy and safety of raltitrexed-loaded DEB-TACE in patients with intermediate-stage HCC.
Design:
The study was conducted as a single-arm prospective study.
Methods:
This study was a prospective, single-arm trial conducted between June 2019 and June 2022. CalliSpheres® beads loaded with raltitrexed were used in the DEB-TACE procedure. The follow-up lasted for at least 1 year or until death. The primary endpoint was overall survival (OS), and the secondary endpoints were time to progression (TTP), progression-free survival (PFS), objective response rate (ORR), and adverse events (AEs).
Results:
The 6-month ORR and disease control rates were 90.1% and 93.8%, respectively. The median OS was 33.0 months. The 1-, 2-, and 3-year survival rates were 95.1%, 82.1%, and 43.6%, respectively. Child–Pugh class and bilobar disease occurrence were identified as independent OS predictors. The median TTP and PFS were 22.7 and 19.8 months, respectively. Eleven (11.5%) patients experienced at least one grade 3 AE, and serious AEs were reported in five participants (5.2%). No patient experienced grade 4 or 5 AEs.
Conclusion:
Raltitrexed-loaded DEB-TACE is feasible, safe, and effective in patients with intermediate-stage HCC.
Trial registration:
This trial was registered at www.chictr.org.cn under the identifier: 1900024097 on 25 June 2019.
Plain language summary
The utility of raltitrexed-loaded CalliSphere® beads in drug-eluting bead transarterial chemoembolization (DEB-TACE) has been demonstrated in in vitro and animal experiments. However, its efficacy and safety in patients with intermediate-stage hepatocellular carcinoma (HCC) remain unclear. Hence, this study aimed to assess the efficacy and safety profiles of DEB-TACE for such patients. We discovered that raltitrexed-loaded DEB-TACE led to a 6-month ORR of 90.1%, a median OS of 33.0 months, a median TTP of 22.7 months, and a median PFS of 19.8 months. The 1-, 2-, and 3-year survival rates were 95.1%, 82.1%, and 43.6%, respectively. Factors such as Child-Pugh class and bilobar disease occurrence were identified as independent predictors of OS. The study also showed acceptable safety profiles, with a low incidence of grade 3 adverse events and no grade 4 or 5 adverse events. The results indicated that raltitrexed-eluting CalliSpheres® beads for TACE can be a viable option for treating patients with intermediate-stage HCC.
Introduction
Worldwide, 905,677 new cases of liver cancer, ranked as the sixth most frequently diagnosed cancer and the third leading cause of cancer-related fatalities, were diagnosed in 2020, leading to 830,180 deaths. 1 Majority of these cases (75–85%) were hepatocellular carcinomas (HCCs). Owing to the lack of early-stage symptoms, most patients with HCC are diagnosed at an intermediate stage [Barcelona Clinic Liver Cancer (BCLC) stage B] or an advanced stage (BCLC stage C), precluding curative therapies such as resection, liver transplantation, and ablation. In patients with intermediate-stage HCC, transarterial chemoembolization (TACE) is the established first-line treatment. 2 In patients with unresectable HCC, TACE improves survival compared with supportive care or systemic chemotherapy.3,4 However, the overall survival (OS) rate remains low and the ideal TACE approach remains undetermined.
Drug-eluting bead transarterial chemoembolization (DEB-TACE) is an emerging therapeutic strategy. Drug-eluting beads (DEBs) facilitate the gradual and continuous release of loaded chemotherapeutic drugs into the tumor region, maintaining a high local drug concentration. CalliSpheres® beads (Jiangsu Hengrui Medicine Co. Ltd, Jiangsu, China) are microspheres used for DAB-TACE with a network structure and negative charge, primarily responsible for the adsorption of positively charged antitumor drugs through ion exchange.
Commonly used chemotherapeutic drugs for DEB-TACE include doxorubicin, epirubicin, pirarubicin, irinotecan, and idarubicin. Raltitrexed, an antimetabolic drug, effectively controls cancer by inhibiting thymidylate synthase (TS) and has shown satisfactory effects in HCC treatment.5–7 Unlike 5-fluorouracil (5-FU), which is a TS inhibitor widely used in conventional transarterial chemoembolization (cTACE), raltitrexed definitively inhibits TS activity in a brief infusion, leading to a more effective dose–response relationship than that by 5-FU, making it a promising candidate for TACE. In both in vitro and animal experiments, CalliSpheres® beads exhibited effective loadability and an acceptable release profile for eluting raltitrexed. 8 However, no study has assessed the efficacy and safety of raltitrexed-loaded DEB-TACE in patients with intermediate-stage HCC. Hence, in this study, we aimed to assess the efficacy and safety of raltitrexed-loaded DEB-TACE in patients with intermediate-stage HCC.
Materials and methods
This single-arm, single-center, prospective study, conducted at the First Affiliated Hospital of Zhengzhou University, was registered on 25 June 2019, at www.chictr.org.cn (ChiCTR 1900024097). The reporting of this study conforms to the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) statement 9 (Supplemental File).
Study population
HCC was diagnosed based on clinical and pathological findings according to the guidelines of the American Association for the Study of Liver Diseases. 10 Patients who were unsuitable for curative therapies (resection, liver transplantation, or ablation) or for whom these therapies were contraindicated were enrolled in this trial. Prior to enrollment, patients were required to meet all inclusion criteria while ensuring that they did not meet the exclusion criteria (Table 1). Patients were enrolled between June 2019 and June 2021, with the last follow-up evaluation conducted in June 2022. During the trial, 143 patients were screened, of whom 47 were excluded: 36 failed to meet the inclusion criteria and 11 declined to participate. Of the 36 patients who failed to meet the inclusion criteria, 15 had tumor nodes ⩾5, 11 had a total tumor size ⩾10 cm, 5 had severe coagulation dysfunction, 3 had severe infection, and 2 had a major arterio-portal shunt. Two patients failed to complete the first TACE cycle: one owing to the inability to catheterize a tumor-feeding artery and the other owing to a high-flow arteriohepatic venous fistula. The remaining 94 patients underwent at least one cycle of raltitrexed-loaded DEB-TACE (Figure 1). The average age during enrollment for these 94 patients was 58.3 ± 9.0 years, with 70.2% being male. Among them, 84 patients (89.4%) were classified as Child–Pugh class A and 10 (10.6%) as class B. The median serum alpha-fetoprotein level was 317.4 ng/mL (2.3–13,902 ng/mL). In approximately 43.6% of patients, both the left and right liver lobes were involved, whereas in the remaining 56.4%, only one lobe was involved. The baseline demographics are summarized in Table 2. At the time of trial closure, two patients underwent curative therapy after downstaging (one resection and one percutaneous thermal ablation), nine patients were lost to follow-up, and two patients withdrew their consent. Finally, 81 of the initial 94 patients were included in the final efficacy analysis.
Inclusion and exclusion criteria.

Major arterio-portal shunt: The main portal vein and its branches are visible in the early arterial phase, but the hepatic artery distal to the fistula is pale or even invisible.
BCLC, Barcelona Clinic Liver Cancer; ECOG, Eastern Cooperative Oncology Group;

Flow chart summary of the study.
Baseline demographic and clinical characteristics.

AFP, alpha-fetoprotein; ALBi grade,albumin-bilirubin grade; ECOG, Eastern Cooperative Oncology Group.
Raltitrexed-loaded DEB-TACE
Prior to the DEB-TACE procedure, the Pharmacy Department aseptically loaded a vial of CalliSpheres® beads with 4 mg of raltitrexed. CalliSpheres® beads ranging 300–500 μm were used for lesions >5 cm and 100–300 μm were used for lesions ⩽5 cm. Following hepatic angiography using a 5F RH (5 French Rosch Hepatic) catheter through the femoral artery, a microcatheter (2.4F–2.8F) was selectively catheterized into the feeding artery of the target tumor. Raltitrexed-eluting CalliSpheres® beads were embolized into the tumor-feeding artery at a rate of no more than 1 mL/min under fluoroscopy guidance. The embolization endpoint was attained when the contrast agent was ambiguous, within three to four heartbeats on angiography. Hepatic angiography was performed again after 5 min to detect incomplete embolization. If the tumor still exhibited a blushed appearance, the embolization procedure was repeated with no more than two DEB vials for each session.
Efficacy and safety analyses
Post-TACE follow-up was necessary for at least 1 year or until death. Postoperatively, the patients received monthly follow-ups for the initial 3 months, which was then shifted to every 3 months. Furthermore, DEB-TACE was performed ‘on-demand.’
Imaging analysis: Imaging was performed at baseline and subsequent follow-up visits using magnetic resonance imaging (MRI) or computed tomography (CT). CT scans were performed in three phases: non-contrast, arterial, and portal. MRI included T1 fat-suppressed images, T2 fat-suppressed sequences, and dynamic fat-suppressed T1 sequences with gadolinium enhancement. All imaging measurements were performed by two radiologists specializing in CT or MRI. In case of discrepancies, a consensus was reached through discussion.
Response analysis: Tumor response was assessed through multiphase MRI or CT using the modified response evaluation criteria in solid tumors (mRECIST). 11 Overall response was categorized as complete response (CR), partial response (PR), stable disease (SD), or progressive disease.
Outcome definition: The disease control rate (DCR) was defined as the sum of the CR, PR, and SD. Objective response rate (ORR) was defined as the percentage of patients who achieved CR or PR. Time to progression (TTP) was calculated from the initial treatment to the date of disease progression. Progression-free survival (PFS) was calculated from the initiation of initial treatment to the date of disease progression or death. OS was defined as the period from initial treatment to death from any cause. The primary endpoint was OS and the secondary endpoints were TTP, PFS, ORR, and adverse events (AEs).
Adverse events: All procedure-related AEs were recorded. Serious adverse events (SAEs) can occur at any point during the observation period, resulting in death, immediate life-threatening conditions, prolonged hospitalization, unscheduled hospital visits, permanent or significant disability, or incapacity. 12 AEs were graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events, version 5.0.
Statistical analysis
Statistical analyses were performed using the statistical package for the social sciences software (version 23.0; IBM, New York, NY, USA). Categorical variables are presented as numbers and percentages, whereas continuous variables are described as median and 95% confidence interval (CI) or (mean ± standard deviation). Survival curves for TTP, PFS, and OS were analyzed using the Kaplan–Meier method and described as medians with 95% CIs. Univariate and multivariate Cox proportional hazard analyses were used to assess the prognostic role of baseline patient characteristics. Variables associated with OS (p < 0.10) in the univariate analysis were included in the multivariate regression model. A two-sided p < 0.05 was considered a significant difference.
Results
Study population
Ninety-four patients underwent 285 sessions of raltitrexed-loaded DEB-TACE in total, with a median of three sessions (range, 1–8). Among these patients, raltitrexed-loaded DEB-TACE sessions were received as follows: 3 (3.2%) had one session, 24 (25.5%) had two sessions, 44 (46.8%) had three sessions, 17 (18.1%) had four sessions, and 6 (6.4%) had more than four sessions, respectively. The bead sizes in the raltitrexed-loaded DEB-TACE sessions were 100–300 μm (88.3%) and 300–500 μm (11.7%). Only two patients (2.1%) required more than one vial of DEBs.
Efficacy
The median follow-up period of the entire series was 22.3 months (range, 4.2–35 months). Based on the mRECIST criteria, the 6-month ORR and DCR were 90.1% and 93.8%, respectively. Of the 81 patients, 10 (12.3%) had CR, 63 (77.8%) had PR, and 3 (3.7%) had SD at the 6-month mark, respectively. Owing to gastrointestinal bleeding, a patient died 4.2 months after the first cycle of raltitrexed-loaded DEB-TACE (Table 3).
Six-month responses.

CR, complete response; DCR, disease control rate; ORR, objective response rate; PD, progressive disease; PR, partial response; SD, stable disease.
In all, 20 patients died during the data collection period. Potential causes of death included liver failure (n = 10), gastrointestinal hemorrhage (n = 4), pulmonary embolism (n = 1), renal failure (n = 1), heart failure (n = 1), and unknown causes (n = 3). The median OS was 33.0 months (95% CI, 29.0–36.9 months) [Figure 2(a)]. The survival rates at 1, 2, and 3 years were 95.1%, 82.1%, and 43.6%, respectively. In univariate regression analysis, Child–Pugh class, bilobar disease occurrence, and a number of nodules were independently and adversely associated with survival (p < 0.1). Among these variables, Child–Pugh class and bilobar disease occurrence were confirmed by multivariate analysis (p < 0.05) (Table 4). Tumor progression was observed in 46 of 81 patients. Five patients had portal vein invasion and eight had extrahepatic spread (four lung metastases, three lymph node metastases, and one adrenal gland metastasis). The median TTP was 22.7 months (95% CI, 19.4–26.0 months) [Figure 2(b)], whereas the median PFS was 19.8 months (95% CI, 15.8–23.8 months) [Figure 2(c)].

Plots depicting the efficacy results in 81 study participants treated with raltitrexed-loaded DEB-TACE. Kaplan–Meier curve and median 95% CI estimates of (a) OS, (b) PFS, and (c) TTP.
Univariate and multivariate regression models on patient survival.
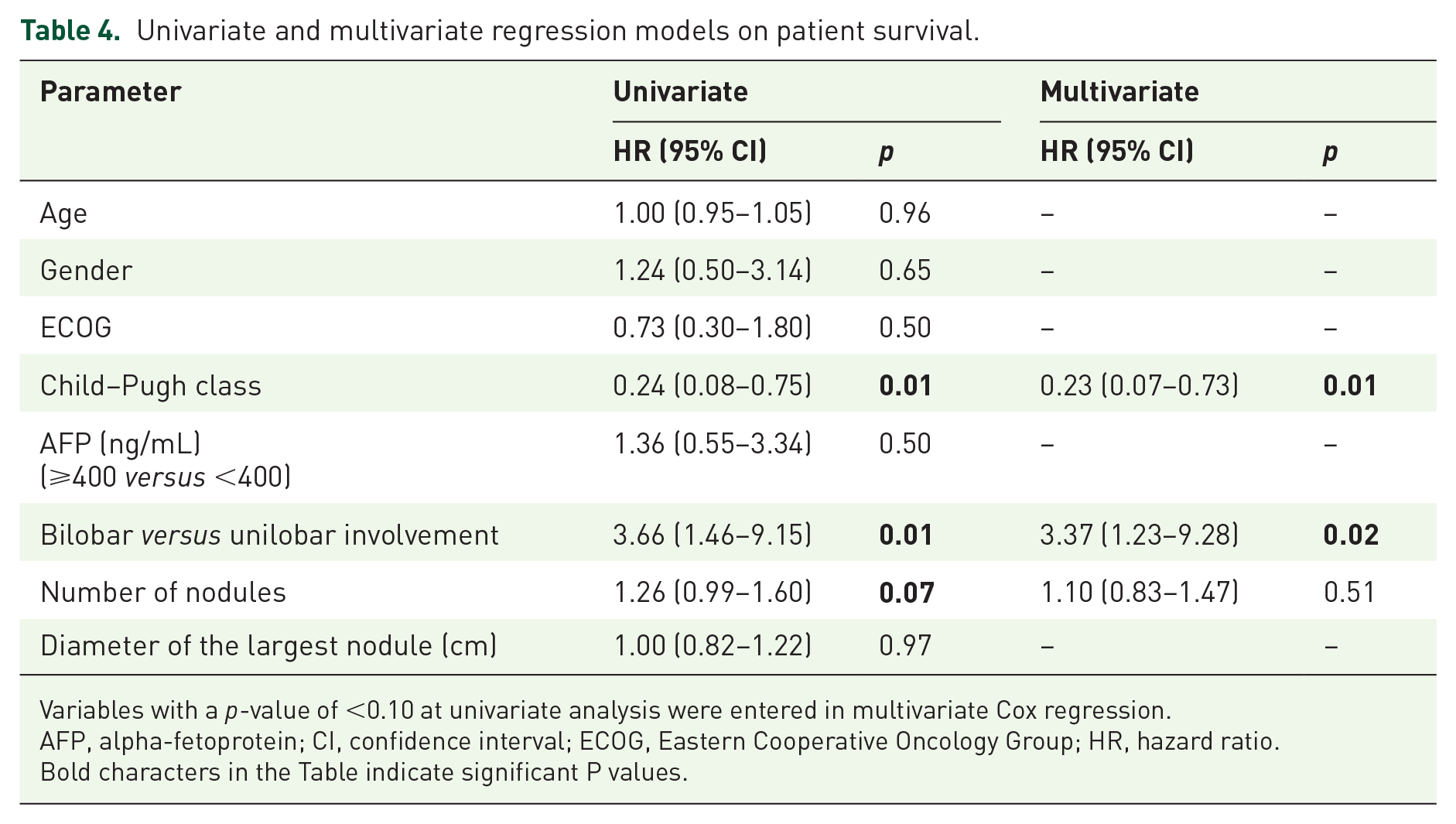
Variables with a p-value of <0.10 at univariate analysis were entered in multivariate Cox regression.
AFP, alpha-fetoprotein; CI, confidence interval; ECOG, Eastern Cooperative Oncology Group; HR, hazard ratio.
Bold characters in the Table indicate significant P values.
Safety
The observed AEs are listed in Table 5. The safety evaluation included 94 patients who underwent at least one cycle of raltitrexed-loaded DEB-TACE. There were no procedure-related mortalities during the 285 TACE sessions. None of the patients withdrew from the study because of AEs. The most common AE was post-embolization syndrome, including abdominal pain (53.2%), nausea or vomiting (28.7%), and fatigue (14.9%). The most common laboratory abnormalities were elevated alanine aminotransferase (ALT) (52.1%) and aspartate aminotransferase (AST) (51.1%) levels. The majority of AEs were of grade 1 or 2. Of the 94 participants, 11 (11.7%) experienced at least one grade 3 AE, potentially attributable to the raltitrexed-loaded DEB-TACE sessions. Notably, no grade 4 or 5 AEs were reported. SAEs were documented in five patients (5.3%), which were attributed to post-embolization syndrome. All these AEs were resolved using conservative treatment.
Adverse events are possibly attributable to TACE sessions.

ALT, alanine aminotransferase; AST, aspartate aminotransferase.
Discussion
In patients with intermediate-stage HCC, we discovered that raltitrexed-loaded DEB-TACE led to a 6-month ORR of 90.1%, a median OS of 33.0 months, a median TTP of 22.7 months, and a median PFS of 19.8 months. The 1-, 2-, and 3-year survival rates were 95.1%, 82.1%, and 43.6%, respectively. The phase III trial conducted by the PRECISION Italia Study Group reported that after doxorubicin-loaded DEB-TACE, the 1- and 2-year survival rates were 86.2% and 56.8%, the median survival was 29 months, and the median TTP was 9 months, respectively. 13 In 2016, a retrospective cohort of 249 patients with unresectable HCC revealed that the doxorubicin-loaded DEB-TACE group achieved an ORR of 74.8% and a median survival of 32 months. 14 In a study by Gomes et al., 15 patients with intermediate-stage HCC treated with doxorubicin-loaded DEB-TACE had a mean survival of 31.9 months. Thus, the results of our study indicate that the efficacy profile of raltitrexed-loaded DEB-TACE is comparable to that of doxorubicin-loaded DEB-TACE.
Regarding the safety of raltitrexed-loaded DEB-TACE, the three most frequently observed AEs were abdominal pain (53.2%) and elevated ALT (52.1%) and AST (51.0%) levels. Eleven (11.7%) participants experienced at least one grade 3 AE, and none experienced grade 4 or 5 AEs. SAEs were reported in five patients (5.3%). In the PRECISION Italia Study Group phase III trial, the most common AEs was post-procedural pain, with a 6% incidence of SAEs in 89 patients who underwent doxorubicin-loaded DEB-TACE. 13 In a single-arm phase II trial by Guiu et al., 16 grade 3 and 4 AEs were observed in 75% of patients who underwent idarubicin-loaded DEB-TACE, with elevated ALT and AST levels being the most common among them. Zhao et al. reported a 12% incidence of grade 3 or 4 AEs in raltitrexed-based cTACE. The most common grade 3 and 4 AEs included elevated AST levels and abdominal pain. 17 The overall incidence of grade 3 and 4 toxicities was lower than that reported in a previous study of DEB-TACE loaded with other drugs and raltitrexed-based cTACE.
TACE has long been used as the standard treatment for patients with intermediate-stage HCC and has evolved steadily over the years. DEB-TACE was developed in the late 2000s and has been widely used as one of the most important treatments for unresectable HCC. 18 Owing to their significantly controlled pharmacokinetics, DEBs enable the sustained local release of drugs, extend their therapeutic effect, reduce peripheral concentrations, and minimize systemic adverse effects. Furthermore, substantial research has been conducted to determine the DEB-TACE values for HCC treatment. The PRECISION V phase II randomized controlled trial demonstrated that DEB-TACE displayed a reduced incidence of systemic toxicity compared with cTACE. A superior objective response was observed in patients with advanced disease (Child–Pugh B, Eastern Cooperative Oncology Group 1, and bilobar or recurrent disease). 19 Greco et al. 20 and Richter et al. 21 achieved similar promising results in terms of tumor response.
Raltitrexed, a next-generation TS inhibitor, is primarily used for the treatment of advanced colorectal cancer. The application of raltitrexed in TACE offers several advantages. (1) Being a direct and specific TS inhibitor, raltitrexed has low toxicity, high efficacy, and mild adverse reactions.22,23 (2) Raltitrexed exhibits a longer half-life and a favorable dose–response relationship than other chemotherapy drugs of the same type. (3) Cardiotoxicity, a well-known AE, is associated with anthracyclines and 5-FU, which are commonly used agents for TACE. Raltitrexed demonstrated no cardiac toxicity and may be more suitable for patients with cardiologic risk factors.24–26 Recent studies have also explored the safety and efficacy of raltitrexed for cTACE. A multicenter real-world study enrolled 184 patients with intermediate and advanced HCC, half of whom were treated with raltitrexed + lobaplatin + pirarubicin-based cTACE, while the others were treated with lobaplatin + pirarubicin-based cTACE. The 6-month, 1-year, and 2-year OS rates in the raltitrexed group were significantly higher than those in the control group (78.2% versus 60.9%, 43.5% versus 22.8%, and 17.4% versus 2.2%), respectively. 6 In a study by Zhao et al., 17 raltitrexed + oxaliplatin-based TACE resulted in a better ORR and OS than 5-FU + oxaliplatin or doxorubicin + oxaliplatin-based TACE in patients with unresectable HCC.
This study has limitations because of its single-center, single-arm nature, and a relatively small study population, potentially introducing the confounding effect of selection bias. The absence of a parallel control group necessitated the use of external historical data to assess the effectiveness and safety comparisons. The research design of this study made it challenging to obtain entirely consistent historical data, leading to the inability to accurately assess these results. Therefore, large, multicenter, prospective, randomized controlled trials are required to validate our results. In addition, clinical trials are required to determine the superiority of raltitrexed in DEB-TACE compared to other chemotherapeutic agents.
Conclusion
In conclusion, this single-center single-arm prospective study showed that raltitrexed-loaded DEB-TACE is safe, feasible, and effective in patients with intermediate-stage HCC. However, additional studies are required to compare the efficacy of raltitrexed-eluting CalliSpheres® beads compared to doxorubicin-eluting beads.
Supplemental Material
sj-docx-1-tam-10.1177_17588359241229661 – Supplemental material for Efficacy and safety of raltitrexed-eluting CalliSpheres® bead transarterial chemoembolization in patients with intermediate-stage hepatocellular carcinoma: a single-arm, prospective study
Supplemental material, sj-docx-1-tam-10.1177_17588359241229661 for Efficacy and safety of raltitrexed-eluting CalliSpheres® bead transarterial chemoembolization in patients with intermediate-stage hepatocellular carcinoma: a single-arm, prospective study by Zhanguo Sun, Dechao Jiao, Yi Fang, Yiming Liu, Kaihao Xu, Chengzhi Zhang, Yuanhao Huang and Xinwei Han in Therapeutic Advances in Medical Oncology
Footnotes
Acknowledgements
We express our sincere gratitude to the patients and their families who participated in this study. We also extend our heartfelt gratitude to our esteemed colleagues in the Departments of Hepatobiliary Surgery, Oncology, Pathology, Radiology, and Hepatology for their support and valuable consultations throughout the patient’s treatment.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
